
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | xuejia kang | -- | 2737 | 2023-06-05 18:52:13 | | | |
| 2 | Peter Tang | + 3 word(s) | 2740 | 2023-06-06 04:41:15 | | | | |
| 3 | Peter Tang | -39 word(s) | 2701 | 2023-06-09 04:31:35 | | |
Video Upload Options
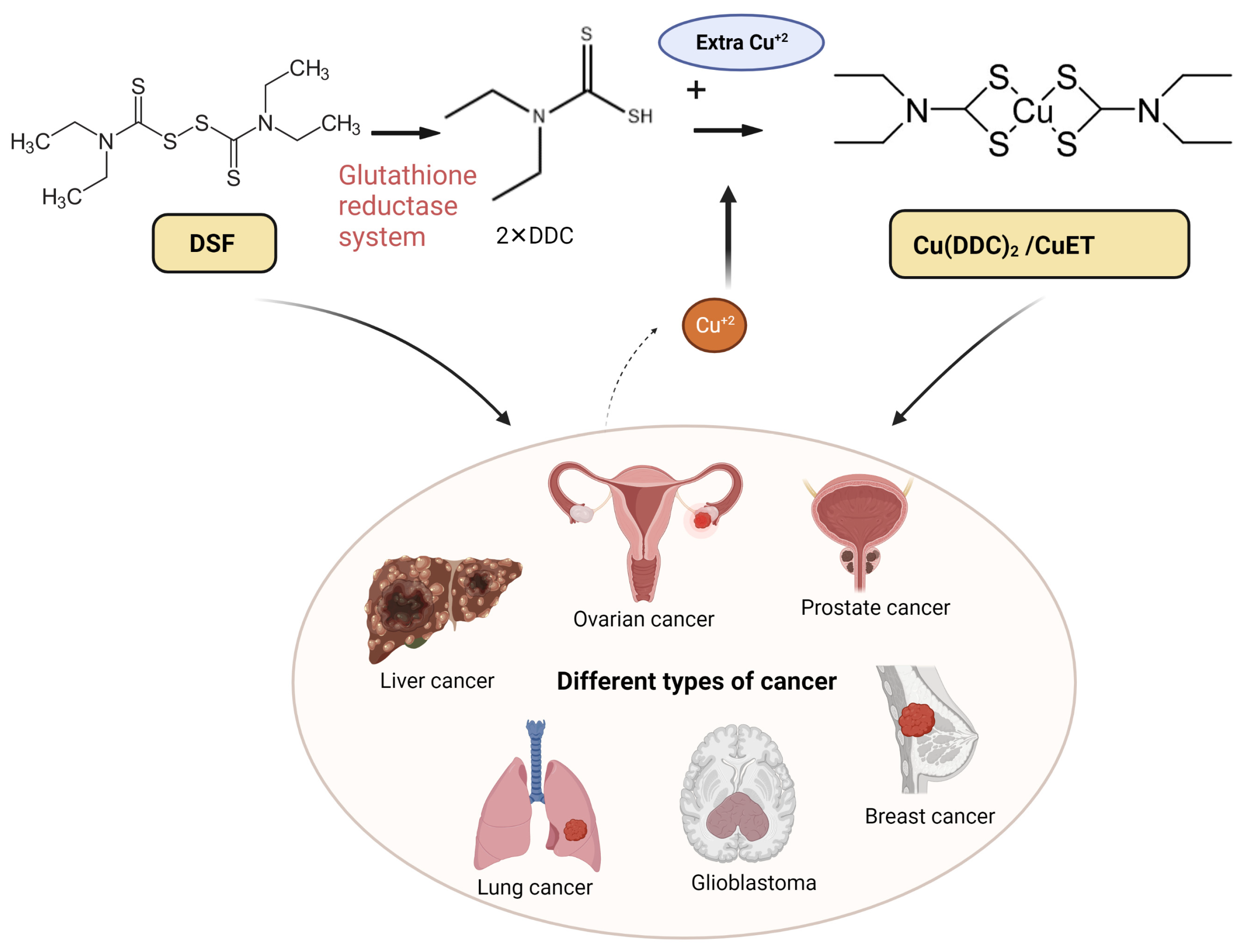
Disulfiram (DSF) is a thiocarbamate based drug. Preclinical studies have shown that DSF has anticancer efficacy, and its supplementation with copper (CuII) significantly potentiates the efficacy of DSF. However, the results of clinical trials have not yielded promising results. The elucidation of the anticancer mechanisms of DSF/Cu (II) will be beneficial in repurposing DSF as a new treatment for certain types of cancer. DSF’s anticancer mechanism is primarily due to its generating reactive oxygen species, inhibiting aldehyde dehydrogenase (ALDH) activity inhibition, and decreasing the levels of transcriptional proteins. DSF also shows inhibitory effects in cancer cell proliferation, the self-renewal of cancer stem cells (CSCs), angiogenesis, drug resistance, and suppresses cancer cell metastasis.
