Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | BALARAM VYSETTI | -- | 1862 | 2023-04-13 05:04:01 |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Balaram, V. Potential Alternative Resources for Rare Earth Elements. Encyclopedia. Available online: https://encyclopedia.pub/entry/43006 (accessed on 05 May 2026).
Balaram V. Potential Alternative Resources for Rare Earth Elements. Encyclopedia. Available at: https://encyclopedia.pub/entry/43006. Accessed May 05, 2026.
Balaram, Vysetti. "Potential Alternative Resources for Rare Earth Elements" Encyclopedia, https://encyclopedia.pub/entry/43006 (accessed May 05, 2026).
Balaram, V. (2023, April 13). Potential Alternative Resources for Rare Earth Elements. In Encyclopedia. https://encyclopedia.pub/entry/43006
Balaram, Vysetti. "Potential Alternative Resources for Rare Earth Elements." Encyclopedia. Web. 13 April, 2023.
Copy Citation
Seventeen elements in the periodic table including fifteen lanthanides (La, Ce, Pr, Nd, Pm, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, and Lu) and Sc, as well as Y are often collectively referred to as rare earth elements (REE). The depleting REE ores and increasing mining costs are prompting us to consider alternative sources for these valuable metals, particularly from waste streams. Although REE concentrations in most of the alternative resources are lower than current REE ores, some sources including marine sediments, coal ash, and industrial wastes, such as red mud, are emerging as promising with significant concentrations of REE.
rare earth elements
deposits
alternative sources
marine sediments
river sediments
phosphorites
red mud
fly ash
acid mine drainage
e-waste
extra-terrestrial
1. Introduction
Seventeen elements in the periodic table including fifteen lanthanides (La, Ce, Pr, Nd, Pm, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, and Lu) and Sc, as well as Y are often collectively referred to as rare earth elements (REE). Promethium is well-known as the only element in the lanthanide series of the periodic table with no stable isotopes and occurs in the Earth’s crust in only tiny amounts in some natural materials, such as some uranium ores. Scandium is geochemically only partly similar to REE. Based on atomic numbers, they are divided into two groups. The lower atomic weight elements from La to Sm, the most abundant ones, with atomic numbers 57–62 are referred to as light REE (LREE), while from Eu to Lu, the least common and the most valuable with atomic numbers 63–71, are known as heavy REE (HREE). In nature, REE do not occur as single native metals, such as gold or silver since they are easily oxidized due to their similar physical and chemical properties. Moreover, they frequently occur together in several geologic formations in many ores or minerals as minor or major constituents.
Due to their unique physical, chemical, mechanical, electronic, magnetic, luminescent, phosphorescent, and catalytic properties, these elements have become exceptionally important during the last two decades, and there has been an explosion in the industrial applications of these elements in different high technology devices, such as smartphones, computers, televisions, light emitting diodes, catalysts for fuel cells, corrosion inhibitors, hard drives, magnets for wind turbines, and other power generating systems. Moreover, they are a crucial element of national security as they are extensively used in several military defence systems [1][2][3]. Their important medical applications include metallic implants, lasers, and magnetic resonance imaging (MRI) measurements [4]. REE are vital, even to the space industry, as they are used in everything from launch vehicles to national defence and commercial communication and observation satellites. Furthermore, REE are used in creating catalysts in several industrial processes, as well as in the fabrication of autocatalytic catalytic converters in transport vehicles [5][6].
Although REE deposits are found practically all over the world, currently, there is a global shortage due to the decreasing number of economically profitable deposits. In addition, our reliance on REE in our high-tech gadget-hungry world is growing with time, but their supply is far from secure. Unfortunately, the conventional ore resources are becoming depleted, as the demand for REE increases, and the industry is paying attention to unconventional resources, such as coal, recycling, and marine sediments as alternative sources for these elements. Moreover, it is very important to maintain a proper supply chain to meet the demand for the development of highly advanced technologies. Even the substitution for the REE is difficult for most applications, although powerful permanent magnets made out of iron and nitrogen (iron nitride, Fe16N2) are helping the automotive and wind energy industries in a small way [7]. Pavel et al. [8] studied the possibilities of the substitution of REE in wind turbines in order to reduce the dependency on REE. However, later, it was found that these claims were impossible without neodymium. As a result, recycling is presently increasing in several countries. Many approaches including physical, chemical, and biological procedures, such as pyrometallurgy, solvent extraction, and membrane separation are available for the efficient extraction of REE from these resources [9]. One very important aspect is that currently extracted ore grades, with an average of 5% REE and running as high as 15%, have significantly higher concentrations than most of the discussed alternative sources [10].
