
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Eric TOUSSIROT | -- | 2515 | 2023-03-14 11:26:52 | | | |
| 2 | Peter Tang | Meta information modification | 2515 | 2023-03-15 02:22:48 | | |
Video Upload Options
Inflammatory rheumatic diseases (IRDs) are complex immune-mediated diseases that are characterized by chronic inflammation of the joints. Rheumatoid arthritis (RA) and spondyloarthritis (SpA), including axial SpA (ax SpA) and psoriatic arthritis (PsA), are the most common forms of IRD. Both RA and ax SpA are characterized by a chronic course with progressive structural modifications, namely, cartilage damage and bone erosions in RA and osteoproliferative changes with spinal ossifications in ax SpA. The adipose tissue is involved in the pathophysiology of IRDs via the release of several proteins, namely, adipokines. Several adipokines with pro-inflammatory effects have been identified, such as leptin, adiponectin, visfatin and resistin.
1. Adipokines: Main Biological and Immunological Properties
1.1. Leptin
1.2. Adiponectin
1.3. Resistin
1.4. Visfatin
2. Adipokines in IRD and Their Effects on Joint Components
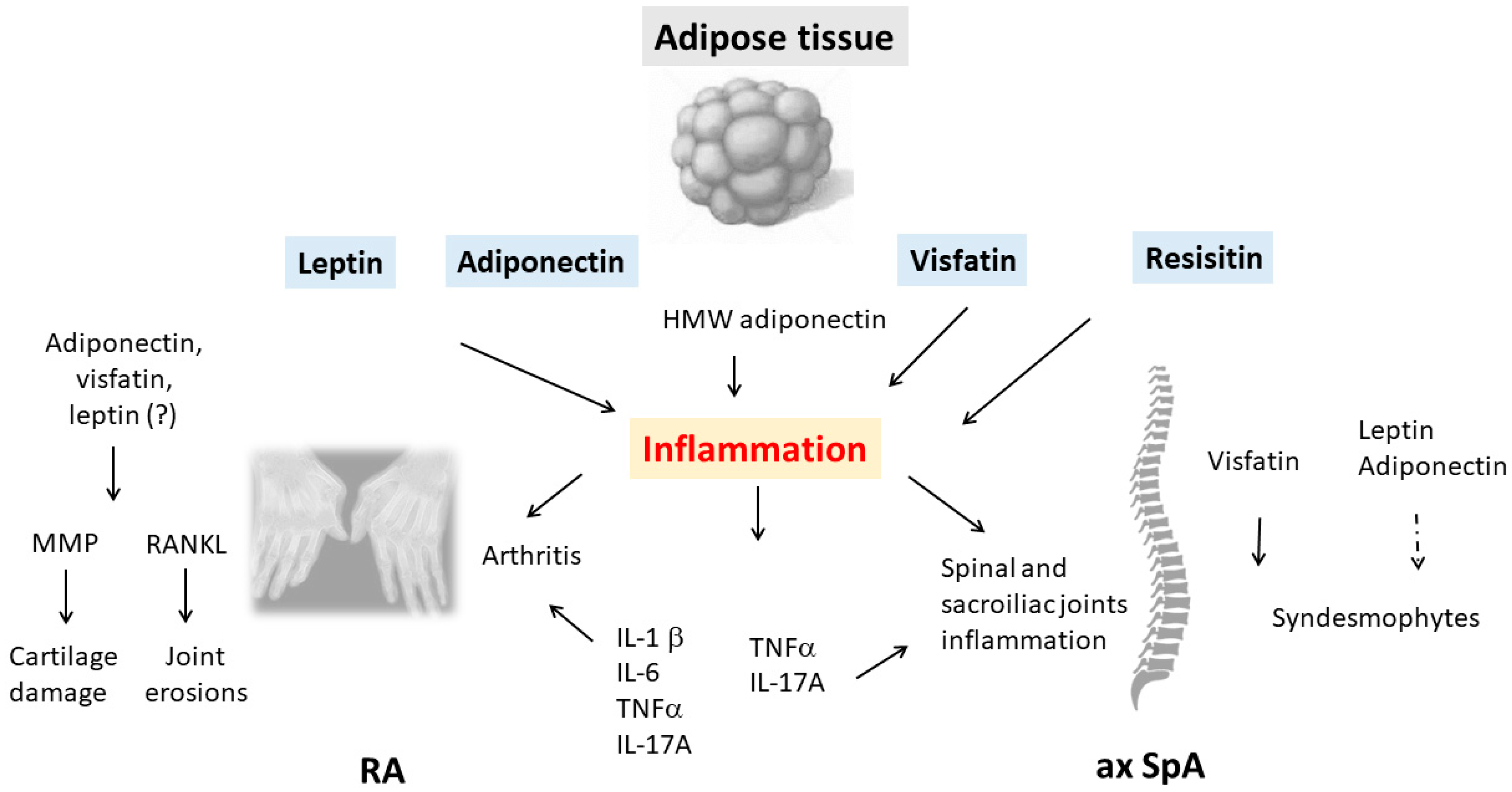
2.1. Rheumatoid Arthritis
2.2. Axial Spondyloarthritis
2.3. Psoriatic Arthritis
References
- Fantuzzi, G. Adipose tissue, adipokines, and inflammation. J. Allergy Clin. Immunol. 2005, 115, 911–919.
- Neumann, E.; Frommer, K.W.; Vasile, M.; Muller-Ladner, U. Adipocytokines as driving forces in rheumatoid arthritis and related inflammatory diseases? Arthritis. Rheum. 2011, 63, 1159–1169.
