Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Pan, D.; Wang, W.; Cheng, T. Antibodies of Varicella-Zoster Virus. Encyclopedia. Available online: https://encyclopedia.pub/entry/43196 (accessed on 26 May 2026).
Pan D, Wang W, Cheng T. Antibodies of Varicella-Zoster Virus. Encyclopedia. Available at: https://encyclopedia.pub/entry/43196. Accessed May 26, 2026.
Pan, Dequan, Wei Wang, Tong Cheng. "Antibodies of Varicella-Zoster Virus" Encyclopedia, https://encyclopedia.pub/entry/43196 (accessed May 26, 2026).
Pan, D., Wang, W., & Cheng, T. (2023, April 18). Antibodies of Varicella-Zoster Virus. In Encyclopedia. https://encyclopedia.pub/entry/43196
Pan, Dequan, et al. "Antibodies of Varicella-Zoster Virus." Encyclopedia. Web. 18 April, 2023.
Copy Citation
Infection with the varicella-zoster virus (VZV) causes chickenpox and shingles, which lead to significant morbidity and mortality globally. The detection of serum VZV-specific antibodies is important for the clinical diagnosis and sero-epidemiological research of VZV infection, and for assessing the effect of VZV vaccine immunization.
varicella-zoster virus (VZV)
antibody detection methods
FAMA
1. Introduction
Varicella-zoster virus (VZV) is a highly contagious alpha-herpesvirus that infects more than 90% of people worldwide [1][2]. Chickenpox (varicella) is the outcome of primary infection with VZV and mainly affects children. As per the report published by the World Health Organization (WHO) in 2014, the minimum of the annual global disease burden of chickenpox was estimated to be 140 million cases, of which 4.2 million have severe complications leading to hospitalization and death [3]. Although usually a mild and self-limiting illness, chickenpox poses a greater risk of severe disease to pregnant women, neonates, VZV-seronegative adults, and immunocompromised individuals [4]. The reactivation of latent VZV causes shingles (herpes zoster), which occurs mainly in people ≥50 years of age and is usually associated with intense neuralgia [5]. It was estimated that about one third of individuals who have had chickenpox will develop shingles [6]. The incidence and severity of shingles increases with age or immunosuppression [7]. Recent studies have shown that COVID-19 vaccination may also increase the risk of VZV reactivation and thus potentially increase the incidence of shingles, especially in the elderly population [8][9][10]. To date, there is still no specific cure for VZV-induced diseases. Vaccination is among the most cost-effective ways for preventing chickenpox and shingles. The most widely used chickenpox vaccines consist of the Oka strain of live-attenuated VZV (vOka), and there are currently two kinds of shingles vaccines on the market, including a live-attenuated vaccine also based on vOka (ZOSTAVAX®, Merck Sharp & Dohme, Rahway, NJ, USA) and a recombinant subunit vaccine based on VZV glycoprotein E (Shingrix®, GlaxoSmithKline, Brentford, Middlesex, UK) [6][11][12]. However, the use of these vaccines is not universal and thus a large proportion of the global population has not been vaccinated against VZV. Consequently, VZV infection is still prevalent and accounts for a significant disease burden worldwide.
Laboratory testing is essential for the diagnosis and surveillance of VZV-induced diseases. Virus isolation was once the “gold standard” for the diagnosis of VZV infection [13][14]. However, this method is time-consuming and not readily accessible, and has thus been replaced by polymerase chain reaction (PCR) for the detection of viral DNA and direct fluorescence assays (DFA) for the detection of viral proteins [15][16]. Most recently, a recombinase-aided amplification-lateral flow system (RAA-LF) has been used for the rapid detection of VZV DNA, which is easier to use and requires no equipment [17]. PCR is currently regarded as the most sensitive and reliable method for VZV detection [14]. Meanwhile, serological assays, which detect VZV-specific antibodies, also represent reliable diagnostic tools in the detection of VZV infection, and have been implemented as complementary approaches to PCR. Furthermore, since the quantitative detection of anti-VZV antibody is necessary for measuring the infection history and evaluating the immune status against VZV in the population, serological assays have been widely used in epidemiological investigations of VZV and evaluations of immune responses to VZV vaccination. These assays include the fluorescent-antibody-to-membrane-antigen (FAMA) test, the complement fixation (CF) test, the immune adherence hemagglutination assay (IAHA), the latex agglutination (LA) test, the radioimmunoassay (RIA), the neutralization assay, the chemiluminescent immunoassay (CLIA), the enzyme-linked immunosorbent assay (ELISA), the immunofluorescence assay (IFA), the time-resolved fluorescence immunoassay (TRFIA), and the lateral flow immunochromatographic assay (LFIA). Among these tests, FAMA and ELISA are the most widely used. On the other hand, the CF test has low sensitivity, while RIA uses harmful radioactive materials; therefore, neither method has been widely used in recent years.
2. Methods for the Detection of Anti-VZV Antibodies
2.1. FAMA
The FAMA test, which was initially developed by Williams et al. [18], is the most extensively validated assay and is considered the “gold standard” for VZV antibody detection [19][20]. This method determines the presence of antibodies specific to viral proteins that distribute on the surface of VZV-infected cells, which correlate with protection from disease. Different human cells, including HFF [21], MRC-5 [22], Vero [23] and Raji [24], have been used to perform the FAMA test in previous studies. According to the standard FAMA procedure (schematic shown in Figure 1A), cells in culture are infected with VZV for 48–72 h and harvested by trypsin digestion until 70–90% of them show a cytopathic effect. Then, the infected cells are resuspended in PBS and incubated with serial dilutions of human sera to bind with VZV-specific antibodies. Following washing, cells are incubated with a fluorescein-conjugated anti-human secondary antibody (specific for IgG, IgM, or IgM and IgG). After a second washing step, the cells are transferred into small wells on glass slides and incubated for certain time to allow the cells to attach. Finally, cells are sealed with 90% glycerin and a cover glass before observation under a fluorescence microscope [18]. When the viral proteins (e.g., VZV glycoproteins) distributed on the surface of VZV-infected cells bind to their specific antibodies in the serum samples, ring-like fluorescence patterns are typically observed. The highest dilution that can still cause a positive ring-like fluorescence reaction is taken as the FAMA titer of the serum sample. Given the false-positive results caused by the non-specific reactivity of undiluted sera, serum samples with a titer of ≥1:2 or 1:4 are generally considered positive [18][25][26]. Several studies have shown that healthy children can be protected from chickenpox infection when they have serum FAMA titers of ≥1:4 for VZV [27][28].