2. REE Deposits
Although REE comprise significant amounts of a wide range of minerals, including oxides, silicates, carbonates, phosphates, and halides, almost all production comes from less than ten minerals, such as apatite, monazite, xenotime, allanite, and bastnäsite [11]. Currently, these deposits are mainly located in China, Brazil, Vietnam, Canada, Russia, Namibia, South Africa, and India. Although China has been dominating in this area, several other countries, such as the USA, Australia, Turkey, and Sweden have successfully discovered new REE deposits recently. In fact, a number of new REE metallogenetic belts can be identified on the basis of age, tectonic setting, lithological association, and known REE enrichments [12]. REE deposits are divided into primary (formed by magmatic, hydrothermal, and/or metamorphic processes) and secondary deposits (formed by weathering and sedimentary transport) depending on their form of occurrence, genetic associations, and mineralogy [13]. Figure 1 presents an illustrative view of different types of REE resources, including the potential alternative future resources.
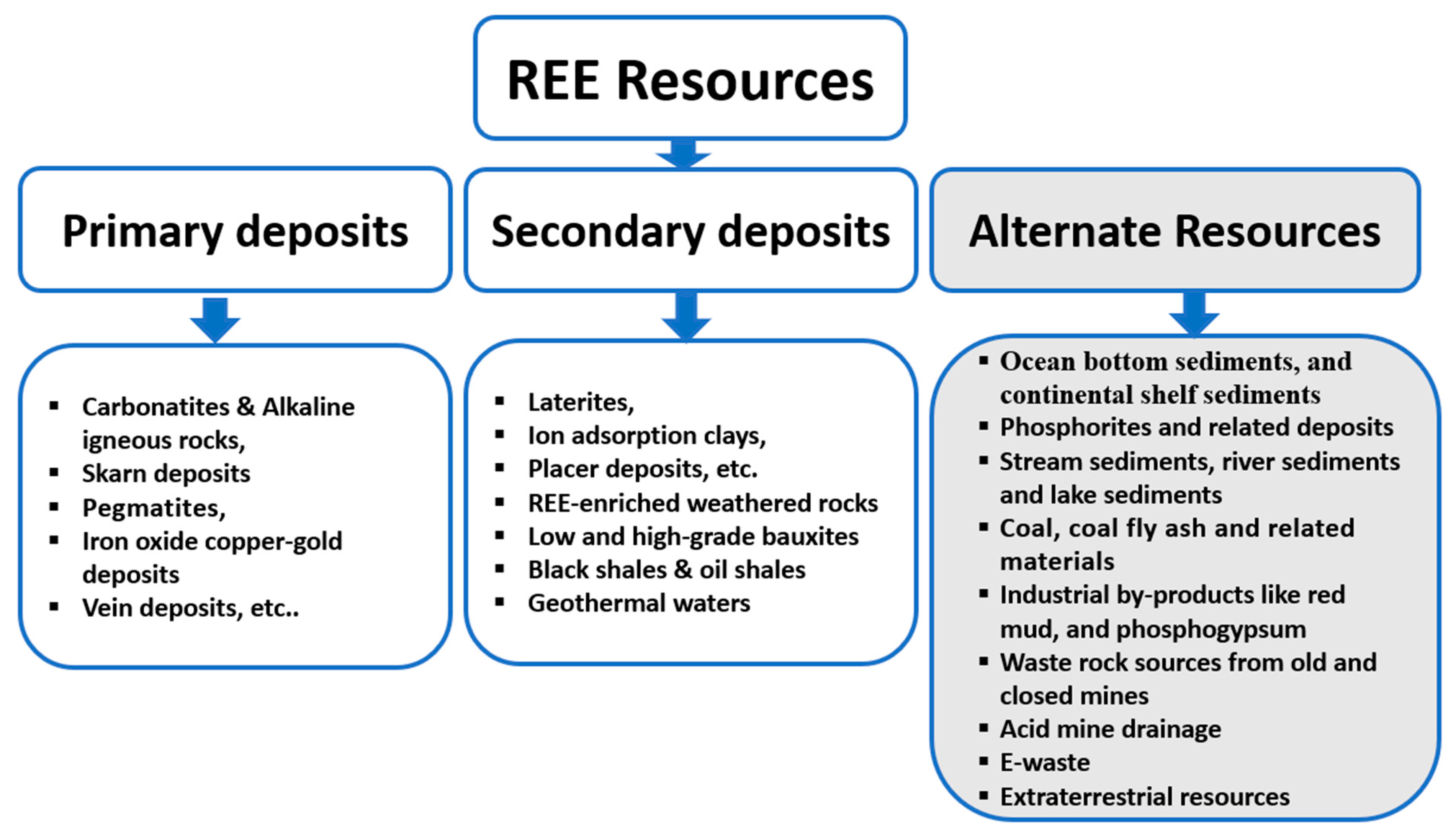
Figure 1. Illustrative view of different types of REE resources, including the potential alternative future resources shaded in light color [14].
