Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mohamed Shanshal | -- | 3657 | 2023-05-29 19:50:22 | | | |
| 2 | Camila Xu | Meta information modification | 3657 | 2023-05-30 02:19:30 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Shanshal, M. T-Cell Engagers in Solid Cancers. Encyclopedia. Available online: https://encyclopedia.pub/entry/44980 (accessed on 24 May 2026).
Shanshal M. T-Cell Engagers in Solid Cancers. Encyclopedia. Available at: https://encyclopedia.pub/entry/44980. Accessed May 24, 2026.
Shanshal, Mohamed. "T-Cell Engagers in Solid Cancers" Encyclopedia, https://encyclopedia.pub/entry/44980 (accessed May 24, 2026).
Shanshal, M. (2023, May 29). T-Cell Engagers in Solid Cancers. In Encyclopedia. https://encyclopedia.pub/entry/44980
Shanshal, Mohamed. "T-Cell Engagers in Solid Cancers." Encyclopedia. Web. 29 May, 2023.
Copy Citation
There are multiple strategies to target cancer cells, and among the rapidly evolving field is the use of bispecific antibodies and T-cell engagers in the treatment of cancers. These drugs work by recruiting and activating T-cells, a type of white blood cell, to recognize and attack cancer cells. These agents consist of two different antibody fragments: one that binds to a tumor antigen on cancer cells and another that binds to the CD3 receptor on T-cells.
bi-/trispecific antibodies
T-cell engagers
solid tumors
1. Introduction
The use of monoclonal antibody (mAb) technology in anti-cancer therapy has evolved rapidly since the Food and Drug Administration first approved the use of the anti-CD20 mAb rituximab in 1997 for treating relapsed CD20-positive B-cell lymphoma. Improvements in gene sequencing, proteomics and computational platforms resulted in the production of antibodies with increasing affinity to antigenic epitopes and efficacy [1]. Targeting tumor-associated antigens (TAA) continues to be a major focus in anti-cancer drug development and has become particularly relevant in the era of immunotherapeutics because these antigens may be targeted to elicit antitumor immune responses [2][3]. However, this approach can be hindered by several resistance mechanisms, including but not limited to downregulation of TAA expression by cancer cells, activation of signaling pathway granting cancer cells resistance to apoptosis as well as tumor microenvironmental factors impeding mAb activity [4]. Furthermore, inducing immune responses that alter antibody-dependent cellular cytotoxicity and complement-dependent cytotoxicity may impair the efficacy of these mAbs [5].
Bispecific antibodies (BsAbs) are a proven anti-cancer drug platform capable of simultaneously targeting multiple TAAs and potentially overcoming these resistance mechanisms, and, in the era of immuno-oncology, they modulate/induce immune cell responses by simultaneously targeting the TAA(s) and antigens/receptors on the effector cells [6]. The majority of BsAbs are designed based on IgG molecules (Figure 1). The antigen-binding site (ABS) is an area between the heavy variable (VH) and light variable (VL) chains and is designed via the re-arrangement of the complementarity-determining regions (CDRs) [7].
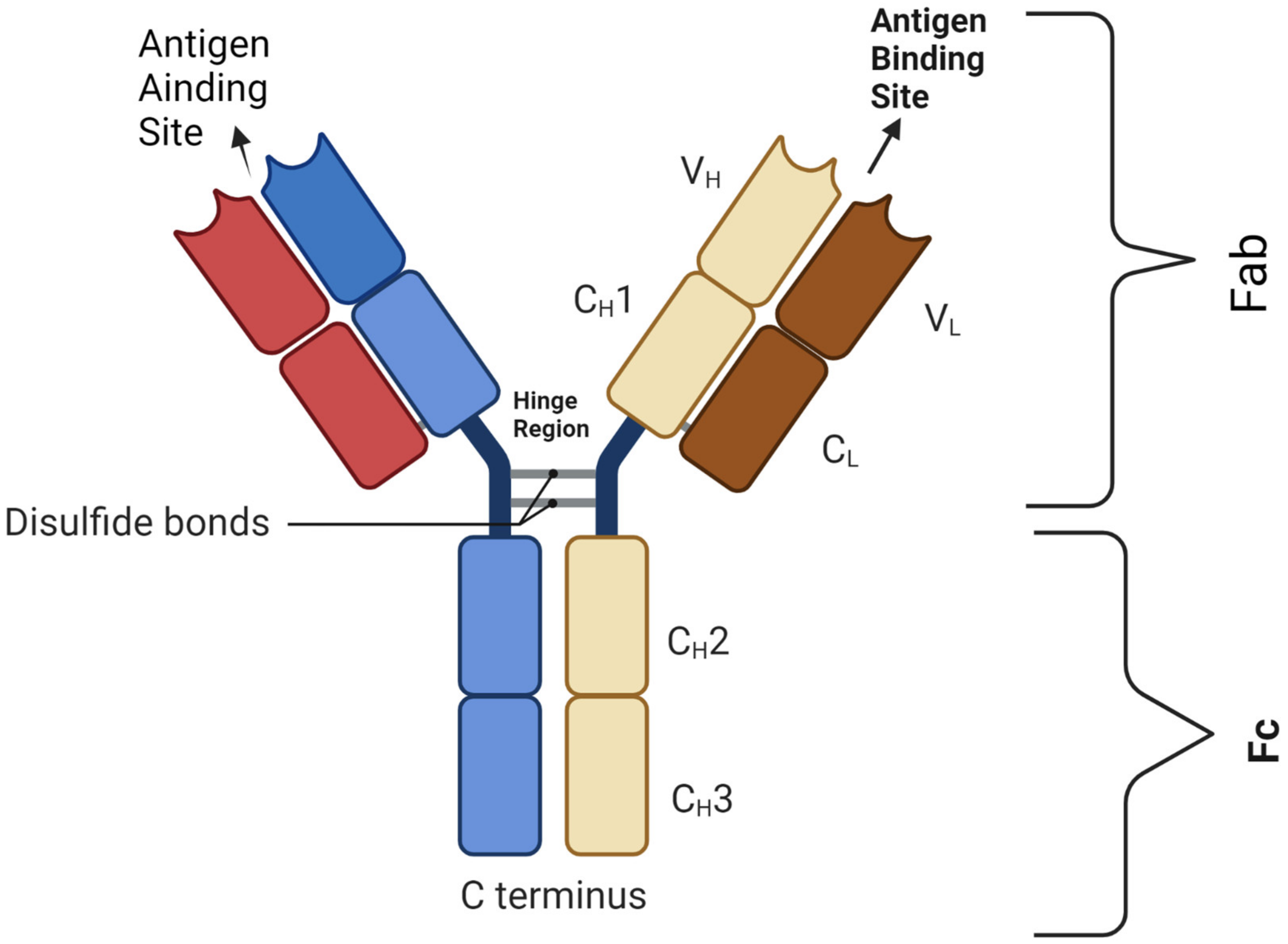
Figure 1. Basic BsAb structure showing the antigen-binding site that harbors the complementarity-determining region (CDR) segment, disulfide bond, light chains (L) and heavy chains (H); C: constant domain; V: variable domain, Fab: fragment antigen-binding domain; Fc: fragment-crystallizable domain. Figure created by BioRender.com (accessed on 14 May 2023).
Optimizing the CDRs is important for higher affinity (on-target efficacy) and avoidance of bystander cells (off-tumor toxicity) [8]. The design of an effective BsAbs involves multiple processes, and four key steps will be summarized here: (A) lead identification, which is the detailed analysis/identification of the antibody-binding site for an antigen e.g., protein docking [9][10]; (B) predicting the antibody structure through proteomic computational methods, e.g., AlphaFold [11]; (C) lead optimization, which is the prediction of the binding affinity between the antibody (paratope) and the antigen (epitope), e.g., the PInet [12]; and finally (D): for the antibody to be effective, it does require good solubility to avoid overt immunogenicity. Lead identification, optimization and assessment of the relative positions of both parts of the paratope–epitope complex are crucial steps for the development of successful therapeutic antibodies [13].
