Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | giandomenico roviello | -- | 2821 | 2023-03-09 10:42:00 | | | |
| 2 | Rita Xu | -1 word(s) | 2820 | 2023-03-09 10:50:32 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Lavacchi, D.; Fancelli, S.; Buttitta, E.; Vannini, G.; Guidolin, A.; Winchler, C.; Caliman, E.; Vannini, A.; Giommoni, E.; Brugia, M.; et al. Perioperative Tailored Treatments for Gastric Cancer. Encyclopedia. Available online: https://encyclopedia.pub/entry/42012 (accessed on 07 February 2026).
Lavacchi D, Fancelli S, Buttitta E, Vannini G, Guidolin A, Winchler C, et al. Perioperative Tailored Treatments for Gastric Cancer. Encyclopedia. Available at: https://encyclopedia.pub/entry/42012. Accessed February 07, 2026.
Lavacchi, Daniele, Sara Fancelli, Eleonora Buttitta, Gianmarco Vannini, Alessia Guidolin, Costanza Winchler, Enrico Caliman, Agnese Vannini, Elisa Giommoni, Marco Brugia, et al. "Perioperative Tailored Treatments for Gastric Cancer" Encyclopedia, https://encyclopedia.pub/entry/42012 (accessed February 07, 2026).
Lavacchi, D., Fancelli, S., Buttitta, E., Vannini, G., Guidolin, A., Winchler, C., Caliman, E., Vannini, A., Giommoni, E., Brugia, M., Cianchi, F., Pillozzi, S., Roviello, G., & Antonuzzo, L. (2023, March 09). Perioperative Tailored Treatments for Gastric Cancer. In Encyclopedia. https://encyclopedia.pub/entry/42012
Lavacchi, Daniele, et al. "Perioperative Tailored Treatments for Gastric Cancer." Encyclopedia. Web. 09 March, 2023.
Copy Citation
Resectable gastric or gastroesophageal (G/GEJ) cancer is a heterogeneous disease with no defined molecularly based treatment strategy. The half of patients experience disease recurrence despite standard treatments (neoadjuvant and/or adjuvant chemotherapy/chemoradiotherapy and surgery).
perioperative GC
HER2
MSI-H
1. Introduction
Gastric cancer (GC) is the fifth most common cancer and the third cause of cancer-related mortality worldwide [1][2]. When diagnosis occurs in metastatic disease, the prognosis is poor and radical surgery is not routinely recommended [3].
In the setting of resectable disease, multimodal treatments, including perioperative chemotherapy, showed a significant survival improvement compared with surgery alone. At first, the Medical Research Council Adjuvant Gastric Infusional Chemotherapy (MAGIC) and FNCLCC/FFCD trials demonstrated the benefit of perioperative chemotherapy in survival and in recurrence-rate reduction. Patients with resectable gastric or gastro-esophageal junction (G/GEJ) adenocarcinoma were assigned to either surgery alone or perioperative chemotherapy (i.e., epirubicin, cisplatin and fluorouracil [ECF] in the MAGIC trial and cisplatin and fluorouracil in the FNCLCC/FFCD trial). Five-year survival rates were higher in the perioperative-chemotherapy group compared to the surgery group (36% vs. 23% in MAGIC, 38% vs. 24% in FNCLCC/FFCD trial) [4][5].
Although docetaxel showed a remarkable efficacy in first- and further-line treatments for metastatic G/GEJ adenocarcinoma, both alone and in combination with cisplatin and fluorouracil [6][7], toxicities associated with combination regimens were so relevant to suggest replacement of cisplatin with oxaliplatin. This new combination, which consisted of perioperative fluorouracil, leucovorin, oxaliplatin and docetaxel (FLOT), was evaluated in the phase III FLOT4-AIO trial that demonstrated the efficacy of FLOT as a perioperative therapy over ECF in G/GEJ adenocarcinoma. The median overall survival (OS) was 50 months in the FLOT arm vs. 35 months in the ECF arm, and the pathological complete regression (pCR) rate was 16% vs. 6%, respectively [8][9]. A recent Italian case series (RealFLOT study) confirmed the feasibility and safety of the FLOT regimen in a less prognostically favorable selected patient population.
In advanced G/GEJ cancer, the introduction of human epidermal growth factor receptor-2 (HER2)-directed therapies and immune checkpoint inhibitors (ICIs) in combination with chemotherapy has improved survival. Moreover, novel targets could significantly change the continuum of care [10][11][12][13][14].
