Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Haimei Chen | -- | 2091 | 2023-05-15 13:23:27 | | | |
| 2 | Rita Xu | Meta information modification | 2091 | 2023-05-16 04:05:53 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Chen, H.; Chen, H.; Kardos, L.; Szabó, V. Contaminations in Ion-Adsorption Type Rare Earths Tailings. Encyclopedia. Available online: https://encyclopedia.pub/entry/44310 (accessed on 07 May 2026).
Chen H, Chen H, Kardos L, Szabó V. Contaminations in Ion-Adsorption Type Rare Earths Tailings. Encyclopedia. Available at: https://encyclopedia.pub/entry/44310. Accessed May 07, 2026.
Chen, Haimei, Haibin Chen, Levente Kardos, Veronika Szabó. "Contaminations in Ion-Adsorption Type Rare Earths Tailings" Encyclopedia, https://encyclopedia.pub/entry/44310 (accessed May 07, 2026).
Chen, H., Chen, H., Kardos, L., & Szabó, V. (2023, May 15). Contaminations in Ion-Adsorption Type Rare Earths Tailings. In Encyclopedia. https://encyclopedia.pub/entry/44310
Chen, Haimei, et al. "Contaminations in Ion-Adsorption Type Rare Earths Tailings." Encyclopedia. Web. 15 May, 2023.
Copy Citation
Rare earth elements, particularly middle and heavy rare earth, are among the most valuable resources in the pursuit of a greener economy. The production of middle and heavy rare earth elements heavily relies on ion adsorption, which constitutes over 80% of global output and is centered in southern China. Unfortunately, the extensive mining activities have led to severe environmental pollution, resource depletion, and risks to human health.
ion adsorption rare earth
biochar
remediation
1. Introduction
1.1. Background
Rare Earth Elements (REE) have received considerable interest due to their vital applications in various areas, including agriculture, electronics, green energy, the national defense industry, and precision equipment [1][2]. They are defined by the International Union of Pure and Applied Chemistry as a group of 17 elements that naturally occur in earth-parent materials, including scandium, yttrium, and the lanthanide series [3]. Although REE are present in more than 250 minerals (silicates, carbonates, oxides, and phosphates), their distribution is unpredictable and scarce, which makes extraction difficult as they are not found in concentrated mineral structures [4][5][6]. The total crustal abundance of REE is 169 mg/kg, with 137.8 mg/kg of light rare earth elements (LREE: La to Gd) and 31.34 mg/kg of heavy rare earth elements (HREE: Tb to Lu, plus Sc and Y), where LREE are 4.39 times higher than HREE [7]. Mid-heavy REE (including Gd) are considered critical resources, and nearly 80% of the world’s supplies are from ion-adsorption type rare-earth (IAT-Res) ores in South China [8][9][10].
IAT-Res deposit is a unique REE resource. REE-oxide are trivalent cations that REE are adsorbed onto clay minerals during weathering [11][12]. In the past few decades, unregulated and intensive extraction for REE in the IAT-Res ore consequence ecological pollutant has been pronounced in these areas [10][13]. Because of the uncertain bioaccumulation mechanisms, there have been only a few successful plant restoration occurrences on IAT-Res mine tailings so far [7]. The crucial lack of HREE resources, however, makes contamination control and IAT-Res recovery from contaminations increasingly crucial.
1.2. Ion-Adsorption Type Rare Earths (IAT-Res)
REE-containing minerals are classified into two main types: mineral type REE (MT-Res) ores and ion adsorption type REE (IAT-Res) ores, with either HREE or LREE dominating [14][15]. The world reserves of different types of REE are shown in Figure 1. The MT-Res ores are rich in LREE and are primarily composed of bastnaesite and monazite, whereas the IAT-Res ores which are weathering profiles developed on protoliths, are primarily composed of middle-heavy REE [1][8][16]. Clay minerals such as kaolinite, halloysite, illite, and montmorillonite are the primary components (40–70%) of the weathering crust throughout the weathering process [12][16]. Meanwhile, REE trivalent cations can bind to clay minerals to form REE-oxide (hydrated or hydroxyl-hydrated ions) [8][14][17]. In this circumstance, an IAT-Res deposit develops.
