
Video Upload Options
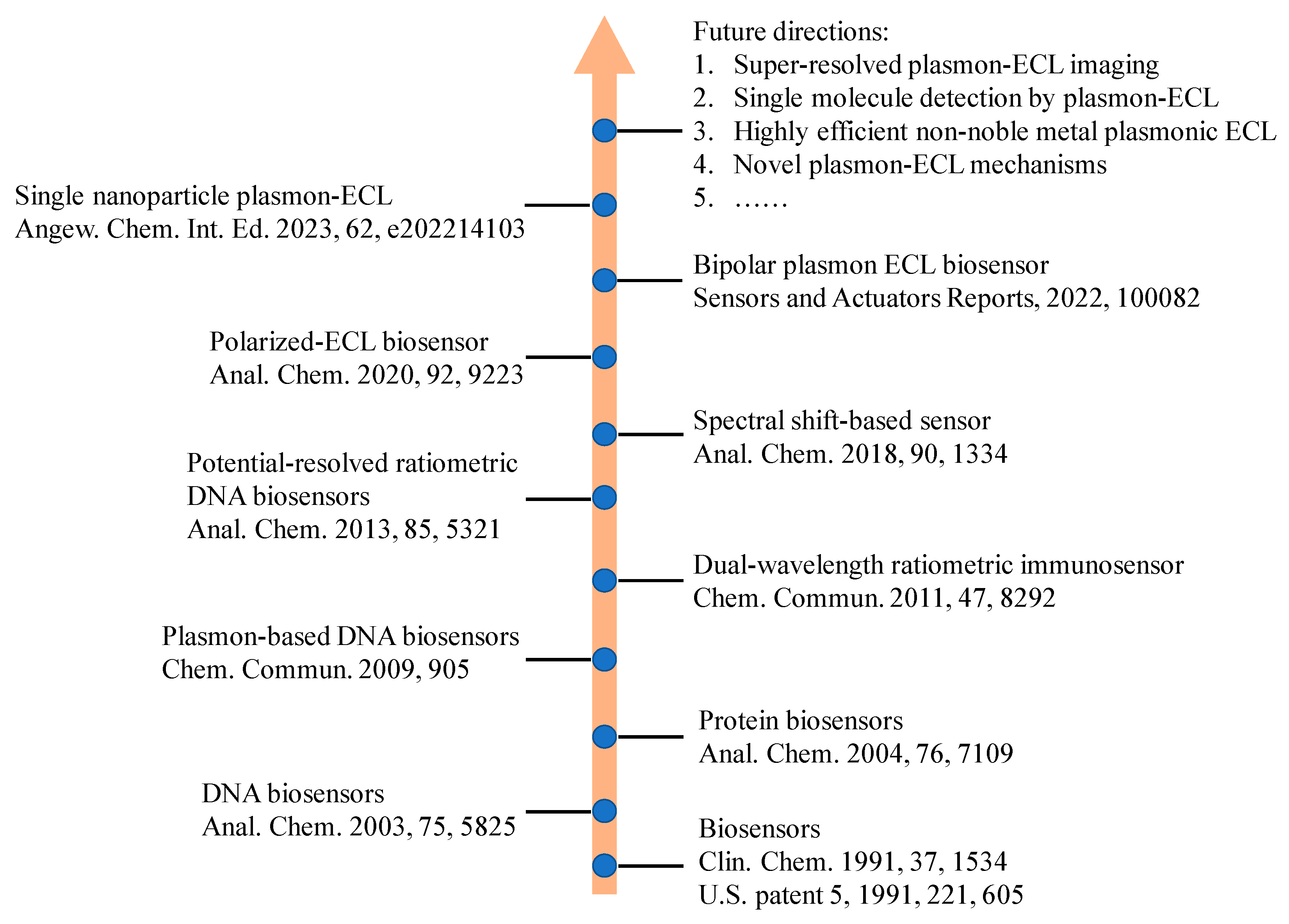
Electrochemiluminescence (ECL) analysis has become a powerful tool in recent biomarker detection and clinic diagnosis due to high sensitivity and broad linear range. To improve the analytical performance of ECL biosensors, various advanced nanomaterials have been introduced to regulate ECL signal such as graphene, gold nanomaterials, quantum dots. Among these nanomaterials, some plasmonic nanostructures play important roles in the fabrication of ECL biosensors. The plasmon effect for ECL signal includes ECL quenching by resonant energy transfer, ECL enhancement by surface plasmon resonance enhancement, and change in the polarized angle of ECL emission. The influence can be regulated by the distance between ECL emitters and plasmonic materials, and the characteristic of polarization-angle-dependent surface plasmon coupling. This research outlines the recent advances of plasmonic based ECL biosensors involving various plasmonic materials include noble metals and semiconductor nanomaterials. The detection targets in these biosensors range from small molecules, proteins, nucleic acids, and cells thanks to the plasmonic effect. In addition to ECL biosensors, ECL microscopy analysis with plasmonic materials is also highlighted because of the enhanced ECL image quality by plasmonic effect. In the end, the future opportunities and challenges are discussed if more plasmonic effect will be introduced into ECL realm.