1. Introduction
|
Cancer |
Status |
Clinical Identifier (Clinicaltrials.Gov) |
|---|---|---|
|
Metastatic melanoma |
Phase I, Terminated |
NCT00571116 |
|
Melanoma |
Phase I/II, Completed |
NCT00256230 |
|
Melanoma |
Phase II, Completed |
NCT02101008 |
|
Prostate Cancer |
Phase I, Completed |
NCT01118741 |
|
Prostate Cancer |
Phase I, Recruiting |
NCT02963051 |
|
Breast Cancer (Metastatic) |
Phase II, Recruiting |
NCT03323346 |
|
Refractory Breast Cancer (Metastatic) |
Phase II, Recruiting |
NCT04265274 |
|
Pancreatic Cancer (Metastatic, Recurrent) |
Phase I, Recruiting |
NCT02671890 |
|
Pancreatic Cancer (Metastatic) |
Phase II, Not Yet Recruiting |
NCT03714555 |
|
Recurrent Glioblastoma |
Phase I, Active, Not Recruiting |
NCT02770378 |
|
Glioma Glioblastoma |
Phase II/III, Recruiting |
NCT02678975 |
|
Glioblastoma Multiforme |
Phase II, Recruiting |
NCT03363659 |
|
Solid Tumors Involving Liver |
Phase I, Completed |
NCT00742911 |
|
Non-small Cell Lung Cancer |
Phase II/III, Completed |
NCT00312819 |
|
Glioblastoma (Recurrent) |
Phase II, Completed |
NCT03034135 |
|
Glioblastoma |
Phase I/II, Recruiting |
NCT02715609 |
|
Glioblastoma |
