
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jussuf Kaifi | + 589 word(s) | 589 | 2020-03-20 10:52:08 | | | |
| 2 | Nicole Yin | + 1 word(s) | 590 | 2020-10-27 07:45:03 | | |
Video Upload Options
The biological nature of the various populations of circulating tumor cells (CTCs) within the blood is still not well understood. Tumor cell fusion with immune cells is a longstanding hypothesis that has caught more attention in recent times. Specifically, fusion of tumor cells with macrophages might lead to the development of metastasis by acquiring features such as genetic and epigenetic heterogeneity, chemotherapeutic resistance, and immune tolerance. A unique circulating cell population has been identified as being potential fusions cells, characterized by distinct, large, polymorphonuclear cancer-associated cells with a dual epithelial and macrophage phenotype. Artificial fusion of tumor cells with macrophages leads to migratory, invasive, and metastatic phenotypes. Further studies might investigate whether these have a potential impact on the immune response towards the cancer. Such fusion cells could be a key component in cancer metastasis, and therefore, evolve as a diagnostic and therapeutic target in cancer precision medicine.
1. Introduction
Recent reports on circulating cancer-associated cells with both epithelial and macrophage/myeloid phenotypes in cancer patients, combined with genetic evidence, have supported the idea that fusion has a critical role in cancer progression (Figure 1)[1][2][3][4].
2. Fusion of Tumor Cells with Macrophages
Macrophage M1 or M2 polarization appears to be critical for various aspects of immune responses to cancer and its progression[5]. Macrophage infiltration of the primary tumor and polarization depend on cytokines in the tumor microenvironment (TME)[6]. Within the TME, tumor-associated macrophage polarization to the M1 phenotype can be triggered through bacterial lipopolysaccharide (LPS) and by T helper 1 (Th1) cytokines, such as IFN-γ and also by TNF-α[7]. The M1 phenotype is associated with anti-tumor properties[8]. M2 phenotype macrophages have pro-tumoral effects, leading to increased cancer cell survival, proliferation, invasiveness, and immunosuppression in favor of the tumor[5]. M2 polarization is induced by T helper 2 cytokines interleukin (IL)-4, IL-13, macrophage colony-stimulating factor (M-CSF), and transforming growth factor (TGF)-β[8]. M2 macrophages are anti-inflammatory, immunosuppressive, and promote cancer progression, chemoresistance, and metastasis[9][10]. M2 macrophages have critical interactions with tumor cells, but also with cells associated with tumor progression, such as Th2 cells, cancer-associated fibroblasts, regulatory T cells (Tregs), and myeloid-derived suppressor cells[5]. M2 polarization phenotypes have also been observed in tumor fusion cells[11]. Importantly, macrophages also have a high fusogenic potential, which is also likely to occur with tumor cells[12][13][8][14]. In vitro and in vivo studies suggest that metastatic cells can be the result of the fusion of tumor cells with cells of hematopoietic/myeloid lineage, specifically with macrophages[15][12][16][13][17][18][19][20] . Importantly, patient-derived tumor-macrophage fusion cells were shown to have M2 macrophage phenotypes[17][18] . In a murine melanoma metastasis model, certain clones of lung metastasis cells had properties of melanoma cell—macrophage fusion cells[21]. Importantly, fusion of tumor cells with macrophages is supported through the observation of these fusion cells in cancer patients[17][18][22][23] . Macrophage fusion receptor DAP12 expression is associated with higher metastatic rates in breast cancer patients[13][24][25] . It remains unclear whether tumor-associated macrophages fuse within the tumor microenvironment at the site of the tumor, in the blood, or in the lymphatic system. Understanding molecular fusion mechanisms between macrophages and tumor cells and the impact that fusion cells have on the immune system is of high interest in identifying therapeutic targets.
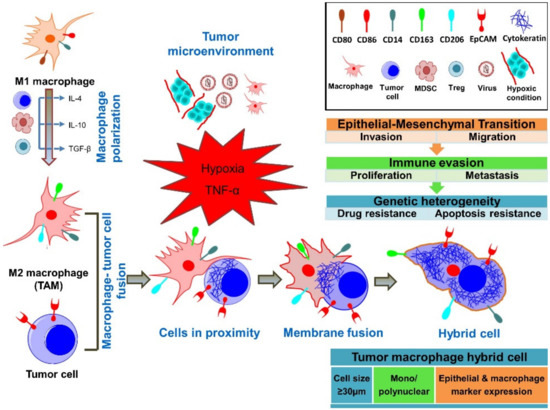
Figure 1. Concepts of fusion between tumor cells and macrophages. It is hypothesized that tumor-associated M2-polarized macrophages (TAMs) fuse their membranes with tumor cells, forming a tumor–macrophage hybrid cell. These fusion cells are large, mononuclear/polynuclear, and express both epithelial and myeloid markers. Importantly, fusion cells exert pro-tumorigenic and pro-metastatic effects through the outlined mechanisms.