- Laurindo, L.F.; de Maio, M.C.; Barbalho, S.M.; Guiguer, E.L.; Araujo, A.C.; de Alvares Goulart, R.; Flato, U.A.P.; Junior, E.B.; Detregiachi, C.R.P.; Dos Santos Haber, J.F.; et al. Organokines in Rheumatoid Arthritis: A Critical Review. Int. J. Mol. Sci. 2022, 23, 6193.
- Toussirot, E.; Streit, G.; Wendling, D. The contribution of adipose tissue and adipokines to inflammation in joint diseases. Curr. Med. Chem. 2007, 14, 1095–1100.
- Neumann, E.; Hasseli, R.; Ohl, S.; Lange, U.; Frommer, K.W.; Muller-Ladner, U. Adipokines and Autoimmunity in Inflammatory Arthritis. Cells 2021, 10, 216.
- Neumann, E.; Junker, S.; Schett, G.; Frommer, K.; Muller-Ladner, U. Adipokines in bone disease. Nat. Rev. Rheumatol. 2016, 12, 296–302.
- Toussirot, E. Mini-Review: The Contribution of Adipokines to Joint Inflammation in Inflammatory Rheumatic Diseases. Front. Endocrinol. 2020, 11, 606560.
- Houseknecht, K.L.; Baile, C.A.; Matteri, R.L.; Spurlock, M.E. The biology of leptin: A review. J. Anim. Sci. 1998, 76, 1405–1420.
- Otero, M.; Lago, R.; Lago, F.; Casanueva, F.F.; Dieguez, C.; Gomez-Reino, J.J.; Gualillo, O. Leptin, from fat to inflammation: Old questions and new insights. FEBS Lett. 2005, 579, 295–301.
- Iikuni, N.; Lam, Q.L.; Lu, L.; Matarese, G.; La Cava, A. Leptin and Inflammation. Curr. Immunol. Rev. 2008, 4, 70–79.
- Deng, J.; Liu, Y.; Yang, M.; Wang, S.; Zhang, M.; Wang, X.; Ko, K.H.; Hua, Z.; Sun, L.; Cao, X.; et al. Leptin exacerbates collagen-induced arthritis via enhancement of Th17 cell response. Arthritis Rheum. 2012, 64, 3564–3573.