Phase II, Not Yet Recruiting |
NCT01777919 |
|
Glioblastoma |
Phase II/III, Recruiting |
NCT02678975 |
|
Glioblastoma |
Early Phase I, Recruiting |
NCT03151772 |
|
Glioblastoma |
Early Phase I, Completed |
NCT01907165 |
|
Germ Cell Tumor |
Phase II, Recruiting |
NCT03950830 |
|
Multiple Myeloma |
Phase I, Terminated |
NCT04521335 |
|
Refractory Sarcomas |
Phase I, Recruiting |
NCT05210374 |
|
Advanced Gastric Cancer |
Phase Not Defined, Not Yet Recruiting |
NCT05667415 |
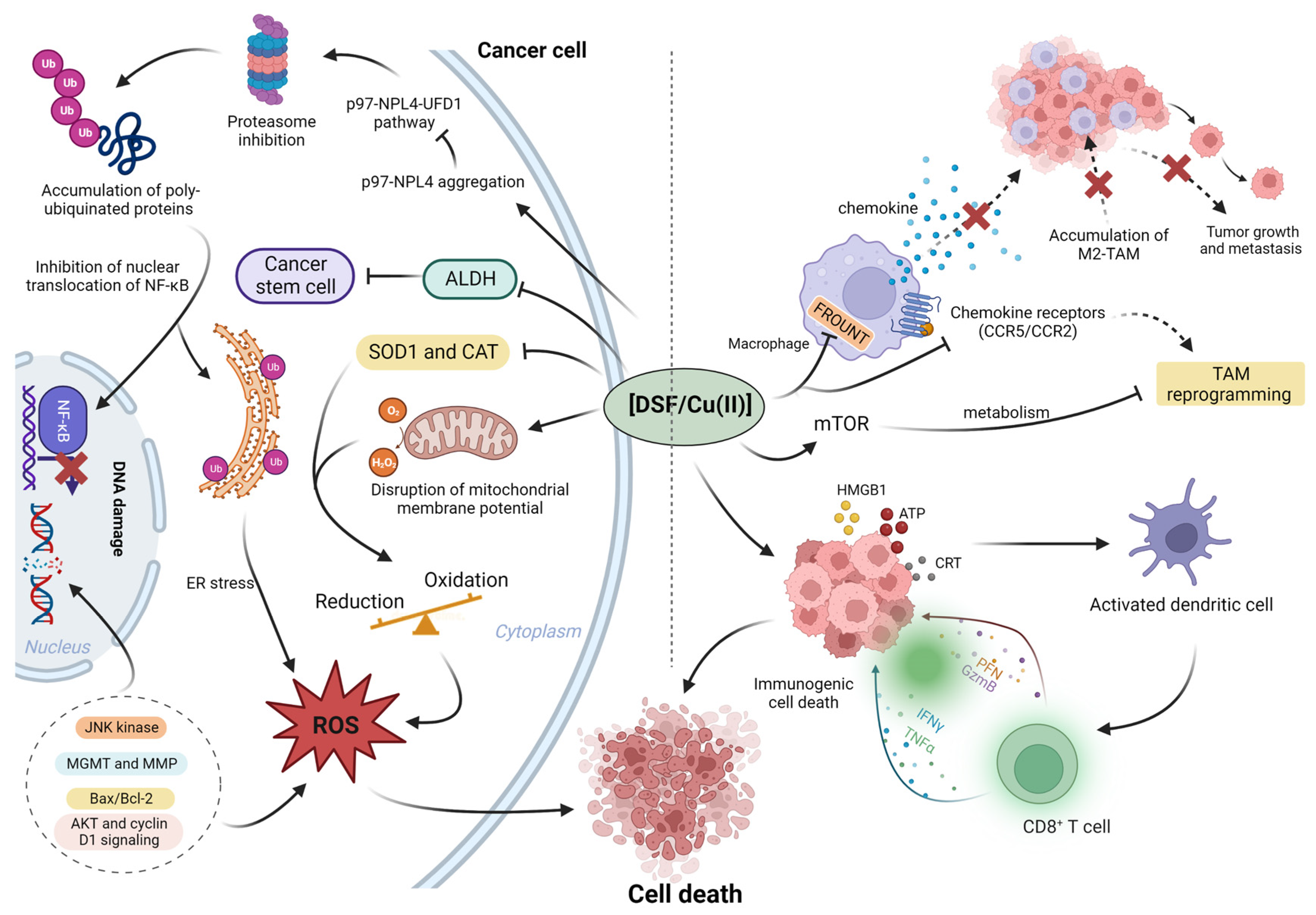
2. Anticancer Mechanisms of DSF/Cu (II)
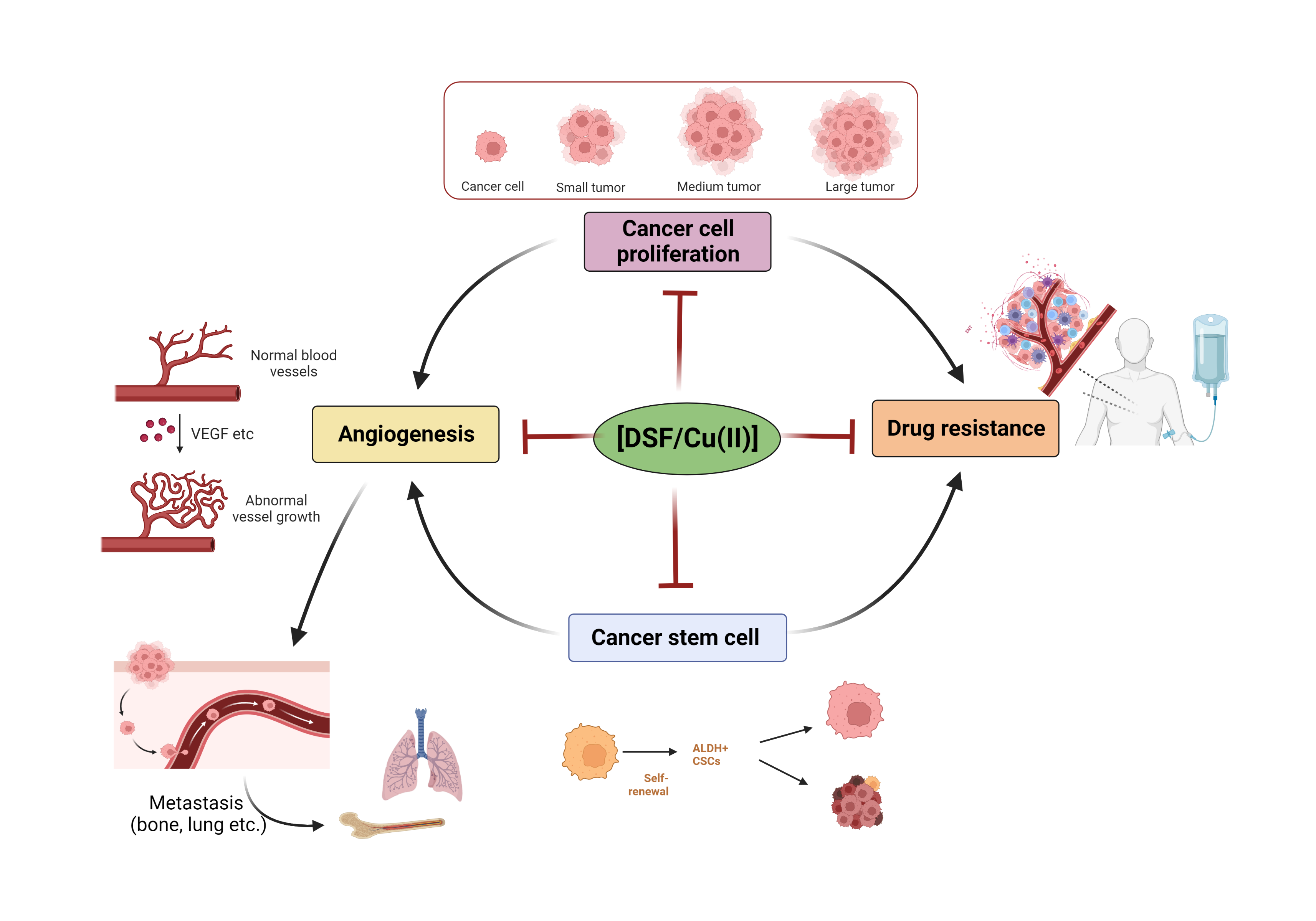
3. The Effect of DSF/Cu (II) on Cancer
3.1. DSF/Cu (II) on the Inhibition of Cancer Proliferation
3.2. DSF/Cu (II) Efficacy in Cancer Stem Cells (CSCs)
3.3. DSF/Cu (II) Effects on the Inhibition of Cancer Angiogenesis
3.4. DSF/Cu (II) Reverses Drug Resistance
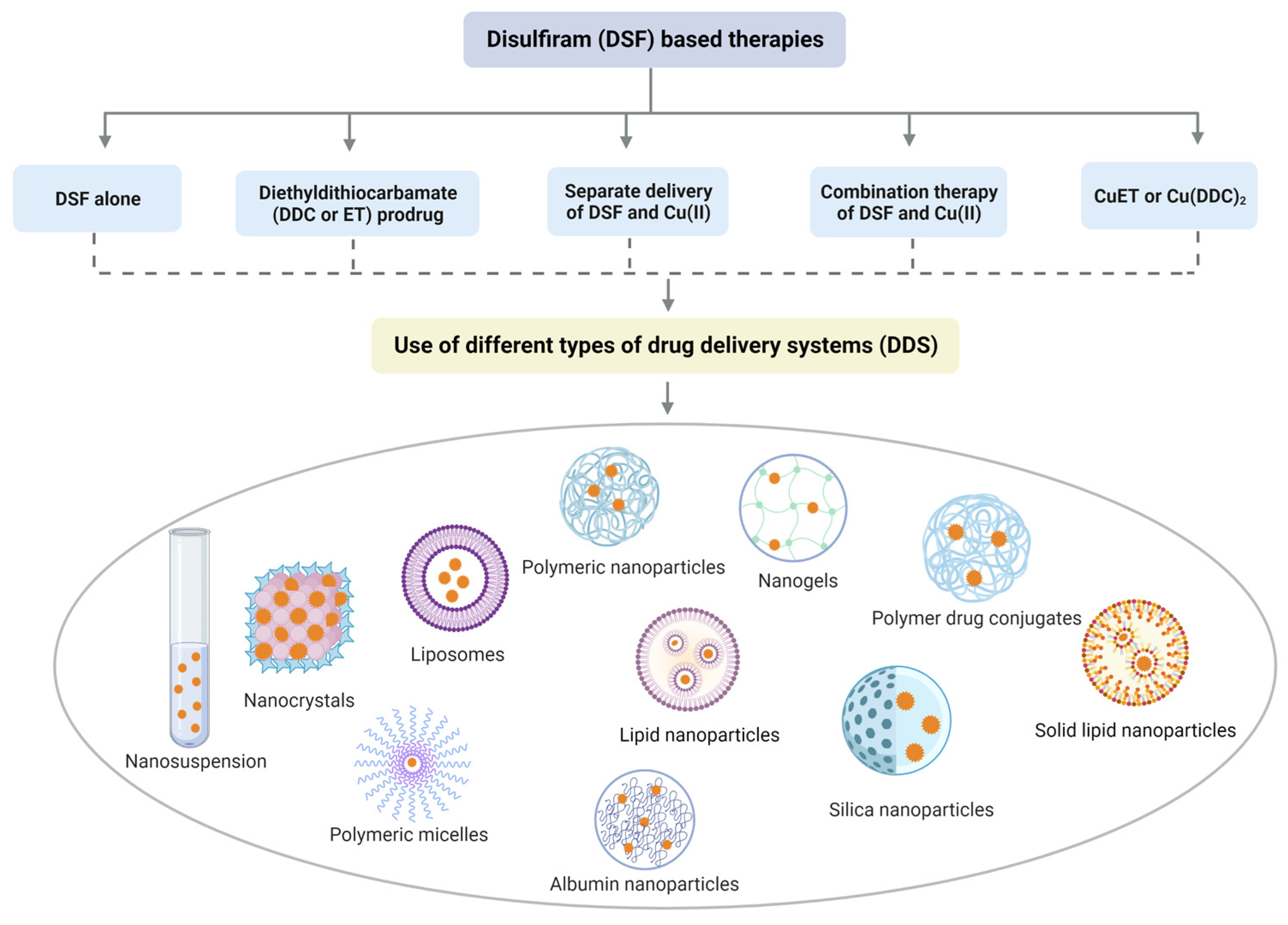
4. DSF-Based Therapies for the Treatment of Cancer
References
- Zhang, Z.; Zhou, L.; Xie, N.; Nice, E.C.; Zhang, T.; Cui, Y.; Huang, C. Overcoming cancer therapeutic bottleneck by drug repurposing. Signal Transduct. Target. Ther. 2020, 5, 113.