BsAbs are designed in a way that one target is the neoepitope of cancer cells (TAA) and the other target site is dedicated to engaging with targets that can facilitate an antineoplastic effect. The antineoplastic activity can be achieved through several mechanisms: (1) direct engagement with surface antigens of immune effector cells, such as CD3 in T-cells, CD16 in NK cells or CD47 in macrophages; (2) engagement with receptors that modulate T-cell response; and (3) interaction with other signaling pathways.
The most common BsAbs are the bispecific T-cell engagers (BiTEs), which act through the simultaneous engagement of TAAs and CD3, resulting in the activation of T-cells irrespective of MHC, with the resultant release of perforins and granzymes [14]. In solid tumors, the success in targeting GP100 through construction of gp100/HLA*0201 fused to anti-CD3 single-chain variable fragments (scFv) led to tebentafusp’s approval for the treatment of uveal melanoma.
BsAbs’ T-cell-modulating targets include those that are immune-inhibitory such as PD-L1/CTLA-4 [15] and those that are immune-stimulatory such as the TNF receptors OX40, CD27 and CD137 (4-1BB) or the T-cell costimulatory receptor CD28. Engagement of one or more of these receptors may enhance the antitumor immune response or activate exhausted tumor-infiltrating lymphocytes in the tumor immune microenvironment [16].
Antibody–drug conjugates are another approach that utilize the mAb platform for targeted delivery of cytotoxic agents [17]. The use of trastuzumab deruxtecan in the treatment of HER2+ breast cancer is a successful example in which the BsAb binds to HER2/CD63 or HER2/PRLR and concurrently facilitates the internalization and lysosomal degradation essential for the action of the cytotoxic payload [18][19].
At the time of the writing of this research, there were a total of seven BsAbs approved for the treatment of different malignant diseases: amivantamab (EGFR/cMET), blinatumomab (CD3/CD19), catumaxomab (CD3/EpCAM), mosunetuzumab (CD3/CD20), tebentafusp (GP100/CD3), teclistamab (CD3/BCMA) and zenocutuzumab (HER2/HER3) [20][21].
2. Structural Mechanism of T-Cell Engagers
Bispecific antibodies (BsAbs) are divided into two major categories based on their structure and mechanism of action.
2.1. T-Cell Engagers without FC Fragment
Characterized by a lack of immune-mediated target cell killing (ADCC), as they lack the FC domain, these agents have lower stability and a short plasma half-life but have better tissue penetration given their lower molecular weight. These are particularly useful in targeting the central nervous system (CNS), and examples include nanobodies and svFC [22][23] (Figure 2A).
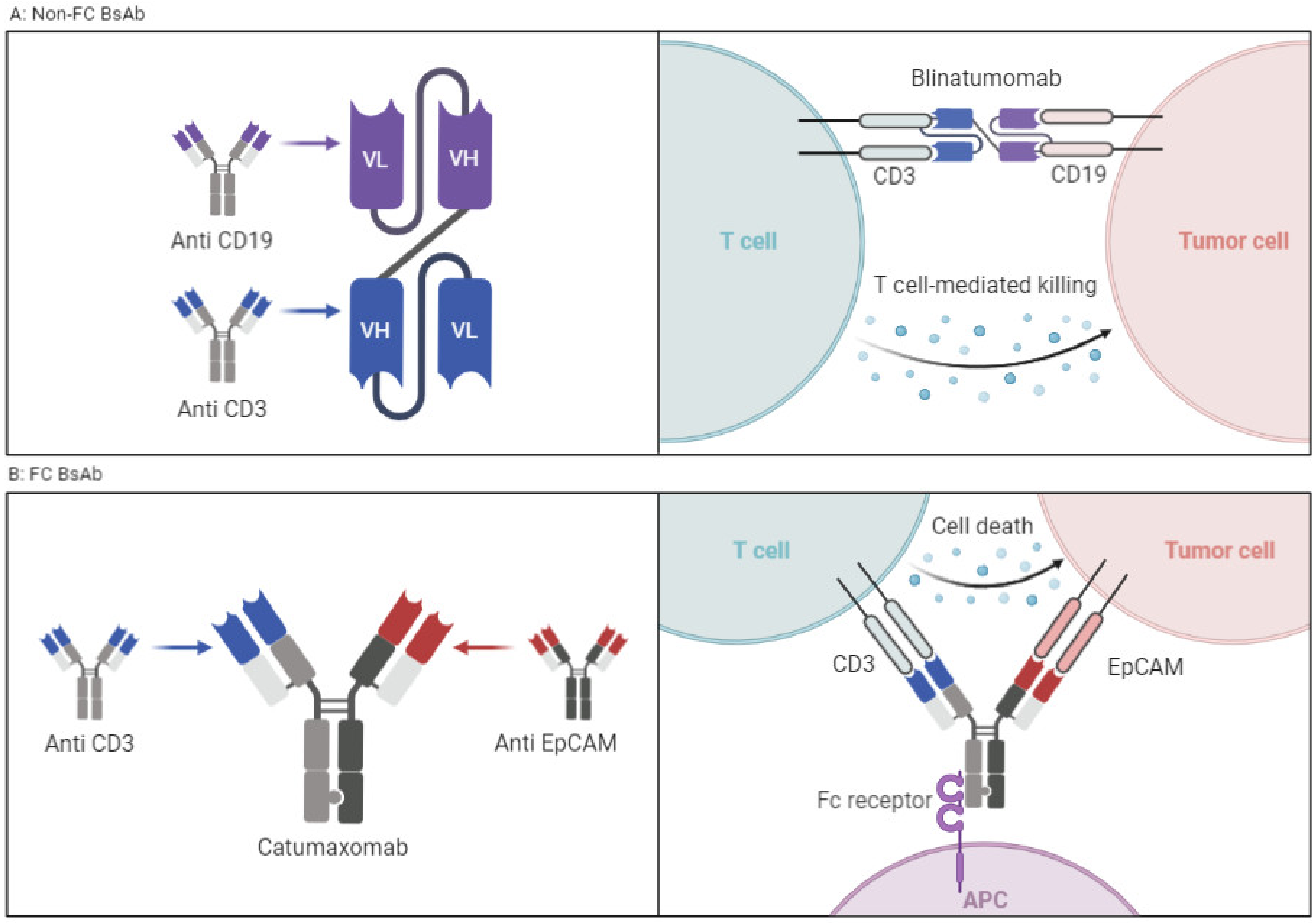
Figure 2. (A) BsAbs without FC portion connected via a linker. Example is blinatumomab. (B) Classic BsAbs with FC portion that requires antigen-presenting cells and activation of which results in ADCC. Example is catumaxomab. Figure created by BioRender.com (accessed on 14 May 2023).
2.2. T-Cell Engagers with FC Fragment
These agents have the potential to exert actions related to their FC fragment, including ADCC. BsAbs with FC fragments are characterized by a longer half-life and higher stability. The design examples include the knob-in-hole technique in which the heavy chain is engineered with a knob, and the other heavy chain consists of a hole [24]. The disadvantages of including an FC fragment on a BsAb are both structural and functional. The structural disadvantage is that the large size of the molecule impedes tissue distribution and access to neoplastic cells. In addition, the non-covalent binding of the two variable domains via a hydrophobic interface is more challenging from the design and manufacturing aspects (Figure 2B). From a functional standpoint, the presence of an intact FC domain decreases T-cell trafficking and limits the antineoplastic activity, though it can be improved by FC silencing via FC fragment modifications [25].
3. T-Cell Engagers in Solid Tumors
Most approved BiTEs are used in the treatment of hematologic malignancies. While TAAs have been identified both in hematologic malignancies and solid tumors, most are also present in normal cellular counterparts, which results in “on-target” toxicities. These on-target toxicities in normal tissues are more manageable in hematologic malignancies than in solid tumors. For example, CD19 targeting results in B-cell aplasia and hypogammaglobulinemia and increases the risk of infection; such infections can be managed by being vigilant clinically and the prompt use of antibiotic(s). On the other hand, targeting epidermal growth factor (EGFR) in lung cancer leads to generalized cutaneous toxicity and cardiotoxicity in the treatment of HER2-positive breast cancer using trastuzumab [26][27].
