Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jolanta Gruszecka | -- | 1078 | 2023-03-24 09:55:17 | | | |
| 2 | Catherine Yang | Meta information modification | 1078 | 2023-03-27 04:17:47 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Jarmakiewicz-Czaja, S.; Gruszecka, J.; Filip, R. Inflammatory Bowel Diseases. Encyclopedia. Available online: https://encyclopedia.pub/entry/42504 (accessed on 10 May 2026).
Jarmakiewicz-Czaja S, Gruszecka J, Filip R. Inflammatory Bowel Diseases. Encyclopedia. Available at: https://encyclopedia.pub/entry/42504. Accessed May 10, 2026.
Jarmakiewicz-Czaja, Sara, Jolanta Gruszecka, Rafał Filip. "Inflammatory Bowel Diseases" Encyclopedia, https://encyclopedia.pub/entry/42504 (accessed May 10, 2026).
Jarmakiewicz-Czaja, S., Gruszecka, J., & Filip, R. (2023, March 24). Inflammatory Bowel Diseases. In Encyclopedia. https://encyclopedia.pub/entry/42504
Jarmakiewicz-Czaja, Sara, et al. "Inflammatory Bowel Diseases." Encyclopedia. Web. 24 March, 2023.
Copy Citation
Liver disease is one of the most common extraintestinal manifestations of inflammatory bowel disease (IBD). IBD cause chronic inflammation and can affect various sections of the gastrointestinal tract. A particular form of inflammatory bowel disease is inflammatory bowel disease (IBD).
Crohn’s disease
inflammatory bowel diseases
liver fibrosis
1. Introduction
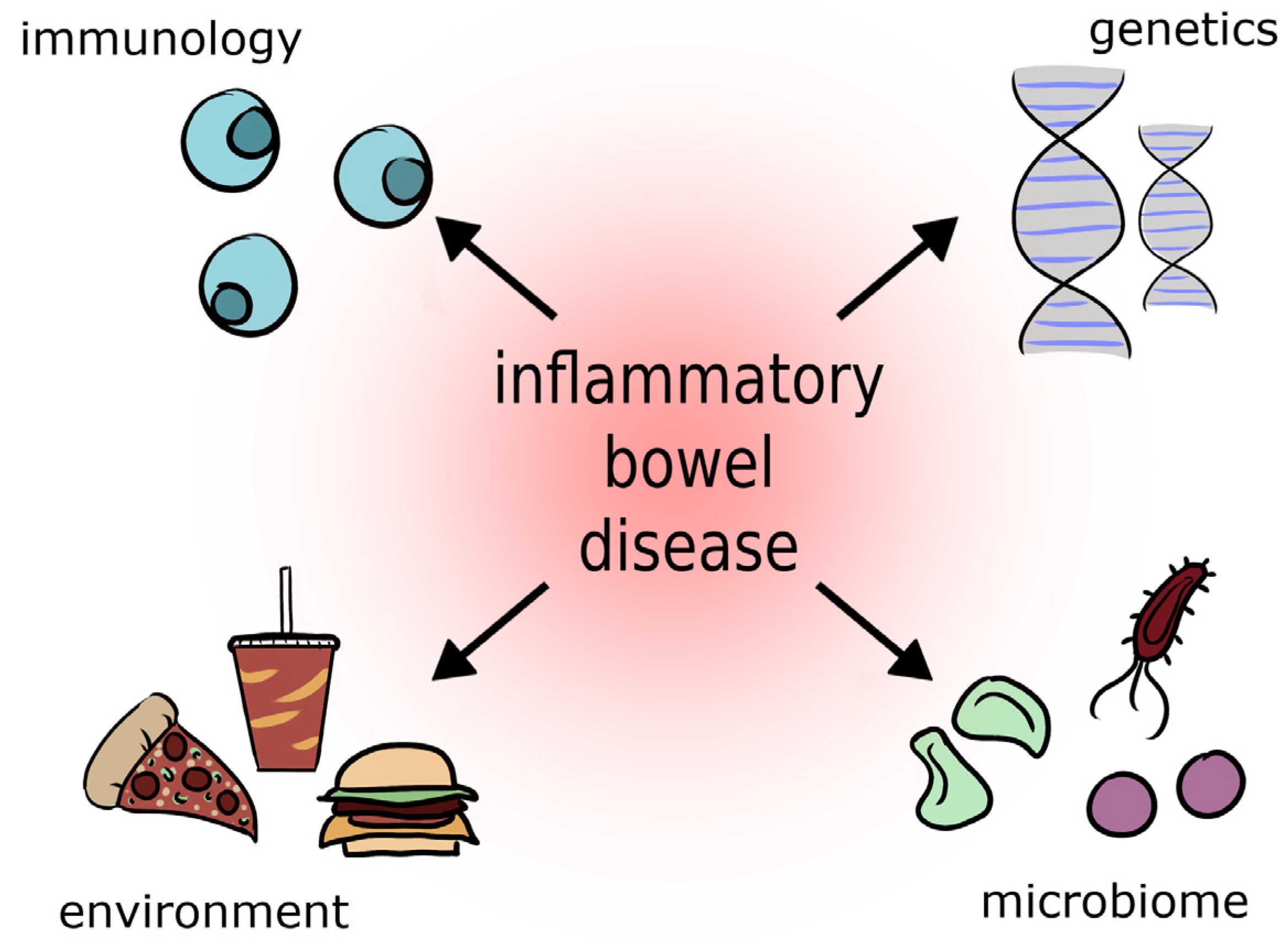
It is estimated that about 2.5–3 million people in Europe struggle with this disease. The prevalence of inflammatory bowel disease in the United States is 70 to 150 cases per 100,000 people and is increasing [1]. In Poland, approximately 50,000 people suffer from inflammatory bowel disease, one in four of whom are minors [2][3]. Inflammatory bowel diseases include CD, UC, indeterminate colitis, and microscopic colitis [2][4][5]. Among the causes that influence the onset of IBD are genetic, immunological, and environmental factors (dietary changes, stress, overuse of antibiotics, especially in the first years of life, smoking, alcohol, progressive environmental degradation, high content of artificial preservatives in food) [1][6] (Figure 1).
An example of immune factors predisposing to IBD is an imbalance of Th17/Treg cells. This balance is influenced by the gut microbiome, T Cell Receptor (TCR) signaling or cytokines, among other factors [7]. Furthermore, changes in the number of T cells can also lead to the development of LF, for example, by indirectly inducing liver damage via Th17 and Tc (cytotoxic T) cells [8]. Many authors also point out intestinal homeostasis disturbances in patients with IBD (Table 1). These conditions affect people of different ages. According to statistics, most patients are adults between the ages of 20 and 40, but a third of patients are adolescents and children. IBD is increasingly affecting older people. Infants are the least likely to suffer from inflammatory bowel disease [2].