- Chong, C.R.; Sullivan, D.J. New uses for old drugs. Nature 2007, 448, 645–646.
- Corsello, S.M.; Nagari, R.T.; Spangler, R.D.; Rossen, J.; Kocak, M.; Bryan, J.G.; Humeidi, R.; Peck, D.; Wu, X.; Tang, A.A. Discovering the anticancer potential of non-oncology drugs by systematic viability profiling. Nat. Cancer 2020, 1, 235–248.
- Ma, C.; Peng, Y.; Li, H.; Chen, W. Organ-on-a-chip: A new paradigm for drug development. Trends Pharmacol. Sci. 2021, 42, 119–133.
- Lowndes, S.A.; Harris, A.L. The role of copper in tumour angiogenesis. J. Mammary Gland. Biol. Neoplasia 2005, 10, 299–310.
- Antoniades, V.; Sioga, A.; Dietrich, E.M.; Meditskou, S.; Ekonomou, L.; Antoniades, K. Is copper chelation an effective anti-angiogenic strategy for cancer treatment? Med. Hypotheses 2013, 81, 1159–1163.
- Wang, X.; Zhou, M.; Liu, Y.; Si, Z. Cope with copper: From copper linked mechanisms to copper-based clinical cancer therapies. Cancer Lett. 2023, 561, 216157.
- da Silva, D.A.; De Luca, A.; Squitti, R.; Rongioletti, M.; Rossi, L.; Machado, C.M.L.; Cerchiaro, G. Copper in tumors and the use of copper-based compounds in cancer treatment. J. Inorg. Biochem. 2022, 226, 111634.
- Tsvetkov, P.; Coy, S.; Petrova, B.; Dreishpoon, M.; Verma, A.; Abdusamad, M.; Rossen, J.; Joesch-Cohen, L.; Humeidi, R.; Spangler, R.D. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science 2022, 375, 1254–1261.
- Ghosh, P.; Vidal, C.; Dey, S.; Zhang, L. Mitochondria targeting as an effective strategy for cancer therapy. Int. J. Mol. Sci. 2020, 21, 3363.
- Oliveri, V. Selective targeting of cancer cells by copper ionophores: An overview. Front. Mol. Biosci. 2022, 9, 841814.
- Xie, J.; Yang, Y.; Gao, Y.; He, J. Cuproptosis: Mechanisms and links with cancers. Mol. Cancer 2023, 22, 46.
- Johansson, B. A review of the pharmacokinetics and pharmacodynamics of disulfiram and its metabolites. Acta Psychiatr. Scand. 1992, 86, 15–26.
- Mittal, M.; Bhagwati, S.; Siddiqi, M.I.; Chattopadhyay, N. A critical assessment of the potential of pharmacological modulation of aldehyde dehydrogenases to treat the diseases of bone loss. Eur. J. Pharmacol. 2020, 886, 173541.
- Swift, R.; Davidson, D. Alcohol hangover: Mechanisms and mediators. Alcohol Health Res. World 1998, 22, 54–60.
- Yoshida, A.; Hsu, L.C.; Davé, V. Retinal oxidation activity and biological role of human cytosolic aldehyde dehydrogenase. Enzyme 1992, 46, 239–244.
- Lewison, E.F. Spontaneous regression of breast cancer. Natl. Cancer Inst. Monogr. 1976, 44, 23–26.
- López-Lázaro, M. Dual role of hydrogen peroxide in cancer: Possible relevance to cancer chemoprevention and therapy. Cancer Lett. 2007, 252, 1–8.
- Schmitt, S.M.; Frezza, M.; Dou, Q.P. New applications of old metal-binding drugs in the treatment of human cancer. Front. Biosci. 2012, 4, 375.
- Wang, W.; McLeod, H.L.; Cassidy, J. Disulfiram-mediated inhibition of NF-κB activity enhances cytotoxicity of 5-fluorouracil in human colorectal cancer cell lines. Int. J. Cancer 2003, 104, 504–511.
- Xu, B.; Shi, P.; Fombon, I.S.; Zhang, Y.; Huang, F.; Wang, W.; Zhou, S. Disulfiram/copper complex activated JNK/c-jun pathway and sensitized cytotoxicity of doxorubicin in doxorubicin resistant leukemia HL60 cells. Blood Cells Mol. Dis. 2011, 47, 264–269.