1. Introduction
2. Fundamentals of Plasmon-Coupled ECL
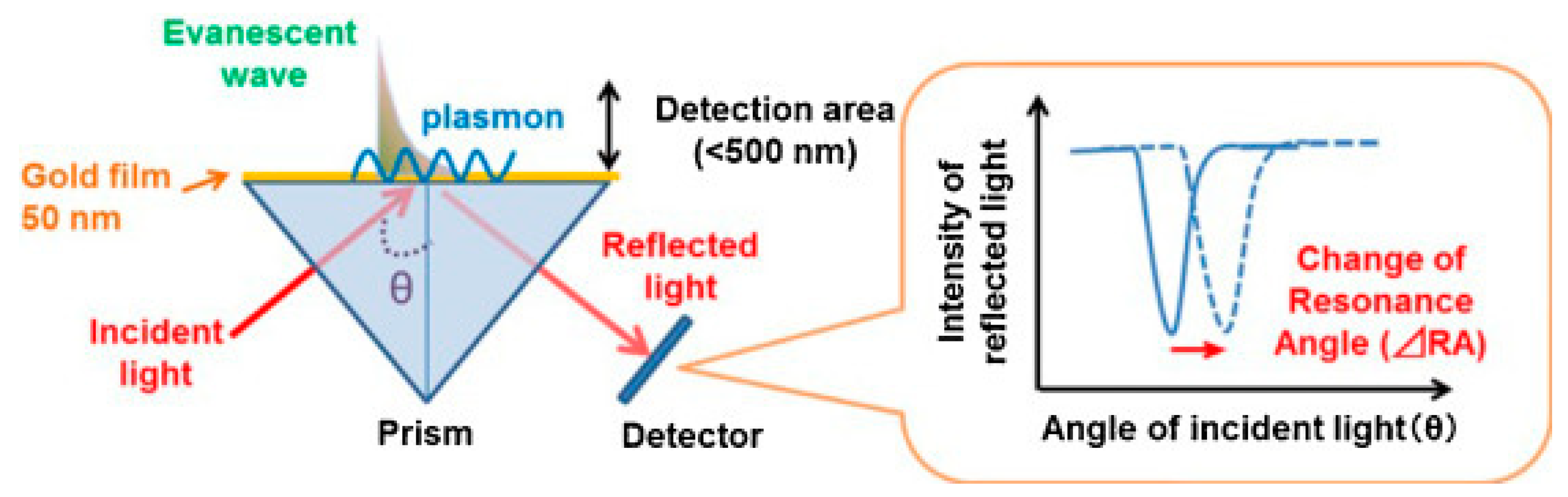
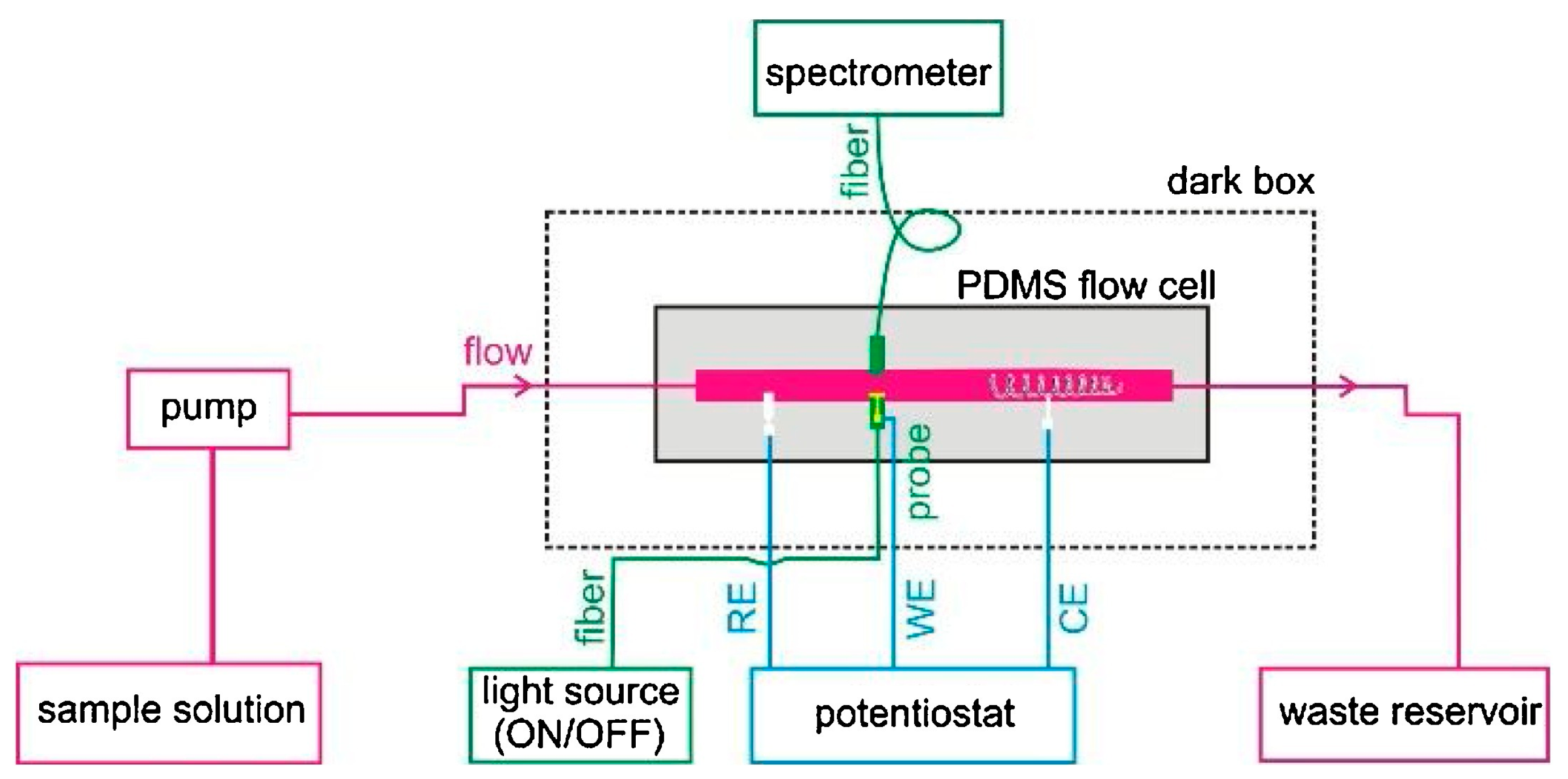
2.1. SPR-Coupled ECL
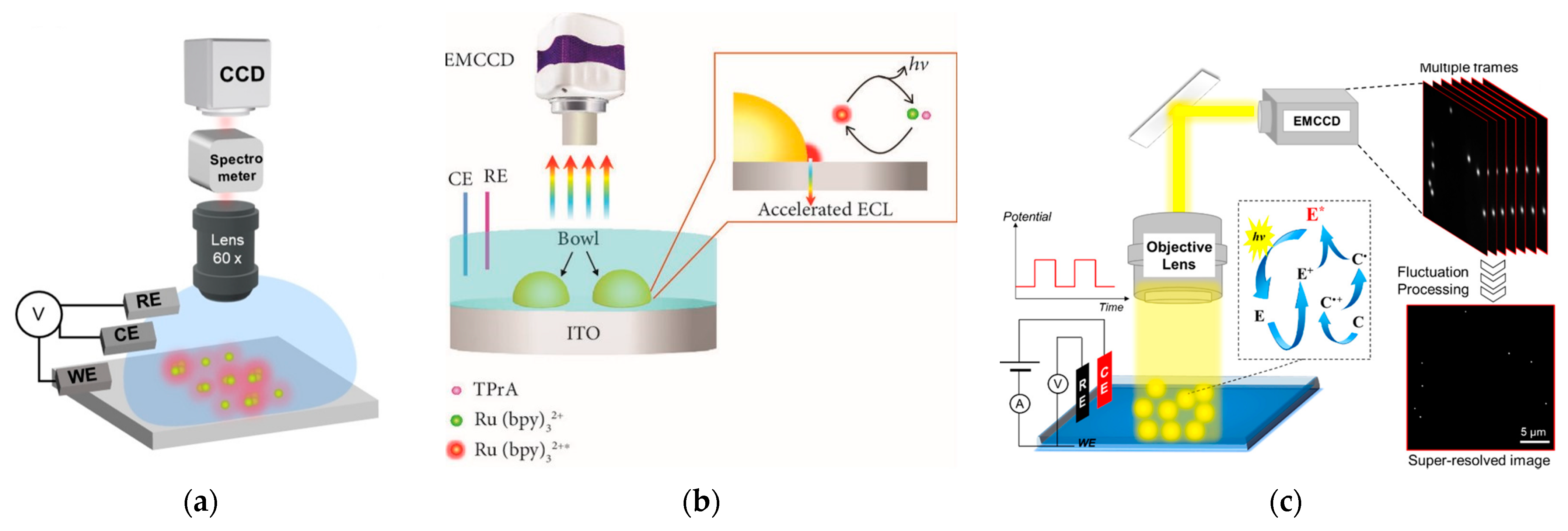
2.2. LSPR-coupled ECL
References
- Ma, C.; Cao, Y.; Gou, X.; Zhu, J.J. Recent Progress in Electrochemiluminescence Sensing and Imaging. Anal. Chem. 2020, 92, 431–454.
- Jiang, J.; Lin, X.; Ding, D.; Diao, G. Graphitic-phase carbon nitride-based electrochemiluminescence sensing analyses: Recent advances and perspectives. RSC Adv. 2018, 8, 19369–19380.
- Valenti, G.; Rampazzo, E.; Kesarkar, S.; Genovese, D.; Fiorani, A.; Zanut, A.; Palomba, F.; Marcaccio, M.; Paolucci, F.; Prodi, L. Electrogenerated chemiluminescence from metal complexes-based nanoparticles for highly sensitive sensors applications. Coord. Chem. Rev. 2018, 367, 65–81.
- Zhang, S.; Liu, Y. Recent Progress of Novel Electrochemiluminescence Nanoprobes and Their Analytical Applications. Front. Chem. 2020, 8, 626243.
- Zanut, A.; Fiorani, A.; Rebeccani, S.; Kesarkar, S.; Valenti, G. Electrochemiluminescence as emerging microscopy techniques. Anal. Bioanal. Chem. 2019, 411, 4375–4382.
- Ding, H.; Su, B.; Jiang, D. Recent Advances in Single Cell Analysis by Electrochemiluminescence. ChemistryOpen 2022, 2022, e202200113.
- Knezevic, S.; Bouffier, L.; Liu, B.; Jiang, D.; Sojic, N. Electrochemiluminescence microscopy: From single objects to living cells. Curr. Opin. Electrochem. 2022, 35, 101096.