2. Tailored Treatments
2.1. HER2-Directed Therapies
Over the past few years, increasing attention has developed around targeted therapies, and substantial changes have been made in therapeutic choices and strategies in most malignant tumors [15]. One of the first molecular targets was HER2. The development of the specific monoclonal antibody (moAb) trastuzumab, the first targeted agent for solid malignancies, led to a drastic change in the clinical course of HER2-positive breast cancer patients [16]. In GC patients, previous studies have reported that the HER2 protein expression level ranged from 7.3% to 22.1% [17]. The association between HER2 status and prognosis has been described in several studies, with a general agreement on the negative prognostic role of HER2 overexpression [18][19][20]. The randomized controlled ToGA trial showed that the addition of trastuzumab to chemotherapy offered a meaningful survival gain in advanced GC patients overexpressing HER2 (mOS 13.8 months vs. 11.1 months, HR 0.74; 95% CI 0.60–0.91; p = 0.0046) [12]. Kurokawa and colleagues later demonstrated that HER2 overexpression was an independent prognostic factor in patients with resectable GC (HR 1.59; 95% CI 1.24–2.02; p = 0.001), and they also showed that HER2 intra-tumoral heterogeneity was frequent and did not affect prognosis. In this study, 180 HER2-positive cases were identified among 1148 GC patients, assessed by immunohistochemistry (IHC) and fluorescence in situ hybridization. The trial showed that HER2-positive GC was more commonly related to intestinal-type adenocarcinoma and upper stomach location. Other factors, including the pT and pN stages, did not show any correlation with HER2 overexpression [21]. As previously described, the results from the AIO-FLOT4 trial have set a new standard of care for resectable GC patients fit for intensive treatment. However, more than half of the patients in the FLOT group experienced disease recurrence within 3 years and the estimated OS at 5 years was 45% [9] (Figure 1).
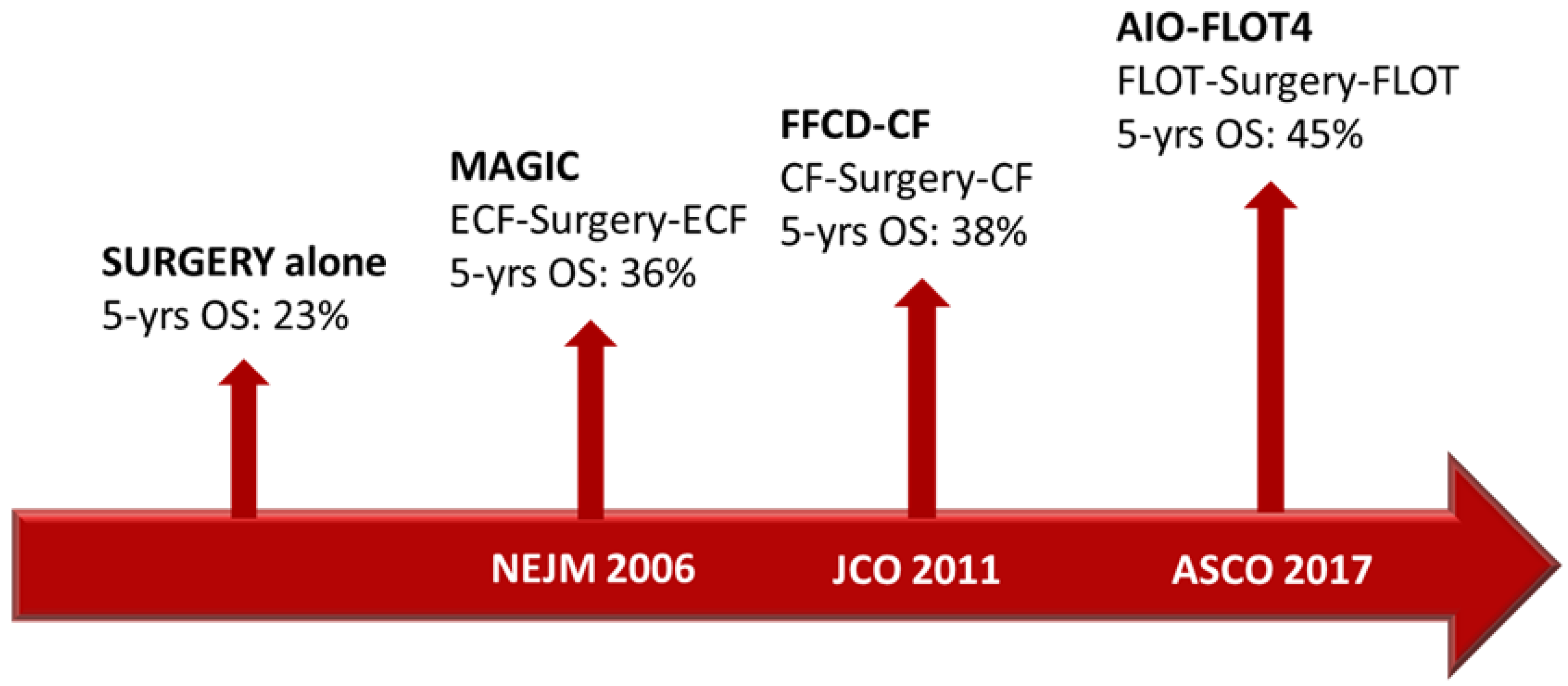
Figure 1. Timeline of the main trials that change the clinical practice in perioperative GC treatment.
Given the results of the AIO-FLOT4 trial in the perioperative setting, the next step was to evaluate the combination of trastuzumab and FLOT as perioperative treatment in patients with HER2-positive, locally advanced esophagogastric adenocarcinomas.
HER-FLOT was a multicenter, phase II study aiming at evaluating the toxicity and activity of trastuzumab in combination with chemotherapy in the perioperative setting. This trial reached the primary endpoint (pCR > 20%) achieving a pCR rate of 21.4%. In fact, 12 out of 56 patients had no viable neoplastic cells at the time of surgery, as assessed by a central pathologist, and 14 patients (25.0%) had subtotal response (<10% residual tumor). The most frequently observed grade (G) 3–4 adverse events (AEs) were quite similar to the FLOT4 trial, and only one case of severe heart failure related to trastuzumab was reported. pCR was also a surrogate of survival outcome. Overall, median disease-free survival (DFS) was 42.5 months and the 3-year OS rate was 82.1% [22].