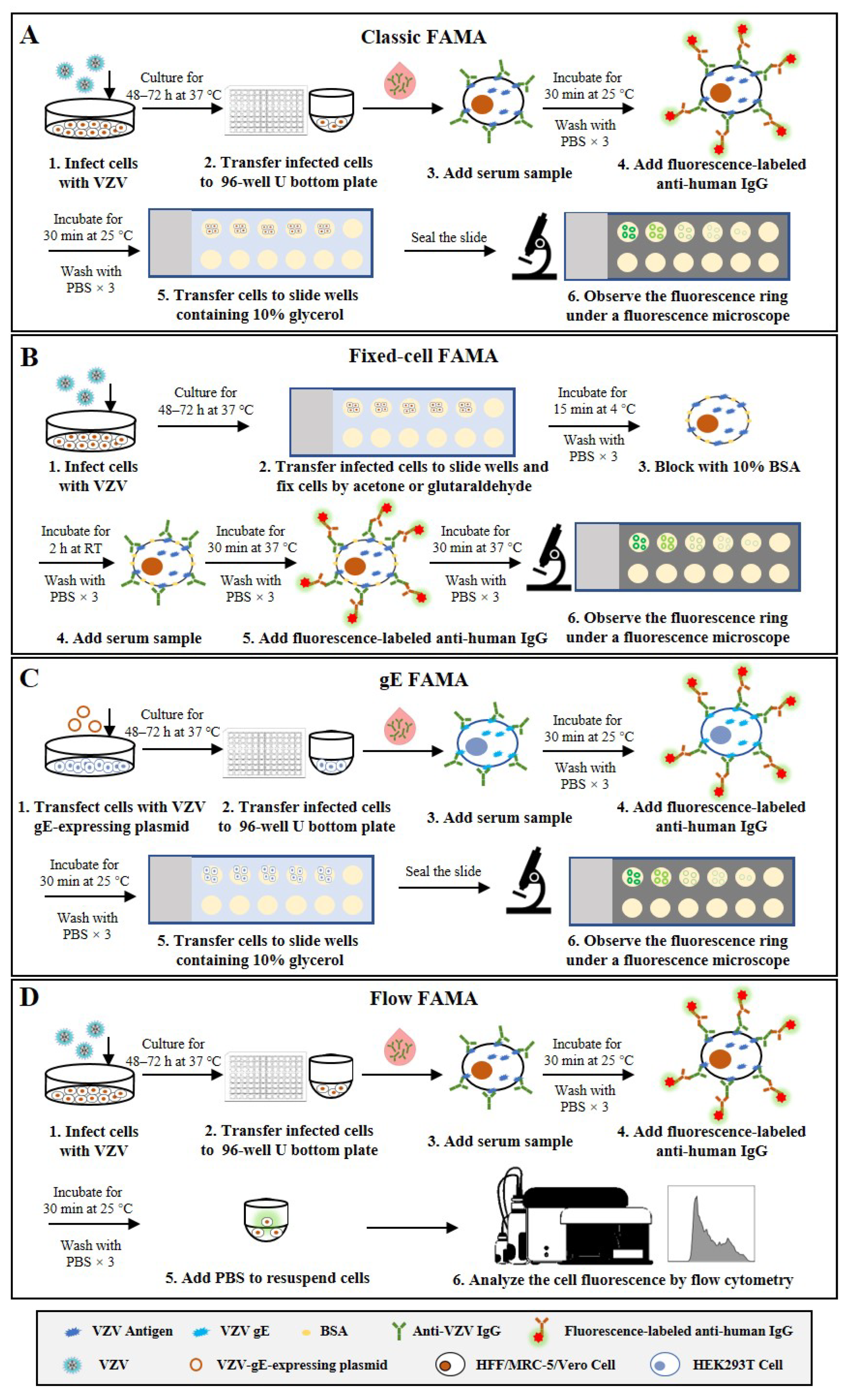
Figure 1. Schematic representation of the basic protocols for four types of FAMA that detect anti-VZV antibodies. (A) Classic FAMA using live VZV-infected cells to capture anti-VZV antibodies. (B) Fixed-cell FAMA using chemically fixed VZV-infected cells to capture anti-VZV antibodies. (C) gE-FAMA using VZV-gE-expressing live cells to capture anti-VZV-gE antibodies. (D) Flow FAMA using flow cytometry to analyze the fluorescence-labeled VZV-infected cells.
The FAMA test can be performed using chemically fixed VZV-infected cells. The fixed-cell FAMA was developed from the classic FAMA to increase throughput and efficiency and has been used to study human immunity to VZV [29][30][31][32]. In this modified FAMA (schematic shown in Figure 1B), VZV-infected cells are first fixed on the slide with cold acetone [31][32] or glutaraldehyde [29][30], while the other steps and the cutoff values (1:2 or 1:4) are similar to or the same as those in the classic live-cell FAMA test. The acetone fixation increases membrane permeability and allows antibody access to the cell cytoplasm and binding to other viral proteins besides membrane antigens [33]. In comparison, glutaraldehyde fixation does not change the permeability of the cell membrane and detects only the membrane antigen, but changes the natural conformation of the antigen, resulting in reduced sensitivity to a certain extent [19]. The fixed-cell FAMA has several advantages over the classic FAMA, e.g., the slides with attached cells can be prepared in large batches in advance and stored in a freezer for a long time, which enables testing at any time and reduces hands-on time. In addition, the used FAMA slides can be kept for re-reading, which creates the possibility of them being inspected by the drug administration. However, the specificity of fixed-cell FAMA could be challenged, since the procedure of fixed-cell FAMA is similar to that of indirect fluorescent antibody test (IFAT), which also uses fixed VZV-infected cells, and previous studies have documented that serum samples from children weakly cross-reacted between VZV and herpes simplex virus (HSV) in IFAT [19][34][35]. Nonetheless, the influence of cell fixation on the specificity of FAMA remains controversial and needs to be further clarified.