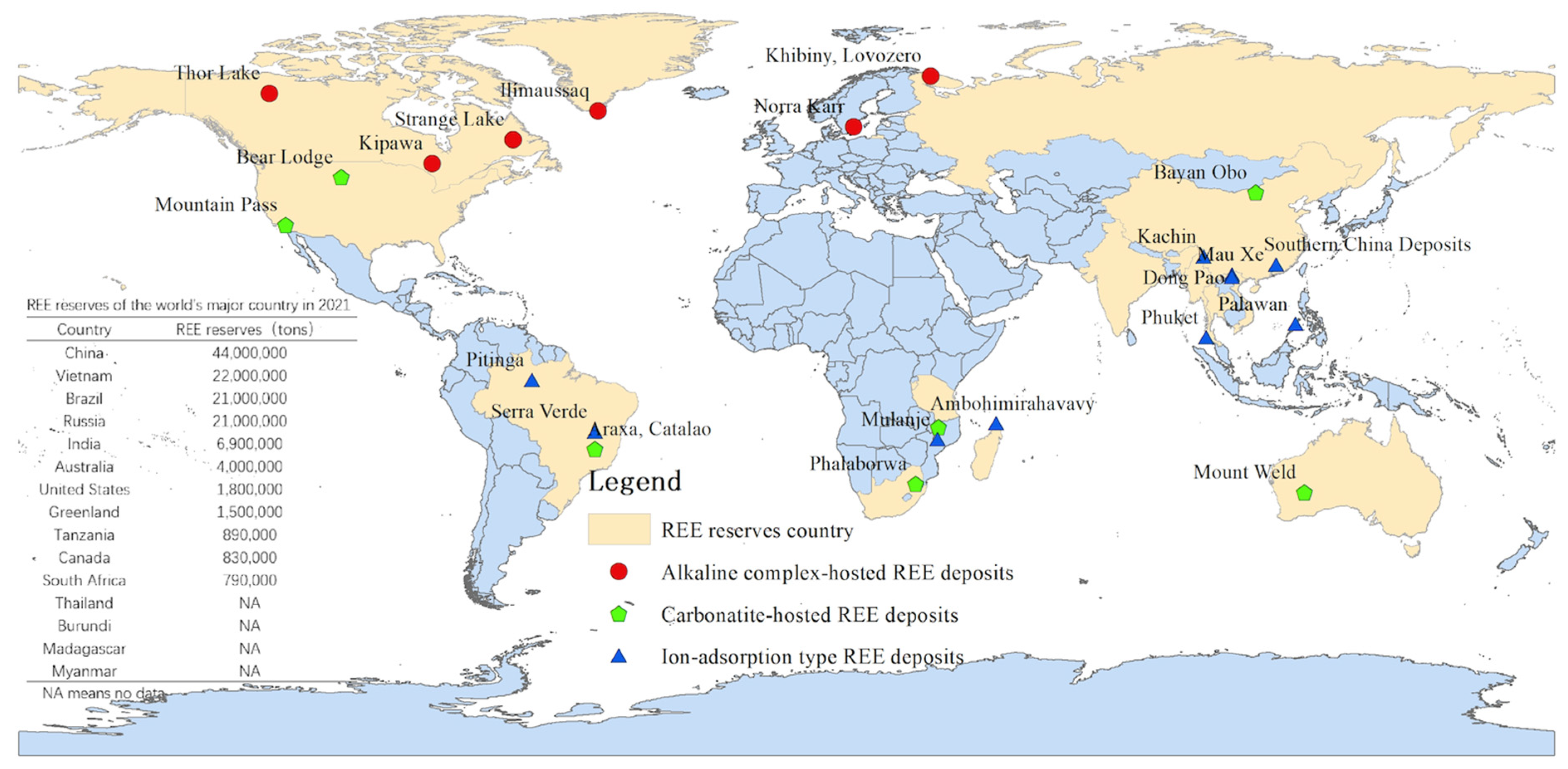
Figure 1. REE types and distributions in the world.
The world’s most important source of HREE are IAT-Res deposits, which are primarily found in the southern Chinese provinces, such as Fujian, Guangdong, Guangxi, Hunan, and Jiangxi [8][18][19]. Since the discovery of IAT-Res ores in Jiangxi, China, in 1969, a series of hydrometallurgy methods, including barrel/pool leaching, heap leaching, and in-situ leaching, have been developed by scientists and engineers [16][20]. Pool leaching is similar to leap leaching. They both require complete stripping of the subsoil in the mining area, causing physical problems with the territory’s geography, such as landslides and loss of vegetation cover, soil erosion, crop production damage, and biodiversity loss [21]. Over the last few decades of exploration, mining for IAT-Res has caused many environmental problems in these areas. Since 2011, China has restricted REE exports because of environmental protection concerns [22]. The exploration of IAT-Res resources has expanded to Brazil, Laos, Thailand, Indonesia, Madagascar, the Philippines, and the United States [23][24][25]. Thus, environmental management in southern China will be a critical example in the developing of IAT-Res mining.
1.3. Usages and Demands
The application of REE takes place in different sectors, such as agriculture and modern technologies. Global REE demand has spiked in recent years and is expected to increase from 167,000 tons in 2020 to 280,000 tons in 2030 [23][26][27]. In agriculture, REE are utilized as micro-fertilizers to promote the growth and yield of plants and livestock [5][28]. Since they are used in so many modern conveniences (including lighter flints, fluorescent lights, batteries, lasers, and permanent magnets), REE are now considered to be strategic components [15][22][29]. REE are applied in relative amounts, yet they are critical components of devices in green economics, high-precision medical equipment, and defense industries [15]. Most REE is used in magnetic field generators, which are in turn used in a broad variety of low-carbon technologies (such as wind turbines and electric and hybrid automobiles) [30][31]. Figure 2 shows a breakdown of their holdings.
