Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Zhang Guozhong | -- | 1443 | 2023-05-22 14:23:40 | | | |
| 2 | Beatrix Zheng | Meta information modification | 1443 | 2023-05-23 08:11:50 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Zhao, J.; Zhao, Y.; Zhang, G. Key Aspects of Coronavirus Avian Infectious Bronchitis Virus. Encyclopedia. Available online: https://encyclopedia.pub/entry/44667 (accessed on 26 April 2026).
Zhao J, Zhao Y, Zhang G. Key Aspects of Coronavirus Avian Infectious Bronchitis Virus. Encyclopedia. Available at: https://encyclopedia.pub/entry/44667. Accessed April 26, 2026.
Zhao, Jing, Ye Zhao, Guozhong Zhang. "Key Aspects of Coronavirus Avian Infectious Bronchitis Virus" Encyclopedia, https://encyclopedia.pub/entry/44667 (accessed April 26, 2026).
Zhao, J., Zhao, Y., & Zhang, G. (2023, May 22). Key Aspects of Coronavirus Avian Infectious Bronchitis Virus. In Encyclopedia. https://encyclopedia.pub/entry/44667
Zhao, Jing, et al. "Key Aspects of Coronavirus Avian Infectious Bronchitis Virus." Encyclopedia. Web. 22 May, 2023.
Copy Citation
Infectious bronchitis virus (IBV) is an enveloped and positive-sense single-stranded RNA virus. IBV was the first coronavirus to be discovered and predominantly causes respiratory disease in commercial poultry worldwide.
coronavirus
avian infectious bronchitis virus
evolution
genetic diversity
1. Genetic Diversity of Avian Infectious Bronchitis Virus
High rates of mutation, short generation times, and large population sizes drive the rapid evolution of RNA viruses, and consequently, most of the viral outbreaks over the last 100 years have been caused by RNA viruses [1]. Among the RNA viruses, positive-sense single-stranded RNA (+ssRNA) viruses such as IBV have the highest mutation rates [2][3]. IBV lacks a viral polymerase proofreading mechanism, and genetic mutation and recombination can continuously occur in the genome, especially in hypervariable regions of the S1 glycoprotein, which enables the continuous emergence of variants, genotypes, or serotypes [4][5][6][7].
IBV strains are clustered into six genotypes (GⅠ–GⅥ) using the complete nucleotide sequences of the S1 gene, and together these contain 34 distinct viral lineages and several inter-lineage recombinants [8]. Of these genetic lineages, GⅠ-1, GⅠ-3, GⅠ-5, GⅠ-6, GⅠ-7, GⅠ-9, GⅠ-11, GⅠ-12, GⅠ-13, GⅠ-15, GⅠ-16, GⅠ-17, GⅠ-18, GⅠ-19, GⅠ-21, GⅠ-23, GⅠ-24, GⅠ-25, GⅡ-1, GⅡ-2,GⅢ-3, GⅤ, GⅥ-1, and several variants exist in many countries. The GⅠ-1 (Mass-type), GI-13 (793B-type), and GI-19 (QX-type) lineages are widely distributed in many countries, and especially QX-like viruses are now globally widespread (Figure 1). GI-7 (Taiwan-I) lineage strains have also been mainly isolated in China in recent years. Notably, live-attenuated vaccines of the Mass-type (e.g., strain H120), 793B-type (e.g., strain 4/91), Delmarva-type (e.g., DMV/1639 in the USA), and QX-type (e.g., QXL87 in China) are commonly used in many farms or countries [9][10][11][12][13][14]. Therefore, the possibility that several IBV strains are responsible for the re-isolation of the vaccine viruses cannot be excluded.
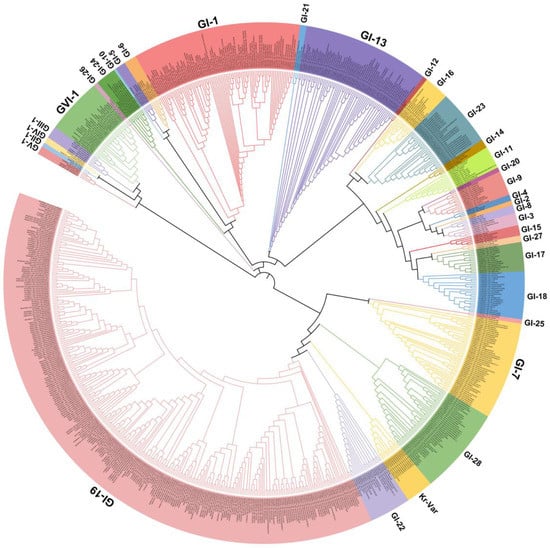
Figure 1. Phylogenetic trees based on the complete S1 glycoprotein gene of the infectious bronchitis virus (IBV). The tree was generated using the neighbor-joining method in MEGA 7 software package with 1000 bootstrap replications. The IBV genotypes were defined as described by Valastro et al. [8].