To avoid the use of infectious viruses, one study has reported a simple and safer FAMA using HEK293T cells transfected with a plasmid encoding VZV glycoprotein E (gE) to replace VZV-infected cells, which is called the gE FAMA (schematic shown in Figure 1C) [36]. The gE FAMA exhibited a similar staining effect to classic FAMA, and the gE-FAMA titers were closely correlated with the gp-ELISA data. However, this assay only detects antibodies against VZV gE, and the abundance of expressed gE on plasmid-transfected cells is different from that of VZV-infected cells; the cutoff value of gE FAMA may be different from that of classic FAMA, and remains to be determined.
All the above-mentioned FAMA assays rely on experienced technicians making result judgments under fluorescence microscopy, which not only leads to subjective bias but also limits the throughput of the detection. Some studies have reported a flow cytometry-adapted FAMA (flow FAMA; schematic shown in Figure 1D), in which flow cytometry is used instead of examination under a microscope to analyze the fluorescence-labeled cells [37][38][39]. The positive cutoff value determined by flow cytometry analysis can make the judgment of FAMA more objective, and the automated measurement can also reduce the complexity of operations and increase the detection throughput of FAMA. In a study involving 62 human serum samples, the detection accuracy of flow FAMA was 90.32% compared with that of standard FAMA [38]. In the flow FAMA, the matter of how to select the appropriate cutoff value remains a key problem. In addition, a flow cytometer is required for the flow FAMA, which limits the application of this method.
Taken together, the classic live-cell FAMA test is regarded as the “gold standard” to detect anti-VZV antibodies because of its high sensitivity and specificity. However, the standard FAMA procedure is semi-quantitative, low-throughput, and labor-intensive, and requires a subjective evaluation by trained, experienced technicians, which limits its widespread use. Fixed-cell FAMA and gE FAMA are modified from the classic live-cell FAMA to improve the throughput and safety, but both of them have their own defects, including possibly lower specificity, a lack of validation, or ambiguous cutoff values.
2.2. ELISA
ELISA is one of the most common antibody detection methods and has been widely used for the quantitative detection of anti-VZV antibodies for epidemiological investigations of VZV infection and for efficacy evaluation of the varicella and zoster vaccines [25][40][41]. Indirect ELISA is the most commonly used type. According to the procedure of indirect ELISA (schematic shown in Figure 2A), VZV antigens are coated on 96-well polystyrene ELISA plates and subsequently blocked with bovine serum albumin or normal goat serum. Diluted serum samples are then added into the plate wells. After incubation and washing, peroxidase- or alkaline-phosphatase-conjugated anti-human antibodies are added to detect the captured anti-VZV antibodies. After another washing step, the substrate solution is added for a chromogenic reaction, which is then terminated with excess acid or base. Finally, the optical density (OD) or absorbance value of appropriate wavelength is measured quantitatively using a spectrophotometer. Within a certain range, the OD or absorbance value is proportional to the number of binding antibodies on the plate, so the standard curve can be drawn through the detection of standard products and achieve quantitative detection of anti-VZV antibodies [42][43][44].
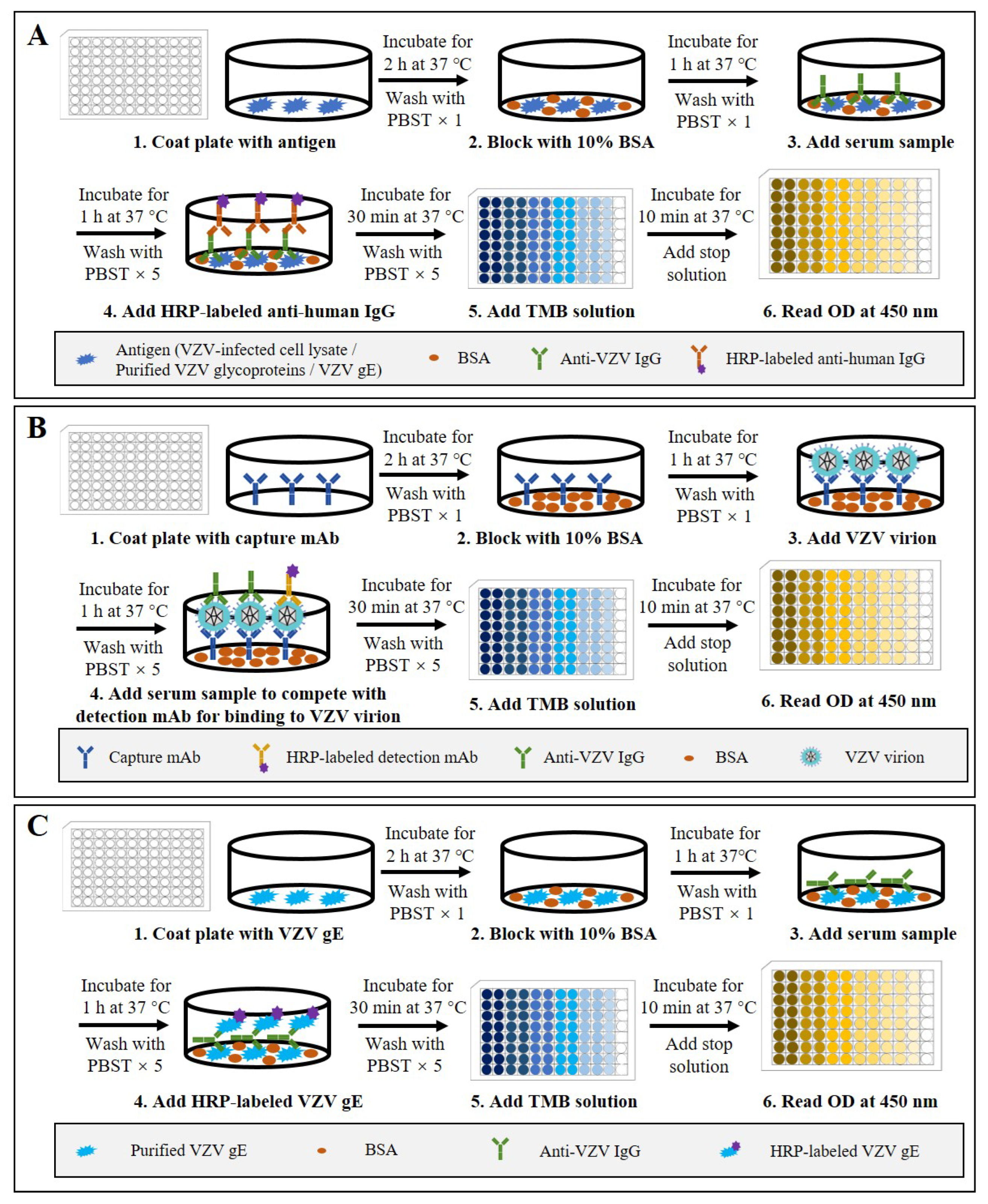
Figure 2. Schematic representation of the basic protocols for three types of ELISA that detect anti-VZV antibodies. (A) Indirect ELISA using VZV-infected cell lysate, purified VZV glycoproteins, or VZV gE as the coating antigen. (B) Double antibody sandwich competitive ELISA using anti-ORF9 antibody as the detection antibody and HRP-labeled anti-gE antibody as the detection antibody. (C) Double gE antigen sandwich ELISA using purified VZV gE as the coating antigen and HRP-labeled gE as the detection antigen.