2.1. Primary Deposits
Primary deposits of REE are commonly associated with carbonatites and alkaline igneous rocks, pegmatites, iron oxide copper-gold deposits, and vein and skarn deposits. Of these carbonatites, peralkaline silicate rocks with appreciable concentrations of REE are the most important REE resources [14][15]. These REE deposits in igneous rocks have played an important role in meeting the industrial demand for decades. Moreover, these igneous mineral deposits can be divided into five distinct categories depending on the provenance, the evolution of the magma, and the rock types hosting mineralization: (i) Carbonatites, (ii) peralkaline silica undersaturated rocks, (iii) peralkaline granites and pegmatites, (iv) pegmatites associated with sub- to meta-luminous granites, and (v) Fe oxide–phosphate deposits [16]. More insights on these primary REE deposits were presented by Weng et al. [17] and Dushyantha et al. [13]. Skarn deposits of REE are important in sustainable economic development across the world [18]. In Fe oxide–phosphate deposits, these elements are concentrated in apatite minerals [19].
2.2. Secondary Deposits
Some of the important secondary deposits of REE are laterites, ion adsorption clays, low- and high-grade bauxites, and placer deposits. Ion-adsorption deposits are formed due to the erosion and weathering of primary deposits within weathering crusts, which supply more than 95% of the global HREE demand. Intensive lateritic weathering of bedrocks under tropical or sub-tropical climatic conditions can form a variety of secondary deposits, which may range in composition from aluminous bauxites to hematite, goethite, and titanium sands, as well as REE [20]. Recently, Jo and Shin [21] reported REE-enriched weathered anorthosite rocks with ΣREE range of 242–857 µg/g. Ionic clays, formed by the natural weathering process of REE minerals and the adsorption of the resulting liberated REE ions on the clay surface, are an important resource for critical REE. These ion-adsorption clay deposits are most suitable for in situ leaching mining [22]. Ion-adsorption REE deposits are mostly developed due to weathering of REE-rich granites, and REE ions are mainly adsorbed on clay minerals [23]. Currently, regolith-hosted REE deposits from areas with abundant REE deposits have become one of the major sources of global REE. Regolith-hosted REE deposits with abundant deposits around Meizhou City, Guangdong Province, China with the highest ΣREE range of 1162 μg/g were reported recently by Lin et al. [24]. The REE concentrations in the regolith-hosted REE deposits in the Chilean coastal range of the central Andes were found to be up to 2000 µg/g [25]. Beach sands are the products of a combination of weathering, fragmentation, and degradation, and are well-known for their economic concentration of heavy minerals, such as monazite, ilmenite, zircon, rutile, allanite, sillimanite, and garnet. REE are abundant in some of these minerals, particularly in monazite and allanite [26]. Black shales can also be considered as a secondary resource for these valuable metals. Recently, El-Anwar et al. [27] reported REE enrichment (average 255.3–325.3 µg/g) in black shales of the Safaga-Qussier sector, Egypt. Another study is reported from the rock-soil-moss system in the black shale area in China, with an average concentration of ∑REE around 245 µg/g [28]. Akhtar et al. [29] reported ∑REE concentrations up to 372 µg/g in Paleoproterozoic black shales from Singhbhum mobile belt, Eastern India. These recent studies, along with the industry standard of ~300 µg/g cut-off grade for mining, prompted more studies on black shale formations worldwide before considering the black shale deposits as an alternative source of REE. In fact, Ketris and Yudovich [30] reported an average worldwide REE concentration of 134.19 in black shales. Li et al. [31] reported moderately high concentrations of REE ranging from 105 to 195 µg/g at an average concentration of 151 µg/g in oil shale samples in Tongchuan City, Southern Ordos Basin, China, prompting the necessity for further investigations to understand the REE potential of these rocks. The value indicated above is significantly below the cut-off grade of 1000 µg/g (ash basis) for coal-hosted REE deposits [32]. In fact, the total REE contents, individual REE compositions, and sizes of different deposits are important for arriving at the cut-off value for the economic recovery of REE.