A timeline with the date of isolation of each presenting IBV strain and used live-attenuated vaccines in China is shown in Figure 2. Suspected IB was first described in 1972 in Guangdong province, China [15]. Early in the 1980s, Mass-like IBV was isolated from Chinese chicken flocks that were genetically similar to that found in the USA. To control the diseases in chickens, the inactivated and live-attenuated vaccines of Mass (H52 and H120) and Conn serotypes were used. The IBV strains from T-type (GI-5), QX-type (GI-19), TC07-2-type (GⅥ-1), and TW-type (GI-7) were successively isolated from the vaccinated chicken flocks [16][17][18]. Currently, GI-19 (QX-type) and GⅥ-1 (TC07-2-type) lineage strains have become the dominant strains of IBV in China [19][20][21].
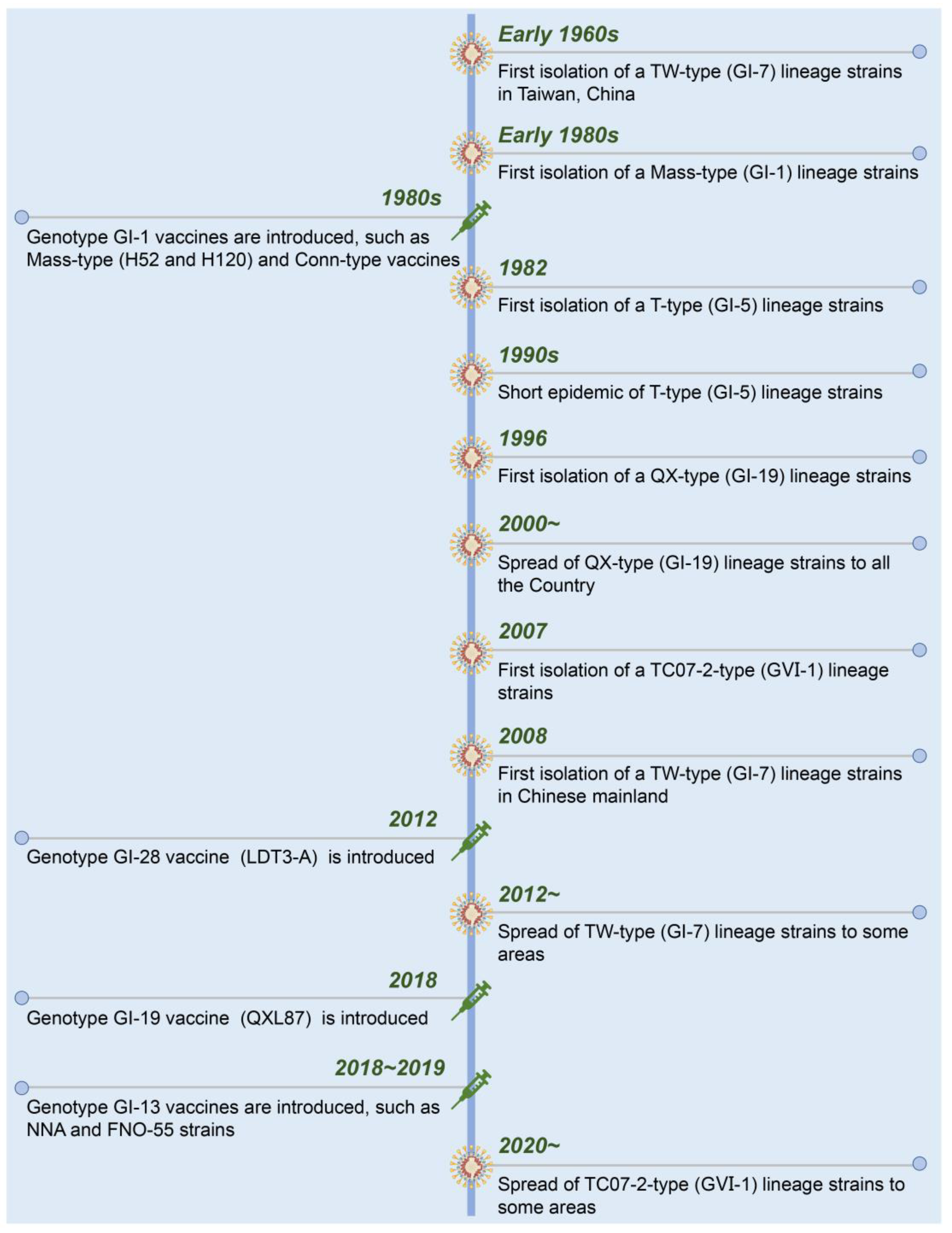
Figure 2. Chronological timeline showing the emergence and spread of the present IBV strains and introduction of live-attenuated vaccines in China.
2. Antigenic Diversity of Avian Infectious Bronchitis Virus
New IBV variants continuously emerge through genetic recombination and mutation, and several exhibit clear antigenicity differences revealed by cross-neutralization tests or monoclonal antibody analysis. The GI-19 lineage (QX-type) strains, for instance, are prevalent in many parts of the world, and are distinct from all other known IBV strains, indicating that these strains belong to different serotypes [22][23][24]. Antigenic variation of IBV is mainly associated with the spike protein because this is a major inducer of neutralizing antibodies and immunoprotection [4][5][20][25]. Other proteins, including structural or nonstructural proteins and accessory proteins, contribute to and influence antigenic differences [4][7]. Thus, antigenic variation of IBV is caused by various factors.