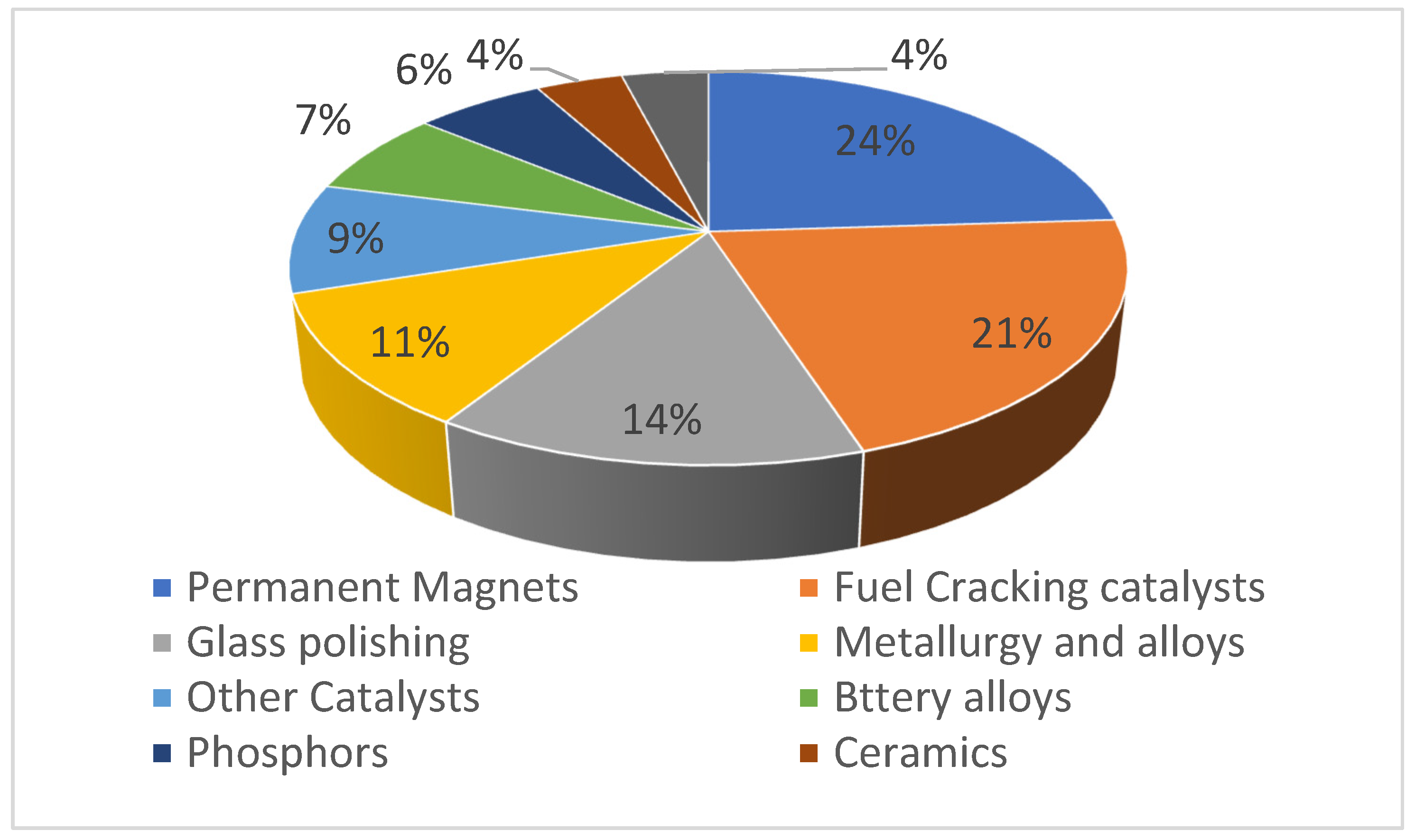
Figure 2. Share of REE application in different sectors.
2. Contaminations in IAT-Res Tailings
In contrast to the fact that REE, especially HREE, are critical resources in many countries, aggressive mining and manufacturing are causing severe environmental contamination and overlaid REE are considered as hazards in these areas [31][32]. Compared to the open-pit mining technologies for bastnaesite (MT-Res), fewer environmental impactions occurred from manufacturing IAT-Res [31][33].
The contamination in the IAT-Res mining areas can be categorized into soil physicochemical properties, ammonium pollution, and REE pollution. Pool and heap leaching methods have been abandoned for IAT-Res mining since 2008, leading to the widespread adoption of in-situ leaching, which does not require the removal of surface vegetation [9][20]. However, contaminations left behind from the early production of IAT-Res by pool or heap leaching are still apparent, trees do not grow at these tailings [21][30]. In-situ leaching, which is considered an eco-friendly method requires a higher amount of ammonium leachate during processing. As the extraction of IAT-Res follows the mechanism, Clay–REE3++3 NH+4→ Clay–(NH+4)3+REE3+
[34][35], the ammonium-containing leakage could further lead to greater terrestrial and aquatic eco-toxicity ratings [20][21][30]. Nevertheless, the toxicity of discharged REE to the environment and humans is usually underestimated because it is commonly evaluated as a group, but their toxicity coefficients can be higher than heavy metals, such as Lu, Eu, and Tb are higher than Pb and Cu [36][37][38].
2.1. Vegetation Loss and Soil Degeneration
IAT-Res only has 0.05–0.3 wt% REO but extracting REE from IAT-Res is simple and inexpensive [14][17][20]. To produce one ton of REO using pool or heap leaching, one needs to strip off 150–200 m2 of vegetation and 1500–2000 m3 of topsoil, leading to 150–200 m2 and 1700 t of tailings [39]. In recent decades, intensive exploration of IAT-Res resources has resulted in more than 100 km2 of REE mine tailings in Southern China [10][17]. Because of the serious environmental consequences of unregulated small-scale extraction for IAT-Res resources, China is stepping up efforts to limit illegal mining and conduct artificial vegetation restoration in mine tailings [22][40][41][42].