Table 1. Intestinal homeostasis disturbances in patients with inflammatory bowel disease [9][10][11][12][13][14][15][16][17][18][19].
| Intestinal Epithelial Barrier Disturbances | Intestinal Microbiota Disturbances |
|---|---|
| -increased expression of claudin-1–reduction of mucus secretion, which increases the risk of enteritis (CD and UC) -increased expression of claudin-2–Increased pores in TJ (CD and UC) -decreased expression of claudin–3,5,8 (CD)–reduction of intestinal barrier properties -decreased expression of claudin–3,4,7 (UC)–reduction of intestinal barrier properties -increased expression of occludin (active UC) -ZO1 dysfunction–decrease in stabilised claudin strands -TNF-α–decrease in normal TJ function and increase in intestinal epithelial apoptosis -IL-1𝛽 i IFN-𝛾–reduction of TJ integrity by affecting occludins and ZO1 -IFN-𝜆–occurrence of Paneth cell defect -dysfunction of ILC3 -secrection of chemokines by EIC–causes and maintains inflammation in IBD (e.g. CXCR1 (+) CXCR2 (+) IL-23) |
Increase in the amount of the following bacteria: -Proteobacteria (degradation of intestinal mucus) np. Haemophilus, Pasteurellaceae -Streptococcus -Fusobacterium (degradation of intestinal mucus) -Enterobacteriaceae np. E. Coli (AIEC) Decrease in the amount of the following bacteria: -Firmicutes np. Faecalibacterium prausnitzii, Roseburia (SCFA producing bacteria) -Euryarchaeota -Bacteroidetes np. Prevotella spp. -Bifidobacterium Increase in the amount of following fungus: -C. albicans -C. parapsilosis -Aspergillus clavatus (CD) -Cryptococcus neoformans (CD) -Ascomycota |
CD-Crohn’s Disease, UC-Ulcerative Colitis, TJ-tight junction, ZO-zonula occludens, TNFα-tumor necrosis factor α, IL-1𝛽- Interleukin-1 beta, IFN-𝛾- Interferon gamma, IFN-𝜆-interferon lambda, ILC3-innate lymphoid cells-3, EIC-intestinal epithelial cells, CXCR- CXC chemokine receptor, SCFA-short-chain fatty acids
2. Symptoms and Diagnostics of Inflammatory Bowel Disease
People with inflammatory digestive disease often experience abdominal pain, nausea, and appetite disorders. Vomiting, diarrhoea, and a subfebrile state are common. These symptoms cause impaired absorption, leading to nutrient deficiencies and hypovitaminosis [20]. Among patients diagnosed with chronic IBD, the incidence rate of depression is 15–30%, with up to 80% of patients experiencing anxiety during disease exacerbation compared to fully healthy individuals. This is caused by the deterioration of quality of life by interfering with normal physical and mental functioning. Patients rarely need psychological support, which is an important part of a holistic approach to their care [21]. IBD sufferers often exclude various foods from their diet, identified with their ailments. Because of this, they may experience hypoproteinemia and low levels of vitamins. In serum, A, D, K, C and B have low concentrations of minerals, iron, zinc, magnesium, resulting in reduced immunity, problems with wound healing, and more frequent infections occur. Inflammatory bowel diseases increase the risk of osteoporosis and osteopenia as a consequence of vitamin D and calcium deficiency, which affect between 3% and 30% of patients [20]. Inflammatory bowel disease leads to serious and life-threatening complications ranging from intestinal perforation, joint, skin, eye, and liver disease to colon cancer [20].