Nowadays, there are several commercial VZV antibody ELISA kits available [19][45]. They are mostly indirect ELISAs and use either whole VZV-infected cell lysate (WC ELISA) [25][44] or purified glycoprotein (gp-ELISA) [46][47] as the antigen to capture anti-VZV antibodies. Different ELISA kits are calibrated according to the first international standard for varicella-zoster immunoglobulin, and results of <50 mIU/mL are considered negative, but the cutoff value varies [25][41][48]. The majority of these commercial ELISAs are designed to measure antibody levels after natural infection and are found to be insufficiently sensitive to measure antibody responses to chickenpox vaccination [19][49][50]. One study compared four commercial ELISAs and showed that their sensitivity ranged from 60.4% to 91.8%, values that are low compared with those of the FAMA test [25]. To address the issue of sensitivity, Merck has developed an in-house, highly sensitive, and specific gp-ELISA that uses lentil lectin-purified VZV glycoproteins, including gE, gB, and gH, from VZV-infected cells as the antigen [51]. The Merck gp-ELISA has been used extensively to evaluate antibody responses in children immunized with the Varivax Oka vaccine and a titer of 5 gp-ELISA units/mL (equivalent to 10 mIU/mL, by the international reference standard) was found to be associated with a high degree of protection against breakthrough infection during seven follow-up years [52][53]. However, the Merck gp-ELISA is not commercially available and is restricted to only a few specialist testing centers. In general, the gp-ELISA has high sensitivity and specificity, and a high consistency with FAMA, and is thus considered to be the most likely alternative to FAMA [54]. In addition, GSK also developed an in-house ELISA to detect VZV gE antibody (gE-ELISA) and applied it to evaluating the immunogenicity of a herpes zoster subunit vaccine Shingrix [55].
In addition to indirect ELISA, some researchers have developed a competitive ELISA and a gE double-antigen sandwich ELISA for VZV antibody detection [56][57]. Firstly, for the competitive ELISA (schematic shown in Figure 2B), an anti-ORF9 antibody is used to capture VZV particles, and then an HRP-labeled anti-gE antibody is used to compete with serum anti-VZV antibodies for virus binding. Using the following formula: PI (%) = 100 × [1 − (positive serum OD450/negative reference serum OD450)], the blocking rate can be calculated to evaluate the VZV antibody levels in serum samples. The study showed that the competitive sandwich ELISA had a sensitivity of 95.6%, a specificity of 99.77%, and coincidence of 97.61% compared to the FAMA test. Secondly, the gE double-antigen sandwich ELISA (schematic shown in Figure 2C) is modified from the gE-based indirect ELISA by replacing the enzyme-conjugated anti-human secondary antibodies with HRP-labeled recombinant gE protein. The study showed that the gE double-antigen sandwich ELISA had sensitivity of 95.08% and specificity of 100% compared to the FAMA test. The findings in these studies suggest that the use of antibody competition or a dual-gE-antigen sandwich could increase the sensitivity and specificity of ELISA for VZV antibody detection. However, to date, there are no reports on the further application and validation of these two methods.
Taken together, to date, many types of ELISAs have been developed for anti-VZV antibody detection. Compared to the FAMA test, these ELISAs are quantitative, easy to use, and high throughput, but are considered to be less sensitive, except for the Merck gp-ELISA, which is regarded as sensitive enough to be an optimal alternative reference assay to FAMA. Recent advances have improved the performance of several ELISAs, achieving sensitivity and specificity similar to FAMA, but these new methods still lack further validation or else are not commercially available.