- Rebeccani, S.; Zanut, A.; Santo, C.I.; Valenti, G.; Paolucci, F. A Guide Inside Electrochemiluminescent Microscopy Mechanisms for Analytical Performance Improvement. Anal. Chem. 2022, 94, 336–348.
- Zhou, J.H.; Zhang, S.Y.; Liu, Y. Electrochemiluminescence Single-cell Analysis on Nanostructured Interface. Electroanal 2022, 34, 937–946.
- Zhang, Z.; Ma, C.; Xu, Q.; Zhu, J.-J. Recent progress in electrochemiluminescence microscopy analysis of single cells. Analyst 2022, 147, 2884–2894.
- Fereja, T.H.; Du, F.; Wang, C.; Snizhko, D.; Guan, Y.; Xu, G. Electrochemiluminescence Imaging Techniques for Analysis and Visualizing. J. Anal. Test. 2020, 4, 76–91.
- Lv, X.; Li, Y.; Cui, B.; Fang, Y.; Wang, L. Electrochemiluminescent sensor based on an aggregation-induced emission probe for bioanalytical detection. Analyst 2022, 147, 2338–2354.
- Wei, X.; Zhu, M.J.; Yan, H.; Lu, C.; Xu, J.J. Recent Advances in Aggregation-Induced Electrochemiluminescence. Chem. A Eur. J. 2019, 25, 12671–12683.
- Chen, Y.; Cao, Y.; Ma, C.; Zhu, J.-J. Carbon-based dots for electrochemiluminescence sensing. Mater. Chem. Front. 2020, 4, 369–385.
- Ma, C.; Wu, W.; Peng, Y.; Wang, M.X.; Chen, G.; Chen, Z.; Zhu, J.J. A Spectral Shift-Based Electrochemiluminescence Sensor for Hydrogen Sulphide. Anal. Chem. 2018, 90, 1334–1339.
- Zou, R.; Teng, X.; Lin, Y.; Lu, C. Graphitic carbon nitride-based nanocomposites electrochemiluminescence systems and their applications in biosensors. TrAC Trends Anal. Chem. 2020, 132, 116054.
- Xue, J.; Zhang, Z.; Zheng, F.; Xu, Q.; Xu, J.; Zou, G.; Li, L.; Zhu, J.-J. Efficient Solid-State Electrochemiluminescence from High-Quality Perovskite Quantum Dot Films. Anal. Chem. 2017, 89, 8212–8216.
- Cao, Y.; Zhu, W.; Wei, H.; Ma, C.; Lin, Y.; Zhu, J.-J. Stable and Monochromatic All-Inorganic Halide Perovskite Assisted by Hollow Carbon Nitride Nanosphere for Ratiometric Electrochemiluminescence Bioanalysis. Anal. Chem. 2020, 92, 4123–4130.
- Han, T.; Cao, Y.; Wang, J.; Jiao, J.; Song, Y.; Wang, L.; Ma, C.; Chen, H.-Y.; Zhu, J.-J. Crystallization-Induced Enhanced Electrochemiluminescence from a New Tris(bipyridine)ruthenium(II) Derivative. Adv. Funct. Mater. 2023, 2023, 2212394.
- Cai, W.-R.; Zeng, H.-B.; Xue, H.-G.; Marks, R.S.; Cosnier, S.; Zhang, X.-J.; Shan, D. Enhanced Electrochemiluminescence of Porphyrin-Based Metal–Organic Frameworks Controlled via Coordination Modulation. Anal. Chem. 2020, 92, 1916–1924.
- Bezuneh, T.T.; Fereja, T.H.; Kitte, S.A.; Li, H.; Jin, Y. Gold nanoparticle-based signal amplified electrochemiluminescence for biosensing applications. Talanta 2022, 248, 123611.
- Chu, Y.; Han, T.; Deng, A.; Li, L.; Zhu, J.-J. Resonance energy transfer in electrochemiluminescent and photoelectrochemical bioanalysis. Trac-Trends Anal. Chem. 2020, 123, 115745.