UC is one of the diseases of the IBD. It involves inflammation of the mucosa of the colon, leading to widespread and shallow ulcerations. Symptoms: diarrhoea, mucus, and blood fragments may appear, abdominal cramps and pain, sudden feeling of pushing on the stool, lack of appetite, significant weight loss. Occasionally, UC often produces extraintestinal symptoms, such as uveitis and scleritis, joint pain, erythema nodosum, alopecia, liver disease, thrombosis, and anemia [1][2][22].
Another disease of inflammatory bowel disease is CD. It is chronic and progresses slowly. Inflammation occupies the intestinal mucosa partially and sometimes even entirely, leading to fistulas, abscesses, and ulcers. The most common symptoms are abdominal pain and cramps of varying severity, vomiting, diarrhoea, nausea, flatulence, and extraintestinal symptoms.
Both of these conditions prevent normal functioning. Patient reports indicate that they sometimes use the bathroom up to 20 times a day [1][2][22].
The diagnosis of IBD is based on invasive and noninvasive methods. For the proper diagnosis of IBD, an endoscopic examination, gastroscopy, and colonoscopy must be performed, which requires adequate preparation, often causing stress and anxiety in patients. It is contraindicated in cases of exacerbation of the disease [23]. Sometimes a histopathological examination of the specimen is necessary. This is followed by a radiographic examination or magnetic resonance imaging of the intestines.
Determining the biomarkers calprotectin and lactoferrin in the stool is helpful for diagnosing IBD [6]. These proteins are released into the gastrointestinal tract as a response to intestinal inflammation. Calprotectin is secreted by immune cells located in the deeper layers of the intestine [20][24][25][26]. The test of a stool sample for the presence of lactoferrin protein also provides valuable information on intestinal status. Lactoferrin is produced by mucosal epithelial cells and is crucial for normal mucosal defence of the gastrointestinal tract [27][28]. Elevated fecal lactoferrin levels signify an increased mucosal immune response to food or bacterial antigens and may indicate a chronic state of inflammatory gastrointestinal disease. If this is the case, a further diagnosis is recommended [24][27][28][29].
IBD have in common that disease exacerbations alternate with moments of remission, when symptoms resolve completely or partially. Treatment should be tailored to the stage of the disease, the patient’s condition, and the specific symptoms present. Complete cure is not possible, so therapy consists of pharmacological maintenance of the remission state and alleviation of symptoms [24][25][26][30].