Currently, IBV exists as a serotype score worldwide, and most of these have poor cross-protection with each other [4][26][27][28][29]. This means that vaccination with a particular serotype of IBV may not provide protection against other serotypes. As a result of the antigenic variation, the disease presents a continuing coexistence of multiple serotyping strains, meaning that the production of new IB vaccines that better match the prevalent virus strains will likely continue for a long time. To address the major challenge of IBV antigenic diversity, researchers have focused on understanding the genetic mechanisms underlying IBV evolution, developing new vaccines with cross-protection, improving biosecurity measures to prevent viral spread, and so on.
3. Multiple System Disease Caused by Avian Infectious Bronchitis Virus
Most IBVs can infect the respiratory, renal, and reproductive systems of chickens. Following the initial infection in the respiratory tract with clinical signs (sneezing, gasping, coughing, tracheal rales, nasal discharge, and dyspnea), IBV is disseminated to other tissues by viremia [27]. One possible mechanism is that some strains could infect blood monocytes, and therefore facilitate the dissemination of IBV to visceral organs [30][31]. IBV dissemination beyond the respiratory tract may involve the lymphatic system and infected macrophages as with several other viruses [32][33]. Compared with other IBV strains, the nephropathogenic strains have received increased attention because of their higher virulence in young birds [34]. The kidneys of nephropathogenic IBV-infected chickens are pale, discolored, and enlarged with obvious urate deposition on post-mortem examination [31][35][36]. IBV infections in young chickens are common between the ages of 1 to 30 days and can cause reproductive tract defects [37][38][39]. The infected chickens develop cystic oviducts, leading to false layer syndrome with distinctly low peak egg production [39][40][41][42]. Infection with IBV in laying hens can negatively influence egg production with poor-quality eggs [43][44][45]. Several IBV strains can infect the nervous system of chickens [46]. These strains contain an extra furin cleavage site upstream of the fusion peptide (S2’ site) enabling them to infect monocytes and neuron cells, thus leading to viremia or encephalitis in chickens [30][46].
Although differences in clinical and pathological outcomes in chickens depend on the infecting IBV strain, many IBV strains can simultaneously infect multiple physiological systems in chickens, which is termed “multiple symptoms with one virus” (Figure 3). First, IBVs can infect the respiratory system of chickens, causing damage to the integrity of the respiratory mucosa, which could increase the susceptibility to secondary viral and bacterial infections [22][47][48][49]. Second, IBVs can infect the renal system, leading to increased mortality [22][35][48][50][51]. Third, IBVs can infect the reproductive system of chickens, reducing egg production with poor shell quality or with false layers [38][39][48][52][53]. Fourth, IBVs can infect the nervous system of chickens, causing significantly increased mortality [30][46]. Studies of IBV and host interactions could help to understand the tropism of IBV in various body systems and act as a model to contribute an invaluable resource for studying other pathogenic CoV diseases.