These “on-target” toxicities can further be mitigated through the use of conditional T-cell engagers (cTCE), which are inactive precursors of TCEs activated by tumor-associated proteases. This induces TCEs to kill tumor cells expressing the target antigen in the tumor microenvironment without affecting distant tissues. The decreased toxicity of cTCEs potentially confers upon them a superior therapeutic index and may allow the administration of a higher dose. An example is ProTriTACs, where a half-life-extending albumin-binding domain masks the CD3-binding domain [28][29].
Intracellular TAA targeting may sidestep the challenges of mAb targeting of shared cell surface TAAs between tumor and normal tissues in solid tumors. This strategy may lead to higher tumor cell killing (on-target/on-tumor effect), decreased risk of damaging normal tissues (on-target/off-tumor toxicity) as well as lower bystander toxicity (off-target/off-tumor toxicity).
However, intracellular TAA targeting is currently challenged by a lack of effective tools to penetrate the cell surface and deliver the antibody arm of TCEs into neoplastic cells. This is an area of active research, and novel approaches include technologies that promote the expression of intracellular TAAs or enhance the delivery of BsAbs into tumor cells.
3.1. Targeting Tumor-Associated Peptides Presented by MHC
Aberrant intracellular proteins can be presented by MHC class 1 molecules on the cell surface (Figure 3E). One example is p53: a small fraction of intracellular p53 is degraded via proteosomes and can be presented by HLA on the cell surface. Specific peptides of mutated p53 (R175H) can bind to HLA-A*02:01 on the surface of cancer cells. Hsiue et al. developed CD3 engagers that bind specifically to the p53-R175H peptide–HLA complex with high affinity and resulted in tumor cell killing [30][31][32].
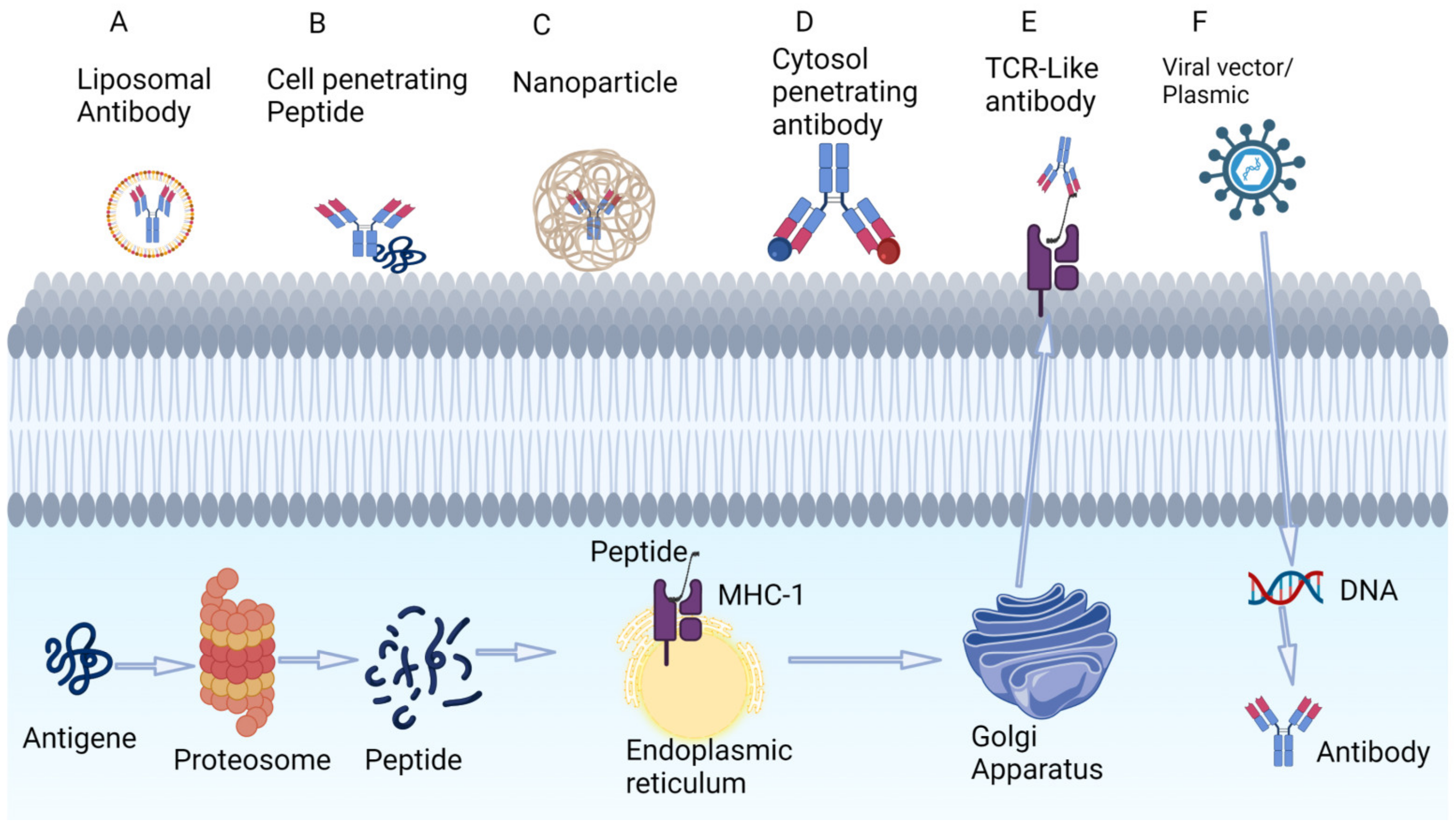
Figure 3. Techniques under investigation for targeting intracellular tumor antigens using antibodies. (A) Liposomal coating of antibodies. (B) Cell-penetrating peptides that adhere to the phospholipid bilayers. (C) Nanoparticles. (D) Cytosol-penetrating antibodies. (E) TCR-like antibodies that can target low-concentration intracellular peptide fragments that are presented to extracellular space via MHC. (F) Viral vectors that incorporate into the genome and produce intracellular antibodies. Figure created by BioRender.com (accessed on 14 May 2023).
A similar strategy is the use of oncolytic vaccines such as MAGE-1 and NY-ESO-1. These are incorporated into the tumor genome and are translated into antigens which, when degraded, are presented by MHC to the extracellular surface of tumor cells, making them recognizable by T-cell engagers. An example is melanoma-associated antigen A4 (MAGE-A4) solid cancers in patients with the HLA-A*02:01 genotype. MAGE-A4 is processed intracellularly, resulting in peptide fragments that are co-presented with HLA-A*02:01 (Figure 3E). Afamitresgene autoleucel is an HLA-restricted autologous T-cell therapy that targets MAGE-A4 that was evaluated in a phase 1 trial in solid tumor patients with HLA*02:01. The overall response rate was 24% (all partial response). All patients experienced grade 3 and above hematologic toxicity; 55% of patients experienced =< grade 2 cytokine release syndrome [33].
3.2. Intracellular Delivery of Antibodies
The delivery of larger antibodies to intracellular targets is an area of intense research, and approaches under investigation include the use of liposomes, cell-penetrating peptides, nanoparticles, cytosol-penetrating antibodies, TCR-Like antibody and the use of viral vectors (Figure 3A–F). The successful targeting of intracellular p53 and KRAS mutants had been demonstrated in preclinical studies [34].
3.3. Successful Examples of T-Cell Engagers in Solid Tumors
This section highlights the importance of T-cell engagers in the treatment of advanced solid tumors. Some of these example are FDA-approved, while others are in later stages of drug development. (Figure 4, summarize therapeutic targets in solid tumors).