- Carrion, M.; Frommer, K.W.; Perez-Garcia, S.; Muller-Ladner, U.; Gomariz, R.P.; Neumann, E. The Adipokine Network in Rheumatic Joint Diseases. Int. J. Mol. Sci. 2019, 20, 4091.
- Ouchi, N.; Walsh, K. Adiponectin as an anti-inflammatory factor. Clin. Chim. Acta 2007, 380, 24–30.
- Okamoto, Y.; Kihara, S.; Funahashi, T.; Matsuzawa, Y.; Libby, P. Adiponectin: A key adipocytokine in metabolic syndrome. Clin. Sci. 2006, 110, 267–278.
- Fatel, E.C.S.; Rosa, F.T.; Simao, A.N.C.; Dichi, I. Adipokines in rheumatoid arthritis. Adv. Rheumatol. 2018, 58, 25.
- Wang, Y.; Liu, R.; Zhao, P.; Zhang, Q.; Huang, Y.; Wang, L.; Lv, C.; Che, N.; Tan, W.; Zhang, M. Blockade of adiponectin receptor 1 signaling inhibits synovial inflammation and alleviates joint damage in collagen-induced arthritis. Clin. Rheumatol. 2022, 41, 255–264.
- Song, H.; Chan, J.; Rovin, B.H. Induction of chemokine expression by adiponectin in vitro is isoform dependent. Transl. Res. 2009, 154, 18–26.
- Bungau, S.; Behl, T.; Tit, D.M.; Banica, F.; Bratu, O.G.; Diaconu, C.C.; Nistor-Cseppento, C.D.; Bustea, C.; Aron, R.A.C.; Vesa, C.M. Interactions between leptin and insulin resistance in patients with prediabetes, with and without NAFLD. Exp. Ther. Med. 2020, 20, 197.
- Carmen Zaha, D.; Vesa, C.; Uivarosan, D.; Bratu, O.; Fratila, O.; Mirela Tit, D.; Pantis, C.; Diaconu, C.; Bungau, S. Influence of inflammation and adipocyte biochemical markers on the components of metabolic syndrome. Exp. Ther. Med. 2020, 20, 121–128.
- Krysiak, R.; Handzlik-Orlik, G.; Okopien, B. The role of adipokines in connective tissue diseases. Eur. J. Nutr. 2012, 51, 513–528.
- Norata, G.D.; Ongari, M.; Garlaschelli, K.; Raselli, S.; Grigore, L.; Catapano, A.L. Plasma resistin levels correlate with determinants of the metabolic syndrome. Eur. J. Endocrinol. 2007, 156, 279–284.
- Nagaev, I.; Bokarewa, M.; Tarkowski, A.; Smith, U. Human resistin is a systemic immune-derived proinflammatory cytokine targeting both leukocytes and adipocytes. PLoS ONE 2006, 1, e31.
- Bokarewa, M.; Nagaev, I.; Dahlberg, L.; Smith, U.; Tarkowski, A. Resistin, an adipokine with potent proinflammatory properties. J. Immunol. 2005, 174, 5789–5795.
- Meier, F.M.; Frommer, K.W.; Peters, M.A.; Brentano, F.; Lefevre, S.; Schroder, D.; Kyburz, D.; Steinmeyer, J.; Rehart, S.; Gay, S.; et al. Visfatin/pre-B-cell colony-enhancing factor (PBEF), a proinflammatory and cell motility-changing factor in rheumatoid arthritis. J. Biol. Chem. 2012, 287, 28378–28385.
- Francisco, V.; Pino, J.; Gonzalez-Gay, M.A.; Mera, A.; Lago, F.; Gomez, R.; Mobasheri, A.; Gualillo, O. Adipokines and inflammation: Is it a question of weight? Br. J. Pharmacol. 2018, 175, 1569–1579.