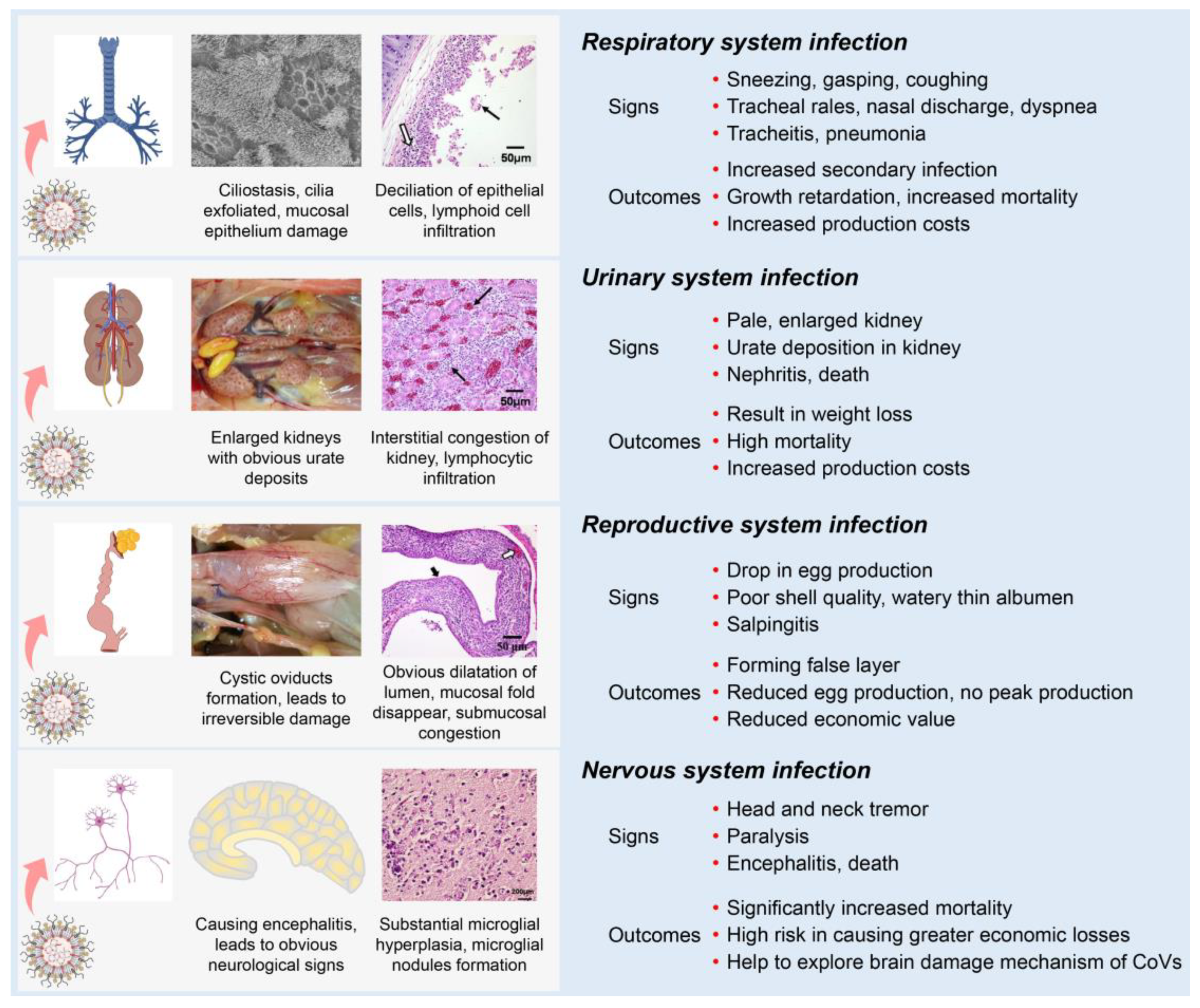
Figure 3. Schematic diagram showing clinical manifestations of IBV infection. Clinical signs and main outcomes are summarized, showing the effects of the virus on different body systems.
4. Prevention and Control of Avian Infectious Bronchitis Virus
In modern poultry farms, an effective management system and biosecurity measures are of primary importance to control infectious diseases. This premise is based entirely on the correct understanding of the factors influencing viral spread, as with the avian influenza virus [54][55][56]. Therefore, a more comprehensive analysis of the epidemiological aspects behind the spread of IBV, especially the spreading determinants, is critical and urgently required [57][58].
Despite drawbacks, including severe reactions to vaccination at the day of age, the likelihood of viral recombination, and frequent vaccine replacement caused by viral mutation, vaccination remains the most effective control measure for IB [59][60][61][62][63]. Efficient and properly performed vaccination may reduce the emergence of clinical signs, infectious pressure, and viral population size [59].
Currently, almost all commercial chicken flocks are vaccinated against IBV. The most effective IB vaccine is the live-attenuated vaccine, and H120 is one of the excellent vaccine strains widely used in the world. The vaccination protocol for IBV can vary depending on the vaccine used and the specific conditions of the poultry operation. In general, chicks are vaccinated at one day old or at hatchery, and then again several times throughout their life to maintain immunity. Booster vaccinations may be given at 7–10 days of age, at 3–4 weeks of age, and then every 5–6 weeks thereafter. Because of the many existing variants worldwide, the concept of protectotype has been increasingly accepted for controlling IB. One of the most commonly applied protectotype vaccination protocols against IB in the EU is the simultaneous or alternate use of Ma5 and 4/91 vaccine strains to provide protection against homologous or heterologous IBV strains [64]. In China, the H120 vaccine combined with an endemic strain, such as QXL87 (QX-like, GI-19), FNO-55 (4/91-like, GI-13), or LDT3-A (YN-like, GI-28), is commonly used to control IB.
Different vaccine platforms have been designed to develop effective vaccines against IBV. However, new vaccine techniques are rarely used because of the relatively complex immune protection mechanism and are currently only in the laboratory research stage [14][65][66][67][68][69][70][71]. Consequently, both inactivated and live-attenuated IBV vaccines are widely available worldwide [72][73][74][75].
References
- Grellet, E.; L’Hôte, I.; Goulet, A.; Imbert, I. Replication of the coronavirus genome: A paradox among positive-strand RNA viruses. J. Biol. Chem. 2022, 298, 101923.
- Echeverría, N.; Moratorio, G.; Cristina, J.; Moreno, P. Hepatitis C virus genetic variability and evolution. World J. Hepatol. 2015, 7, 831–845.
- Thébaud, G.; Chadoeuf, J.; Morelli, M.J.; McCauley, J.W.; Haydon, D.T. The relationship between mutation frequency and replication strategy in positive-sense single-stranded RNA viruses. Proc. Biol. Sci. 2010, 277, 809–817.
- Cavanagh, D. Coronavirus avian infectious bronchitis virus. Vet. Res. 2007, 38, 281–297.