References
- de Zoeten, E.F.; Pasternak, B.A.; Mattei, P.; Kramer, R.E.; Kader, H.A. Diagnosis and treatment of perianal Crohn disease: NASPGHAN clinical report and consensus statement. J. Pediatr. Gastroenterol. Nutr. 2013, 57, 401–412.
- Radwan, P.; Radwan, K. Nieswoiste choroby zapalne jelit u osób w podeszłym wieku. Gastroenterol. Klin. 2016, 8, 27–35.
- Haac, B.; Palmateer, N.; Seaton, M.; VanYPeren, R.; Fraser, C.; Bafford, A. A Distinct Gut Microbiota Exists Within Crohn’s Disease–Related Perianal Fistulae. Gastrointestinal 2019, 242, 118–128.
- Fiocchi, C. Inflammatory bowel disease: Etiology and pathogenesis. Gastroenterology 1998, 115, 182–205.
- Chreptowicz, A. Rośnie liczba osób z rozpoznaniem mikroskopowego zapalenia jelit—Co nowego w leczeniu? Gastroenterol. Klin. 2016, 8, 107–112.
- Soubieres, A.A.; Poullis, A. Emerging role of novel biomarkers in the diagnosis of inflammatory bowel disease. World J. Gastrointest. Pharmacol. Ther. 2016, 7, 41–50.
- Yan, J.B.; Luo, M.M.; Chen, Z.Y.; He, B.H. The Function and Role of the Th17 (T helper 17)/Treg (T regulatory) Cell Balance in Inflammatory Bowel Disease. J. Immunol. Res. 2020, 2020, 8813558.
- Van Herck, M.A.; Weyler, J.; Kwanten, W.J.; Dirinck, E.L.; De Winter, B.Y.; Francque, S.M.; Vonghia, L. The Differential Roles of T Cells in Non-alcoholic Fatty Liver Disease and Obesity. Front. Immunol. 2019, 10, 82.
- De la Fuente, M.; MacDonald, T.T.; Hermoso, M.A. Intestinal Homeostasis and Disease: A Complex Partnership Between Immune Cells, Non-Immune Cells, and the Microbiome. Front. Immunol. 2019, 10, 2775.
- Garcia-Hernandez, V.; Quiros, M.; Nusrat, A. Intestinal epithelial claudins: Expression and regulation in homeostasis and inflammation. Ann. N. Y. Acad. Sci. 2017, 1397, 66–79.
- Hu, C.A.; Hou, Y.; Yi, D.; Qiu, Y.; Wu, G.; Kong, X.; Yin, Y. Autophagy and tight junction proteins in the intestine and intestinal diseases. Anim. Nutr. 2015, 1, 123–127.
- Yamamoto-Furusho, J.K.; Mendivil, E.J.; Fonseca-Camarillo, G. Differential expression of occludin in patients with ulcerative colitis and healthy controls. Inflamm. Bowel Dis. 2012, 18, E1999.
- Van Itallie, C.M.; Tietgens, A.J.; Anderson, J.M. Visualizing the dynamic coupling of claudin strands to the actin cytoskeleton through ZO-1. Mol. Biol. Cell 2017, 28, 524–534.
- Sugita, K.; Kabashima, K. Tight junctions in the development of asthma, chronic rhinosinusitis, atopic dermatitis, eosinophilic esophagitis, and inflammatory bowel diseases. J. Leukoc. Biol. 2020, 107, 749–762.
- Østvik, A.E.; Svendsen, T.D.; Granlund, A.V.B.; Doseth, B.; Skovdahl, H.K.; Bakke, I.; Thorsvik, S.; Afroz, W.; Walaas, G.A.; Mollnes, T.E.; et al. Intestinal epithelial cells express immunomodulatory ISG15 during active ulcerative colitis and Crohn’s disease. J. Crohns Colitis 2020, 14, 920–934.
- Danilova, N.A.; Abdulkhakov, S.R.; Grigoryeva, T.V.; Markelova, M.I.; Vasilyev, I.Y.; Boulygina, E.A.; Ardatskaya, M.D.; Pavlenko, A.V.; Tyakht, A.V.; Odintsova, A.K.; et al. Markers of dysbiosis in patients with ulcerative colitis and Crohn’s disease. Ter. Arkh. 2019, 91, 17–24.