- Toussirot, E.; Michel, F.; Binda, D.; Dumoulin, G. The role of leptin in the pathophysiology of rheumatoid arthritis. Life Sci. 2015, 140, 29–36.
- Otero, M.; Lago, R.; Gomez, R.; Lago, F.; Dieguez, C.; Gomez-Reino, J.J.; Gualillo, O. Changes in plasma levels of fat-derived hormones adiponectin, leptin, resistin and visfatin in patients with rheumatoid arthritis. Ann. Rheum. Dis. 2006, 65, 1198–1201.
- Harle, P.; Pongratz, G.; Weidler, C.; Buttner, R.; Scholmerich, J.; Straub, R.H. Possible role of leptin in hypoandrogenicity in patients with systemic lupus erythematosus and rheumatoid arthritis. Ann. Rheum. Dis. 2004, 63, 809–816.
- Toussirot, E.; Nguyen, N.U.; Dumoulin, G.; Aubin, F.; Cedoz, J.P.; Wendling, D. Relationship between growth hormone-IGF-I-IGFBP-3 axis and serum leptin levels with bone mass and body composition in patients with rheumatoid arthritis. Rheumatology 2005, 44, 120–125.
- Tian, G.; Liang, J.N.; Pan, H.F.; Zhou, D. Increased leptin levels in patients with rheumatoid arthritis: A meta-analysis. Ir. J. Med. Sci. 2014, 183, 659–666.
- Lee, Y.H.; Bae, S.C. Circulating leptin level in rheumatoid arthritis and its correlation with disease activity: A meta-analysis. Z. Rheumatol. 2016, 75, 1021–1027.
- Anders, H.J.; Rihl, M.; Heufelder, A.; Loch, O.; Schattenkirchner, M. Leptin serum levels are not correlated with disease activity in patients with rheumatoid arthritis. Metabolism 1999, 48, 745–748.
- Muraoka, S.; Kusunoki, N.; Takahashi, H.; Tsuchiya, K.; Kawai, S. Leptin stimulates interleukin-6 production via janus kinase 2/signal transducer and activator of transcription 3 in rheumatoid synovial fibroblasts. Clin. Exp. Rheumatol. 2013, 31, 589–595.
- Rodriguez, J.; Lafaurie, G.I.; Bautista-Molano, W.; Chila-Moreno, L.; Bello-Gualtero, J.M.; Romero-Sanchez, C. Adipokines and periodontal markers as risk indicators of early rheumatoid arthritis: A cross-sectional study. Clin. Oral. Investig. 2021, 25, 1685–1695.
- Ramos-Casallas, A.; De Avila, J.; Chaparro-Sanabria, A.; Chalem-Choueka, P.; Bello-Gualtero, J.M.; Chila-Moreno, L.; Pacheco-Tena, C.; Bautista-Molano, W.; Romero-Sanchez, C. Adipokine profile on joint and periodontal conditions in first-degree relatives of patients with rheumatoid arthritis. Curr. Rheumatol. Rev. 2022.
- Gomez, R.; Scotece, M.; Conde, J.; Gomez-Reino, J.J.; Lago, F.; Gualillo, O. Adiponectin and leptin increase IL-8 production in human chondrocytes. Ann. Rheum. Dis. 2011, 70, 2052–2054.
- Yaykasli, K.O.; Hatipoglu, O.F.; Yaykasli, E.; Yildirim, K.; Kaya, E.; Ozsahin, M.; Uslu, M.; Gunduz, E. Leptin induces ADAMTS-4, ADAMTS-5, and ADAMTS-9 genes expression by mitogen-activated protein kinases and NF-kB signaling pathways in human chondrocytes. Cell. Biol. Int. 2015, 39, 104–112.