- Yip, N.C.; Fombon, I.S.; Liu, P.; Brown, S.; Kannappan, V.; Armesilla, A.L.; Xu, B.; Cassidy, J.; Darling, J.L.; Wang, W. Disulfiram modulated ROS–MAPK and NFκB pathways and targeted breast cancer cells with cancer stem cell-like properties. Br. J. Cancer 2011, 104, 1564–1574.
- Roudi, R.; Korourian, A.; Shariftabrizi, A.; Madjd, Z. Differential expression of cancer stem cell markers ALDH1 and CD133 in various lung cancer subtypes. Cancer Investig. 2015, 33, 294–302.
- Koh, H.K.; Seo, S.Y.; Kim, J.H.; Kim, H.J.; Chie, E.K.; Kim, S.-K.; Kim, I.H. Disulfiram, a re-positioned aldehyde dehydrogenase inhibitor, enhances radiosensitivity of human glioblastoma cells in vitro. Cancer Res. Treat. Off. J. Korean Cancer Assoc. 2019, 51, 696–705.
- Chen, W.; Yang, W.; Chen, P.; Huang, Y.; Li, F. Disulfiram copper nanoparticles prepared with a stabilized metal ion ligand complex method for treating drug-resistant prostate cancers. ACS Appl. Mater. Interfaces 2018, 10, 41118–41128.
- Wang, N.-n.; Wang, L.-H.; Li, Y.; Fu, S.-Y.; Xue, X.; Jia, L.-N.; Yuan, X.-Z.; Wang, Y.-T.; Tang, X.; Yang, J.-Y. Targeting ALDH2 with disulfiram/copper reverses the resistance of cancer cells to microtubule inhibitors. Exp. Cell Res. 2018, 362, 72–82.
- Liu, X.; Wang, L.; Cui, W.; Yuan, X.; Lin, L.; Cao, Q.; Wang, N.; Li, Y.; Guo, W.; Zhang, X. Targeting ALDH1A1 by disulfiram/copper complex inhibits non-small cell lung cancer recurrence driven by ALDH-positive cancer stem cells. Oncotarget 2016, 7, 58516–58530.
- Denoyer, D.; Pearson, H.B.; Clatworthy, S.A.S.; Smith, Z.M.; Francis, P.S.; Llanos, R.M.; Volitakis, I.; Phillips, W.A.; Meggyesy, P.M.; Masaldan, S. Copper as a target for prostate cancer therapeutics: Copper-ionophore pharmacology and altering systemic copper distribution. Oncotarget 2016, 7, 37064–37080.
- Orlov, A.P.; Orlova, M.A.; Trofimova, T.P.; Kalmykov, S.N.; Kuznetsov, D.A. The role of zinc and its compounds in leukemia. JBIC J. Biol. Inorg. Chem. 2018, 23, 347–362.
- Gupte, A.; Mumper, R.J. Elevated copper and oxidative stress in cancer cells as a target for cancer treatment. Cancer Treat. Rev. 2009, 35, 32–46.
- Chen, D.; Dou, Q.P. New uses for old copper-binding drugs: Converting the pro-angiogenic copper to a specific cancer cell death inducer. Expert Opin. Ther. Targets 2008, 12, 739–748.
- Chang, Y.; Jiang, J.; Chen, W.; Yang, W.; Chen, L.; Chen, P.; Shen, J.; Qian, S.; Zhou, T.; Wu, L. Biomimetic metal-organic nanoparticles prepared with a 3D-printed microfluidic device as a novel formulation for disulfiram-based therapy against breast cancer. Appl. Mater. Today 2020, 18, 100492.
- Lu, C.; Li, X.; Ren, Y.; Zhang, X. Disulfiram: A novel repurposed drug for cancer therapy. Cancer Chemother. Pharmacol. 2021, 87, 159–172.
- Skrott, Z.; Mistrik, M.; Andersen, K.K.; Friis, S.; Majera, D.; Gursky, J.; Ozdian, T.; Bartkova, J.; Turi, Z.; Moudry, P. Alcohol-abuse drug disulfiram targets cancer via p97 segregase adaptor NPL4. Nature 2017, 552, 194–199.
- Brar, S.S.; Grigg, C.; Wilson, K.S.; Holder, W.D., Jr.; Dreau, D.; Austin, C.; Foster, M.; Ghio, A.J.; Whorton, A.R.; Stowell, G.W. Disulfiram inhibits activating transcription factor/cyclic AMP-responsive element binding protein and human melanoma growth in a metal-dependent manner in vitro, in mice and in a patient with metastatic disease. Mol. Cancer Ther. 2004, 3, 1049–1060.
- Wickström, M.; Danielsson, K.; Rickardson, L.; Gullbo, J.; Nygren, P.; Isaksson, A.; Larsson, R.; Lövborg, H. Pharmacological profiling of disulfiram using human tumor cell lines and human tumor cells from patients. Biochem. Pharmacol. 2007, 73, 25–33.
- Li, H.; Wang, J.; Wu, C.; Wang, L.; Chen, Z.-S.; Cui, W. The combination of disulfiram and copper for cancer treatment. Drug Discov. Today 2020, 25, 1099–1108.
- Ren, X.; Li, Y.; Zhou, Y.; Hu, W.; Yang, C.; Jing, Q.; Zhou, C.; Wang, X.; Hu, J.; Wang, L.; et al. Overcoming the compensatory elevation of NRF2 renders hepatocellular carcinoma cells more vulnerable to disulfiram/copper-induced ferroptosis. Redox Biol. 2021, 46, 102122.
- Guo, F.; Yang, Z.; Kulbe, H.; Albers, A.E.; Sehouli, J.; Kaufmann, A.M. Inhibitory effect on ovarian cancer ALDH+ stem-like cells by Disulfiram and Copper treatment through ALDH and ROS modulation. Biomed. Pharmacother. 2019, 118, 109371.