- Ali, A.; Ojkic, D.; Elshafiee, E.A.; Shany, S.; El-Safty, M.M.; Shalaby, A.A.; Abdul-Careem, M.F. Genotyping and in silico analysis of Delmarva (DMV/1639) infectious bronchitis virus (IBV) spike 1 (S1) glycoprotein. Genes 2022, 13, 1617.
- Webb, I.; Keep, S.; Littolff, K.; Stuart, J.; Freimanis, G.; Britton, P.; Davidson, A.D.; Maier, H.J.; Bickerton, E. The genetic stability, replication kinetics and cytopathogenicity of recombinant avian coronaviruses with a T16A or an A26F mutation within the E protein is cell-type dependent. Viruses 2022, 14, 1784.
- Kumar, S.; Thambiraja, T.S.; Karuppanan, K.; Subramaniam, G. Omicron and Delta variant of SARS-CoV-2: A comparative computational study of spike protein. J. Med. Virol. 2022, 94, 1641–1649.
- Valastro, V.; Holmes, E.C.; Britton, P.; Fusaro, A.; Jackwood, M.W.; Cattoli, G.; Monne, I. S1 gene-based phylogeny of infectious bronchitis virus: An attempt to harmonize virus classification. Infect. Genet. Evol. 2016, 39, 349–364.
- Bande, F.; Arshad, S.S.; Omar, A.R.; Hair-Bejo, M.; Mahmuda, A.; Nair, V. Global distributions and strain diversity of avian infectious bronchitis virus: A review. Anim. Health Res. Rev. 2017, 18, 70–83.
- Jung, J.S.; Lee, R.; Yoon, S.I.; Lee, G.S.; Sung, H.W.; Kwon, H.M.; Park, J. Genetic and immunological characterization of commercial infectious bronchitis virus vaccines used in Korea. Arch. Virol. 2022, 167, 2123–2132.
- Tizard, I.R. Vaccination against coronaviruses in domestic animals. Vaccine 2020, 38, 5123–5130.
- Abozeid, H.H.; Naguib, M.M. Infectious bronchitis virus in Egypt: Genetic diversity and vaccination strategies. Vet. Sci. 2020, 7, 204.
- Hassan, M.S.H.; Buharideen, S.M.; Ali, A.; Najimudeen, S.M.; Goldsmith, D.; Coffin, C.S.; Cork, S.C.; van der Meer, F.; Abdul-Careem, M.F. Efficacy of commercial infectious bronchitis vaccines against Canadian Delmarva (DMV/1639) infectious bronchitis virus infection in layers. Vaccines 2022, 10, 1194.
- Qin, Y.; Teng, Q.; Feng, D.; Pei, Y.; Zhao, Y.; Zhang, G. Development of a nanoparticle multiepitope DNA vaccine against virulent infectious bronchitis virus challenge. J. Immunol. 2022, 208, 1396–1405.
- Xin, C.A.; Chen, T.J. The research of chicken infectious bronchitis-I. Isolation and identification of chicken infectious bronchitis virus in Guangzhou. J. S. China Agric. Coll. 1982, 3, 90–98.
- Liu, S.; Wang, Y.; Ma, Y.; Han, Z.; Zhang, Q.; Shao, Y.; Chen, J.; Kong, X. Identification of a newly isolated avian infectious bronchitis coronavirus variant in China exhibiting affinity for the respiratory tract. Avian Dis. 2008, 52, 306–314.
- Xu, G.; Liu, X.Y.; Zhao, Y.; Chen, Y.; Zhao, J.; Zhang, G.Z. Characterization and analysis of an infectious bronchitis virus strain isolated from southern China in 2013. Virol. J. 2016, 13, 40.
- Wang, Y.D.; Wang, Y.L.; Zhang, Z.C. Isolation and identification of glandular stomach type IBV (QX IBV) in chickens. Chin. J. Anim. Quar. 1998, 15, 1–3.
- Bo, Z.; Chen, S.; Zhang, C.; Guo, M.; Cao, Y.; Zhang, X.; Wu, Y. Pathogenicity evaluation of GVI-1 lineage infectious bronchitis virus and its long-term effects on reproductive system development in SPF hens. Front. Microbiol. 2022, 13, 1049287.
- Luo, H.; Qin, J.; Chen, F.; Xie, Q.; Bi, Y.; Cao, Y.; Xue, C. Phylogenetic analysis of the S1 glycoprotein gene of infectious bronchitis viruses isolated in China during 2009–2010. Virus Genes 2012, 44, 19–23.
- Zhao, Y.; Zhang, H.; Zhao, J.; Zhong, Q.; Jin, J.H.; Zhang, G.Z. Evolution of infectious bronchitis virus in China over the past two decades. J. Gen. Virol. 2016, 97, 1566–1574.
- Cheng, J.; Huo, C.; Zhao, J.; Liu, T.; Li, X.; Yan, S.; Wang, Z.; Hu, Y.; Zhang, G. Pathogenicity differences between QX-like and Mass-type infectious bronchitis viruses. Vet. Microbiol. 2018, 213, 129–135.