2.3. Neutralization Assay
The neutralization assay measures the titers of neutralizing antibodies that confer protection from VZV infections. However, the sensitivity of the earliest neutralization assay was low, making it difficult to detect anti-VZV neutralizing antibodies in individuals many years after infection [58]. Some studies have reported that the addition of guinea pig complement and anti-immunoglobulin antibodies can make the sensitivity of the neutralization assay for VZV 2 to 16 fold and 7 to 100 fold higher, respectively, and thus the enhanced neutralization assay is more frequently used for the evaluation of antibody responses to VZV infection, compared to the original method [59][60][61][62][63].
According to the procedure of the complement-enhanced neutralization assay for VZV (schematic shown in Figure 3) [59][64][65], which is modified from the universal plaque-reduction neutralization test (PRNT), hundreds of PFUs of cell-free VZV are mixed with diluted heat-inactivated serum samples (at 56 °C for 30 min) and guinea pig complement, and co-incubated at 37 °C for 1 h before being added into the cultured cells (e.g., MRC-5). After incubation for 5–7 days, the number of virus plaques is directly counted under an inverted microscope, and the highest dilutions of serum that result in ≥50% reduction in plaque counts are defined as the neutralization titers. In this experiment, cells can be stained with dyes such as crystal violet to make the plaques easier to observe. The neutralization assay requires viral plaque formation and takes about one week to complete. To reduce the testing time, an indirect immunoperoxidase assay (IPA) was used to stain VZV-infected cells and shortened the test period to 72 h [66][67]. Another study established a neutralization test basing on an enzyme-linked immunosorbent spot (Elispot) assay with VZV-gK protein as the detection target, and shortened the test period to 36 h [68].
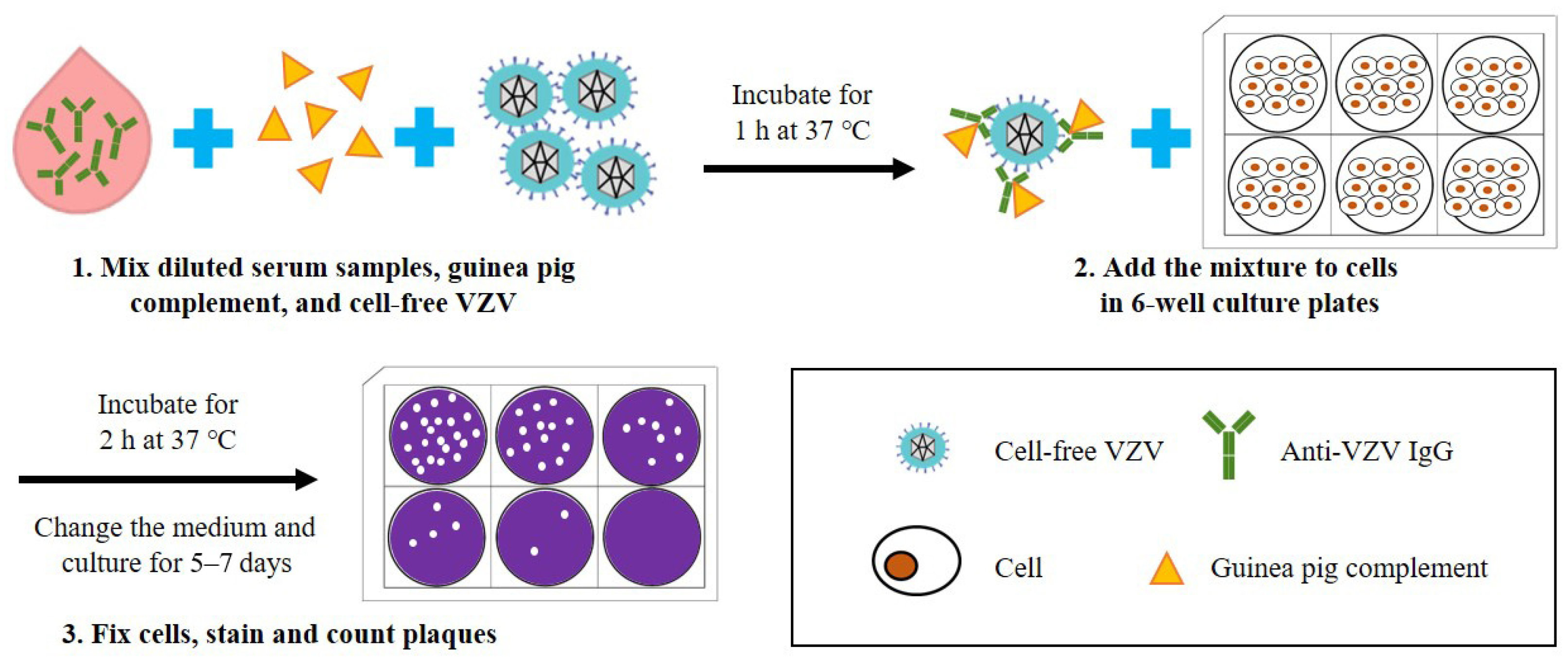
Figure 3. Schematic diagram depicting the basic steps of a neutralization assay for the detection of anti-VZV antibodies. (1) Diluted serum samples, guinea pig complement, and cell-free VZV are mixed and incubated for 1 h at 37 °C. (2) The mixture is added to cells in 6-well culture plates and incubated for 2 h at 37 °C, and then the culture medium is replaced. (3) After culturing for 5–7 days, the cells are fixed and stained to visualize the plaques, and the neutralization titer of serum samples can be calculated according to the number of plaques.
References
- Gershon, A.A.; Breuer, J.; Cohen, J.I.; Cohrs, R.J.; Gershon, M.D.; Gilden, D.; Grose, C.; Hambleton, S.; Kennedy, P.G.; Oxman, M.N.; et al. Varicella zoster virus infection. Nat. Rev. Dis. Primers 2015, 1, 15016.
- Arvin, A.M.; Gilden, D. Varicella-zoster virus. In Fields Virology, 6th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; pp. 2015–2057.