The PETRARCA trial explored the role of dual HER2 blockade (trastuzumab and pertuzumab) in combination with chemotherapy (FLOT) compared to chemotherapy alone in HER2-positive GC patients who were amenable to a perioperative strategy. Trastuzumab and pertuzumab were administered every 3 weeks for 3 preoperative and 3 postoperative cycles, followed by 9 maintenance cycles. The primary endpoint was the pCR rate that was achieved in 35% of patients (n = 14) in the experimental arm vs. 12% of patients (n = 5) in the control group (p = 0.019). Among secondary endpoints, node-negativity and radical-surgery (i.e., R0) rates were met. The 2-year OS rate was 84% in the experimental arm and 77% in the control arm. Median DFS was not reached in the experimental arm vs. 26 months in the FLOT arm (HR 0.58, 95% CI 0.28–1.19, p = 0.130), with 2-year DFS of 70% and 54%, respectively. The safety profile of the experimental group was characterized by a predominance of diarrhea, neutropenia and leukopenia as high G AEs [23].
The negative results from the JACOB trial, which evaluated the addition of dual anti-HER2 blockade to a taxane-free polychemotherapy regimen, led to a premature stop of the PETRARCA trial and failure to transition to a phase III study [24]. However, PETRARCA’s investigators pointed out the high pCR and pN0 rates in the experimental arm at the cost of slightly higher gastrointestinal and hematopoietic toxicity.
The ongoing, international, randomized phase II EORTC INNOVATION trial has the purpose to evaluate the addition of trastuzumab or trastuzumab plus pertuzumab to chemotherapy in the perioperative treatment of patients with HER2-positive G/GEJ adenocarcinoma. Preliminary results are expected in 2023 [25].
In support of the anti-HER2 blockade strategy, results from some retrospective and prospective experiences conducted in Asian patients have recently been published.
Data from 45 Asian patients with HER2-positive stage II-III GC were retrospectively analyzed. Twenty-nine patients received trastuzumab plus FLOT and sixteen received chemotherapy alone. The primary endpoint was the objective response rate (ORR). In the trastuzumab + FLOT arm, the ORR was 72.4% and the disease control rate (DCR) was 89.7%, while in the FLOT group the ORR and DCR were 43.8% and 87.5%, respectively. The 2-year OS rates were 78.1% and 73.9%, respectively (p = 0.932). Although the study did not reach statistical significance in primary and ancillary endpoints, the addition of trastuzumab provided a numerically high tumor response rate and a promising pathological regression compared to the control group: the tumor regression rate to grade Ia/Ib was obtained in 44.8% of patients [26].
The Japan Clinical Oncology Group (JCOG) conducted the multi-institutional two-arm open label randomized phase II Trigger trial to evaluate the efficacy and safety of S-1-cisplatin in combination with trastuzumab vs. S-1-cisplatin alone for patients with HER2-positive locally advanced GC. The primary endpoint was OS. The study enrolled 46 patients of the preplanned 130 due to slow accrual and was prematurely ended in 2021. The ORR was higher in the experimental group than in the control group (66.7% vs. 36.4%, p = 0.08), even though the difference was not statistically significant. pCR rates were 50.0% and 22.7% (p = 0.07), respectively, and the percentages of patients who experienced pathological downstage were 22.7% and 50.0% (p = 0.07), respectively [27]. Although of interest, this study did not add conclusive data on the role of anti-HER2 blockade in patients with locally advanced disease.
After several disappointing or inconclusive results about the use of anti-HER2 agents other than trastuzumab, the recent results from the phase II DESTINY-Gastric01 trial with the antibody-drug conjugate trastuzumab deruxtecan (T-DXd) renewed interest in advanced GC. The excellent performance of T-DXt in terms of ORR (51.3% vs. 14.3%) and OS (median 12.5 vs. 8.4 months; IC 95% 0.39–0.88, HR 0.59; p = 0.01) compared with physicians’ choice of chemotherapy has led to planning the phase II EPOC2003 study (NCT05034887) with the aim to evaluate T-DXd as a neoadjuvant treatment for patients with HER2-positive G/GEJ adenocarcinoma in six Japanese centers [28][29].
Finally, the randomized phase III RTOG 1010 study included a total of 202 patients diagnosed with locally advanced HER2-positive esophageal adenocarcinoma. Patients were randomized to receive either chemoradiotherapy (CROSS scheme) plus trastuzumab or chemoradiotherapy alone. The primary endpoint was DFS, which was not met. Median DFS was 19.6 months (95% CI 13.5–26.2) with chemoradiotherapy plus trastuzumab compared to 14.2 months (10.5–23.0) for chemoradiotherapy alone (HR 0.99, 95% CI 0.71–1.39, p = 0.97). In conclusion, although well tolerated, the addition of trastuzumab to the neoadjuvant trimodality treatment did not result in any benefit, neither in terms of pCR nor of DFS [30].
2.2. Immune Checkpoint Blockade
As in many solid tumors, the standard of care in G/GEJ cancer is about to change with the introduction of ICIs in combination with chemotherapy as the first line of treatment [14].