- Yan, S.; Liu, X.; Zhao, J.; Xu, G.; Zhao, Y.; Zhang, G. Analysis of antigenicity and pathogenicity reveals major differences among QX-like infectious bronchitis viruses and other serotypes. Vet. Microbiol. 2017, 203, 167–173.
- Xu, G.; Cheng, J.; Ma, S.; Jia, W.; Yan, S.; Zhang, G. Pathogenicity differences between a newly emerged TW-like strain and a prevalent QX-like strain of infectious bronchitis virus. Vet. Microbiol. 2018, 227, 20–28.
- Ekiri, A.B.; Armson, B.; Adebowale, K.; Endacott, I.; Galipo, E.; Alafiatayo, R.; Horton, D.L.; Ogwuche, A.; Bankole, O.N.; Galal, H.M.; et al. Evaluating disease threats to sustainable poultry production in Africa: Newcastle disease, infectious bursal disease, and avian infectious bronchitis in commercial poultry flocks in Kano and Oyo States, Nigeria. Front. Vet. Sci. 2021, 8, 730159.
- Kariithi, H.M.; Volkening, J.D.; Leyson, C.M.; Afonso, C.L.; Christy, N.; Decanini, E.L.; Lemiere, S.; Suarez, D.L. Genome sequence variations of infectious bronchitis virus serotypes from commercial chickens in Mexico. Front. Vet. Sci. 2022, 9, 931272.
- Ghetas, A.M. Infectious bronchitis virus genotypes in the Middle East. Avian Dis. 2021, 65, 647–651.
- De Wit, J.J.; de Wit, M.K.; Cook, J.K.A. Infectious bronchitis virus types affecting european countries—A review. Avian Dis. 2021, 65, 643–648.
- Raj, G.D.; Jones, R.C. Infectious bronchitis virus: Immunopathogenesis of infection in the chicken. Avian Pathol. 1997, 26, 677–706.
- Cheng, J.; Zhao, Y.; Hu, Y.; Zhao, J.; Xue, J.; Zhang, G. The furin-S2’ site in avian coronavirus plays a key role in central nervous system damage progression. J. Virol. 2021, 95, e02447-20.
- Reddy, V.R.; Trus, I.; Desmarets, L.M.; Li, Y.; Theuns, S.; Nauwynck, H.J. Productive replication of nephropathogenic infectious bronchitis virus in peripheral blood monocytic cells, a strategy for viral dissemination and kidney infection in chickens. Vet. Res. 2016, 47, 70.
- Amarasinghe, A.; Abdul-Cader, M.S.; Nazir, S.; De Silva Senapathi, U.; van der Meer, F.; Cork, S.C.; Gomis, S.; Abdul-Careem, M.F. Infectious bronchitis corona virus establishes productive infection in avian macrophages interfering with selected antimicrobial functions. PLoS ONE 2017, 12, e0181801.
- Barrow, A.D.; Burgess, S.C.; Baigent, S.J.; Howes, K.; Nair, V.K. Infection of macrophages by a lymphotropic herpesvirus: A new tropism for Marek’s disease virus. J. Gen. Virol. 2003, 84, 2635–2645.
- Feng, J.; Hu, Y.; Ma, Z.; Yu, Q.; Zhao, J.; Liu, X.; Zhang, G. Virulent avian infectious bronchitis virus, People’s Republic of China. Emerg. Infect. Dis. 2012, 8, 1994–2001.
- Chong, K.T.; Apostolov, K. The pathogenesis of nephritis in chickens induced by infectious bronchitis virus. J. Comp. Pathol. 1982, 92, 199–211.
- Tang, X.; Qi, J.; Sun, L.; Zhao, J.; Zhang, G.; Zhao, Y. Pathological effect of different avian infectious bronchitis virus strains on the bursa of Fabricius of chickens. Avian Pathol. 2022, 51, 339–348.
- Broadfoot, D.I.; Pomeroy, B.S.; Smith, W.M., Jr. Effects of infectious bronchitis in baby chicks. Poult. Sci. 1956, 35, 757–762.
- Crinion, R.A.P.; Hofstad, M.S. Pathogenicity of four serotypes of avian infectious bronchitis virus for the oviduct of young chickens of various ages. Avian Dis. 1972, 16, 351–363.
- Zhong, Q.; Hu, Y.X.; Jin, J.H.; Zhao, Y.; Zhao, J.; Zhang, G.Z. Pathogenicity of virulent infectious bronchitis virus isolate YN on hen ovary and oviduct. Vet. Microbiol. 2016, 193, 100–105.
- Shao, L.; Zhao, J.; Li, L.; Huang, X.; Yang, H.; Cheng, J.; Liu, C.; Zhang, G. Pathogenic characteristics of a QX-like infectious bronchitis virus strain SD in chickens exposed at different ages and protective efficacy of combining live homologous and heterologous vaccination. Vet. Res. 2020, 51, 86.
- Cook, J.K.; Jackwood, M.; Jones, R.C. The long view: 40 years of infectious bronchitis research. Avian Pathol. 2012, 41, 239–250.
- Jones, R.C.; Jordan, F.T. Persistence of virus in the tissues and development of the oviduct in the fowl following infection at day old with infectious bronchitis virus. Res. Vet. Sci. 1972, 13, 52–60.