- Zhao, P.; Zhang, J.; Wu, A.; Zhang, M.; Zhao, Y.; Tang, Y.; Wang, B.; Chen, T.; Li, F.; Zhao, Q. Biomimetic codelivery overcomes osimertinib-resistant NSCLC and brain metastasis via macrophage-mediated innate immunity. J. Control. Release 2021, 329, 1249–1261.
- Zheng, Z.; Zhang, J.; Jiang, J.; He, Y.; Zhang, W.; Mo, X.; Kang, X.; Xu, Q.; Wang, B.; Huang, Y. Remodeling tumor immune microenvironment (TIME) for glioma therapy using multi-targeting liposomal codelivery. J. Immunother. Cancer 2020, 8, e000207.
- Morrison, B.W.; Doudican, N.A.; Patel, K.R.; Orlow, S.J. Disulfiram induces copper-dependent stimulation of reactive oxygen species and activation of the extrinsic apoptotic pathway in melanoma. Melanoma Res. 2010, 20, 11–20.
- Liu, P.; Brown, S.; Goktug, T.; Channathodiyil, P.; Kannappan, V.; Hugnot, J.P.; Guichet, P.O.; Bian, X.; Armesilla, A.L.; Darling, J.L. Cytotoxic effect of disulfiram/copper on human glioblastoma cell lines and ALDH-positive cancer-stem-like cells. Br. J. Cancer 2012, 107, 1488–1497.
- Nie, D.; Chen, C.; Li, Y.; Zeng, C. Disulfiram, an aldehyde dehydrogenase inhibitor, works as a potent drug against sepsis and cancer via NETosis, pyroptosis, apoptosis, ferroptosis, and cuproptosis. Blood Sci. 2022, 4, 152–154.
- Huang, J.; Chaudhary, R.; Cohen, A.L.; Fink, K.; Goldlust, S.; Boockvar, J.; Chinnaiyan, P.; Wan, L.; Marcus, S.; Campian, J.L. A multicenter phase II study of temozolomide plus disulfiram and copper for recurrent temozolomide-resistant glioblastoma. J. Neuro-Oncol. 2019, 142, 537–544.
- Huang, J.; Campian, J.L.; Gujar, A.D.; Tsien, C.; Ansstas, G.; Tran, D.D.; DeWees, T.A.; Lockhart, A.C.; Kim, A.H. Final results of a phase I dose-escalation, dose-expansion study of adding disulfiram with or without copper to adjuvant temozolomide for newly diagnosed glioblastoma. J. Neuro-Oncol. 2018, 138, 105–111.
- Nechushtan, H.; Hamamreh, Y.; Nidal, S.; Gotfried, M.; Baron, A.; Shalev, Y.I.; Nisman, B.; Peretz, T.; Peylan-Ramu, N. A phase IIb trial assessing the addition of disulfiram to chemotherapy for the treatment of metastatic non-small cell lung cancer. Oncologist 2015, 20, 366–367.
- Wang, L.; Yu, Y.; Zhou, C.; Wan, R.; Li, Y. Anticancer effects of disulfiram: A systematic review of in vitro, animal, and human studies. Syst. Rev. 2022, 11, 109.
- Oberley, T.D. Oxidative damage and cancer. Am. J. Pathol. 2002, 160, 403–408.
- Cen, D.; Gonzalez, R.I.; Buckmeier, J.A.; Kahlon, R.S.; Tohidian, N.B.; Meyskens, F.L., Jr. Disulfiram induces apoptosis in human melanoma cells: A redox-related process. Mol. Cancer Ther. 2002, 1, 197–204.
- Wu, X.; Xue, X.; Wang, L.; Wang, W.; Han, J.; Sun, X.; Zhang, H.; Liu, Y.; Che, X.; Yang, J. Suppressing autophagy enhances disulfiram/copper-induced apoptosis in non-small cell lung cancer. Eur. J. Pharmacol. 2018, 827, 1–12.
- Zhang, X.; Hu, P.; Ding, S.-Y.; Sun, T.; Liu, L.; Han, S.; DeLeo, A.B.; Sadagopan, A.; Guo, W.; Wang, X. Induction of autophagy-dependent apoptosis in cancer cells through activation of ER stress: An uncovered anti-cancer mechanism by anti-alcoholism drug disulfiram. Am. J. Cancer Res. 2019, 9, 1266–1281.
- Yang, Y.; Deng, Q.; Feng, X.; Sun, J. Use of the disulfiram/copper complex for breast cancer chemoprevention in MMTV-erbB2 transgenic mice. Mol. Med. Rep. 2015, 12, 746–752.
- Cheriyan, V.T.; Wang, Y.; Muthu, M.; Jamal, S.; Chen, D.; Yang, H.; Polin, L.A.; Tarca, A.L.; Pass, H.I.; Dou, Q.P. Disulfiram suppresses growth of the malignant pleural mesothelioma cells in part by inducing apoptosis. PLoS ONE 2014, 9, e93711.
- Lin, Y.; Bai, L.; Chen, W.; Xu, S. The NF-κB activation pathways, emerging molecular targets for cancer prevention and therapy. Expert Opin. Ther. Targets 2010, 14, 45–55.
- Li, Y.; Wang, L.H.; Zhang, H.T.; Wang, Y.T.; Liu, S.; Zhou, W.L.; Yuan, X.Z.; Li, T.Y.; Wu, C.F.; Yang, J.Y. Disulfiram combined with copper inhibits metastasis and epithelial–mesenchymal transition in hepatocellular carcinoma through the NF-κB and TGF-β pathways. J. Cell. Mol. Med. 2018, 22, 439–451.
- Bao, B.; Wang, Z.; Ali, S.; Ahmad, A.; Azmi, A.S.; Sarkar, S.H.; Banerjee, S.; Kong, D.; Li, Y.; Thakur, S. Metformin inhibits cell proliferation, migration and invasion by attenuating CSC function mediated by deregulating miRNAs in pancreatic cancer cells. Cancer Prev. Res. 2012, 5, 355–364.