- Yang, W.H.; Tsai, C.H.; Fong, Y.C.; Huang, Y.L.; Wang, S.J.; Chang, Y.S.; Tang, C.H. Leptin induces oncostatin M production in osteoblasts by downregulating miR-93 through the Akt signaling pathway. Int. J. Mol. Sci. 2014, 15, 15778–15790.
- Liu, G.Y.; Liang, Q.H.; Cui, R.R.; Liu, Y.; Wu, S.S.; Shan, P.F.; Yuan, L.Q.; Liao, E.Y. Leptin promotes the osteoblastic differentiation of vascular smooth muscle cells from female mice by increasing RANKL expression. Endocrinology 2014, 155, 558–567.
- Cirmanova, V.; Bayer, M.; Starka, L.; Zajickova, K. The effect of leptin on bone: An evolving concept of action. Physiol. Res. 2008, 57, S143–S151.
- Toussirot, E.; Grandclement, E.; Gaugler, B.; Michel, F.; Wendling, D.; Saas, P.; Dumoulin, G.; Cbt. Serum adipokines and adipose tissue distribution in rheumatoid arthritis and ankylosing spondylitis. A comparative study. Front. Immunol. 2013, 4, 453.
- Rho, Y.H.; Solus, J.; Sokka, T.; Oeser, A.; Chung, C.P.; Gebretsadik, T.; Shintani, A.; Pincus, T.; Stein, C.M. Adipocytokines are associated with radiographic joint damage in rheumatoid arthritis. Arthritis Rheum. 2009, 60, 1906–1914.
- Giles, J.T.; Allison, M.; Bingham, C.O., 3rd; Scott, W.M., Jr.; Bathon, J.M. Adiponectin is a mediator of the inverse association of adiposity with radiographic damage in rheumatoid arthritis. Arthritis Rheum. 2009, 61, 1248–1256.
- Giles, J.T.; van der Heijde, D.M.; Bathon, J.M. Association of circulating adiponectin levels with progression of radiographic joint destruction in rheumatoid arthritis. Ann. Rheum. Dis. 2011, 70, 1562–1568.
- Klein-Wieringa, I.R.; van der Linden, M.P.; Knevel, R.; Kwekkeboom, J.C.; van Beelen, E.; Huizinga, T.W.; van der Helm-van Mil, A.; Kloppenburg, M.; Toes, R.E.; Ioan-Facsinay, A. Baseline serum adipokine levels predict radiographic progression in early rheumatoid arthritis. Arthritis Rheum. 2011, 63, 2567–2574.
- Senolt, L.; Pavelka, K.; Housa, D.; Haluzik, M. Increased adiponectin is negatively linked to the local inflammatory process in patients with rheumatoid arthritis. Cytokine 2006, 35, 247–252.
- Ebina, K.; Fukuhara, A.; Ando, W.; Hirao, M.; Koga, T.; Oshima, K.; Matsuda, M.; Maeda, K.; Nakamura, T.; Ochi, T.; et al. Serum adiponectin concentrations correlate with severity of rheumatoid arthritis evaluated by extent of joint destruction. Clin. Rheumatol. 2009, 28, 445–451.
- Ozgen, M.; Koca, S.S.; Dagli, N.; Balin, M.; Ustundag, B.; Isik, A. Serum adiponectin and vaspin levels in rheumatoid arthritis. Arch. Med. Res. 2010, 41, 457–463.
- Tan, W.; Wang, F.; Zhang, M.; Guo, D.; Zhang, Q.; He, S. High adiponectin and adiponectin receptor 1 expression in synovial fluids and synovial tissues of patients with rheumatoid arthritis. Semin. Arthritis Rheum. 2009, 38, 420–427.
- Chen, X.; Lu, J.; Bao, J.; Guo, J.; Shi, J.; Wang, Y. Adiponectin: A biomarker for rheumatoid arthritis? Cytokine Growth Factor Rev. 2013, 24, 83–89.