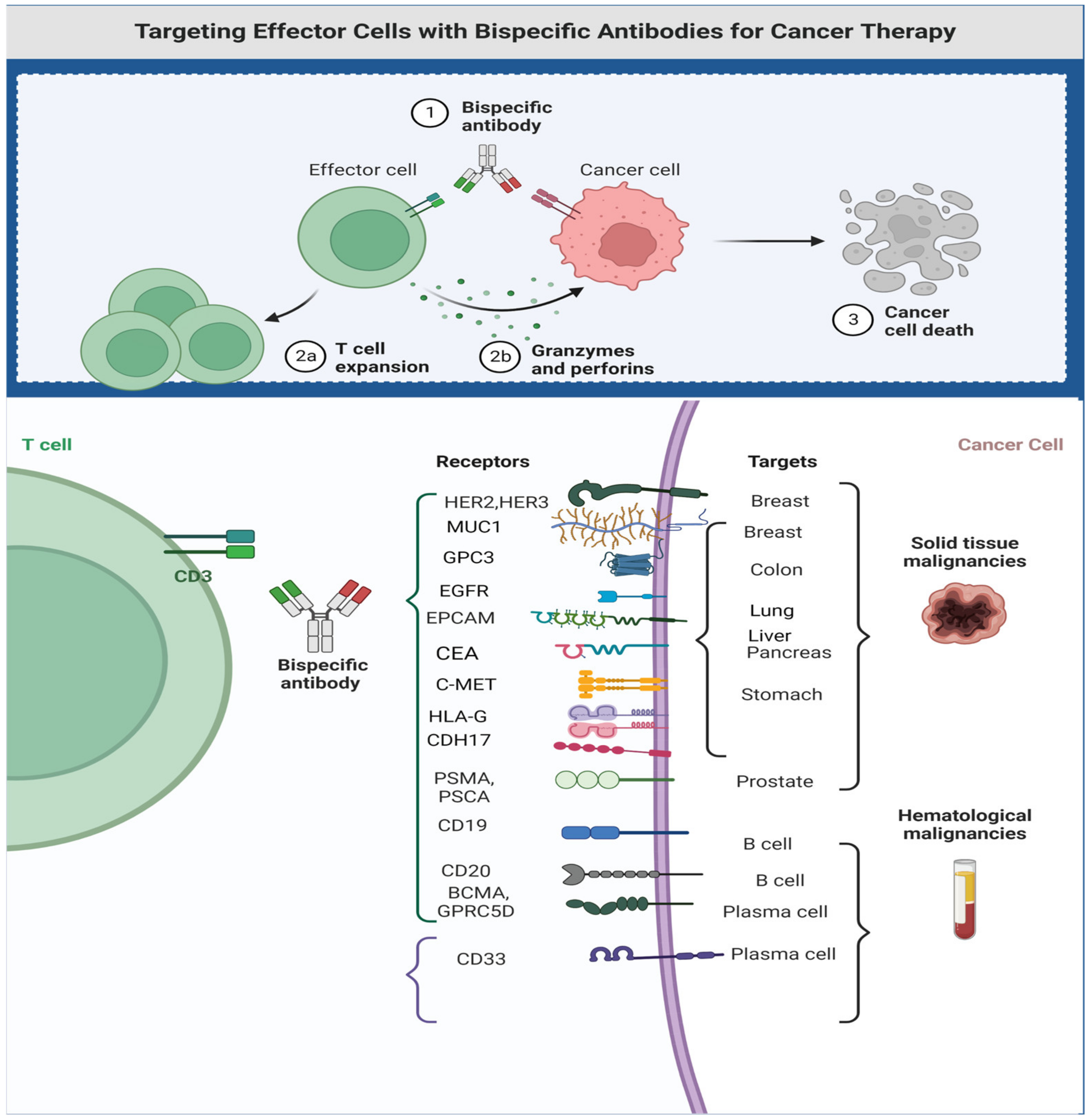
Figure 4. Therapeutic targets for bi- and trispecific antibodies. Figure created by BioRender.com (accessed on 14 May 2023).
-
GP100
Melanoma-associated antigen (gp100) is a membrane-bound protein expressed on the surface of melanocytes and most malignant melanoma. T-lymphocytes recognize gp100 presented by HLA proteins such as HLA-A*02:01. Vaccination with gp100 peptide vaccine was evaluated in a phase II randomized trial in the treatment of metastatic melanoma in combination with IL2; the overall response rate (ORR) was 16%, and overall survival (OS) was 17 months [35]. This suggested gp100 as a potential TAA, which led to designer BiTEs’ with a high-affinity TCR-binding domain and an anti-CD3 T-cell-engaging domain. Such a construct facilitates and redirects T-cells to attack gp100-expressing tumor cells. Tebentafusp is a gp100/CD3 BsAb and was evaluated in a phase 1 study of 42 patients with metastatic uveal melanoma. The study found that tebentafusp was generally well-tolerated, with the most common adverse events (AEs) being fever (91%), rash and pruritis (83%), fatigue (71%) and chills (69%). The overall response rate was 11.9% (95% CI, 4.0 to 25.6). The median overall survival was 25.5 months (range, 0.89–31.1 months), and the 1-year overall survival rate was 67% [36]. The subsequent phase 3 trial in HLA-A*02:01-expressing patients with treatment-naive metastatic uveal and cutaneous melanoma compared tebentafusp against the investigator’s choice of treatment (pembrolizumab, ipilimumab or dacarbazine). At a median follow-up of 14 months, the tebentafusp-containing arm achieved superior survival compared to the investigator’s choice (median OS 22 months versus 16 months; 6-month PFS 31% versus 19%, respectively). A 1-year overall survival rate of 65% was reported for both patient cohorts [37][38]. This pivotal trial led to Food and Drug Administration (FDA) approval of tebentafusp in April 2022. Cytokine release syndrome (CRS) occurred in 89% of patients, though the adverse events improved during subsequent dosing. Only 2% of patients discontinued treatment due to treatment-related side effects, and there were no treatment-related deaths.
-
EPCAM
Catumaxomab is a T-cell engager with specificity for CD3 and epithelial cell adhesion molecule (EpCAM). Interestingly, this agent can be considered “trifunctional” because the mAb has a functional FC receptor capable of engaging FCγ receptors that induce immune reaction [39]. Catumaxomab was approved by the European Medical Agency in 2009 for treatment of EpCAM-positive carcinomas and malignant ascites. However, catumaxomab was withdrawn from the market in 2017 due to unacceptable CRS and high-grade liver toxicity which was fatal in some patients [40]. The severity of the toxicities was likely due to the high immunogenicity and wide expression of EpCAM in normal tissues such as the Kupffer cells of the liver.
-
PSMA
Pasotuxizumab, a CD3/PSMA T-cell engager, was evaluated in a phase 1 trial of patients with metastatic castrate-resistant prostate cancer (mCRPC). The BsAb was administered as a continuous IV infusion over 12 weeks. Of the 16 patients treated, 13 (81%) experienced grade ≥ 3 adverse events (AEs). The most frequent all-grade AEs were flu-like illness and fatigue, whereas the most frequent grade ≥ 3 AEs were decreased lymphocytes and infections (44%). Three patients had a reduction of >50% in serum PSA levels, two of them had long-term responses, and one achieved a near CR as assessed by PSMA PET imaging [41]. The development of pasotuxizumab was halted after this study in favor of acapatamab.
Acapatamab is T-cell engager targeting CD3/PSMA but has a longer half-life than pasotuxizumab. Acapatamab was evaluated alone or in combination with pembrolizumab in a phase 1 trial involving patients with mCRPC. In the monotherapy cohort, a ‘confirmed ≥ 30% PSA reduction’ was achieved in 27.6% of patients, a partial response in 20% and stable disease in 53.3%. However, 60.5% had grade 2 CRS, and 25.6% had grade 3 CRS. [42][43]. The CD3/PSMA T-cell engager is being compared to enzalutamide and abiraterone in patients with mCRPC in a phase 2 trial (NCT04631601). The study has completed accrual, and the results are pending.