Geothermal fluids are potentially significant sources of valuable minerals and metals. Smith et al. [33] provided a rough estimate of total REE concentrations of 0.17 µg/mL in geothermal waters. Kurzawa et al. [34], in an attempt to understand the REE concentrations in the mineral and thermal waters in Polish Lowlands, found that a high temperature (T > 60 °C) favored the release of the more easily soluble REE from rocks into the water. Wei et al. [35] recently reported the total REE contents of the geothermal water in the range of 0.059–0.547 ng/mL, in the Ganzi–Litang fault, western Sichuan, China. The REE contents in geothermal waters are related to the reservoir lithology and are significantly influenced by pH and HCO3−, Na+, and Mn minerals. Although the REE content in geothermal waters is not considerable, if the abundant geothermal waters in places, such as Iceland are taken into consideration, this source could be one of the most promising ones for REE with an efficient extraction technology. Recently, significant efforts have been made to develop technologies for the extraction of REE from geothermal brines since the geothermal brine can be a cost-effective mineral resource. REE extraction from geothermal brine only seems feasible at present if extracted with other co-products, such as silica and lithium [33].
2.3. Different Types of Potential Alternative Deposits
To date, the identified primary and secondary REE resources are not able to meet the global demand and requirements. Discovering and establishing alternative resources for REE have been a topic of high interest for the past decade. Therefore, a large number of studies are carried out for the identification of potential alternative deposits to meet the growing demand worldwide. Powerful analytical techniques, such as inductively coupled plasma mass spectrometry (ICP-MS), inductively coupled plasma optical emission spectrometry (ICP-AES), scanning electron microscopy with energy dispersive X-ray spectrometry (SEM-EDX), X-ray fluorescence spectrometry (XRF), X-ray diffractometry (XRD), and integrated mineral analysis (IMA), are being utilized to understand the future potential of various alternative REE resources [36][37][38].
References
- Balaram, V. Rare Earth Elements: Sources and Applications. In Environmental Technologies to Treat Rare Earth Elements Pollution: Principles and Engineering; Sinharoy, A., Lens, P., Eds.; IWA Publishers: London, UK, 2021.
- Mendez, J.A.C.; Vong, Y.M.; Bueno, J.J.P. Cerium and Other Rare Earth Salts as Corrosion Inhibitors—A Review. In Protection of Metals and Physical Chemistry of Surfaces; Springer: Berlin/Heidelberg, Germany, 2022; pp. 1–10.
- Zhu, S.; Yang, L.; Bai, J.; Chu, Y.; Liu, J.; Jin, Z.; Liu, C.; Ge, J.; Xing, W. Ultra-stable Pt La intermetallic compound towards highly efficient oxygen reduction reaction. Nano Res. 2022, 16, 2035–2040.
- Raju, C.S.K.; Cossmer, A.; Scharf, H.; Panne, U.; Lück, D. Speciation of gadolinium-based MRI contrast agents in environmental water samples using hydrophilic interaction chromatography hyphenated with inductively coupled plasma mass spectrometry. J. Anal. At. Spectrom. 2010, 25, 55e61.