- Li, L.; Chen, Y.; Zhu, J.J. Recent Advances in Electrochemiluminescence Analysis. Anal. Chem. 2017, 89, 358–371.
- Cao, Y.; Ma, C.; Zhu, J.-J. DNA Technology-assisted Signal Amplification Strategies in Electrochemiluminescence Bioanalysis. J. Anal. Test. 2021, 5, 95–111.
- Ma, C.; Wu, S.; Zhou, Y.; Wei, H.-F.; Zhang, J.; Chen, Z.; Zhu, J.-J.; Lin, Y.; Zhu, W. Bio-Coreactant-Enhanced Electrochemiluminescence Microscopy of Intracellular Structure and Transport. Angew. Chem. 2021, 60, 4907–4914.
- Guo, W.; Zhou, P.; Sun, L.; Ding, H.; Su, B. Microtube Electrodes for Imaging the Electrochemiluminescence Layer and Deciphering the Reaction Mechanism. Angew. Chem. 2021, 60, 2089–2093.
- Wang, Q.; Ren, Z.H.; Zhao, W.M.; Wang, L.; Yan, X.; Zhu, A.S.; Qiu, F.M.; Zhang, K.K. Research advances on surface plasmon resonance biosensors. Nanoscale 2022, 14, 564–591.
- Pan, S.; Li, X.; Yadav, J. Single-nanoparticle spectroelectrochemistry studies enabled by localized surface plasmon resonance. Phys. Chem. Chem. Phys. 2021, 23, 19120–19129.
- Mouloua, D.; Kotbi, A.; Deokar, G.; Kaja, K.; El Marssi, M.; El Khakani, M.A.; Jouiad, M. Recent Progress in the Synthesis of MoS(2) Thin Films for Sensing, Photovoltaic and Plasmonic Applications: A Review. Materials 2021, 14, 3283.
- Gong, L.; Feng, L.; Zheng, Y.; Luo, Y.; Zhu, D.; Chao, J.; Su, S.; Wang, L. Molybdenum Disulfide-Based Nanoprobes: Preparation and Sensing Application. Biosensors 2022, 12, 87.
- Dao, T.D.; Han, G.; Arai, N.; Nabatame, T.; Wada, Y.; Hoang, C.V.; Aono, M.; Nagao, T. Plasmon-mediated photocatalytic activity of wet-chemically prepared ZnO nanowire arrays. Phys. Chem. Chem. Phys. 2015, 17, 7395–7403.
- Guo, L.; Yin, H.; Xu, M.; Zheng, Z.; Fang, X.; Chong, R.; Zhou, Y.; Xu, L.; Xu, Q.; Li, J.; et al. In Situ Generated Plasmonic Silver Nanoparticle-Sensitized Amorphous Titanium Dioxide for Ultrasensitive Photoelectrochemical Sensing of Formaldehyde. ACS Sens. 2019, 4, 2724–2729.
- Zheng, Z.; Murakami, N.; Liu, J.; Teng, Z.; Zhang, Q.; Cao, Y.; Cheng, H.; Ohno, T. Development of Plasmonic Photocatalyst by Site-selective Loading of Bimetallic Nanoparticles of Au and Ag on Titanium(IV) Oxide. ChemCatChem 2020, 12, 3783–3792.
- Li, M.; Singh, R.; Soares, M.S.; Marques, C.; Zhang, B.; Kumar, S. Convex fiber-tapered seven core fiber-convex fiber (CTC) structure-based biosensor for creatinine detection in aquaculture. Opt. Express 2022, 30, 13898–13914.
- Wang, Y.; Singh, R.; Chaudhary, S.; Zhang, B.; Kumar, S. 2-D Nanomaterials Assisted LSPR MPM Optical Fiber Sensor Probe for Cardiac Troponin I Detection. IEEE Trans. Instrum. Meas. 2022, 71, 1–9.