Programmed cell death ligand 1 (PD-L1) is a protein expressed in different immune system cells (e.g., T lymphocytes, epithelial cells, endothelial cells, macrophages, dendritic cells). Cancer cells also use PD-L1 to evade the anti-tumor immune response inhibiting cytotoxic T-cell activity. Moreover, neoplastic cells have the ability to upregulate anti-cytotoxic T-lymphocyte-associated antigen-4 (CTLA-4), leading to the formation of a co-inhibitory pathway to avoid host immune responses. Antibodies against the checkpoint proteins programmed death-1 (PD-1) (nivolumab, pembrolizumab), PD-L1 (atezolizumab, avelumab, durvalumab) and CTLA-4 (ipilimumab, tremelimumab) were shown to be effective in reactivating protective T-cell activity [31].
One of the most important predictors of benefit from immunotherapy is the deficient mismatch repair/microsatellite instability-high (dMMR/MSI-H) status, leading to the agnostic approval of pembrolizumab for patients with dMMR advanced tumor [32]. Moreover, several pieces of evidence showed no benefit or harm from peri-/post-operative chemotherapies in resectable MSI-H G/GEJ cancers.
In this regard, an exploratory subgroup analysis from the MAGIC trial showed that patients with an MSI-H/dMMR tumor had a better prognosis (mOS not reached 95% CI, 11.5-NR months) compared to those with an MSS tumor in the surgery-alone arm (mOS 20.5 months; 95% CI, 16.7–27.8 months; HR, 0.42; p = 0.09) [33]. Similarly, a post hoc analysis from the ITACA-S trial suggested that MSI-H status represents an independent prognostic factor, being associated with better DFS (p = 0.02) and OS (p = 0.01) [34]. It is noteworthy that a post hoc analysis from the CLASSIC trial showed that MSI-H (p = 0.008) and PD-L1 (p = 0.044) were independent prognostic factors and no DFS benefit was achieved by adjuvant chemotherapy for patients with an MSI-H tumor (5-year DFS 83.9% vs. 85.7%; p = 0.931) [35]. Finally, a meta-analysis focusing on patients with an MSI-H tumor including MAGIC, CLASSIC, ARTIST and ITACA-S trials was recently conducted. The authors described an advantage in DFS (5-year DFS 71.8% vs. 52.3%; p < 0.001) and OS (5-year OS 77.5% vs. 59.3%; p < 0.001) for patients with an MSI-H tumor compared to those with a microsatellite stable (MSS)/MSI-low tumor [36].
Immunotherapy in Perioperative Treatment of G/GEJ Cancer
The turning point of adjuvant therapy in esophageal or GEJ cancer was represented by the phase III, global, randomized, double-blind, placebo-controlled arm CheckMate 577 trial. The study included patients with stage II/III esophageal or GEJ cancer. After chemoradiotherapy and radical surgery, patients with at least ypT1 and/or ypN1 histologically confirmed cancer were randomized to receive nivolumab 240 mg every 2 weeks for 16 weeks, then 480 mg every 4 weeks (up to 1 year of therapy) or placebo. The primary endpoint was DFS, which was found to be superior in the experimental arm for all the intention-to-treat (ITT) population (22.4 vs. 11.0 months; HR, 0.69; 95% CI, 0.56–0.86; p < 0.001). The benefit was observed in all subgroups, regardless of histological type and PD-L1 level. However, in the subgroup of GEJ patients, the benefit was less marked, with an mDFS of 22.4 months in the nivolumab arm vs. 20.6 months in the placebo arm (HR 0.87, IC 95% 0.63–1.21) as compared to that obtained in esophageal cancer (mDFS 24.0, vs. 8.3, respectively, HR 0.61) [37].
In the phase II PERFECT trial, 40 patients with resectable esophageal adenocarcinoma received atezolizumab in combination with chemoradiotherapy followed by surgery in 83% of cases. Immune-related AEs were observed in 15% (n = 6) of patients in the experimental arm. Ten patients achieved a partial response (PR). Despite being a single-arm study, this represented the first trial to propose the combination of the CROSS scheme and an anti-PD-L1 agent, and it proved the feasibility of adding atezolizumab to CROSS-based neoadjuvant chemoradiotherapy [38].
The efficacy of atezolizumab in the perioperative setting was also evaluated in the randomized multicenter phase IIb DANTE trial from the German and Swiss groups in which patients with resectable G/GEJ adenocarcinoma were assigned to receive atezolizumab in combination with FLOT or FLOT alone. The primary endpoint was progression-free survival (PFS)/DFS after 5 years of observation. Secondary endpoints were recently summarized in an interim analysis presented at the 2022 ASCO meeting, reporting atezolizumab plus FLOT as feasible and safe in the perioperative setting. Surgical mobility and mortality, and R0 rates were comparable between the two arms, while a higher rate of downsizing and pathological regression for the experimental arm (pT0, 23% vs. 15%; pN0, 68% vs. 54%) was reached, especially in patients whose tumor had a high PD-L1 expression or MSI-H status. Indeed, in the MSI-H and PD-L1 combined positive score (CPS) ≥ 10, tumor regression grade (TRG) 1a/b, assessed by central pathologists, was observed in 70–71% of cases in the experimental arm vs. 47–52% in the control arm. These data surely provide the rationale for implementation towards a phase III study [39][40].