- Balaram, V. Environmental Impact of Pt, Pd and Rh Emissions from Autocatalytic Converters—A Brief Review of the Latest Developments. In Handbook of Environmental Materials Management; Hussain, C.M., Ed.; Springer: Cham, Switzerland, 2020.
- Gong, Y.; Li, H.; Li, C.; Bao, X.; Hosono, H.; Wang, J. Insight into rare-earth-incorporated catalysts: The chance for a more efficient ammonia synthesis. J. Adv. Ceram. 2022, 11, 1499–1529.
- Molla, S.; Farrok, O.; Islam, M.R.; Muttaqi, K.M. Application of iron nitride compound as alternative permanent magnet for designing linear generators to harvest oceanic wave energy. IET Electr. Power Appl. 2020, 14, 762–770.
- Pavel, C.C.; Arántegui, R.L.; Marmier, A.; Schüler, D.; Tzimas, E.; Buchert, M.; Jenseit, W.; Blagoeva, D. Substitution strategies for reducing the use of rare earths in wind turbines. Resour. Policy 2017, 52, 349–357.
- Balaram, V. Rare Earth Elements, Resources, Extraction Technologies, Limitations, and Global Trade—A Comprehensive Review. In Treatise on Geochemistry, 3rd ed.; Filippelli, G., Anbar, A., Weis, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2024; in press.
- Deng, B.; Wang, X.; Luong, D.X.; Carter, R.A.; Wang, Z.; Tomson, M.B.; Tour, J.M. Rare earth elements from waste. Sci. Adv. 2022, 8, eabm3132.
- Balaram, V.; Sawant, S.S. Indicator Minerals, Pathfinder Elements, and Portable Analytical Instruments in Mineral Exploration Studies. Minerals 2022, 12, 394.
- Goodenough, K.M.; Schilling, J.; Jonsson, E.; Kalvig, P.; Charles, N.; Tuduri, J.; Deady, E.A.; Sadeghi, M.; Schiellerup, H.; Müller, A.; et al. Europe’s rare earth element resource potential: An overview of REE metallogenetic provinces and their geodynamic setting. Ore Geol. Rev. 2016, 72, 838–856.
- Dushyantha, N.; Batapola, N.; Ilankoon, I.M.S.K.; Rohitha, S.; Premasiri, R.; Abeysinghe, B.; Ratnayake, N.; Dissanayake, K. The story of rare earth elements (REEs): Occurrences, global distribution, genesis, geology, mineralogy and global production. Ore Geol. Rev. 2020, 122, 103521.
- Balaram, V. Rare Earth Element Deposits—Sources, and Exploration Strategies. J. Geol. Soc. India 2022, 98, 1210–1216.
- Dostal, J. Rare Earth Element Deposits of Alkaline Igneous Rocks. Resources 2017, 6, 34.
- Chakhmouradian, A.R.; Zaitsev, A.N. Rare Earth Mineralization in Igneous Rocks: Sources and Processes. Elements 2012, 8, 347–353.
- Weng, Z.; Jowitt, S.M.; Mudd, G.M.; Haque, N. A Detailed Assessment of Global Rare Earth Element Resources: Opportunities and Challenges. Econ. Geol. 2015, 110, 1925–1952.
- Elsharif, I.A. Geological and Geochemical exploration methods for mineral resources (skarn deposits and rare earth elements). News Ural State Min. Univ. Earth Sci. 2022, 2, 7–15.
- Aali, A.A.; Khakmardan, S.; Shirazi, A.; Nazerian, H. A Review of Mineralization of Rare Earth Elements in Iran. Int. J. Sci. Eng. Appl. 2022, 11, 92–99.
- Cocker, M.D. Lateritic, supergene rare earth element (REE) deposits. In Proceedings of the 48th Annual Forum on the Geology of Industrial Minerals, Phoenix, AZ, USA, 30 April–4 May 2012; pp. 1–18.
- Jo, J.; Shin, D. Geochemical characteristics of REE-enriched weathered anorthosite complex in Hadong district, South Korea. Geochem. J. 2023, 57, 13–27.
- Zhong, W.; Ouyang, J.; Yang, D.; Wang, X.; Guo, Z.; Hu, K. Effect of the in situ leaching solution of ion-absorbed rare earth on the mechanical behavior of basement rock. J. Rock Mech. Geotech. Eng. 2022, 14, 1210e1220.