As IAT-Res are scattered across the distribution, surface mining has become the predominant method. This has changed how land is used and caused soil degradation in Southern China. Further contamination caused irreparable harm to the ecosystem, including soil erosion, air pollution, biodiversity loss, and human health issues [17][38]. Moreover, red soil predominates in these regions, making natural restoration after extensive vegetation loss problematic. For example, Figure 3 shows a satellite image of IAT-Res mine regions and mine tailings where no grass grows even after many years of rehabilitation.
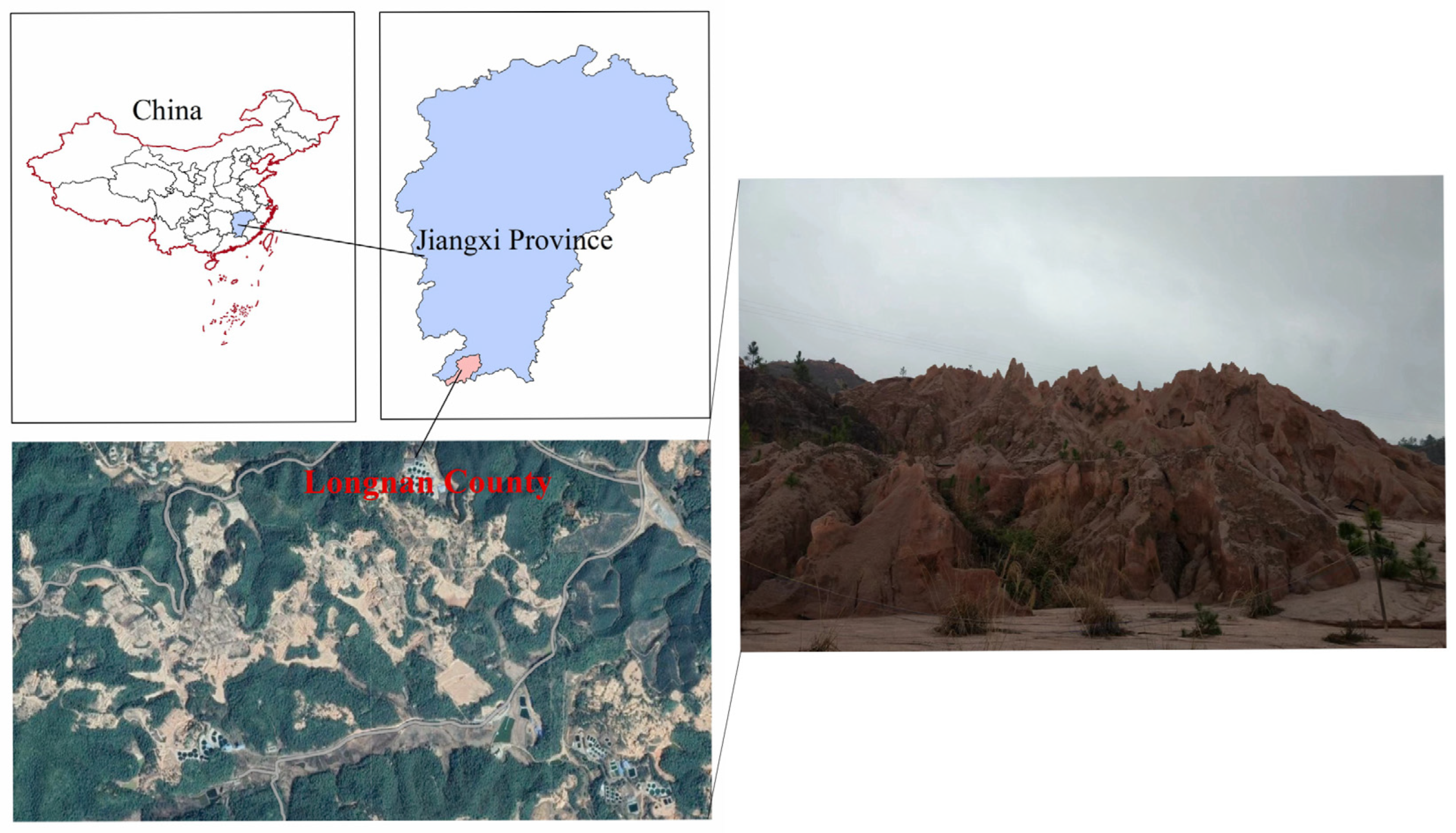
Figure 3. IAT-Res mine tailing in Southern China.