The randomized, double-blind, phase III MATTERHORN trial aims at assessing the efficacy and safety of durvalumab in combination with perioperative FLOT, followed by adjuvant durvalumab monotherapy in patients with resectable G/GEJ cancer. The study is currently ongoing, with an estimated sample size of 900 patients at approximately 180 sites worldwide. The primary endpoint is event-free survival; secondary endpoints include OS and the pCR rate. Safety assessment will be evaluated [41].
A remarkable topic is the combination of immune checkpoint therapy, based on the possible synergistic effect of blocking both the PD-1/PD-L1 and the CTLA-4 pathways [42].
To this purpose, the phase II/III trial EA 2174, of the ECOG-ACRIN Cancer Research Group, is evaluating the perioperative treatment with nivolumab and ipilimumab in addition to standard chemotherapy and radiation therapy in patients with locoregional esophageal and GEJ adenocarcinoma. The primary neoadjuvant endpoint is the pCR rate, and the primary adjuvant endpoint is DFS. The safety run-in phase enrolled 31 patients divided into the two arms with no disproportional difference in safety or number of patients who proceeded to surgery [43].
The phase II VESTIGE trial has the primary objective to assess if the combination of nivolumab plus ipilimumab as an adjuvant treatment after neoadjuvant chemotherapy with FLOT and surgery improves DFS in patients with G/GEJ adenocarcinoma [44].
The immunotherapy combination strategy is also under evaluation in patients selected for biomarkers of response. The phase II NEONIPIGA trial enrolled 32 patients with resectable MSI-H/dMMR G/GEJ adenocarcinoma to receive neoadjuvant nivolumab in combination with ipilimumab for six cycles, followed by surgery and subsequent adjuvant treatment with single-agent nivolumab for 9 months. All patients received neoadjuvant treatment, and 29 of them proceeded to surgery. After a median follow-up of 12 months, 30 patients were free from progressive disease (PD), 1 patient died due to PD after five cycles of neoadjuvant therapy, and 1 patient died without relapse. Neoadjuvant therapy with dual checkpoint inhibition led to a pCR rate of 58.6%, meeting the primary endpoint. The combination of nivolumab and ipilimumab is promising as a neoadjuvant treatment in patients with MSI-H tumor and highlights the possibility of delaying or even avoiding surgery in highly selected subgroups of patients [45]. Furthermore, the Italian multicenter, non-randomized, single-arm, multi-cohort, open-label, phase II INFINITY study is evaluating the efficacy and tolerability of the combination of tremelimumab and durvalumab as a neoadjuvant (cohort 1) treatment or as a definitive treatment (cohort 2) in patients with MSI-H/dMMR resectable G/GEJ adenocarcinoma [46]. This study may provide new evidence on the therapeutic management of resectable G/GEJ cancer in patients selected for biomarkers highly predictive of response, particularly in regard to the possibility of non-surgical management.
Among the ongoing trials, the phase II, randomized IMAGINE trial (NCT04062656) is evaluating various immunotherapy or chemo-immunotherapy treatments (nivolumab, nivolumab plus ipilimumab, and nivolumab plus relatlimab, an anti-LAG3 antibody) in the perioperative set, while the KEYNOTE-585 trial (NCT03221426) is evaluating perioperative pembrolizumab plus chemotherapy for resectable G/GEJ adenocarcinoma. Notably, as there is no international standard adjuvant chemotherapy, pembrolizumab can be administered with cisplatin plus capecitabine or 5-fluorouracil, or, in a separate safety cohort, with the FLOT regimen.
The phase II, multicenter, 4-cohort, IMHOTEP trial (NCT04795661) is also evaluating the use of pembrolizumab in the neoadjuvant phase. Patients with resectable MSI-H/dMMR or EBV-positive GC will receive a single dose of pembrolizumab 400 mg 6 weeks before the surgery and a clinician’s choice adjuvant therapy. The primary endpoint will be pCR.
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424.
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30.
- Li, X.; Huang, Q.; Lei, Y.; Zheng, X.; Dai, S.; Leng, W.; Liu, M. Locally advanced gastroesophageal junction cancer with pathological complete response to neoadjuvant therapy: A case report and literature review. Ann. Transl. Med. 2021, 9, 513.
- Cunningham, D.; Allum, W.H.; Stenning, S.P.; Thompson, J.N.; Van de Velde, C.J.; Nicolson, M.; Scarffe, J.H.; Lofts, F.J.; Falk, S.J.; Iveson, T.J.; et al. Perioperative chemotherapy versus surgery alone for resectable gastro-esophageal cancer. N. Engl. J. Med. 2006, 355, 11–20.
- Ychou, M.; Boige, V.; Pignon, J.P.; Conroy, T.; Bouché, O.; Lebreton, G.; Ducourtieux, M.; Bedenne, L.; Fabre, J.-M.; Saint-Aubert, B.; et al. Perioperative chemotherapy compared with surgery alone for resectable gastro-esophageal adenocarcinoma: An FNCLCC and FFCD multicenter phase III trial. J. Clin. Oncol. 2011, 29, 1715–1721.
- Van Cutsem, E.; Moiseyenko, V.M.; Tjulandin, S.; Majlis, A.; Constenla, M.; Boni, C.; Rodrigues, A.; Fodor, M.; Chao, Y.; Voznyi, E.; et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: A report of the V325 Study Group. J. Clin. Oncol. 2006, 24, 4991–4997.