- Bisgaard, M. The influence of infectious bronchitis virus on egg production, fertility, hatchability and mortality rate in chickens. Nord. Vet. Med. 1976, 28, 368–376.
- Sevoian, M.; Levine, P.P. Effects of infectious bronchitis on the reproductive tracts, egg production, and egg quality of laying chickens. Avian Dis. 1957, 1, 136–164.
- Zhao, Y.; Xie, D.; Zhang, K.; Cheng, J.; Xu, G.; Zhang, G. Pathogenicity of a GI-22 genotype infectious bronchitis virus isolated in China and protection against it afforded by GI-19 vaccine. Virus Res. 2019, 267, 59–66.
- Cheng, J.; Zhao, Y.; Xu, G.; Zhang, K.; Jia, W.; Sun, Y.; Zhao, J.; Xue, J.; Hu, Y.; Zhang, G. The S2 subunit of QX-type infectious bronchitis coronavirus spike protein is an essential determinant of neurotropism. Viruses 2019, 11, 972.
- Hoerr, F.J. The pathology of infectious bronchitis. Avian Dis. 2021, 65, 600–611.
- Zhang, X.; Yan, K.; Zhang, C.; Guo, M.; Chen, S.; Liao, K.; Bo, Z.; Cao, Y.; Wu, Y. Pathogenicity comparison between QX-type and Mass-type infectious bronchitis virus to different segments of the oviducts in laying phase. Virol. J. 2022, 9, 62.
- Amarasinghe, A.; De Silva Senapathi, U.; Abdul-Cader, M.S.; Popowich, S.; Marshall, F.; Cork, S.C.; van der Meer, F.; Gomis, S.; Abdul-Careem, M.F. Comparative features of infections of two Massachusetts (Mass) infectious bronchitis virus (IBV) variants isolated from Western Canadian layer flocks. BMC Vet. Res. 2018, 14, 391.
- Li, N.; Huang, C.; Chen, W.; Li, Z.; Hu, G.; Li, G.; Liu, P.; Hu, R.; Zhuang, Y.; Luo, J.; et al. Nephropathogenic infectious bronchitis virus mediates kidney injury in chickens via the TLR7/NF-κB signaling Axis. Front. Cell Infect. Microbiol. 2022, 12, 865283.
- Khanh, N.P.; Tan, S.W.; Yeap, S.K.; Lee, H.J.; Choi, K.S.; Hair-Bejo, M.; Bich, T.N.; Omar, A.R. Comparative pathogenicity of Malaysian QX-like and variant infectious bronchitis virus strains in chickens at different age of exposure to the viruses. J. Comp. Pathol. 2018, 161, 43–54.
- Mueller Slay, A.; Franca, M.; Jackwood, M.; Jordan, B. Infection with IBV DMV/1639 at a young age leads to increased incidence of cystic oviduct formation associated with false layer syndrome. Viruses 2022, 14, 852.
- Hassan, M.S.H.; Ali, A.; Buharideen, S.M.; Goldsmith, D.; Coffin, C.S.; Cork, S.C.; van der Meer, F.; Boulianne, M.; Abdul-Careem, M.F. Pathogenicity of the Canadian Delmarva (DMV/1639) infectious bronchitis virus (IBV) on female reproductive tract of chickens. Viruses 2021, 13, 2488.
- Boender, G.J.; Hagenaars, T.J.; Bouma, A.; Nodelijk, G.; Elbers, A.R.; de Jong, M.C.; van Boven, M. Risk maps for the spread of highly pathogenic avian influenza in poultry. PLoS Comput. Biol. 2007, 3, e71.
- Mannelli, A.; Busani, L.; Toson, M.; Bertolini, S.; Marangon, S. Transmission parameters of highly pathogenic avian influenza (H7N1) among industrial poultry farms in northern Italy in 1999–2000. Prev. Vet. Med. 2007, 81, 318–322.
- Singh, M.; Toribio, J.A.; Scott, A.B.; Groves, P.; Barnes, B.; Glass, K.; Moloney, B.; Black, A.; Hernandez-Jover, M. Assessing the probability of introduction and spread of avian influenza (AI) virus in commercial Australian poultry operations using an expert opinion elicitation. PLoS ONE 2018, 13, e0193730.
- Franzo, G.; Tucciarone, C.M.; Moreno, A.; Legnardi, M.; Massi, P.; Tosi, G.; Trogu, T.; Ceruti, R.; Pesente, P.; Ortali, G.; et al. Phylodynamic analysis and evaluation of the balance between anthropic and environmental factors affecting IBV spreading among Italian poultry farms. Sci. Rep. 2020, 10, 7289.
- Najimudeen, S.M.; Hassan, M.S.H.; Cork, S.C.; Abdul-Careem, M.F. Infectious bronchitis coronavirus infection in chickens: Multiple system disease with immune suppression. Pathogens 2020, 9, 779.