- World Health Organization. Varicella and herpes zoster vaccines: WHO position paper, June 2014. Relev. Epidemiol. Hebd. 2014, 89, 265–287.
- Heininger, U.; Seward, J.F. Varicella. Lancet 2006, 368, 1365–1376.
- Cohen, J.I. Clinical practice: Herpes zoster. N. Engl. J. Med. 2013, 369, 255–263.
- Harpaz, R.; Ortega-Sanchez, I.R.; Seward, J.F. Prevention of herpes zoster: Recommendations of the Advisory Committee on Immunization Practices (ACIP). Morb. Mortal. Wkly. Rep. Recomm. Rep. 2008, 57, 1–30.
- Sampathkumar, P.; Drage, L.A.; Martin, D.P. Herpes zoster (shingles) and postherpetic neuralgia. Mayo Clin. Proc. 2009, 84, 274–280.
- Crooke, S.N.; Ovsyannikova, I.G.; Poland, G.A.; Kennedy, R.B. Immunosenescence and human vaccine immune responses. Immun. Ageing 2019, 16, 25.
- Kechagias, K.; Giannos, P. Varicella Zoster Virus Reactivation Following COVID-19 Vaccination: A Systematic Review of Case Reports. Vaccines 2021, 9, 1013.
- Psichogiou, M.; Samarkos, M.; Mikos, N.; Hatzakis, A. Reactivation of varicella zoster virus after vaccination for SARS-CoV-2. Vaccines 2021, 9, 572.
- Gershon, A.; Gershon, M.; Shapiro, E.D. Live Attenuated Varicella Vaccine: Prevention of Varicella and of Zoster. J. Infect. Dis. 2021, 224, S387–S397.
- Harbecke, R.; Cohen, J.; Oxman, M.N. Herpes Zoster Vaccines. J. Infect. Dis. 2021, 224, S429–S442.
- Kennedy, P.G.E.; Gershon, A.A. Clinical Features of Varicella-Zoster Virus Infection. Viruses 2018, 10, 609.
- Guarner, J.; Del Rio, C.; Malani, P.N. Monkeypox in 2022-What Clinicians Need to Know. JAMA 2022, 328, 139–140.
- McCollum, A.M.; Damon, I.K. Human monkeypox. Clin. Infect. Dis. 2014, 58, 260–267.
- MacNeil, A.; Reynolds, M.G.; Carroll, D.S.; Karem, K.; Braden, Z.; Lash, R.; Moundeli, A.; Mombouli, J.-V.; Jumaan, A.O.; Schmid, D.S.; et al. Monkeypox or varicella? Lessons from a rash outbreak investigation in the Republic of the Congo. Am. J. Trop. Med. Hyg. 2009, 80, 503–507.
- Bienes, K.M.; Mao, L.; Selekon, B.; Gonofio, E.; Nakoune, E.; Wong, G.; Berthet, N. Rapid Detection of the Varicella-Zoster Virus Using a Recombinase-Aided Amplification-Lateral Flow System. Diagnostics 2022, 12, 2957.
- Williams, V.; Gershon, A.; Brunell, P.A. Serologic response to varicella-zoster membrane antigens measured by indirect immunofluorescence. J. Infect. Dis. 1974, 130, 669–672.
- Breuer, J.; Schmid, D.S.; Gershon, A.A. Use and limitations of varicella-zoster virus-specific serological testing to evaluate breakthrough disease in vaccinees and to screen for susceptibility to varicella. J. Infect. Dis. 2008, 197, S147–S151.
- Gershon, A.A.; Gershon, M.D. Perspectives on vaccines against varicella-zoster virus infections. In Varicella-Zoster Virus; Springer: Berlin/Heidelberg, Germany, 2010; pp. 359–372.
- Landry, M.L.; Cohen, S.; Mayo, D.; Fong, C.; Andiman, W.A. Comparison of fluorescent-antibody-to-membrane-antigen test, indirect immunofluorescence assay, and a commercial enzyme-linked immunosorbent assay for determination of antibody to varicella-zoster virus. J. Clin. Microbiol. 1987, 25, 832–835.
- Keller, P.M.; Neff, B.J.; Ellis, R.W. Three major glycoprotein genes of varicella-zoster virus whose products have neutralization epitopes. J. Virol. 1984, 52, 293–297.
- Baba, K.; Yoshida, M.; Tawa, A.; Yabuuchi, H.; Maeda, K.; Takahashi, M. A simplified immunofluorescence technique for antibody to varicella-zoster membrane antigen (FAMA). Biken J. 1984, 27, 23–29.
- Iltis, J.; Castellano, G.; Gerber, P.; Le, C.; Vujcic, L.; Quinnan, G.V., Jr. Comparison of the Raji cell line fluorescent antibody to membrane antigen test and the enzyme-linked immunosorbent assay for determination of immunity to varicella-zoster virus. J. Clin. Microbiol. 1982, 16, 878–884.
- de Ory, F.; Echevarría, J.M.; Kafatos, G.; Anastassopoulou, C.; Andrews, N.; Backhouse, J.; Berbers, G.; Bruckova, B.; Cohen, D.I.; de Melker, H.; et al. European seroepidemiology network 2: Standardisation of assays for seroepidemiology of varicella zoster virus. J. Clin. Virol. 2006, 36, 111–118.
- Maple, P.; Haedicke, J.; Quinlivan, M.; Steinberg, S.; Gershon, A.; Brown, K.; Breuer, J. The differences in short-and long-term varicella-zoster virus (VZV) immunoglobulin G levels following varicella vaccination of healthcare workers measured by VZV fluorescent-antibody-to-membrane-antigen assay (FAMA), VZV time-resolved fluorescence immunoassay and a VZV purified glycoprotein enzyme immunoassay. Epidemiol. Infect. 2016, 144, 2345–2353.