- Ford, H.E.; Marshall, A.; Bridgewater, J.A.; Janowitz, T.; Coxon, F.Y.; Wadsley, J.; Mansoor, W.; Fyfe, D.; Madhusudan, S.; Middleton, G.W.; et al. Docetaxel versus active symptom control for refractory oesophagogastric adenocarcinoma (COUGAR-02): An open-label, phase 3 randomised controlled trial. Lancet Oncol. 2014, 15, 78–86.
- Al-Batran, S.E.; Hofheinz, R.D.; Pauligk, C.; Kopp, H.-G.; Haag, G.M.; Luley, K.B.; Meiler, J.; Homann, N.; Lorenzen, S.; Schmalenberg, H.; et al. Histopathological regression after neoadjuvant docetaxel, oxaliplatin, fluorouracil, and leucovorin versus epirubicin, cisplatin, and fluorouracil or capecitabine in patients with resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4-AIO): Results from the phase 2 part of a multicentre, open-label, randomised phase 2/3 trial. Lancet Oncol. 2016, 17, 1697–1708.
- Al-Batran, S.E.; Homann, N.; Pauligk, C.; Goetze, T.O.; Meiler, J.; Kasper, S.; Kopp, H.-G.; Mayer, F.; Haag, G.M.; Luley, K.; et al. Perioperative chemotherapy with fluorouracil plus leucovorin, oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus cisplatin and epirubicin for locally advanced, resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4): A randomised, phase 2/3 trial. Lancet 2019, 393, 1948–1957.
- Wagner, A.D.; Grothe, W.; Haerting, J.; Kleber, G.; Grothey, A.; Fleig, W.E. Chemotherapy in advanced gastric cancer: A systematic review and meta-analysis based on aggregate data. J. Clin. Oncol. 2006, 24, 2903–2909.
- Gravalos, C.; Jimeno, A. HER2 in gastric cancer: A new prognostic factor and a novel therapeutic target. Ann. Oncol. 2008, 19, 1523–1529.
- Bang, Y.J.; Van Cutsem, E.; Feyereislova, A.; Chung, H.C.; Shen, L.; Sawaki, A.; Lordick, F.; Ohtsu, A.; Omuro, Y.; Satoh, T.; et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): A phase 3, open-label, randomised controlled trial. Lancet 2010, 376, 687–697.
- Chao, J.; Fuchs, C.S.; Shitara, K.; Tabernero, J.; Muro, K.; Van Cutsem, E.; Bang, Y.-J.; De Vita, F.; Landers, G.; Yen, C.-J.; et al. Assessment of Pembrolizumab Therapy for the Treatment of Microsatellite Instability-High Gastric or Gastroesophageal Junction Cancer among Patients in the KEYNOTE-059, KEYNOTE-061, and KEYNOTE-062 Clinical Trials. JAMA Oncol. 2021, 7, 895–902.
- Janjigian, Y.Y.; Shitara, K.; Moehler, M.; Garrido, M.; Salman, P.; Shen, L.; Wyrwicz, L.; Yamaguchi, K.; Skoczylas, T.; Bragagnoli, A.C.; et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): A randomised, open-label, phase 3 trial. Lancet 2021, 398, 27–40.
- Lavacchi, D.; Roviello, G.; D’Angelo, A. Tumor-Agnostic Treatment for Cancer: When How is Better than Where. Clin. Drug Investig. 2020, 40, 519–527.
- Hudis, C.A. Trastuzumab—Mechanism of action and use in clinical practice. N. Engl. J. Med. 2007, 357, 39–51.
- Pereira, M.A.; Ramos, M.F.K.P.; Dias, A.R.; Faraj, S.F.; e Ribeiro, R.R.; de Castria, T.B.; Zilberstein, B.; Alves, V.A.F.; Ribeiro, U.; de Mello, E.S. Expression profile of markers for targeted therapy in gastric cancer patients: HER-2, microsatellite instability and PD-L1. Mol. Diagn. Ther. 2019, 23, 761–771.
- Wang, Y.L.; Sheu, B.S.; Yang, H.B.; Lin, P.W.; Chang, Y.C. Overexpression of c-erb-B2 proteins in tumor and non-tumor parts of gastric adenocarcinoma—Emphasis on its relation to H. pylori infection and clinicohistological characteristics. Hepato-Gastro-enterology 2002, 49, 1172–1176.
- Begnami, M.D.; Fukuda, E.; Fregnani, J.H.; Nonogaki, S.; Montagnini, A.L.; de Costa, W.L., Jr.; Soares, F.A. Prognostic implications of altered human epidermal growth factor receptors (HERs) in gastric carcinomas: HER2 and HER3 are predictors of poor outcome. J. Clin. Oncol. 2011, 29, 3030–3036.
- Kim, K.C.; Koh, Y.W.; Chang, H.M.; Kim, T.H.; Yook, J.H.; Kim, B.S.; Jang, S.J.; Park, Y.S. Evaluation of HER2 protein expression in gastric carcino- mas: Comparative analysis of 1414 cases of whole-tissue sections and 595 cases of tissue microarrays. Ann. Surg. Oncol. 2011, 18, 2833–2840.
- Kurokawa, Y.; Matsuura, N.; Kimura, Y.; Adachi, S.; Fujita, J.; Imamura, H.; Kobayashi, K.; Yokoyama, Y.; Shaker, M.N.; Takiguchi, S.; et al. Multicenter large-scale study of prognostic impact of HER2 expression in patients with resectable gastric cancer. Gastric Cancer 2015, 18, 691–697.