- Szumilas, K.; Szumilas, P.; Sluczanowska-Glabowska, S.; Zgutka, K.; Pawlik, A. Role of Adiponectin in the Pathogenesis of Rheumatoid Arthritis. Int. J. Mol. Sci. 2020, 21, 8265.
- Neumeier, M.; Weigert, J.; Schaffler, A.; Wehrwein, G.; Muller-Ladner, U.; Scholmerich, J.; Wrede, C.; Buechler, C. Different effects of adiponectin isoforms in human monocytic cells. J. Leukoc. Biol. 2006, 79, 803–808.
- Zhang, Q.; Wang, L.; Jiang, J.; Lin, S.; Luo, A.; Zhao, P.; Tan, W.; Zhang, M. Critical Role of AdipoR1 in Regulating Th17 Cell Differentiation Through Modulation of HIF-1alpha-Dependent Glycolysis. Front. Immunol. 2020, 11, 2040.
- Sun, X.; Feng, X.; Tan, W.; Lin, N.; Hua, M.; Wei, Y.; Wang, F.; Li, N.; Zhang, M. Adiponectin exacerbates collagen-induced arthritis via enhancing Th17 response and prompting RANKL expression. Sci. Rep. 2015, 5, 11296.
- Qian, J.; Xu, L.; Sun, X.; Wang, Y.; Xuan, W.; Zhang, Q.; Zhao, P.; Wu, Q.; Liu, R.; Che, N.; et al. Adiponectin aggravates bone erosion by promoting osteopontin production in synovial tissue of rheumatoid arthritis. Arthritis Res. Ther. 2018, 20, 26.
- Lee, Y.H.; Bae, S.C. Circulating adiponectin and visfatin levels in rheumatoid arthritis and their correlation with disease activity: A meta-analysis. Int. J. Rheum. Dis. 2018, 21, 664–672.
- Moschen, A.R.; Geiger, S.; Gerner, R.; Tilg, H. Pre-B cell colony enhancing factor/NAMPT/visfatin and its role in inflammation-related bone disease. Mutat. Res. 2010, 690, 95–101.
- Venkateshaiah, S.U.; Khan, S.; Ling, W.; Bam, R.; Li, X.; van Rhee, F.; Usmani, S.; Barlogie, B.; Epstein, J.; Yaccoby, S. NAMPT/PBEF1 enzymatic activity is indispensable for myeloma cell growth and osteoclast activity. Exp. Hematol. 2013, 41, 547–557.e2.
- Hasseli, R.; Frommer, K.W.; Schwarz, M.; Hulser, M.L.; Schreiyack, C.; Arnold, M.; Diller, M.; Tarner, I.H.; Lange, U.; Pons-Kuhnemann, J.; et al. Adipokines and Inflammation Alter the Interaction Between Rheumatoid Arthritis Synovial Fibroblasts and Endothelial Cells. Front. Immunol. 2020, 11, 925.
- Li, X.; Islam, S.; Xiong, M.; Nsumu, N.N.; Lee, M.W., Jr.; Zhang, L.Q.; Ueki, Y.; Heruth, D.P.; Lei, G.; Ye, S.Q. Epigenetic regulation of NfatC1 transcription and osteoclastogenesis by nicotinamide phosphoribosyl transferase in the pathogenesis of arthritis. Cell Death Discov. 2019, 5, 62.
- Schaffler, A.; Ehling, A.; Neumann, E.; Herfarth, H.; Tarner, I.; Scholmerich, J.; Muller-Ladner, U.; Gay, S. Adipocytokines in synovial fluid. JAMA 2003, 290, 1709–1710.
- Huang, Q.; Tao, S.S.; Zhang, Y.J.; Zhang, C.; Li, L.J.; Zhao, W.; Zhao, M.Q.; Li, P.; Pan, H.F.; Mao, C.; et al. Serum resistin levels in patients with rheumatoid arthritis and systemic lupus erythematosus: A meta-analysis. Clin. Rheumatol. 2015, 34, 1713–1720.