-
CD33 (MDSC-targeting)
CD33 is a transmembrane receptor expressed on the surface of various myeloid cells, including myeloid-derived suppressor cells (MDSCs), and is known to promote tumor growth and suppress antitumor immune response in solid tumors [44]. AMV564 is a novel CD3/CD33 T-cell engager which induces antitumor immune response via T-cell-directed lysis of CD33 cells [45]. The agent was evaluated in a phase 1 trial which included 30 patients with advanced solid tumors. AMV564 was given via subcutaneous injections on days 1–5 and 8–12 of a 21-day cycle, either alone (20 patients) or in combination with pembrolizumab (10 patients) at a dose of 200 mg IV q3w. The monotherapy cohort dosing was 15, 50 and 75 mcg/day, while the combination therapy cohort dosing was 5, 15 and 50 mcg/day. Complete response was achieved in patients with ovarian cancer in the monotherapy cohort. The agent was well tolerated, with 2 cases of grade 2 CRS at 75 mcg/day and no dose-limiting toxicities. The most common side effects were injection site reactions, fever, fatigue, anemia, hypotension and nausea. Subcutaneous injection of AMV564 resulted in relevant plasma exposure [46].
3.4. Toxicity Profile and Management of Toxicity of T-Cell Engagers in Solid Tumors
The major advantages of T-cell-engaging immunotherapeutics such as BsAbs and T-cell engagers over cellular therapeutics, e.g., CAR-T, include their “off-the-shelf” availability and their requiring no lymphodepletion prior to administration. Both treatment modalities (T-cell engagers and CAR-T-cells) have similar toxicity profiles, including CRS and immune effector-associated neurotoxicity syndrome (ICANS). These toxicities are less severe or less frequent in T-cell engagers and may be related to their dosing (Table 1 and Table 3) [47][48]. While CAR-T-cell treatment is often administered in a single dose, T-cell engagers may be administered repeatedly. As such, T-cell engager administration includes a period of step-up dosing to mitigate the risk of these toxicities and is followed by the full doses about 1 to 2 weeks after.
Table 1. Unique toxicities based on therapeutic targets of T-cell engagers.
| Target Antigen | Specific Toxicity |
|---|---|
| EPCAM | Immune-mediated hepatotoxicity |
| HER2 | Hypotension, hypertension, tachycardia |
| PSMA | Hepatotoxicity |
| DLL3 | Pneumonitis |
| Myeloid-derived suppressor cells (MDSC) | Anemia, hypotension, pruritis |
| EGFRvIII | Dermatologic toxicities, SJS, TEN |
| CEA | Hepatotoxicity |
-
Cytokine release syndrome
The immune-mediated toxicities associated with treatment using CAR-T-cells and T-cell engagers are significant challenges requiring close attention. The binding of the target antigen induces T-cell expansion and the release of proinflammatory cytokines, which then leads to the recruitment of other endogenous immune cells such as macrophages and further escalation of the immune response [49]. The overactive and uncontrolled immune response results in CRS, which is characterized by fever and multiple-end-organ dysfunction/damage. This systemic toxicity is experienced by approximately 15% of patients treated using blinatumomab. The incidence is dependent on the BiTEs’ structure, affinity for CD3 engagement and TAA abundance, e.g., grade 3 and above CRS being reported in 2% of patients with MUC17/CD3 (AMG199) and 25% of patients with PSMA/CD3 (acapatamab) [42].
The risk can be mitigated by close monitoring and premedication with corticosteroids. Step-up dosing can also reduce CRS risk by starting at a lower dose and escalating during the first cycle of treatment. The use of a subcutaneous route has been proposed to decrease the risk of CRS [50]. A recent computational study showed that the co-administration of the interleukin (IL)-6 receptor-blocking mAb tocilizumab and/or anti-TNFα may reduce the incidence without compromising antitumor activity [51].
The management of low-grade CRS includes the use of antihistamines, antipyretics and intravenous fluids. CRS of grade 2 and above requires admission to the hospital and may require pressors for hypotension, as well as supplemental oxygen for respiratory distress [52][53]. Severe CRS will require intensive care unit care with early initiation of high-dose corticosteroids—for example, methylprednisone at 1 mg/kg/day and tocilizumab at 8 to 12 mg/kg based on body weight. Tocilizumab can be repeated after an interval of 8 h, and it should not exceed 4 doses in total [54]. Supportive therapies include the use of intravenous vasopressors or mechanical ventilation to assist with respiratory distress.
-
Neurotoxicity
Immune effector-associated neurotoxicity syndrome (ICANS) with T-cell engager treatment may manifest as mild confusion, headache, dysgraphia and, in more severe cases, encephalopathy. The pathophysiology of ICANS is not yet fully understood. Cerebral microvasculature damage from activated immune cells, endothelial damage, blood–brain barrier disruption and direct effects of cytokines have been hypothesized as underlying mechanisms [55].
The neurotoxicity risk from T-cell engagers may be mitigated by step-up dosing and pre-treatment with corticosteroids. Once it has occurred, management consists primarily of aggressive supportive care and corticosteroids. Tocilizumab, however, is not recommended for neurotoxicity, as it can increase IL-6 plasma concentration and exacerbate ICANS.
The management of ICANS often involves supportive care measures such as hydration, electrolyte balancing, the use of steroids and seizure management. For patients with more severe symptoms, more aggressive interventions may be indicated including mechanical ventilation or intracranial pressure monitoring. A neurologist or other specialist with experience in managing neurological complications of immunotherapy should be consulted if one has not been already.
The prophylactic use of corticosteroids has been shown to be effective in ameliorating the side effects of CRS and ICANS. The ZUMA-1 study showed the superiority of prophylactic use of dexamethasone at 10 mg for 3 days before axicabtagene ciloleucel (no grade 3 CRS) compared to the 13% incidence of CRS among patients who received steroids after an infusion of axicabtagene ciloleucel. The efficacy was not compromised in this approach, though there was a higher rate of ICANS [56].
Based on the above information, clinicians should be aware of the potential side effects of CRS and ICANS when using T-cell engagers. Early identification and prompt management are key to preventing the development of severe side effects. A multidisciplinary team with experience in immunotoxicity provides the best outcome.
-
TAA-specific toxicities
Other less common but important side effects include hepatoxicity and cardiac toxicity, which are mostly managed with supportive care and interventions described above. Table 1 lists toxicities described for solid tumor T-cell engagers.
-
T-cell engagers undergoing clinical evaluation in solid tumors
Publicly available databases including PubMed, meeting abstracts of major scientific meetings and ClinicalTrials.gov were searched for T-cell engagers that are in the early phases clinical testing (summarized in Table 2).
Table 2. Summary of T-cell engagers in development against solid tumors.