- Bleau, A.-M.; Hambardzumyan, D.; Ozawa, T.; Fomchenko, E.I.; Huse, J.T.; Brennan, C.W.; Holland, E.C. PTEN/PI3K/Akt Pathway Regulates the Side Population Phenotype and ABCG2 Activity in Glioma Tumor Stem-like Cells. Cell Stem Cell 2009, 4, 226–235.
- Venere, M.; Hamerlik, P.; Wu, Q.; Rasmussen, R.D.; Song, L.A.; Vasanji, A.; Tenley, N.; Flavahan, W.A.; Hjelmeland, A.B.; Bartek, J.; et al. Therapeutic targeting of constitutive PARP activation compromises stem cell phenotype and survival of glioblastoma-initiating cells. Cell Death Differ. 2014, 21, 258–269.
- Petersen, E.N. The pharmacology and toxicology of disulfiram and its metabolites. Acta Psychiatr. Scand. 1992, 86, 7–13.
- Ginestier, C.; Hur, M.H.; Charafe-Jauffret, E.; Monville, F.; Dutcher, J.; Brown, M.; Jacquemier, J.; Viens, P.; Kleer, C.G.; Liu, S. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell 2007, 1, 555–567.
- Moreb, J.S. Aldehyde dehydrogenase as a marker for stem cells. Curr. Stem Cell Res. Ther. 2008, 3, 237–246.
- Duan, X.; Xiao, J.; Yin, Q.; Zhang, Z.; Yu, H.; Mao, S.; Li, Y. Multi-targeted inhibition of tumor growth and lung metastasis by redox-sensitive shell crosslinked micelles loading disulfiram. Nanotechnology 2014, 25, 125102.
- Chiba, T.; Suzuki, E.; Yuki, K.; Zen, Y.; Oshima, M.; Miyagi, S.; Saraya, A.; Koide, S.; Motoyama, T.; Ogasawara, S. Disulfiram eradicates tumor-initiating hepatocellular carcinoma cells in ROS-p38 MAPK pathway-dependent and-independent manners. PLoS ONE 2014, 9, e84807.
- Hothi, P.; Martins, T.J.; Chen, L.; Deleyrolle, L.; Yoon, J.-G.; Reynolds, B.; Foltz, G. High-throughput chemical screens identify disulfiram as an inhibitor of human glioblastoma stem cells. Oncotarget 2012, 3, 1124–1136.
- Triscott, J.; Lee, C.; Hu, K.; Fotovati, A.; Berns, R.; Pambid, M.; Luk, M.; Kast, R.E.; Kong, E.; Toyota, E. Disulfiram, a drug widely used to control alcoholism, suppresses self-renewal of glioblastoma and overrides resistance to temozolomide. Oncotarget 2012, 3, 1112–1123.
- Mimeault, M.; Batra, S.K. Recent advances in the development of novel anti-cancer drugs targeting cancer stem/progenitor cells. Drug Dev. Res. 2008, 69, 415–430.
- Manders, P.; Beex, L.; Tjan-Heijnen, V.C.G.; Geurts-Moespot, J.; Van Tienoven, T.; Foekens, J.A.; Sweep, C.G.J. The prognostic value of vascular endothelial growth factor in 574 node-negative breast cancer patients who did not receive adjuvant systemic therapy. Br. J. Cancer 2002, 87, 772–778.
- Schreier, S.M.; Muellner, M.K.; Steinkellner, H.; Hermann, M.; Esterbauer, H.; Exner, M.; Gmeiner, B.M.K.; Kapiotis, S.; Laggner, H. Hydrogen sulfide scavenges the cytotoxic lipid oxidation product 4-HNE. Neurotox. Res. 2010, 17, 249–256.
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxidative Med. Cell. Longev. 2014, 2014, 360438.
- Chapple, S.J.; Cheng, X.; Mann, G.E. Effects of 4-hydroxynonenal on vascular endothelial and smooth muscle cell redox signaling and function in health and disease. Redox Biol. 2013, 1, 319–331.
- Camaré, C.; Vanucci-Bacqué, C.; Augé, N.; Pucelle, M.; Bernis, C.; Swiader, A.; Baltas, M.; Bedos-Belval, F.; Salvayre, R.; Nègre-Salvayre, A. 4-Hydroxynonenal contributes to angiogenesis through a redox-dependent sphingolipid pathway: Prevention by hydralazine derivatives. Oxidative Med. Cell. Longev. 2017, 2017, 9172741.
- Liu, X.; Sun, X.; Liao, H.; Dong, Z.; Zhao, J.; Zhu, H.; Wang, P.; Shen, L.; Xu, L.; Ma, X. Mitochondrial aldehyde dehydrogenase 2 regulates revascularization in chronic ischemia: Potential impact on the development of coronary collateral circulation. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 2196–2206.
- Marikovsky, M.; Nevo, N.; Vadai, E.; Harris-Cerruti, C. Cu/Zn superoxide dismutase plays a role in angiogenesis. Int. J. Cancer 2002, 97, 34–41.
- Shian, S.-G.; Kao, Y.-R.; Wu, F.Y.-H.; Wu, C.-W. Inhibition of invasion and angiogenesis by zinc-chelating agent disulfiram. Mol. Pharmacol. 2003, 64, 1076–1084.
- Kast, R.E.; Halatsch, M.-E. Matrix Metalloproteinase-2 and-9 in glioblastoma: A trio of old drugs—Captopril, disulfiram and nelfinavir—Are inhibitors with potential as adjunctive treatments in glioblastoma. Arch. Med. Res. 2012, 43, 243–247.
- Li, Y.; Fu, S.-Y.; Wang, L.-H.; Wang, F.-Y.; Wang, N.-N.; Cao, Q.; Wang, Y.-T.; Yang, J.-Y.; Wu, C.-F. Copper improves the anti-angiogenic activity of disulfiram through the EGFR/Src/VEGF pathway in gliomas. Cancer Lett. 2015, 369, 86–96.
- Zhang, L.; Zhou, Y.; Sun, X.; Zhou, J.; Yang, P. CXCL12 overexpression promotes the angiogenesis potential of periodontal ligament stem cells. Sci. Rep. 2017, 7, 10286.