2.2. Ammonium Contamination
In-situ leaching has replaced pool leaching and heap leaching in IAT-Res mining since 2008, as it does not result in severe soil deterioration, such as landslides and soil erosion, nor does it leave behind a barren wasteland [43][44]. Nevertheless, in-situ leaching requires a substantial quantity of the leaching reagent ammonium sulfate , which, after leaching, enters the soil and groundwater [10][45]. Around seven to ten tons of ammonium sulfate are required to manufacture one ton of REO; hence, the excessive amount of ammonium sulfate at a mining site leads to changes in the surface soil and groundwater characteristics [17][43][46]. In the IAT-Res mine tailing soil profiles, the ammonia nitrogen concentration ranged from 49.8 to 77.7 mg/kg, whereas it was between 5 and 6 mg/kg in the original mine soil [47]. According to the Pollution Discharge Standards for the Rare Earth Industry, the limits for water or air in the form of ammonia pollution should not be higher than 25 mg/L [31][48]. However, after in-situ leaching, the wastewater in the IAT-Res mine tailings was found to contain ammonia-nitrogen up to 3000–4000 mg/L [10]. Ammonium pollution extends to nearby habitats; Hao et al. (2016) [49] evaluated different surface and shallow aquatic systems from the REE mining area in southern Jiangxi Province. They stated that the concentration of NH+4 paddy field water (1.28 mg/L), pond water (4.53 mg/L), and stream water (8.31 mg/L) all exceeded the standard (0.02 mg/L). In addition, the leachate produces a large amount of ions and lead to soil acidification [50]. In conclusion, leaching solutions containing ammonium sulfate not only lead to severe ammonia-nitrogen pollution and soil acidification, but they also contain low levels of REE and flow off into rivers when it rains, resulting in incalculable REE loss and contamination.
2.3. REE Contamination
REE enter the environment through various pathways, including mining, atmospheric deposition, and end-life product disposal [5]. In the IAT-Res resources reserved surroundings, mining is the majority means of exceeding REE in the ecological environment and is considered an emerging contaminant. Mining for the IAT-Res in Southern China has left a large area of tailings that contain 409–1035 mg/kg of REE [51][52]. In the majority of these sites, the REE level exceeds the mining-grade threshold (500 mg/kg) [53]. For instance, the total REE level of 976.94 mg/kg found by Jin et al. (2019) [2] in a mine tailing in China’s Jiangxi province, is 4.53 times and 5.09 times greater than the soil in Jiangxi province and the entire country, respectively. Liu et al. (2019) [52] also found the total REE in a mine tailing soil (392 mg/kg) in Jiangxi province was two times higher than the control sites (192 mg/kg). Similarly, soil samples from 118 locations around Fujian province were analyzed for their Rare Earth Element (REE) composition by Chen et al. (2019) [54]. With an average of 255.34 mg/kg, the total REE content was significantly higher than the Fujian province background value of 186.76 mg/kg and the HREE (77.07 mg/kg) was two times higher than the background value. This also agreed with the study of Li et al. (2013) [38] in Hetian County, Fujian; the soil REE (242.92 mg/kg) was higher than the background soil (135.85 mg/kg). Studies that claim soil REE contamination in IAT-Res mining tailings are listed in Table 1. Nevertheless, the content of REE in the soil is also determined by the soil pH, sorption capacity, and salt content and studies focusing on REE contaminations are spare, thus more systematic studies and comprehensive information still need to be conducted.
Table 1. REE content in the soil of IAT-Res mining tailings (mg/kg).
Despite the soil contaminations, overloaded REE also spread to the surrounding water system. The concentration of REE in rivers in the mining area is 55.72 mg/L, which is 8974.7 times higher than the contrast area [2]. Studies by Liu et al. (2019) [52] and Hao et al. (2016) [49] also found that the total REE concentrations in the stream water (4.46 mg/L and 1.596 mg/L), pond water (0.069 mg/L), paddy field water (0.031 mg/L), spring water (0.035 mg/L), and well water (0.03 mg/L) all exceeded the recommended contents (0.02 mg/L). Although it is applied in agriculture as a micronutrient, studies show that REEs also cause adverse effects in plants and animals [55][56][57]. Furthermore, Li et al. (2013) [38] documented higher REE in human blood and hair in the mine tailing surrounding areas than in control sites. To date, the determination of REE in the environment is limited because more sensitive analytical techniques allow a quantitative assessment of the trace concentrations in various biological materials [4].
2.4. Current Problems
The shortage of REE is a global problem. They are considered the most critical raw materials group with the highest supply risk by the European Commission, the US Department of Energy and the British Geological Survey [23][58][59]. Although the only REE element estimated to have a resource life of fewer than 1000 years is Eu, which has a resource life of about 600 years [15], the dramatic growing consumption of particular REE still marks the importance of the recovery potential.
On the other hand, the extensive mining for IAT-Res has led to severe environmental contamination and toxic effects on surrounding habits. REE mine tailings contain high concentrations of REE even after years of abandonment, and these REE are highly exchangeable (>20% of total REE) [10][52][54]. The contradiction between limited resources and resource demand growth has shifted governments’ awareness of the importance of resource conservation. A new concept has been promoted: from waste to valuable resources. However, in contrast to the emerging studies focusing on REE pollution and biochar as promising adsorbents for toxic contamination, the information on biochar application in IAT-Res tailings remains sparse.
References
- Balaram, V. Rare Earth Elements: A Review of Applications, Occurrence, Exploration, Analysis, Recycling, and Environmental Impact. Geosci. Front. 2019, 10, 1285–1303.
- Jin, S.; Hu, Z.; Man, B.; Pan, H.; Kong, X.; Jin, D. Application of Phosphate-Containing Materials Affects Bioavailability of Rare Earth Elements and Bacterial Community in Soils. Sci. China Technol. Sci. 2019, 62, 1616–1627.
- International Union of Pure and Applied Chemistry (IUPAC). Nomenclature of Inorganic Chemistry. Chem. Int. Newsmag. IUPAC 2005, 27.