References
- John M. Pawelek; Ashok K. Chakraborty; Chapter 10 The Cancer Cell–Leukocyte Fusion Theory of Metastasis. Advances in Cancer Research 2008, 101, 397-444, 10.1016/s0065-230x(08)00410-7.
- Clawson, G.A. Cancer. Fusion for moving. Science 2013, 342, 699–700.
- Ramakrishnan, M.; Mathur, S.R.; Mukhopadhyay, A. Fusion-derived epithelial cancer cells express hematopoietic markers and contribute to stem cell and migratory phenotype in ovarian carcinoma. Cancer Res. 2013, 73, 5360–5370.
- Germana Rappa; Javier Mercapide; Aurelio Lorico; Spontaneous Formation of Tumorigenic Hybrids between Breast Cancer and Multipotent Stromal Cells Is a Source of Tumor Heterogeneity. The American Journal of Pathology 2012, 180, 2504-2515, 10.1016/j.ajpath.2012.02.020.
- Masoud Najafi; Nasser Hashemi Goradel; Bagher Farhood; Eniseh Salehi; Maryam Shabani Nashtaei; Neda Khanlarkhani; Zahra Khezri; Jamal Majidpoor; Morteza Abouzaripour; Mohsen Habibi; et al.Iraj Ragerdi KashaniKeywan Mortezaee Macrophage polarity in cancer: A review. Journal of Cellular Biochemistry 2018, 120, 2756-2765, 10.1002/jcb.27646.
- Ding, J.; Jin, W.; Chen, C.; Shao, Z.; Wu, J.; Tumor associated macrophage x cancer cell hybrids may acquire cancer stem cell properties in breast cancer. PLoS ONE 2012, 7, e41942.
- Olga S. Blomberg; Lorenzo Spagnuolo; Karin E. De Visser; Immune regulation of metastasis: mechanistic insights and therapeutic opportunities. Disease Models & Mechanisms 2018, 11, dmm036236, 10.1242/dmm.036236.
- Bruno Sainz; Emily Carron; Mireia Vallespinós; Heather L. Machado; Cancer Stem Cells and Macrophages: Implications in Tumor Biology and Therapeutic Strategies. Mediators of Inflammation 2016, 2016, 1-15, 10.1155/2016/9012369.
- Svensson, J.; Jenmalm, M.C.; Matussek, A.; Geffers, R.; Berg, G.; Ernerudh, J. Macrophages at the fetal-maternal interface express markers of alternative activation and are induced by M-CSF and IL-10. J. Immunol. 2011, 187, 3671–3682.
- Mitchem, J.B.; Brennan, D.J.; Knolhoff, B.L.; Belt, B.A.; Zhu, Y.; Sanford, D.E.; Belaygorod, L.; Carpenter, D.; Collins, L.; Piwnica-Worms, D.; et al. Targeting tumor-infiltrating macrophages decreases tumor-initiating cells, relieves immunosuppression, and improves chemotherapeutic responses. Cancer Res. 2013, 73, 1128–1141.
- Kazuya Kuwada; Shunsuke Kagawa; Ryuichi Yoshida; Shuichi Sakamoto; Atene Ito; Megumi Watanabe; Takeshi Ieda; Shinji Kuroda; Satoru Kikuchi; Hiroshi Tazawa; et al.Toshiyoshi Fujiwara The epithelial-to-mesenchymal transition induced by tumor-associated macrophages confers chemoresistance in peritoneally disseminated pancreatic cancer. Journal of Experimental & Clinical Cancer Research 2018, 37, 1-10, 10.1186/s13046-018-0981-2.