- Senolt, L.; Housa, D.; Vernerova, Z.; Jirasek, T.; Svobodova, R.; Veigl, D.; Anderlova, K.; Muller-Ladner, U.; Pavelka, K.; Haluzik, M. Resistin in rheumatoid arthritis synovial tissue, synovial fluid and serum. Ann. Rheum. Dis. 2007, 66, 458–463.
- Yoshino, T.; Kusunoki, N.; Tanaka, N.; Kaneko, K.; Kusunoki, Y.; Endo, H.; Hasunuma, T.; Kawai, S. Elevated serum levels of resistin, leptin, and adiponectin are associated with C-reactive protein and also other clinical conditions in rheumatoid arthritis. Intern. Med. 2011, 50, 269–275.
- Sato, H.; Muraoka, S.; Kusunoki, N.; Masuoka, S.; Yamada, S.; Ogasawara, H.; Imai, T.; Akasaka, Y.; Tochigi, N.; Takahashi, H.; et al. Resistin upregulates chemokine production by fibroblast-like synoviocytes from patients with rheumatoid arthritis. Arthritis Res. Ther. 2017, 19, 263.
- Sari, I.; Demir, T.; Kozaci, L.D.; Akar, S.; Kavak, T.; Birlik, M.; Onen, F.; Akkoc, N. Body composition, insulin, and leptin levels in patients with ankylosing spondylitis. Clin. Rheumatol. 2007, 26, 1427–1432.
- Toussirot, E.; Streit, G.; Nguyen, N.U.; Dumoulin, G.; Le Huede, G.; Saas, P.; Wendling, D. Adipose tissue, serum adipokines, and ghrelin in patients with ankylosing spondylitis. Metabolism 2007, 56, 1383–1389.
- Park, M.C.; Chung, S.J.; Park, Y.B.; Lee, S.K. Pro-inflammatory effect of leptin on peripheral blood mononuclear cells of patients with ankylosing spondylitis. Jt. Bone Spine 2009, 76, 170–175.
- Park, M.C.; Lee, S.W.; Choi, S.T.; Park, Y.B.; Lee, S.K. Serum leptin levels correlate with interleukin-6 levels and disease activity in patients with ankylosing spondylitis. Scand. J. Rheumatol. 2007, 36, 101–106.
- Yang, J.; Zhang, X.; Ma, Y.; Wu, M.; Hu, X.; Han, R.; Yuan, Y.; Wang, M.; Chen, M.; Jiang, S.; et al. Serum levels of leptin, adiponectin and resistin in patients with ankylosing spondylitis: A systematic review and meta-analysis. Int. Immunopharmacol. 2017, 52, 310–317.
- Mei, Y.J.; Wang, P.; Chen, L.J.; Li, Z.J. Plasma/Serum Leptin Levels in Patients with Ankylosing Spondylitis: A Systematic Review and Meta-analysis. Arch. Med. Res. 2016, 47, 111–117.
- Xie, H.; Tang, S.Y.; Luo, X.H.; Huang, J.; Cui, R.R.; Yuan, L.Q.; Zhou, H.D.; Wu, X.P.; Liao, E.Y. Insulin-like effects of visfatin on human osteoblasts. Calcif. Tissue Int. 2007, 80, 201–210.
- Laiguillon, M.C.; Houard, X.; Bougault, C.; Gosset, M.; Nourissat, G.; Sautet, A.; Jacques, C.; Berenbaum, F.; Sellam, J. Expression and function of visfatin (Nampt), an adipokine-enzyme involved in inflammatory pathways of osteoarthritis. Arthritis Res. Ther. 2014, 16, R38.
- Karsenty, G.; Khosla, S. The crosstalk between bone remodeling and energy metabolism: A translational perspective. Cell. Metab. 2022, 34, 805–817.