- Wang, Z.; Singh, R.; Marques, C.; Jha, R.; Zhang, B.; Kumar, S. Taper-in-taper fiber structure-based LSPR sensor for alanine aminotransferase detection. Opt. Express 2021, 29, 43793–43810.
- Wang, Z.; Singh, R.; Zhang, B.; Kumar, S. SMF tapered fiber/AuNPs/ZnO based sensor for detection of acetylcholine. In Optics in Health Care and Biomedical Optics XI; Luo, Q., Li, X., Gu, Y., Zhu, D., Eds.; Spie-Int Soc Optical Engineering: Bellingham, WA, USA, 2021; Volune 11900, p. 119003D.
- Kumar, S.; Singh, R.; Kaushik, B.K.; Chen, N.-k.; Yang, Q.S.; Zhang, X. LSPR-Based Cholesterol Biosensor Using Hollow Core Fiber Structure. IEEE Sens. J. 2019, 19, 7399–7406.
- Singh, L.; Singh, R.; Zhang, B.; Cheng, S.; Kumar Kaushik, B.; Kumar, S. LSPR based uric acid sensor using graphene oxide and gold nanoparticles functionalized tapered fiber. Opt. Fiber Technol. 2019, 53, 102043.
- Kumar, S.; Wang, Y.; Li, M.Y.; Wang, Q.L.; Malathi, S.; Marques, C.; Singh, R.; Zhang, B.Y. Plasmon-Based Tapered-in-Tapered Fiber Structure for p-Cresol Detection: From Human Healthcare to Aquaculture Application. Ieee Sens. J. 2022, 22, 18493–18500.
- Su, Y.; Xu, S.; Zhang, J.; Chen, X.; Jiang, L.-P.; Zheng, T.; Zhu, J.-J. Plasmon Near-Field Coupling of Bimetallic Nanostars and a Hierarchical Bimetallic SERS “Hot Field”: Toward Ultrasensitive Simultaneous Detection of Multiple Cardiorenal Syndrome Biomarkers. Anal. Chem. 2019, 91, 864–872.
- Wen, S.; Su, Y.; Dai, C.; Jia, J.; Fan, G.-C.; Jiang, L.-P.; Song, R.-B.; Zhu, J.-J. Plasmon Coupling-Enhanced Raman Sensing Platform Integrated with Exonuclease-Assisted Target Recycling Amplification for Ultrasensitive and Selective Detection of microRNA-21. Anal. Chem. 2019, 91, 12298–12306.
- Wang, Z.; Xue, J.; Bi, C.; Xin, H.; Wang, Y.; Cao, X. Quantitative and specific detection of cancer-related microRNAs in living cells using surface-enhanced Raman scattering imaging based on hairpin DNA-functionalized gold nanocages. Analyst 2019, 144, 7250–7262.
- Lu, D.; Xia, J.; Deng, Z.; Cao, X. Detection of squamous cell carcinoma antigen in cervical cancer by surface-enhanced Raman scattering-based immunoassay. Anal. Methods 2019, 11, 2809–2818.
- Su, Y.-w.; Wang, W. Surface plasmon resonance sensing: From purified biomolecules to intact cells. Anal. Bioanal. Chem. 2018, 410, 3943–3951.
- Li, J.; Qi, H.; Wang, H.; Yang, Z.; Zhu, P.; Diao, G. Fluorescence energy transfer-based multiplexed hybridization assay using gold nanoparticles and quantum dot conjugates on photonic crystal beads. Microchim. Acta 2014, 181, 1109–1115.
- Cui, C.; Jin, R.; Jiang, D.; Zhang, J.; Zhu, J. Visualization of an Accelerated Electrochemical Reaction under an Enhanced Electric Field. Research 2021, 2021, 1742919.
- Chen, M.M.; Xu, C.H.; Zhao, W.; Chen, H.Y.; Xu, J.J. Super-Resolution Electrogenerated Chemiluminescence Microscopy for Single-Nanocatalyst Imaging. J. Am. Chem. Soc. 2021, 143, 18511–18518.