- Gershon, A.A.; LaRussa, P.; Steinberg, S. Detection of antibodies to varicella-zoster virus using a latex agglutination assay. Clin. Diagn. Virol. 1994, 2, 271–277.
- Kim, Y.H.; Hwang, J.Y.; Shim, H.M.; Lee, E.; Park, S.; Park, H. Evaluation of a commercial glycoprotein enzyme-linked immunosorbent assay for measuring vaccine immunity to varicella. Yonsei Med. J. 2014, 55, 459–466.
- Zaia, J.A.; Oxman, M.N. Antibody to varicella-zoster virus-induced membrane antigen: Immunofluorescence assay using monodisperse glutaraldehyde-fixed target cells. J. Infect. Dis. 1977, 136, 519–530.
- Wutzler, P.; Färber, I.; Wagenpfeil, S.; Bisanz, H.; Tischer, A. Seroprevalence of varicella-zoster virus in the German population. Vaccine 2001, 20, 121–124.
- Sauerbrei, A.; Färber, I.; Brandstädt, A.; Schacke, M.; Wutzler, P. Immunofluorescence test for sensitive detection of varicella-zoster virus-specific IgG: An alternative to fluorescent antibody to membrane antigen test. J. Virol. Methods 2004, 119, 25–30.
- Han, S.; Kang, K.; Huh, D.; Lee, H.; Kim, J.; Kang, J.; Ma, S.H. Seroepidemiology of varicella-zoster virus in Korean adolescents and adults using fluorescent antibody to membrane antigen test. Epidemiol. Infect. 2015, 143, 1643–1650.
- Larussa, P.; Steinberg, S.; Waithe, E.; Hanna, B.; Holzman, R. Comparison of five assays for antibody to varicella-zoster virus and the fluorescent-antibody-to-membrane-antigen test. J. Clin. Microbiol. 1987, 25, 2059–2062.
- Cradock-Watson, J.; Ridehalgh, M.K.; Bourne, M.S. Specific immunoglobulin responses after varicella and herpes zoster. Epidemiol. Infect. 1979, 82, 319–336.
- Kitamura, K.; Namazue, J.; Campo-Vera, H.; Ogino, T.; Yamanishi, K. Induction of neutralizing antibody against varicella-zoster virus (VZV) by VZV gp3 and cross-reactivity between VZV gp3 and herpes simplex viruses gB. Virology 1986, 149, 74–82.
- Park, R.; Hwang, J.; Lee, K.; Namkoong, S.; Choi, S.; Park, S.; Park, H.; Park, J. Measurement of antibodies to varicella-zoster virus using a virus-free fluorescent-antibody-to-membrane-antigen (FAMA) test. J. Microbiol. Biotechnol. 2015, 25, 268–273.
- Litwin, V.; Sandor, M.; Grose, C. Cell surface expression of the varicella-zoster virus glycoproteins and Fc receptor. Virology 1990, 178, 263–272.
- Lafer, M.M.; Weckx, L.; de Moraes-Pinto, M.; Garretson, A.; Steinberg, S.; Gershon, A.; LaRussa, P.S. Comparative study of the standard fluorescent antibody to membrane antigen (FAMA) assay and a flow cytometry-adapted FAMA assay to assess immunity to varicella-zoster virus. Clin. Vaccine Immunol. 2011, 18, 1194–1197.
- Tourtelot, E.; Quataert, S.; Glantz, J.C.; Perlis, L.; Muthukrishnan, G.; Mosmann, T. Women who received varicella vaccine versus natural infection have different long-term T cell immunity but similar antibody levels. Vaccine 2020, 38, 1581–1585.
- Smith-Norowitz, T.A.; Saadia, T.A.; Norowitz, K.B.; Joks, R.; Durkin, H.G.; Kohlhoff, S. Negative IgG varicella zoster virus antibody status: Immune responses pre and post re-immunization. Infect. Dis. Ther. 2018, 7, 175–181.
- Gillard, P.; Povey, M.; Carryn, S. Clinically-versus serologically-identified varicella: A hidden infection burden. A ten-year follow-up from a randomized study in varicella-endemic countries. Hum. Vaccines Immunother. 2021, 17, 3747–3756.
- Chenghua, P.; Xiaolin, L.; Peixia, P.; Guiqiu, W.; Ming, D.; Yongji, W. Detection of VZV IgG by indirect ELISA. Chin. J. Biol. 1995, 8, 34–36.
- Enders, G. Serodiagnosis of Varicella-Zoster virus infection in pregnancy and standardization of the ELISA IgG and IgM antibody tests. Dev. Biol. Stand. 1982, 52, 221–236.
- Ono, E.; Lafer, M.M.; Weckx, L.Y.; Granato, C.; Moraes-Pinto, M.I.D. A simple and cheaper in house varicella zoster virus antibody indirect ELISA. Rev. Do Inst. De Med. Trop. De São Paulo 2004, 46, 165–168.
- Sauerbrei, A.; Schäfler, A.; Hofmann, J.; Schacke, M.; Gruhn, B.; Wutzler, P. Evaluation of three commercial varicella-zoster virus IgG enzyme-linked immunosorbent assays in comparison to the fluorescent-antibody-to-membrane-antigen test. Clin. Vaccine Immunol. 2012, 19, 1261–1268.
- Sauerbrei, A.; Wutzler, P. Serological detection of varicella-zoster virus-specific immunoglobulin G by an enzyme-linked immunosorbent assay using glycoprotein antigen. J. Clin. Microbiol. 2006, 44, 3094–3097.
- Sauerbrei, A.; Wutzler, P. Serological detection of specific IgG to varicella-zoster virus by novel ELISA based on viral glycoprotein antigen. Clin. Lab. 2009, 55, 1–7.
- Heininger, U.; Desgrandchamps, D.; Schaad, U.B. Seroprevalence of varicella-zoster virus IgG antibodies in Swiss children during the first 16 months of age. Vaccine 2006, 24, 3258–3260.