- Gupta, C.K.; Krishnamurthy, N. Extractive Metallurgy of Rare Earths; CRC Press: Boca Raton, FL, USA, 2005; ISBN 978-0-415-33340-5.
- Hu, Z.; Haneklaus, S.; Sparovek, G.; Schnug, E. Rare Earth Elements in Soils. Commun. Soil Sci. Plant Anal. 2006, 37, 1381–1420.
- Jordens, A.; Cheng, Y.P.; Waters, K.E. A Review of the Beneficiation of Rare Earth Element Bearing Minerals. Miner. Eng. 2013, 41, 97–114.
- Zhou, B.; Li, Z.; Chen, C. Global Potential of Rare Earth Resources and Rare Earth Demand from Clean Technologies. Minerals 2017, 7, 203.
- Borst, A.M.; Smith, M.P.; Finch, A.A.; Estrade, G.; Villanova-de-Benavent, C.; Nason, P.; Marquis, E.; Horsburgh, N.J.; Goodenough, K.M.; Xu, C.; et al. Adsorption of Rare Earth Elements in Regolith-Hosted Clay Deposits. Nat. Commun. 2020, 11, 4386.
- Chi, R.; Tian, J.; Li, Z.; Peng, C.; Wu, Y.; Li, S.; Wang, C.; Zhou, Z. Existing State and Partitioning of Rare Earth on Weathered Ores. J. Rare Earths 2005, 23, 756.
- Liu, J.; Wu, X.; Huang, K.; Liu, W.; Zhao, Z.; Liu, H. Instability Behavior of Bubble Supported Organic Liquid Membrane in Extraction of Low-Concentration Rare Earths from in-Situ Leaching Solutions of Ion-Adsorption Ores. Miner. Eng. 2020, 159, 106645.
- Ou, X.; Chen, Z.; Chen, X.; Li, X.; Wang, J.; Ren, T.; Chen, H.; Feng, L.; Wang, Y.; Chen, Z.; et al. Redistribution and Chemical Speciation of Rare Earth Elements in an Ion–Adsorption Rare Earth Tailing, Southern China. Sci. Total Environ. 2022, 821, 153369.
- Xu, Q.; Yang, L.; Wang, D.; Hou, X.; Sun, Y.; Zhou, X.; Zhou, X.; Li, Y. Evaluating the Fractionation of Ion-Adsorption Rare Earths for in-Situ Leaching and Metallogenic Mechanism. J. Rare Earths 2018, 36, 1333–1341.
- Luo, C.; Deng, Y.; Inubushi, K.; Liang, J.; Zhu, S.; Wei, Z.; Guo, X.; Luo, X. Sludge Biochar Amendment and Alfalfa Revegetation Improve Soil Physicochemical Properties and Increase Diversity of Soil Microbes in Soils from a Rare Earth Element Mining Wasteland. Int. J. Environ. Res. Public Health 2018, 15, 965.
- Feng, X.; Onel, O.; Council-Troche, M.; Noble, A.; Yoon, R.-H.; Morris, J.R. A Study of Rare Earth Ion-Adsorption Clays: The Speciation of Rare Earth Elements on Kaolinite at Basic PH. Appl. Clay Sci. 2021, 201, 105920.
- Haque, N.; Hughes, A.; Lim, S.; Vernon, C. Rare Earth Elements: Overview of Mining, Mineralogy, Uses, Sustainability and Environmental Impact. Resources 2014, 3, 614–635.
- Nie, W.; Zhang, R.; He, Z.; Zhou, J.; Wu, M.; Xu, Z.; Chi, R.; Yang, H. Research Progress on Leaching Technology and Theory of Weathered Crust Elution-Deposited Rare Earth Ore. Hydrometallurgy 2020, 193, 105295.
- Yang, X.J.; Lin, A.; Li, X.-L.; Wu, Y.; Zhou, W.; Chen, Z. China’s Ion-Adsorption Rare Earth Resources, Mining Consequences and Preservation. Environ. Dev. 2013, 8, 131–136.
- Kynicky, J.; Smith, M.P.; Xu, C. Diversity of Rare Earth Deposits: The Key Example of China. Elements 2012, 8, 361–367.
- Xiao, Y.; Feng, Z.; Hu, G.; Huang, L.; Huang, X.; Chen, Y.; Long, Z. Reduction Leaching of Rare Earth from Ion-Adsorption Type Rare Earths Ore with Ferrous Sulfate. J. Rare Earths 2016, 34, 917–923.
- Huang, X.-W.; Long, Z.-Q.; Wang, L.-S.; Feng, Z.-Y. Technology Development for Rare Earth Cleaner Hydrometallurgy in China. Rare Met. 2015, 34, 215–222.
- Chen, H.B.; Chen, H.M.; Chen, Z.B.; Chen, Z.Q. The Ecological Impacts of Residues from the Heap Leaching of Ion-Adsorption Rare Earth Clays. Int. J. Environ. Sci. Technol. 2023.
- Mancheri, N.A.; Sprecher, B.; Bailey, G.; Ge, J.; Tukker, A. Effect of Chinese Policies on Rare Earth Supply Chain Resilience. Resour. Conserv. Recycl. 2019, 142, 101–112.
- United States Geological Survey (USGS). Mineral Commodity Summaries; United States Geological Survey (USGS): Reston, VA, USA, 2022; 202p.