- Chen, M.M.; Zhao, W.; Zhu, M.J.; Li, X.L.; Xu, C.H.; Chen, H.Y.; Xu, J.J. Spatiotemporal imaging of electrocatalytic activity on single 2D gold nanoplates via electrogenerated chemiluminescence microscopy. Chem. Sci. 2019, 10, 4141–4147.
- Wang, P.; Zhao, J.; Wang, Z.; Liang, Z.; Nie, Y.; Xu, S.; Ma, Q. Polarization-Resolved Electrochemiluminescence Sensor Based on the Surface Plasmon Coupling Effect of a Au Nanotriangle-Patterned Structure. Anal. Chem. 2021, 93, 15785–15793.
- Xia, J.; Zhou, J.; Zhang, R.; Jiang, D.; Jiang, D. Gold-coated polydimethylsiloxane microwells for high-throughput electrochemiluminescence analysis of intracellular glucose at single cells. Anal. Bioanal. Chem. 2018, 410, 4787–4792.
- Villani, E.; Valenti, G.; Marcaccio, M.; Mattarozzi, L.; Barison, S.; Garoli, D.; Cattarin, S.; Paolucci, F. Coreactant electrochemiluminescence at nanoporous gold electrodes. Electrochim. Acta 2018, 277, 168–175.
- Dinel, M.P.; Tartaggia, S.; Wallace, G.Q.; Boudreau, D.; Masson, J.F.; Polo, F. The Fundamentals of Real-Time Surface Plasmon Resonance/Electrogenerated Chemiluminescence. Angew. Chem. Int. Ed. 2019, 58, 18202–18206.
- Yu, J.; Jia, P.; Wang, S.; Ebendorff-Heidepriem, H.; Abell, A.D. Electrochemical plasmonic optical fiber probe for real-time insight into coreactant electrochemiluminescence. Sens. Actuators B Chem. 2020, 321, 128469.
- Perez-Tejeda, P.; Grueso, E.; Marin-Gordillo, A.; Torres-Marquez, C.; Giraldez-Perez, R.M. Aqueous Gold Nanoparticle Solutions for Improved Efficiency in Electrogenerated Chemiluminescent Reactions. Acs. Appl. Nano. Mater. 2018, 1, 5307–5315.
- Pan, S.; Liu, J.; Hill, C.M. Observation of Local Redox Events at Individual Au Nanoparticles Using Electrogenerated Chemiluminescence Microscopy. J. Phys. Chem. C 2015, 119, 27095–27103.
- Wei, Y.; Zhang, Y.; Pan, J.; Chen, T.; Xing, X.; Zhang, W.; Lu, Z. Plasmon-Enhanced Electrochemiluminescence at the Single-Nanoparticle Level. Angew. Chem. 2022, 2022, e202214103.
- Heiderscheit, T.S.; Gallagher, M.J.; Baiyasi, R.; Collins, S.S.E.; Jebeli, S.A.H.; Scarabelli, L.; Al-Zubeidi, A.; Flatebo, C.; Chang, W.-S.; Landes, C.F.; et al. Nanoelectrode-emitter spectral overlap amplifies surface enhanced electrogenerated chemiluminescence. J. Chem. Phys. 2019, 151, 144712.
- Heiderscheit, T.S.; Oikawa, S.; Sanders, S.; Minamimoto, H.; Searles, E.K.; Landes, C.F.; Murakoshi, K.; Manjavacas, A.; Link, S. Tuning Electrogenerated Chemiluminescence Intensity Enhancement Using Hexagonal Lattice Arrays of Gold Nanodisks. J. Phys. Chem. Lett. 2021, 12, 2516–2522.
- Wilson, A.J.; Marchuk, K.; Willets, K.A. Imaging Electrogenerated Chemiluminescence at Single Gold Nanowire Electrodes. Nano Lett. 2015, 15, 6110–6115.
- Dong, J.; Xu, Y.; Zhang, Z.; Feng, J. Operando Imaging of Chemical Activity on Gold Plates with Single-Molecule Electrochemiluminescence Microscopy. Angew. Chem. 2022, 2022, e202200187.