- Hofheinz, R.D.; Hegewisch-Becker, S.; Kunzmann, V.; Thuss-Patience, P.; Fuchs, M.; Homann, N.; Graeven, U.; Schulte, N.; Merx, K.; Pohl, M.; et al. Trastuzumab in combination with 5-fluorouracil, leucovorin, oxaliplatin and docetaxel as perioperative treatment for patients with human epidermal growth factor receptor 2-positive locally advanced esophagogastric adenocarcinoma: A phase II trial of the Arbeitsgemeinschaft Internistische Onkologie Gastric Cancer Study Group. Int. J. Cancer 2021, 149, 1322–1331.
- Hofheinz, R.D.; Merx, K.; Haag, G.M.; Springfeld, C.; Ettrich, T.; Borchert, K.; Kretzschmar, A.; Teschendorf, C.; Siegler, G.; Ebert, M.P.; et al. FLOT Versus FLOT/Trastuzumab/Pertuzumab Perioperative Therapy of Human Epidermal Growth Factor Receptor 2-Positive Resectable Esophagogastric Adenocarcinoma: A Randomized Phase II Trial of the AIO EGA Study Group. J. Clin. Oncol. 2022, 40, 3750–3761.
- Tabernero, J.; Hoff, P.M.; Shen, L.; Ohtsu, A.; Shah, M.A.; Cheng, K.; Song, C.; Wu, H.; Eng-Wong, J.; Kim, K.; et al. Pertuzumab plus trastuzumab and chemotherapy for HER2-positive metastatic gastric or gastro-oesophageal junction cancer (JACOB): Final analysis of a double-blind, randomised, placebo-controlled phase 3 study. Lancet Oncol. 2018, 19, 1372–1384.
- Wagner, A.D.; Grabsch, H.I.; Mauer, M.; Marreaud, S.; Caballero, C.; Thuss-Patience, P.; Mueller, L.; Elme, A.; Moehler, M.H.; Martens, U.; et al. EORTC-1203-GITCG—The “INNOVATION”-trial: Effect of chemotherapy alone versus chemotherapy plus trastuzumab, versus chemotherapy plus trastuzumab plus pertuzumab, in the perioperative treatment of HER2 positive, gastric and gastroesophageal junction adenocarcinoma on pathologic response rate: A randomized phase II-intergroup trial of the EORTC-Gastrointestinal Tract Cancer Group, Korean Cancer Study Group and Dutch Upper GI-Cancer group. BMC Cancer 2019, 19, 494.
- Tong, G.; Li, S.; Lin, L.; He, L.; Wang, L.; Lv, G.; Zheng, R.; Wang, S. Trastuzumab with FLOT Regimen for the Perioperative Treatment of Resectable HER2 + Advanced Gastric Cancer: A Retrospective Study. Cancer Manag. Res. 2020, 12, 2481–2489.
- Tokunaga, M.; Machida, N.; Mizusawa, J.; Yabusaki, H.; Yasui, H.; Hirao, M.; Watanabe, M.; Yasuda, T.; Kinoshita, T.; Imamura, H.; et al. A randomized phase II trial of preoperative chemotherapy of S-1/CDDP with or without trastuzumab followed by surgery in HER2 positive advanced gastric or esophagogastric junction adenocarcinoma with extensive lymph node metastasis: Japan Clinical Oncology Group study JCOG1301C (Trigger Study). J. Clin. Oncol. 2020, 40 (Suppl. S4), 285.
- Shitara, K.; Bang, Y.J.; Iwasa, S.; Sugimoto, N.; Ryu, M.H.; Sakai, D.; Chung, H.C.; Kawakami, H.; Yabusaki, H.; Lee, J.; et al. Trastuzumab Deruxtecan in Previously Treated HER2-Positive Gastric Cancer. N. Engl. J. Med. 2020, 382, 2419–2430.
- Takahari, D.; Kawazoe, A.; Machida, N.; Minashi, K.; Yamagata, Y.; Hara, H.; Wakabayashi, M.; Komura, Y.; Sato, A.; Kuwata, T.; et al. Phase 2 study of trastuzumab deruxtecan in the neoadjuvant treatment for patients with HER2-positive gastric and gastroesophageal junction adenocarcinoma (EPOC2003). J. Clin. Oncol. 2022, 40 (Suppl. S16), TPS4161.
- Safran, H.P.; Winter, K.; Ilson, D.H.; Wigle, D.; DiPetrillo, T.; Haddock, M.G.; Hong, T.S.; Leichman, L.P.; Rajdev, L.; Resnick, M.; et al. Trastuzumab with trimodality treatment for oesophageal adenocarcinoma with HER2 overexpression (NRG Oncology/RTOG 1010): A multicentre, randomised, phase 3 trial. Lancet Oncol. 2022, 23, 259–269.
- Bonotto, M.; Garattini, S.K.; Basile, D.; Ongaro, E.; Fanotto, V.; Cattaneo, M.; Cortiula, F.; Iacono, D.; Cardellino, G.G.; Pella, N.; et al. Immunotherapy for gastric cancers: Emerging role and future perspectives. Expert Rev. Clin. Pharmacol. 2017, 10, 609–619.
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bartlett, B.R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Luber, B.S.; et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 2017, 357, 409–413.