- da Costa, T.B.; Silva, M.G.C.d.; Vieira, M.G.A. Recovery of Rare-Earth Metals from Aqueous Solutions by Bio/Adsorption Using Non-Conventional Materials: A Review with Recent Studies and Promising Approaches in Column Applications. J. Rare Earths 2020, 38, 339–355.
- Estrade, G.; Marquis, E.; Smith, M.; Goodenough, K.; Nason, P. REE Concentration Processes in Ion Adsorption Deposits: Evidence from the Ambohimirahavavy Alkaline Complex in Madagascar. Ore Geol. Rev. 2019, 112, 103027.
- Dushyantha, N.; Batapola, N.; Ilankoon, I.M.S.K.; Rohitha, S.; Premasiri, R.; Abeysinghe, B.; Ratnayake, N.; Dissanayake, K. The Story of Rare Earth Elements (REEs): Occurrences, Global Distribution, Genesis, Geology, Mineralogy and Global Production. Ore Geol. Rev. 2020, 122, 103521.
- Goodenough, K.M.; Wall, F.; Merriman, D. The Rare Earth Elements: Demand, Global Resources, and Challenges for Resourcing Future Generations. Nat. Resour. Res. 2018, 27, 201–216.
- Tommasi, F.; Thomas, P.J.; Pagano, G.; Perono, G.A.; Oral, R.; Lyons, D.M.; Toscanesi, M.; Trifuoggi, M. Review of Rare Earth Elements as Fertilizers and Feed Additives: A Knowledge Gap Analysis. Arch. Environ. Contam. Toxicol. 2021, 81, 531–540.
- Anastopoulos, I.; Bhatnagar, A.; Lima, E.C. Adsorption of Rare Earth Metals: A Review of Recent Literature. J. Mol. Liq. 2016, 221, 954–962.
- Asadollahzadeh, M.; Torkaman, R.; Torab-Mostaedi, M. Extraction and Separation of Rare Earth Elements by Adsorption Approaches: Current Status and Future Trends. Sep. Purif. Rev. 2021, 50, 417–444.
- Bailey, G.; Joyce, P.J.; Schrijvers, D.; Schulze, R.; Sylvestre, A.M.; Sprecher, B.; Vahidi, E.; Dewulf, W.; Van Acker, K. Review and New Life Cycle Assessment for Rare Earth Production from Bastnäsite, Ion Adsorption Clays and Lateritic Monazite. Resour. Conserv. Recycl. 2020, 155, 104675.
- Adeel, M.; Lee, J.Y.; Zain, M.; Rizwan, M.; Nawab, A.; Ahmad, M.A.; Shafiq, M.; Yi, H.; Jilani, G.; Javed, R.; et al. Cryptic Footprints of Rare Earth Elements on Natural Resources and Living Organisms. Environ. Int. 2019, 127, 785–800.
- Schreiber, A.; Marx, J.; Zapp, P.; Hake, J.-F.; Voßenkaul, D.; Friedrich, B. Environmental Impacts of Rare Earth Mining and Separation Based on Eudialyte: A New European Way. Resources 2016, 5, 32.
- Liu, L.; Wang, X.; Wen, Q.; Jia, Q.; Liu, Q. Interspecific Associations of Plant Populations in Rare Earth Mining Wasteland in Southern China. Int. Biodeterior. Biodegrad. 2017, 118, 82–88.
- Qiu, T.; Zhu, D.; Fang, X.; Zeng, Q.; Gao, G.; Zhu, H. Leaching Kinetics of Ionic Rare-Earth in Ammonia-Nitrogen Wastewater System Added with Impurity Inhibitors. J. Rare Earths 2014, 32, 1175–1183.
- Chen, H.; Chen, Z.; Chen, Z.; Ou, X.; Chen, J. Calculation of Toxicity Coefficient of Potential Ecological Risk Assessment of Rare Earth Elements. Bull. Environ. Contam. Toxicol. 2020, 104, 582–587.
- Chen, Z. The Biological Hormesis Effects of Rare Earths and Potential Effects of Application of Rare Earths on Agricultural Eco-Environment. Rural Eco-Environ. 2004, 20, 1–5. (In Chinese)
- Li, X.; Chen, Z.; Chen, Z.; Zhang, Y. A Human Health Risk Assessment of Rare Earth Elements in Soil and Vegetables from a Mining Area in Fujian Province, Southeast China. Chemosphere 2013, 93, 1240–1246.
- Gao, Z.; Zhou, Q. Contamination from Rare Earth Ore Strip Mining and Its Impacts on Resources and Ecovironment. Chin. J. Ecol. 2011, 30, 2915–2922. (In Chinese)
- Chen, Z.; Chen, Z. Effects of Ecological Restoration Measures on the Distribution of Dicranopteris Dichotoma at the Microscale in the Red Soil Hilly Region of China. PLoS ONE 2018, 13, e0204743.
- Ou, Z.; Pang, S.; He, Q.; Peng, Y.; Huang, X.; Shen, W. Effects of Vegetation Restoration and Environmental Factors on Understory Vascular Plants in a Typical Karst Ecosystem in Southern China. Sci. Rep. 2020, 10, 12011.
- Szamałek, K.; Konopka, G.; Zglinicki, K.; Marciniak-Maliszewska, B. New Potential Source of Rare Earth Elements. Gospod. Surowcami Min. 2013, 29, 59–76.