| Target Population | Phase of Study/References | Number of Patients | Results | Clinical Trial Number | |
|---|---|---|---|---|---|
| CD3/HER2 | Advanced HER2-positive breast cancer | Phase 2 [57] | 32 patients, 8 patients had stable disease. | The median OS was 13.1, 15.2 and 12.3 months for the entire group, HER2-HR+ and TNBC patients, respectively. Plan for phase 3. | NCT03272334 |
| EGFRvIII/CD3 (AMG596), (CX-904) EGFR/CD3. |
EGFRvIII-positive GBM or malignant glioma | Phase 1/1b [58] | Total 14 patients. | 1 partial response, 2 stable disease. | NCT03296696 |
| Tumor expression EGFR | Early phase 1 | Plan for 100 patients | Not published | NCT05387265 | |
| Tyrosinase Related Protein 1 (TYRP1) (RO7293583) | Melanoma | Phase 1 | 20 patients | Not published | NCT04551352 |
| MUC17/CD3 | Advanced gastric, GE junction, CRC and pancreatic (AMG199) | Phase 1 [59] | Total 64 patients. | 13 had PR, 17 SD. CRS > grade 3 occurred in 2% | NCT04117958 |
| Advanced liver cancer | Phase 2 [60] | 11 Patients | Median PFS 4 months, median OS 13.2 months; 5 discontinued treatments due to severe side effects | NCT03146637 | |
| DLL3/CD3 | Small cell lung cancer | Phase 1 [61] | Confirmed partial responses in 20% of patients and duration of response of 8.7 months | Phase 2 ongoing | NCT05060016 |
| CEA (MEDI-565) |
Advanced GI cancers | Phase 1 | Total 39 patients, | 11 patients have stable disease as best response. | NCT01284231 |
| PSMA | Prostate cancer | Phase ½ | LAVA-1207 | Not published | NCT05369000 |
| Phase 1 [62] | AMG 509 | Not published | NCT04221542 | ||
| Phase 1 | BAY 2010112 | 47 patients, 12 patients had >50% decrease in PSA | NCT01723475 | ||
| EpCAM | Advanced solid tumors | Solitomab: Phase 1 Catumoximab: Phase 2 [63] |
Catumoximab and Solitomab: for malignant ascites both associated with sever toxicities precluding development of Solitomab. | Solitomab DLT in phase-limiting escalation. Catumoximab, withdrawn from market due to toxicities. |
NCT00635596 NCT00836654 |
| Myeloid-derived suppressor cells (MDSC) | Advanced solid tumors. with and without pembrolizumab |
Phase 1 | 20 patients in monotherapy arm. 10 in combination arm. |
Not fully published, study mentioned One CR. Study is going to phase 2. | NCT04128423 |
| CLDN18.2 (AMG 910) |
Gastric and gastroesophageal junction (G/GEJ) adenocarcinoma | Phase 1 [64] | Plan recruitment 34 patients | Not finished | (NCT04260191) |
| HLA-G | Advanced solid tumors | Phase 1 | Actively recruiting | Not finished | NCT04991740 |
References
- Oostindie, S.C.; Lazar, G.A.; Schuurman, J.; Parren, P. Avidity in antibody effector functions and biotherapeutic drug design. Nat. Rev. Drug. Discov. 2022, 21, 715–735.
- Tran, E.; Robbins, P.F.; Rosenberg, S.A. ‘Final common pathway’ of human cancer immunotherapy: Targeting random somatic mutations. Nat. Immunol. 2017, 18, 255–262.
- Schumacher, T.N.; Schreiber, R.D. Neoantigens in cancer immunotherapy. Science 2015, 348, 69–74.
- Torka, P.; Barth, M.; Ferdman, R.; Hernandez-Ilizaliturri, F.J. Mechanisms of Resistance to Monoclonal Antibodies (mAbs) in Lymphoid Malignancies. Curr. Hematol. Malig. Rep. 2019, 14, 426–438.
- Pierpont, T.M.; Limper, C.B.; Richards, K.L. Past, Present, and Future of Rituximab-The World’s First Oncology Monoclonal Antibody Therapy. Front. Oncol. 2018, 8, 163.
- Ma, J.; Mo, Y.; Tang, M.; Shen, J.; Qi, Y.; Zhao, W.; Huang, Y.; Xu, Y.; Qian, C. Bispecific Antibodies: From Research to Clinical Application. Front. Immunol. 2021, 12, 626616.
- Abanades, B.; Georges, G.; Bujotzek, A.; Deane, C.M. ABlooper: Fast accurate antibody CDR loop structure prediction with accuracy estimation. Bioinformatics 2022, 38, 1877–1880.
- Pantazes, R.J.; Maranas, C.D. OptCDR: A general computational method for the design of antibody complementarity determining regions for targeted epitope binding. Protein Eng. Des. Sel. 2010, 23, 849–858.
- Guest, J.D.; Vreven, T.; Zhou, J.; Moal, I.; Jeliazkov, J.R.; Gray, J.J.; Weng, Z.; Pierce, B.G. An expanded benchmark for antibody-antigen docking and affinity prediction reveals insights into antibody recognition determinants. Structure 2021, 29, 606–621.e5.
- Ambrosetti, F.; Jiménez-García, B.; Roel-Touris, J.; Bonvin, A.M.J.J. Modeling Antibody-Antigen Complexes by Information-Driven Docking. Structure 2019, 28, 119–129.
- Tunyasuvunakool, K.; Adler, J.; Wu, Z.; Green, T.; Zielinski, M.; Žídek, A.; Bridgland, A.; Cowie, A.; Meyer, C.; Laydon, A.; et al. Highly accurate protein structure prediction for the human proteome. Nature 2021, 596, 590–596.
- Dai, B.; Bailey-Kellogg, C. Protein Interaction Interface Region Prediction by Geometric Deep Learning. Bioinformatics 2021, 37, 2580–2588.
- Kuroda, D.; Tsumoto, K. Antibody Affinity Maturation by Computational Design. Methods Mol. Biol. 2018, 1827, 15–34.
- Garrido, F. HLA class-I expression and cancer immunotherapy. In MHC Class-I Loss and Cancer Immune Escape; Springer: Berlin/Heidelberg, Germany, 2019; pp. 79–90.
- Dovedi, S.; Mazor, Y.; Elder, M.; Hasani, S.; Wang, B.; Mosely, S.; Jones, D.; Hansen, A.; Yang, C.; Wu, Y.; et al. Abstract 2776: MEDI5752: A novel bispecific antibody that preferentially targets CTLA-4 on PD-1 expressing T-cells. Cancer Res. 2018, 78, 2776.
- Halim, L.; Das, K.K.; Larcombe-Young, D.; Ajina, A.; Candelli, A.; Benjamin, R.; Dillon, R.; Davies, D.M.; Maher, J. Engineering of an Avidity-Optimized CD19-Specific Parallel Chimeric Antigen Receptor That Delivers Dual CD28 and 4-1BB Co-Stimulation. Front. Immunol. 2022, 13, 836549.
- Marei, H.E.; Cenciarelli, C.; Hasan, A. Potential of antibody-drug conjugates (ADCs) for cancer therapy. Cancer Cell. Int. 2022, 22, 255.
- de Goeij, B.E.; Vink, T.; Ten Napel, H.; Breij, E.C.; Satijn, D.; Wubbolts, R.; Miao, D.; Parren, P.W. Efficient Payload Delivery by a Bispecific Antibody-Drug Conjugate Targeting HER2 and CD63. Mol. Cancer Ther. 2016, 15, 2688–2697.
- Andreev, J.; Thambi, N.; Perez Bay, A.E.; Delfino, F.; Martin, J.; Kelly, M.P.; Kirshner, J.R.; Rafique, A.; Kunz, A.; Nittoli, T.; et al. Bispecific Antibodies and Antibody-Drug Conjugates (ADCs) Bridging HER2 and Prolactin Receptor Improve Efficacy of HER2 ADCs. Mol. Cancer Ther. 2017, 16, 681–693.
- Esfandiari, A.; Cassidy, S.; Webster, R.M. Bispecific antibodies in oncology. Nat. Rev. Drug. Discov. 2022, 21, 411–412.
- Schram, A.M.; Goto, K.; Kim, D.-W.; Martin-Romano, P.; Ou, S.-H.I.; O’Kane, G.M.; O’Reilly, E.M.; Umemoto, K.; Duruisseaux, M.; Neuzillet, C.; et al. Efficacy and safety of zenocutuzumab, a HER2 x HER3 bispecific antibody, across advanced NRG1 fusion (NRG1+) cancers. J. Clin. Oncol. 2022, 40, 105.
- Kariolis, M.S.; Wells, R.C.; Getz, J.A.; Kwan, W.; Mahon, C.S.; Tong, R.; Kim, D.J.; Srivastava, A.; Bedard, C.; Henne, K.R.; et al. Brain delivery of therapeutic proteins using an Fc fragment blood-brain barrier transport vehicle in mice and monkeys. Sci. Transl. Med. 2020, 12, eaay1359.
- Yin, W.; Zhao, Y.; Kang, X.; Zhao, P.; Fu, X.; Mo, X.; Wang, Y.; Huang, Y. BBB-penetrating codelivery liposomes treat brain metastasis of non-small cell lung cancer with EGFR(T790M) mutation. Theranostics 2020, 10, 6122–6135.