- Chen, X.X.; Yang, T. Roles of leptin in bone metabolism and bone diseases. J. Bone. Miner. Metab. 2015, 33, 474–485.
- Ducy, P.; Amling, M.; Takeda, S.; Priemel, M.; Schilling, A.F.; Beil, F.T.; Shen, J.; Vinson, C.; Rueger, J.M.; Karsenty, G. Leptin inhibits bone formation through a hypothalamic relay: A central control of bone mass. Cell 2000, 100, 197–207.
- Reseland, J.E.; Syversen, U.; Bakke, I.; Qvigstad, G.; Eide, L.G.; Hjertner, O.; Gordeladze, J.O.; Drevon, C.A. Leptin is expressed in and secreted from primary cultures of human osteoblasts and promotes bone mineralization. J. Bone Miner. Res. 2001, 16, 1426–1433.
- Holloway, W.R.; Collier, F.M.; Aitken, C.J.; Myers, D.E.; Hodge, J.M.; Malakellis, M.; Gough, T.J.; Collier, G.R.; Nicholson, G.C. Leptin inhibits osteoclast generation. J. Bone Miner. Res. 2002, 17, 200–209.
- Luo, X.H.; Guo, L.J.; Yuan, L.Q.; Xie, H.; Zhou, H.D.; Wu, X.P.; Liao, E.Y. Adiponectin stimulates human osteoblasts proliferation and differentiation via the MAPK signaling pathway. Exp. Cell. Res. 2005, 309, 99–109.
- Luo, X.H.; Guo, L.J.; Xie, H.; Yuan, L.Q.; Wu, X.P.; Zhou, H.D.; Liao, E.Y. Adiponectin stimulates RANKL and inhibits OPG expression in human osteoblasts through the MAPK signaling pathway. J. Bone Miner. Res. 2006, 21, 1648–1656.
- Thommesen, L.; Stunes, A.K.; Monjo, M.; Grosvik, K.; Tamburstuen, M.V.; Kjobli, E.; Lyngstadaas, S.P.; Reseland, J.E.; Syversen, U. Expression and regulation of resistin in osteoblasts and osteoclasts indicate a role in bone metabolism. J. Cell. Biochem. 2006, 99, 824–834.
- Russolillo, A.; Iervolino, S.; Peluso, R.; Lupoli, R.; Di Minno, A.; Pappone, N.; Di Minno, M.N. Obesity and psoriatic arthritis: From pathogenesis to clinical outcome and management. Rheumatology 2013, 52, 62–67.
- Toussirot, E.; Aubin, F.; Desmarets, M.; Wendling, D.; Auge, B.; Gillard, J.; Messica, O.; Guillot, X.; Laheurte, C.; Monnet, E.; et al. Visceral adiposity in patients with psoriatic arthritis and psoriasis alone and its relationship with metabolic and cardiovascular risk. Rheumatology 2021, 60, 2816–2825.
- Xue, Y.; Jiang, L.; Cheng, Q.; Chen, H.; Yu, Y.; Lin, Y.; Yang, X.; Kong, N.; Zhu, X.; Xu, X.; et al. Adipokines in psoriatic arthritis patients: The correlations with osteoclast precursors and bone erosions. PLoS ONE 2012, 7, e46740.
- Eder, L.; Jayakar, J.; Pollock, R.; Pellett, F.; Thavaneswaran, A.; Chandran, V.; Rosen, C.F.; Gladman, D.D. Serum adipokines in patients with psoriatic arthritis and psoriasis alone and their correlation with disease activity. Ann. Rheum. Dis. 2013, 72, 1956–1961.
- Dikbas, O.; Tosun, M.; Bes, C.; Tonuk, S.B.; Aksehirli, O.Y.; Soy, M. Serum levels of visfatin, resistin and adiponectin in patients with psoriatic arthritis and associations with disease severity. Int. J. Rheum. Dis. 2016, 19, 672–677.