- Greggory S. Laberge; Eric Duvall; Zachary Grasmick; Kay Haedicke; John M Pawelek; A Melanoma Lymph Node Metastasis with a Donor-Patient Hybrid Genome following Bone Marrow Transplantation: A Second Case of Leucocyte-Tumor Cell Hybridization in Cancer Metastasis. PLoS ONE 2017, 12, e0168581, 10.1371/journal.pone.0168581.
- Ivan Shabo; Kristine Midtbö; Henrik Andersson; Emma Åkerlund; Hans Olsson; Pia Wegman; Cecilia Gunnarsson; Annelie Lindström; Macrophage traits in cancer cells are induced by macrophage-cancer cell fusion and cannot be explained by cellular interaction. BMC Cancer 2015, 15, 1-11, 10.1186/s12885-015-1935-0.
- Elizabeth H. Chen; Eric N. Olson; Unveiling the Mechanisms of Cell-Cell Fusion. Science 2005, 308, 369-373, 10.1126/science.1104799.
- Rossitza Lazova; Greggory S. Laberge; Eric Duvall; Nicole Spoelstra; Vincent Klump; Mario Sznol; Dennis Cooper; Richard A. Spritz; Joseph T. Chang; John M. Pawelek; et al. A Melanoma Brain Metastasis with a Donor-Patient Hybrid Genome following Bone Marrow Transplantation: First Evidence for Fusion in Human Cancer. PLOS ONE 2013, 8, e66731, 10.1371/journal.pone.0066731.
- Dominik M. Duelli; Hesed M. Padilla-Nash; David Berman; Kathleen M. Murphy; Thomas Ried; Yuri Lazebnik; A Virus Causes Cancer by Inducing Massive Chromosomal Instability through Cell Fusion. Current Biology 2007, 17, 431-437, 10.1016/j.cub.2007.01.049.
- Clawson, G.A.; Matters, G.L.; Xin, P.; McGovern, C.; Wafula, E.; dePamphilis, C.; Meckley, M.; Wong, J.; Stewart, L.; D’Jamoos, C.; et al. “Stealth dissemination” of macrophage-tumor cell fusions cultured from blood of patients with pancreatic ductal adenocarcinoma. PLoS ONE 2017, 12, e0184451.
- Clawson, G.A.; Matters, G.L.; Xin, P.; Imamura-Kawasawa, Y.; Du, Z.; Thiboutot, D.M.; Helm, K.F.; Neves, R.I.; Abraham, T. Macrophage-tumor cell fusions from peripheral blood of melanoma patients. PLoS ONE 2015, 10, e0134320.
- John M Pawelek; Cancer cell fusion with migratory bone marrow-derived cells as an explanation for metastasis: new therapeutic paradigms. Future Oncology 2008, 4, 449-452, 10.2217/14796694.4.4.449.
- A Chakraborty; R Lazova; S Davies; H Bäckvall; F Ponten; D Brash; J Pawelek; Donor DNA in a renal cell carcinoma metastasis from a bone marrow transplant recipient. Bone Marrow Transplantation 2004, 34, 183-186, 10.1038/sj.bmt.1704547.
- Ashok Chakraborty; Stefano Sodi; M Rachkovsky; N Kolesnikova; J T Platt; J L Bolognia; J M Pawelek; A spontaneous murine melanoma lung metastasis comprised of host x tumor hybrids.. Cancer Research 2000, 60, 2512–2519.
- Clawson, G.A.; Kimchi, E.; Patrick, S.D.; Xin, P.; Harouaka, R.; Zheng, S.; Berg, A.; Schell, T.; Staveley-O’Carroll, K.F.; Neves, R.I.; et al. Circulating tumor cells in melanoma patients. PLoS ONE 2012, 7, e41052.
- Kaifi, J.T.; Kunkel, M.; Das, A.; Harouaka, R.A.; Dicker, D.T.; Li, G.; Zhu, J.; Clawson, G.A.; Yang, Z.; Reed, M.F.; et al. Circulating tumor cell isolation during resection of colorectal cancer lung and liver metastases: A prospective trial with different detection techniques. Cancer Biol. Ther. 2015, 16, 699–708.
- Shabo, I.; Olsson, H.; Stal, O.; Svanvik, J. Breast cancer expression of DAP12 is associated with skeletal and liver metastases and poor survival. Clin. Breast Cancer 2013, 13, 371–377.
- Shabo, I.; Svanvik, J. Expression of macrophage antigens by tumor cells. Adv. Exp. Med. Biol. 2011, 714, 141–150.