- Wang, X.; Cao, Y.; Zhang, S.; Chen, Z.; Fan, L.; Shen, X.; Zhou, S.; Chen, D. Stem cell autocrine CXCL12/CXCR4 stimulates invasion and metastasis of esophageal cancer. Oncotarget 2017, 8, 36149–36160.
- Ponti, D.; Costa, A.; Zaffaroni, N.; Pratesi, G.; Petrangolini, G.; Coradini, D.; Pilotti, S.; Pierotti, M.A.; Daidone, M.G. Isolation and in vitro propagation of tumorigenic breast cancer cells with stem/progenitor cell properties. Cancer Res. 2005, 65, 5506–5511.
- Beckermann, B.M.; Kallifatidis, G.; Groth, A.; Frommhold, D.; Apel, A.; Mattern, J.; Salnikov, A.V.; Moldenhauer, G.; Wagner, W.; Diehlmann, A. VEGF expression by mesenchymal stem cells contributes to angiogenesis in pancreatic carcinoma. Br. J. Cancer 2008, 99, 622–631.
- O’Brien, A.; Barber, J.E.B.; Reid, S.; Niknejad, N.; Dimitroulakos, J. Enhancement of cisplatin cytotoxicity by disulfiram involves activating transcription factor 3. Anticancer. Res. 2012, 32, 2679–2688.
- Olmo, F.; Urbanová, K.; Rosales, M.J.; Martín-Escolano, R.; Sánchez-Moreno, M.; Marín, C. An in vitro iron superoxide dismutase inhibitor decreases the parasitemia levels of Trypanosoma cruzi in BALB/c mouse model during acute phase. Int. J. Parasitol. Drugs Drug Resist. 2015, 5, 110–116.
- Schmidtova, S.; Kalavska, K.; Gercakova, K.; Cierna, Z.; Miklikova, S.; Smolkova, B.; Buocikova, V.; Miskovska, V.; Durinikova, E.; Burikova, M. Disulfiram overcomes cisplatin resistance in human embryonal carcinoma cells. Cancers 2019, 11, 1224.
- Lun, X.; Wells, J.C.; Grinshtein, N.; King, J.C.; Hao, X.; Dang, N.-H.; Wang, X.; Aman, A.; Uehling, D.; Datti, A. Disulfiram when Combined with Copper Enhances the Therapeutic Effects of Temozolomide for the Treatment of GlioblastomaDisulfiram/Copper Enhance Temozolomide Treatment for Glioblastoma. Clin. Cancer Res. 2016, 22, 3860–3875.
- Thiery, J.P. Epithelial–mesenchymal transitions in development and pathologies. Curr. Opin. Cell Biol. 2003, 15, 740–746.
- Thiery, J.P.; Acloque, H.; Huang, R.Y.J.; Nieto, M.A. Epithelial-mesenchymal transitions in development and disease. Cell 2009, 139, 871–890.
- Kang, X.; Wang, J.; Huang, C.-H.; Wibowo, F.S.; Amin, R.; Chen, P.; Li, F. Diethyldithiocarbamate copper nanoparticle overcomes resistance in cancer therapy without inhibiting P-glycoprotein. Nanomed. Nanotechnol. Biol. Med. 2023, 47, 102620.
- Han, D.; Wu, G.; Chang, C.; Zhu, F.; Xiao, Y.; Li, Q.; Zhang, T.; Zhang, L. Disulfiram inhibits TGF-β-induced epithelial-mesenchymal transition and stem-like features in breast cancer via ERK/NF-κB/Snail pathway. Oncotarget 2015, 6, 40907–40919.
- Krasnovskaya, O.; Naumov, A.; Guk, D.; Gorelkin, P.; Erofeev, A.; Beloglazkina, E.; Majouga, A. Copper Coordination Compounds as Biologically Active Agents. Int. J. Mol. Sci. 2020, 21, 3965.
- Kang, X.-j.; Wang, H.-y.; Peng, H.-g.; Chen, B.-f.; Zhang, W.-y.; Wu, A.-h.; Xu, Q.; Huang, Y.-z. Codelivery of dihydroartemisinin and doxorubicin in mannosylated liposomes for drug-resistant colon cancer therapy. Acta Pharmacol. Sin. 2017, 38, 885–896.
- Alisi, A.; Cho, W.C.; Locatelli, F.; Fruci, D. Multidrug resistance and cancer stem cells in neuroblastoma and hepatoblastoma. Int. J. Mol. Sci. 2013, 14, 24706–24725.
- Siddique, M.R. Improving Leukaemia Diagnosis and Management with Selected Ion Flow Tube Mass Spectrometry and Vibrational Spectroscopy Techniques. Ph.D. Thesis, Keele University, Newcastle, UK, 2017.
- Mafficini, A.; Scarpa, A. Genetics and Epigenetics of Gastroenteropancreatic Neuroendocrine Neoplasms. Endocr. Rev. 2019, 40, 506–536.
- Farooq, M.A.; Aquib, M.; Khan, D.H.; Hussain, Z.; Ahsan, A.; Baig, M.M.F.A.; Wande, D.P.; Ahmad, M.M.; Ahsan, H.M.; Jiajie, J. Recent advances in the delivery of disulfiram: A critical analysis of promising approaches to improve its pharmacokinetic profile and anticancer efficacy. DARU J. Pharm. Sci. 2019, 27, 853–862.
- Banerjee, P.; Geng, T.; Mahanty, A.; Li, T.; Zong, L.; Wang, B. Integrating the drug, disulfiram into the vitamin E-TPGS-modified PEGylated nanostructured lipid carriers to synergize its repurposing for anti-cancer therapy of solid tumors. Int. J. Pharm. 2019, 557, 374–389.
- Zhong, Y.; Sun, R.; Geng, Y.; Zhou, Q.; Piao, Y.; Xie, T.; Zhou, R.; Shen, Y. N-Oxide polymer–cupric ion nanogels potentiate disulfiram for cancer therapy. Biomater. Sci. 2020, 8, 1726–1733.