- Ceroni, A.; Sibani, S.; Baiker, A.; Pothineni, V.R.; Bailer, S.M.; LaBaer, J.; Haas, J.; Campbell, C.J. Systematic analysis of the IgG antibody immune response against varicella zoster virus (VZV) using a self-assembled protein microarray. Mol. Biosyst. 2010, 6, 1604–1610.
- Rolando, L.; Schneider, W.J.; Steinberg, S.; Low, S.; Stiles, J.; Gomez, L.; Gershon, A.A.; Brown, A.E. Effect of varicella-zoster virus (VZV) fluorescent-antibody-to-membrane-antigen (FAMA) testing on sensitivity of determining VZV immunity in healthcare workers and on furlough days. Infect. Control. Hosp. Epidemiol. 2010, 31, 972–974.
- Wasmuth, E.H.; Miller, W.J. Sensitive enzyme-linked immunosorbent assay for antibody to varicella-zoster virus using purified VZV glycoprotein antigen. J. Med. Virol. 1990, 32, 189–193.
- Li, S.; CHAN IV, A.S.; Matthews, H.; Heyse, J.F.; Chan, C.Y.; Kuter, B.J.; Kaplan, K.M.; Vessey, S.R.; Sadoff, J.C. Inverse relationship between six week postvaccination varicella antibody response to vaccine and likelihood of long term breakthrough infection. Pediatr. Infect. Dis. J. 2002, 21, 337–342.
- Mitra, M.; Faridi, M.; Ghosh, A.; Shah, N.; Shah, R.; Chaterjee, S.; Narang, M.; Bhattacharya, N.; Bhat, G.; Choudhury, H.; et al. Safety and immunogenicity of single dose live attenuated varicella vaccine (VR 795 Oka strain) in healthy Indian children: A randomized controlled study. Hum. Vaccines Immunother. 2015, 11, 443–449.
- Otani, N.; Tanaka, M.; Maeda, K.; Gomi, Y.; Nakajima, K.; Tanimura, S.; Takesue, Y.; Shima, M.; Okuno, T. Varicella zoster virus antibody detection: A comparison of four commonly used techniques. J. Infect. Chemother. 2016, 22, 225–228.
- Muchtar, E.; Koehler, A.B.; Johnson, M.J.; Rabe, K.G.; Ding, W.; Call, T.G.; Leis, J.F.; Kenderian, S.S.; Hayman, S.R.; Wang, Y.; et al. Humoral and cellular immune responses to recombinant herpes zoster vaccine in patients with chronic lymphocytic leukemia and monoclonal B cell lymphocytosis. Am. J. Hematol. 2022, 97, 90–98.
- Liu, J.; Ye, X.; Jia, J.; Zhu, R.; Wang, L.; Chen, C.; Yang, L.; Wang, Y.; Wang, W.; Ye, J.; et al. Serological evaluation of immunity to the varicella-zoster virus based on a novel competitive enzyme-linked immunosorbent assay. Sci. Rep. 2016, 6, 20577.
- Liu, J.; Chen, C.; Zhu, R.; Ye, X.; Jia, J.; Yang, L.; Wang, Y.; Wang, W.; Ye, J.; Li, Y.; et al. Evaluation of immunity to varicella zoster virus with a novel double antigen sandwich enzyme-linked immunosorbent assay. Appl. Microbiol. Biotechnol. 2016, 100, 9321–9329.
- Caunt, A.E.; Shaw, D.G. Neutralization tests with varicella-zoster virus. Epidemiol. Infect. 1969, 67, 343–352.
- Grose, C.; Edmond, B.J.; Brunell, P.A. Complement-enhanced neutralizing antibody response to varicella-zoster virus. J. Infect. Dis. 1979, 139, 432–437.
- Asano, Y.; Takahashi, M. Clinical and serologic testing of a live varicella vaccine and two-year follow-up for immunity of the vaccinated children. Pediatrics 1977, 60, 810–814.
- Asano, Y.; Albrecht, P.; Stagno, S.; Takahashi, M. Potentiation of neutralization of varicella-zoster virus by antibody to immunoglobulin. J. Infect. Dis. 1982, 146, 524–529.
- Krah, D.L.; Provost, P.J.; Ellis, R.W. Combined use of complement and anti-immunoglobulin in an enhanced neutralization assay for antibodies to varicella-zoster virus. J. Virol. Methods 1995, 53, 176–187.
- Asano, Y.; Nakayama, H.; Yazaki, T.; Ito, S.; Isomura, S. Protective efficacy of vaccination in children in four episodes of natural varicella and zoster in the ward. Pediatrics 1977, 59, 8–12.
- Schmidt, N.J.; Lennette, E.H. Neutralizing antibody responses to varicella-zoster virus. Infect. Immun. 1975, 12, 606–613.
- Asano, Y.; Takahashi, M. Studies on neutralization of varicella-zoster virus and serological follow-up of cases of varicella and zoster. Biken J. 1978, 21, 15–23.
- Gerna, G.; Chambers, R.W. Varicella-zoster plaque assay and plaque reduction neutralization test by the immunoperoxidase technique. J. Clin. Microbiol. 1976, 4, 437–442.
- Schmidtmayerova, H.; Mayer, V.; Zachar, V. Focus assay for varicella-zoster virus in human embryo cells stained with immunoperoxidase method. Acta Virol. 1986, 30, 468–474.
- Chen, L.; Liu, J.; Wang, W.; Ye, J.; Wen, L.; Zhao, Q.; Zhu, H.; Cheng, T.; Xia, N. Development of a varicella-zoster virus neutralization assay using a glycoprotein K antibody enzyme-linked immunosorbent spot assay. J. Virol. Methods 2014, 200, 10–14.
More
Information
Subjects:
Biotechnology & Applied Microbiology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
2 times
(View History)
Update Date:
19 Apr 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