- Franzo, G.; Tucciarone, C.M.; Blanco, A.; Nofrarías, M.; Biarnés, M.; Cortey, M.; Majó, N.; Catelli, E.; Cecchinato, M. Effect of different vaccination strategies on IBV QX population dynamics and clinical outbreaks. Vaccine 2016, 34, 5670–5676.
- Moreno, A.; Franzo, G.; Massi, P.; Tosi, G.; Blanco, A.; Antilles, N.; Biarnes, M.; Majó, N.; Nofrarías, M.; Dolz, R.; et al. A novel variant of the infectious bronchitis virus resulting from recombination events in Italy and Spain. Avian Pathol. 2017, 46, 28–35.
- Sjaak de Wit, J.J.; Cook, J.K.A. Factors influencing the outcome of infectious bronchitis vaccination and challenge experiments. Avian Pathol. 2014, 43, 485–497.
- Alluwaimi, A.M.; Alshubaith, I.H.; Al-Ali, A.M.; Abohelaika, S. The coronaviruses of animals and birds: Their zoonosis, vaccines, and models for SARS-CoV and SARS-CoV2. Front. Vet. Sci. 2020, 7, 582287.
- Ji, W.; Wang, W.; Zhao, X.; Zai, J.; Li, X. Cross-species transmission of the newly identified coronavirus 2019-nCoV. J. Med. Virol. 2020, 92, 433–440.
- Smialek, M.; Tykalowski, B.; Dziewulska, D.; Stenzel, T.; Koncicki, A. Immunological aspects of the efficiency of protectotype vaccination strategy against chicken infectious bronchitis. BMC Vet Res. 2017, 13, 44.
- Bande, F.; Arshad, S.S.; Bejo, M.H.; Moeini, H.; Omar, A.R. Progress and challenges towards the development of vaccines against avian infectious bronchitis. J. Immunol. Res. 2015, 2015, 424860.
- Johnson, M.A.; Pooley, C.; Ignjatovic, J.; Tyack, S.G. A recombinant fowl adenovirus expressing the S1 gene of infectious bronchitis virus protects against challenge with infectious bronchitis virus. Vaccine 2003, 21, 2730–2736.
- Kapczynski, D.R.; Hilt, D.A.; Shapiro, D.; Sellers, H.S.; Jackwood, M.W. Protection of chickens from infectious bronchitis by in ovo and intramuscular vaccination with a DNA vaccine expressing the S1 glycoprotein. Avian Dis. 2003, 47, 272–285.
- Li, H.; Wang, Y.; Han, Z.; Wang, Y.; Liang, S.; Jiang, L.; Hu, Y.; Kong, X.; Liu, S. Recombinant duck enteritis viruses expressing major structural proteins of the infectious bronchitis virus provide protection against infectious bronchitis in chickens. Antivir. Res. 2016, 130, 19–26.
- Lv, L.; Li, X.; Liu, G.; Li, R.; Liu, Q.; Shen, H.; Wang, W.; Xue, C.; Cao, Y. Production and immunogenicity of chimeric virus-like particles containing the spike glycoprotein of infectious bronchitis virus. J. Vet. Sci. 2014, 15, 209–216.
- Shi, X.M.; Zhao, Y.; Gao, H.B.; Jing, Z.; Wang, M.; Cui, H.Y.; Tong, G.Z.; Wang, Y.F. Evaluation of recombinant fowlpox virus expressing infectious bronchitis virus S1 gene and chicken interferon-g gene for immune protection against heterologous strains. Vaccine 2011, 29, 1576–1582.
- Yang, T.; Wang, H.N.; Wang, X.; Tang, J.N.; Lu, D.; Zhang, Y.F.; Guo, Z.C.; Li, Y.L.; Gao, R.; Kang, R.M. The protective immune response against infectious bronchitis virus induced by multi-epitope based peptide vaccines. Biosci. Biotech. Biochem. 2009, 73, 1500–1504.
- Bijlenga, G.; Cook, J.K.; Gelb, J., Jr.; de Wit, J.J. Development and use of the H strain of avian infectious bronchitis virus from the Netherlands as a vaccine: A review. Avian Pathol. 2004, 33, 550–557.
- Chhabra, R.; Forrester, A.; Lemiere, S.; Awad, F.; Chantrey, J.; Ganapathy, K. Mucosal, cellular, and humoral immune responses induced by different live infectious bronchitis virus vaccination regimes and protection conferred against infectious bronchitis virus Q1 strain. Clin. Vaccine Immunol. 2015, 22, 1050–1059.
- Finney, P.M.; Box, P.G.; Holmes, H.C. Studies with a bivalent infectious bronchitis killed virus vaccine. Avian Pathol. 1990, 19, 435–450.
- Lee, H.J.; Youn, H.N.; Kwon, J.S.; Lee, Y.J.; Kim, J.H.; Lee, J.B.; Park, S.Y.; Choi, I.S.; Song, C.S. Characterization of a novel live attenuated infectious bronchitis virus vaccine candidate derived from a Korean nephropathogenic strain. Vaccine 2010, 28, 2887–2894.
More
Information
Subjects:
Veterinary Sciences
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
705
Revisions:
2 times
(View History)
Update Date:
23 May 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