- Chen, S.; Zha, X.; Bai, Y.; Wang, L. Evaluation of Soil Erosion Vulnerability on the Basis of Exposure, Sensitivity, and Adaptive Capacity: A Case Study in the Zhuxi Watershed, Changting, Fujian Province, Southern China. Catena 2019, 177, 57–69.
- Wen, X.; Duan, C.; Zhang, D. Effect of Simulated Acid Rain on Soil Acidification and Rare Earth Elements Leaching Loss in Soils of Rare Earth Mining Area in Southern Jiangxi Province of China. Environ. Earth Sci. 2013, 69, 843–853.
- Yang, L.; Wang, D.; Li, C.; Sun, Y.; Zhou, X.; Li, Y. Searching High Efficiency and Environmentally Benign Leaching Reagent for Ion-Adsorption Rare Earth Based on the Zeta Potential of Clay Particles. Green Chem. 2016, 18, 1839–1854.
- Xiao, Y.; Chen, Y.; Feng, Z.; Huang, X.; Huang, L.; Long, Z.; Cui, D. Leaching Characteristics of Ion-Adsorption Type Rare Earths Ore with Magnesium Sulfate. Trans. Nonferrous Met. Soc. China 2015, 25, 3784–3790.
- Zhang, Q.; Ren, F.; Li, F.; Chen, G.; Yang, G.; Wang, J.; Du, K.; Liu, S.; Li, Z. Ammonia Nitrogen Sources and Pollution along Soil Profiles in an In-Situ Leaching Rare Earth Ore. Environ. Pollut. 2020, 267, 115449.
- GB 26451-2011; China Rare Earth Emission Standards of Pollutants from Rare Earths Industry. National Standard of the People’s Republic of China: Beijing, China, 2011. Available online: https://std.samr.gov.cn/gb/search/gbDetailed?id=71F772D7DFD1D3A7E05397BE0A0AB82A (accessed on 8 May 2023). (In Chinese)
- Hao, X.; Wang, D.; Wang, P.; Wang, Y.; Zhou, D. Evaluation of Water Quality in Surface Water and Shallow Groundwater: A Case Study of a Rare Earth Mining Area in Southern Jiangxi Province, China. Environ. Monit. Assess. 2016, 188, 24.
- Zhang, Q.; Wan, G.; Zhou, C.; Luo, J.; Lin, J.; Zhao, X. Rehabilitation Effect of the Combined Application of Bamboo Biochar and Coal Ash on Ion-Adsorption-Type Rare Earth Tailings. J. Soils Sediments 2020, 20, 3351–3357.
- Chao, Y.; Liu, W.; Chen, Y.; Chen, W.; Zhao, L.; Ding, Q.; Wang, S.; Tang, Y.-T.; Zhang, T.; Qiu, R.-L. Structure, Variation, and Co-Occurrence of Soil Microbial Communities in Abandoned Sites of a Rare Earth Elements Mine. Environ. Sci. Technol. 2016, 50, 11481–11490.
- Liu, W.-S.; Guo, M.-N.; Liu, C.; Yuan, M.; Chen, X.-T.; Huot, H.; Zhao, C.-M.; Tang, Y.-T.; Morel, J.L.; Qiu, R.-L. Water, Sediment and Agricultural Soil Contamination from an Ion-Adsorption Rare Earth Mining Area. Chemosphere 2019, 216, 75–83.
- Ding, J.; Deng, G. Main Problems in the Current Ionic Adsorption Rare Earth Exploration Specifications and Their Amendment Proposals. Nonferrous Met. Sci. Eng. 2013, 4, 96–102. (In Chinese)
- Chen, H.; Chen, Z.; Chen, Z.; Ma, Q.; Zhang, Q. Rare Earth Elements in Paddy Fields from Eroded Granite Hilly Land in a Southern China Watershed. PLoS ONE 2019, 14, e0222330.
- Martinez, R.E.; Pourret, O.; Faucon, M.-P.; Dian, C. Effect of Rare Earth Elements on Rice Plant Growth. Chem. Geol. 2018, 489, 28–37.
- Pagano, G.; Guida, M.; Tommasi, F.; Oral, R. Health Effects and Toxicity Mechanisms of Rare Earth Elements—Knowledge Gaps and Research Prospects. Ecotoxicol. Environ. Saf. 2015, 115, 40–48.
- Thomas, P.J.; Carpenter, D.; Boutin, C.; Allison, J.E. Rare Earth Elements (REEs): Effects on Germination and Growth of Selected Crop and Native Plant Species. Chemosphere 2014, 96, 57–66.
- British Geological Survey. An Update to the Supply Risk Index for Elements or Element Groups That Are of Economic Value; British Geological Survey: Nottingham, UK, 2015. Available online: https://www2.bgs.ac.uk/mineralsuk/download/statistics/risk_list_2015.pdf (accessed on 8 May 2023).
- European Commission. Critical Raw Materials Resilience: Charting a Path towards Greater Security and Sustainability; Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions. Brussels, 3.9.2020 COM(2020) 474 Final. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52020DC0474 (accessed on 8 May 2023).
More
Information
Subjects:
Environmental Sciences
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
2.7K
Revisions:
2 times
(View History)
Update Date:
16 May 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