- Glassner, K.L.; Abraham, B.P.; Quigley, E.M.M. The microbiome and inflammatory bowel disease. J. Allergy Clin. Immunol. 2020, 145, 16–27.
- Zuo, T.; Ng, S.C. The Gut Microbiota in the Pathogenesis and Therapeutics of Inflammatory Bowel Disease. Front. Microbiol. 2018, 9, 2247.
- Chu, Y.; Jiang, M.Z.; Xu, B.; Wang, W.J.; Chen, D.; Li, X.W.; Zhang, Y.J.; Liang, J. Specific changes of enteric mycobiota and virome in inflammatory bowel disease. J. Dig. Dis. 2018, 19, 2–7.
- Gonciarz, M.; Szkudłapski, D.; Mularczyk, A.; Radwan, P.; Kłopocka, M.; Bartnik, W.; Rydzewska, G. Wytyczne postępowania z chorymi na nieswoiste choroby zapalne jelit w praktyce lekarza rodzinnego. Lekarz POZ 2017, 3, 1–11.
- Nowakowski, J.; Chrobak, A.; Dudek, D. Psychiatric illnesses in inflammatory bowel diseases—psychiatric comorbidity and biological underpinnings. Psychiatr. Pol. 2016, 50, 1157–1166.
- Man, S.; Kaakoush, N.; Mitchell, H. The role of bacteria and pattern-recognition receptors in Crohn’s disease. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 152–168.
- Bartnik, W. Wytyczne postępowania w nieswoistych chorobach zapalnych jelit. Przegląd Gastroenterol. 2007, 2, 215–229.
- Eder, P. Przydatność biomarkerów w ocenie aktywności nieswoistych chorób zapalnych jelit -wskazówki praktyczne. Varia Med. 2018, 2, 371–380.
- Arai, T.; Takeuchi, K.; Miyamura, M.; Ishikawa, R.; Yamada, A.; Katsumata, M.; Igarashi, Y.; Suzuki, Y. Level of Fecal Calprotectin Correlates With Severity of Small Bowel Crohn’s Disease, Measured by Balloon-assisted Enteroscopy and Computed Tomography Enterography. Clin. Gastroenterol. Hepatol. 2017, 15, 56–62.
- Molander, P.; Färkkilä, M.; Ristimäki, A.; Salminen, K.; Kemppainen, H.; Blomster, T.; Koskela, R.; Jussila, A.; Rautiainen, H.; Nissinen, M.; et al. Does fecal calprotectin predict short-term relapse after stopping TNFα-blocking agents in inflammatory bowel disease patients in deep remission? J. Crohn’s. Colitis 2015, 9, 33–40.
- Hao, L.; Shan, Q.; Wei, J.; Ma, F.; Sun, P. Lactoferrin: Major Physiological Functions and Applications. Curr. Protein Pept. Sci. 2019, 20, 139–144.
- Niaz, B.; Saeed, F.; Ahmed, A.; Imran, M.; Maan, A.A.; Khan, M.K.I.; Tufail, T.; Anjum, F.M.; Hussain, S.; Suleria, H.A.R. Lactoferrin (LF): A natural antimicrobial protein. Int. J. Food Prop. 2019, 22, 1626–1641.
- Pan, S.; Weng, H.; Hu, G.; Wang, S.; Zhao, T.; Yao, X.; Liao, L.; Zhu, X.; Ge, Y. Lactoferrin may inhibit the development of cancer via its immunostimulatory and immunomodulatory activities (Review). Int. J. Oncol. 2021, 59, 85.
- Boyapati, R.K.; Torres, J.; Palmela, C.; Parker, C.E.; Silverberg, O.M.; Upadhyaya, S.D.; Nguyen, T.M.; Colombel, J.F. Withdrawal of immunosuppressant or biologic therapy for patients with quiescent Crohn’s disease. Cochrane Database Syst. Rev. 2018, 5, CD012540.
More
Information
Subjects:
Gastroenterology & Hepatology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.2K
Revisions:
2 times
(View History)
Update Date:
27 Mar 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No