- Wang, H.; He, H.; Bao, W.Y.Z.; Liang, X.; Zhu, J.; Ma, L.M.; Huang, Y. Zircon texture and composition fingerprint HREE enrichment in muscovite granite bedrock of the Dabu ion-adsorption REE deposit, South China. Chem. Geol. 2023, 616, 121231.
- Lin, Z.; Wei, G.; Zhang, J.; Liang, X.; Huang, G. Origin and distribution of rare earth elements (REEs) in the soils of Meizhou City, southern China with high abundance of regolith-hosted REEs. Appl. Geochem. 2022, 147, 105514.
- Bustos, N.; Marquardt, C.; Belmar, A.; Cordeiro, P. Regolith-hosted rare earth exploration in the Chilean Coastal Range of the Central Andes. J. Geochem. Explor. 2022, 234, 106934.
- Sengupta, D.; van Gosen, B. Placer-Type Rare Earth Element Deposits. Rev. Econ. Geol. 2016, 18, 81–100.
- El-Anwar, E.A.A.; Abdelhafiz, M.A.; Salman, S.A. Rare earth and trace elements enrichment and implications in black shales of Safaga-Qussier sector, Egypt. J. Afr. Earth Sci. 2022, 188, 104482.
- Xu, Y.; Luo, C.; Gao, L.; Long, J.; Xu, H.; Yang, R. Anomalous concentrations and environmental implications of rare earth elements in the rock-soil-moss system in the black shale area. Chemosphere 2022, 307, 135770.
- Akhtar, R.M.; Zubair, A.B.; Shabber, H.A.; Balaram, V. Geochemistry of black shales from Singhbhum mobile belt, Eastern India: Implications for paleo-weathering and provenance. Himal. Geol. 2015, 36, 126–133.
- Ketris, M.P.; Yudovich, Y.E. Estimations of Clarkes for Carbonaceous biolithes: World averages for trace element contents in black shales and coals. Int. J. Coal Geol. 2009, 78, 135–148.
- Li, D.; Li, R.; Xue, T.; Wang, B.; Liu, F.; Zhao, B.; Zhao, D. Characteristic and Geological Implications of Major Elements and Rare Earth Elements of Triassic Chang 7 Oil Shale in Tongchuan City, Southern Ordos Basin (China). Minerals 2018, 8, 157.
- Dai, S.; Finkelman, R.B.; French, D.; Hower, J.C.; Graham, I.T.; Zhao, F. Modes of occurrence of elements in coal: A critical evaluation. Earth-Science Rev. 2021, 222, 103815.
- Smith, Y.R.; Kumar, P.; McLennan, J.D. On the Extraction of Rare Earth Elements from Geothermal Brines. Resources 2017, 6, 39.
- Kaczor-Kurzawa, D.; Wysocka, I.; Porowski, A.; Drzewicz, P.; Vassileva, E. The occurrence and distribution of rare earth elements in mineral and thermal waters in the Polish Lowlands. J. Geochem. Explor. 2022, 237, 106984.
- Wei, S.; Liu, F.; Zhang, W.; Zhang, H.; Zhao, J.; Liao, Y.; Yan, X. Typical geothermal waters in the Ganzi–Litang fault, western Sichuan, China: Hydro chemical processes and the geochemical characteristics of rare-earth elements. Environ. Earth Sci. 2022, 81, 538.
- Balaram, V. Recent trends in the instrumental analysis of rare earth elements in geological and industrial materials. TrAC Trends Anal. Chem. 1996, 15, 475–486.
- Balaram, V. Current and emerging analytical techniques for geochemical and geochronological studies. Geol. J. 2021, 56, 2300–2359.
- Van Rythoven, A.D.; Pfaff, K.; Clark, J.G. Use of QEMSCAN® to characterize oxidized REE ore from the Bear Lodge carbonatite, Wyoming, USA. Ore Energy Resour. Geology. 2020, 2–3, 10005.
More
Information
Subjects:
Mineralogy
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revision:
1 time
(View History)
Update Date:
13 Apr 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