- Wang, Q.; Chen, Y.; Park, J.; Liu, X.; Hu, Y.; Wang, T.; McFarland, K.; Betenbaugh, M.J. Design and Production of Bispecific Antibodies. Antibodies 2019, 8, 43.
- Wang, L.; Hoseini, S.S.; Xu, H.; Ponomarev, V.; Cheung, N.K. Silencing Fc Domains in T cell-Engaging Bispecific Antibodies Improves T-cell Trafficking and Antitumor Potency. Cancer Immunol. Res. 2019, 7, 2013–2024.
- Ellerman, D. Bispecific T-cell engagers: Towards understanding variables influencing the in vitro potency and tumor selectivity and their modulation to enhance their efficacy and safety. Methods 2019, 154, 102–117.
- Middelburg, J.; Kemper, K.; Engelberts, P.J.; Labrijn, A.F.; Schuurman, J.; van Hall, T. Overcoming Challenges for CD3-Bispecific Antibody Therapy in Solid Tumors. Cancers 2021, 13, 287.
- Baeuerle, P.A. Abstract IAP0301: Bispecific T cell engagers (TCEs) for treatment of solid tumors: Challenges and opportunities. Mol. Cancer Ther. 2021, 20, IAP0301.
- Lin, S.J.; Rocha, S.S.; Kwant, K.; Dayao, M.R.; Ng, T.M.; Banzon, R.R.; Thothathri, S.; Aaron, W.; Callihan, E.; Hemmati, G.; et al. Abstract 933: ProTriTAC is a modular and robust T cell engager prodrug platform with therapeutic index expansion observed across multiple tumor targets. Cancer Res. 2021, 81, 933.
- Weidle, U.H.; Maisel, D.; Klostermann, S.; Schiller, C.; Weiss, E.H. Intracellular proteins displayed on the surface of tumor cells as targets for therapeutic intervention with antibody-related agents. Cancer Genom. Proteom. 2011, 8, 49–63.
- Thura, M.; Al-Aidaroos, A.Q.O.; Yong, W.P.; Kono, K.; Gupta, A.; Lin, Y.B.; Mimura, K.; Thiery, J.P.; Goh, B.C.; Tan, P.; et al. PRL3-zumab, a first-in-class humanized antibody for cancer therapy. JCI Insight 2016, 1, e87607.
- Hsiue, E.H.; Wright, K.M.; Douglass, J.; Hwang, M.S.; Mog, B.J.; Pearlman, A.H.; Paul, S.; DiNapoli, S.R.; Konig, M.F.; Wang, Q.; et al. Targeting a neoantigen derived from a common TP53 mutation. Science 2021, 371, eabc8697.
- Hong, D.S.; Van Tine, B.A.; Biswas, S.; McAlpine, C.; Johnson, M.L.; Olszanski, A.J.; Clarke, J.M.; Araujo, D.; Blumenschein, G.R., Jr.; Kebriaei, P.; et al. Autologous T cell therapy for MAGE-A4(+) solid cancers in HLA-A*02(+) patients: A phase 1 trial. Nat. Med. 2023, 29, 104–114.
- Shin, S.-M.; Kim, J.-S.; Park, S.-W.; Jun, S.-Y.; Kweon, H.-J.; Choi, D.-K.; Lee, D.; Cho, Y.B.; Kim, Y.-S. Direct targeting of oncogenic RAS mutants with a tumor-specific cytosol-penetrating antibody inhibits RAS mutant–driven tumor growth. Sci. Adv. 2020, 6, eaay2174.
- Sosman, J.A.; Carrillo, C.; Urba, W.J.; Flaherty, L.; Atkins, M.B.; Clark, J.I.; Dutcher, J.; Margolin, K.A.; Mier, J.; Gollob, J.; et al. Three phase II cytokine working group trials of gp100 (210M) peptide plus high-dose interleukin-2 in patients with HLA-A2-positive advanced melanoma. J. Clin. Oncol. 2008, 26, 2292–2298.
- Carvajal, R.D.; Nathan, P.; Sacco, J.J.; Orloff, M.; Hernandez-Aya, L.F.; Yang, J.; Luke, J.J.; Butler, M.O.; Stanhope, S.; Collins, L.; et al. Phase I Study of Safety, Tolerability, and Efficacy of Tebentafusp Using a Step-Up Dosing Regimen and Expansion in Patients With Metastatic Uveal Melanoma. J. Clin. Oncol. 2022, 40, 1939–1948.
- Middleton, M.R.; McAlpine, C.; Woodcock, V.K.; Corrie, P.; Infante, J.R.; Steven, N.M.; Evans, T.R.J.; Anthoney, A.; Shoushtari, A.N.; Hamid, O.; et al. Tebentafusp, A TCR/Anti-CD3 Bispecific Fusion Protein Targeting gp100, Potently Activated Antitumor Immune Responses in Patients with Metastatic Melanoma. Clin. Cancer Res. 2020, 26, 5869–5878.
- Nathan, P.; Hassel, J.C.; Rutkowski, P.; Baurain, J.F.; Butler, M.O.; Schlaak, M.; Sullivan, R.J.; Ochsenreither, S.; Dummer, R.; Kirkwood, J.M.; et al. Overall Survival Benefit with Tebentafusp in Metastatic Uveal Melanoma. N. Engl. J. Med. 2021, 385, 1196–1206.
- Linke, R.; Klein, A.; Seimetz, D. Catumaxomab: Clinical development and future directions. mAbs 2010, 2, 129–136.
- Borlak, J.; Länger, F.; Spanel, R.; Schöndorfer, G.; Dittrich, C. Immune-mediated liver injury of the cancer therapeutic antibody catumaxomab targeting EpCAM, CD3 and Fcγ receptors. Oncotarget 2016, 7, 28059–28074.
- Hummel, H.-D.; Kufer, P.; Grüllich, C.; Deschler-Baier, B.; Chatterjee, M.; Goebeler, M.-E.; Miller, K.; Santis, M.D.; Loidl, W.C.; Buck, A.; et al. Phase 1 study of pasotuxizumab (BAY 2010112), a PSMA-targeting Bispecific T cell Engager (BiTE) immunotherapy for metastatic castration-resistant prostate cancer (mCRPC). J. Clin. Oncol. 2019, 37, 5034.
- Tran, B.; Horvath, L.G.; Dorff, T.B.; Greil, R.; Machiels, J.-P.H.; Roncolato, F.T.; Autio, K.A.; Rettig, M.B.; Fizazi, K.; Lolkema, M.P.; et al. Phase I study of AMG 160, a half-life extended bispecific T-cell engager (HLE BiTE) immune therapy targeting prostate-specific membrane antigen (PSMA), in patients with metastatic castration-resistant prostate cancer (mCRPC). J. Clin. Oncol. 2020, 38, TPS5590.
- ESMO Virtual Congress 2020: Novel Immunotherapy for Prostate Cancer—AMG 160—PSMA-Targeted, Bispecific T-Cell Engager (BiTE®) Immune Therapy for Metastatic Castration-Resistant Prostate Cancer—Invited Discussan. Available online: https://www.urotoday.com/conference-highlights/esmo-2020/prostate-cancer/124635-esmo-virtual-congress-2020-novel-immunotherapy-for-prostate-cancer-amg-160-psma-targeted-bispecific-t-cell-engager-bite-immune-therapy-for-metastatic-castration-resistant-prostate-cancer-invited-discussant.html (accessed on 14 May 2023).
- Tolcher, A.W.; Gordon, M.; Mahoney, K.M.; Seto, A.; Zavodovskaya, M.; Hsueh, C.H.; Zhai, S.; Tarnowski, T.; Jürgensmeier, J.M.; Stinson, S.; et al. Phase 1 first-in-human study of dalutrafusp alfa, an anti-CD73-TGF-β-trap bifunctional antibody, in patients with advanced solid tumors. J. Immunother. Cancer 2023, 11, e005267.
- Cheng, P.; Chen, X.; Dalton, R.; Calescibetta, A.; So, T.; Gilvary, D.; Ward, G.; Smith, V.; Eckard, S.; Fox, J.A.; et al. Immunodepletion of MDSC by AMV564, a novel bivalent, bispecific CD33/CD3 T cell engager, ex vivo in MDS and melanoma. Mol. Ther. 2022, 30, 2315–2326.
- Mettu, N.B.; Starodub, A.; Piha-Paul, S.A.A.; Abdul-Karim, R.M.; Tinoco, G.; Shafique, M.R.; Smith, V.; Baccei, C.; Chun, P.Y. Results of a phase 1 dose-escalation study of AMV564, a novel T-cell engager, alone and in combination with pembrolizumab in patients with relapsed/refractory solid tumors. J. Clin. Oncol. 2021, 39, 2555.
- Yan, Z.; Zhang, H.; Cao, J.; Zhang, C.; Liu, H.; Huang, H.; Cheng, H.; Qiao, J.; Wang, Y.; Wang, Y.; et al. Characteristics and Risk Factors of Cytokine Release Syndrome in Chimeric Antigen Receptor T Cell Treatment. Front. Immunol. 2021, 12, 611366.
- Weddell, J. Mechanistically modeling peripheral cytokine dynamics following bispecific dosing in solid tumors. CPT Pharmacomet. Syst. Pharmacol. 2023. Online ahead of print.
- Morris, E.C.; Neelapu, S.S.; Giavridis, T.; Sadelain, M. Cytokine release syndrome and associated neurotoxicity in cancer immunotherapy. Nat. Rev. Immunol. 2022, 22, 85–96.
- Lesokhin, A.M.; Levy, M.Y.; Dalovisio, A.P.; Bahlis, N.J.; Solh, M.; Sebag, M.; Jakubowiak, A.; Jethava, Y.S.; Costello, C.L.; Chu, M.P.; et al. Preliminary Safety, Efficacy, Pharmacokinetics, and Pharmacodynamics of Subcutaneously (SC) Administered PF-06863135, a B-Cell Maturation Antigen (BCMA)-CD3 Bispecific Antibody, in Patients with Relapsed/Refractory Multiple Myeloma (RRMM). Blood 2020, 136, 8–9.
- Selvaggio, G.; Parolo, S.; Bora, P.; Leonardelli, L.; Harrold, J.; Mehta, K.; Rock, D.A.; Marchetti, L. Computational Analysis of Cytokine Release Following Bispecific T-Cell Engager Therapy: Applications of a Logic-Based Model. Front. Oncol. 2022, 12, 818641.
- Neelapu, S.S.; Tummala, S.; Kebriaei, P.; Wierda, W.; Gutierrez, C.; Locke, F.L.; Komanduri, K.V.; Lin, Y.; Jain, N.; Daver, N.; et al. Chimeric antigen receptor T-cell therapy—Assessment and management of toxicities. Nat. Rev. Clin. Oncol. 2018, 15, 47–62.
- Lee, D.W.; Santomasso, B.D.; Locke, F.L.; Ghobadi, A.; Turtle, C.J.; Brudno, J.N.; Maus, M.V.; Park, J.H.; Mead, E.; Pavletic, S.; et al. ASTCT Consensus Grading for Cytokine Release Syndrome and Neurologic Toxicity Associated with Immune Effector Cells. Biol. Blood Marrow Transplant. 2019, 25, 625–638.
- Si, S.; Teachey, D.T. Spotlight on Tocilizumab in the Treatment of CAR-T-Cell-Induced Cytokine Release Syndrome: Clinical Evidence to Date. Ther. Clin. Risk Manag. 2020, 16, 705–714.
- Gust, J.; Hay, K.A.; Hanafi, L.A.; Li, D.; Myerson, D.; Gonzalez-Cuyar, L.F.; Yeung, C.; Liles, W.C.; Wurfel, M.; Lopez, J.A.; et al. Endothelial Activation and Blood-Brain Barrier Disruption in Neurotoxicity after Adoptive Immunotherapy with CD19 CAR-T Cells. Cancer Discov. 2017, 7, 1404–1419.
- Oluwole, O.O.; Bouabdallah, K.; Muñoz, J.; De Guibert, S.; Vose, J.M.; Bartlett, N.L.; Lin, Y.; Deol, A.; McSweeney, P.A.; Goy, A.H.; et al. Prophylactic corticosteroid use in patients receiving axicabtagene ciloleucel for large B-cell lymphoma. Br. J. Haematol. 2021, 194, 690–700.
- Lum, L.G.; Al-Kadhimi, Z.; Deol, A.; Kondadasula, V.; Schalk, D.; Tomashewski, E.; Steele, P.; Fields, K.; Giroux, M.; Liu, Q.; et al. Phase II clinical trial using anti-CD3 × anti-HER2 bispecific antibody armed activated T cells (HER2 BATs) consolidation therapy for HER2 negative (0-2+) metastatic breast cancer. J. Immunother. Cancer 2021, 9, e002194.
- Sternjak, A.; Lee, F.; Thomas, O.; Balazs, M.; Wahl, J.; Lorenczewski, G.; Ullrich, I.; Muenz, M.; Rattel, B.; Bailis, J.M.; et al. Preclinical Assessment of AMG 596, a Bispecific T-cell Engager (BiTE) Immunotherapy Targeting the Tumor-specific Antigen EGFRvIII. Mol. Cancer Ther. 2021, 20, 925–933.
- Chao, J.; Buxó, E.; Cervantes, A.; Dayyani, F.; Lima, C.M.S.P.R.; Greil, R.; Laarhoven, H.W.M.V.; Lorenzen, S.; Heinemann, V.; Kischel, R.; et al. Trial in progress: A phase I study of AMG 199, a half-life extended bispecific T-cell engager (HLE BiTE) immune therapy, targeting MUC17 in patients with gastric and gastroesophageal junction (G/GEJ) cancer. J. Clin. Oncol. 2020, 38, TPS4649.
- Lu, Y.Y.; Yu, H.; Tang, Y. Efficacy and safety of MUC1 targeted CIK cells for the treatment of advanced liver cancer. J. Clin. Oncol. 2021, 39, e16278.
- Wermke, M.; Felip, E.; Gambardella, V.; Kuboki, Y.; Morgensztern, D.; Oum’Hamed, Z.; Geng, J.; Studeny, M.; Owonikoko, T.K. A phase I, open-label, dose-escalation trial of BI 764532, a DLL3/CD3 bispecific antibody, in patients (pts) with small cell lung carcinoma (SCLC) or other neuroendocrine neoplasms expressing DLL3. J. Clin. Oncol. 2021, 39, TPS8588.
- Danila, D.C.; Waterhouse, D.M.; Appleman, L.J.; Pook, D.W.; Matsubara, N.; Dorff, T.B.; Lee, J.-L.; Armstrong, A.J.; Kim, M.; Horvath, L.; et al. A phase 1 study of AMG 509 in patients (pts) with metastatic castration-resistant prostate cancer (mCRPC). J. Clin. Oncol. 2022, 40, TPS5101.
- Heiss, M.M.; Murawa, P.; Koralewski, P.; Kutarska, E.; Kolesnik, O.O.; Ivanchenko, V.V.; Dudnichenko, A.S.; Aleknaviciene, B.; Razbadauskas, A.; Gore, M.; et al. The trifunctional antibody catumaxomab for the treatment of malignant ascites due to epithelial cancer: Results of a prospective randomized phase II/III trial. Int. J. Cancer 2010, 127, 2209–2221.
- Lordick, F.; Chao, J.; Buxò, E.; van Laarhoven, H.; Lima, C.; Lorenzen, S.; Dayyani, F.; Heinemann, V.; Greil, R.; Stienen, S. 1496TiP Phase I study evaluating safety and tolerability of AMG 910, a half-life extended bispecific T cell engager targeting claudin-18.2 (CLDN18. 2) in gastric and gastroesophageal junction (G/GEJ) adenocarcinoma. Ann. Oncol. 2020, 31, S928–S929.
More
Information
Subjects:
Oncology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
2 times
(View History)
Update Date:
30 May 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