- Smyth, E.C.; Wotherspoon, A.; Peckitt, C.; Gonzalez, D.; Hulkki-Wilson, S.; Eltahir, Z.; Fassan, M.; Rugge, M.; Valeri, N.; Okines, A.; et al. Mismatch Repair Deficiency, Microsatellite Instability, and Survival: An Exploratory Analysis of the Medical Research Council Adjuvant Gastric Infusional Chemotherapy (MAGIC) Trial. JAMA Oncol. 2017, 3, 1197–1203, Erratum in JAMA Oncol. 2022, 8, 1359.
- Di Bartolomeo, M.; Morano, F.; Raimondi, A.; Miceli, R.; Corallo, S.; Tamborini, E.; Perrone, F.; Antista, M.; Niger, M.; Pellegrinelli, A.; et al. Prognostic and Predictive Value of Microsatellite Instability, Inflammatory Reaction and PD-L1 in Gastric Cancer Patients Treated with Either Adjuvant 5-FU/LV or Sequential FOLFIRI Followed by Cisplatin and Docetaxel: A Translational Analysis from the ITACA-S Trial. Oncologist 2020, 25, e460–e468.
- Choi, Y.Y.; Kim, H.; Shin, S.-J.; Kim, H.Y.; Lee, J.; Yang, H.-K.; Kim, W.H.; Kim, Y.-W.; Kook, M.-C.; Park, Y.K.; et al. Microsatellite Instability and Programmed Cell Death-Ligand 1 Expression in Stage II/III Gastric Cancer: Post Hoc Analysis of the CLASSIC Randomized Controlled study. Ann. Surg. 2019, 270, 309–316.
- Pietrantonio, F.; Miceli, R.; Raimondi, A.; Kim, Y.W.; Kang, W.K.; Langley, R.E.; Choi, Y.Y.; Kim, K.-M.; Nankivell, M.G.; Morano, F.; et al. Individual Patient Data Meta-Analysis of the Value of Microsatellite Instability as a Biomarker in Gastric Cancer. J. Clin. Oncol. 2019, 37, 3392–3400.
- Kelly, R.J.; Ajani, J.A.; Kuzdzal, J.; Zander, T.; Van Cutsem, E.; Piessen, G.; Mendez, G.; Feliciano, J.; Motoyama, S.; Lièvre, A.; et al. Adjuvant Nivolumab in Resected Esophageal or Gastroesophageal Junction Cancer. N. Engl. J. Med. 2021, 384, 1191–1203.
- van den Ende, T.; de Clercq, N.C.; van Berge Henegouwen, M.I.; Gisbertz, S.S.; Geijsen, E.D.; Verhoeven, R.H.A.; Meijer, S.L.; Schokker, S.; Dings, M.P.G.; Bergman, J.J.G.H.M.; et al. Neoadjuvant Chemoradiotherapy Combined with Atezolizumab for Resectable Esophageal Adenocarcinoma: A Single-Arm Phase II Feasibility Trial (PERFECT). Clin. Cancer Res. 2021, 27, 3351–3359.
- Al-Batran, S.-E.; Pauligk, C.; Hofheinz, R.; Lorenzen, S.; Wicki, A.; Siebenhuener, A.R.; Schenk, M.; Welslau, M.; Heuer, V.; Goekkurt, E.; et al. Perioperative atezolizumab in combination with FLOT versus FLOT alone in patients with resectable esophagogastric adenocarcinoma: DANTE, a randomized, open-label phase II trial of the German Gastric Group of the AIO and the SAKK. J. Clin. Oncol. 2019, 37 (Suppl. S15), TPS4142.
- Al-Batran, S.; Lorenzen, S.; Thuss-Patience, P.C.; Homann, N.; Schenk, M.; Lindig, U.; Heuer, V.; Kretzschmar, A.; Goekkurt, E.; Haag, G.M.; et al. Surgical and pathological outcome, and pathological regression, in patients receiving perioperative atezolizumab in combination with FLOT chemotherapy versus FLOT alone for resectable esophagogastric adenocarcinoma: Interim results from DANTE, a randomized, multicenter, phase IIb trial of the FLOT-AIO German Gastric Cancer Group and Swiss SAKK. J. Clin. Oncol. 2022, 40 (Suppl. S16), 4003.
- Janjigian, Y.Y.; Van Cutsem, E.; Muro, K.; Wainberg, Z.A.; Al-Batran, S.-E.; Hyung, W.J.; Molena, D.; Evans, B.; Ruscica, D.; Robbins, S.H.; et al. MATTERHORN: Efficacy and safety of neoadjuvant-adjuvant durvalumab and FLOT chemotherapy in resectable gastric and gastroesophageal junction cancer—A randomized, double-blind, placebo-controlled, phase 3 study. J. Clin. Oncol. 2021, 39 (Suppl. S15), TPS4151.
- Pardoll, D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264.
- Eads, J.R.; Weitz, M.; Catalano, P.J.; Gibson, M.K.; Rajdev, L.; Khullar, O.; Lin, S.H.; Gatsonis, C.; Wistuba, I.I.; Sanjeevaiah, A.; et al. A phase II/III study of perioperative nivolumab and ipilimumab in patients (pts) with locoregional esophageal (E) and gastroesophageal junction (GEJ) adenocarcinoma: Results of a safety run-in—A trial of the ECOG-ACRIN Cancer Research Group (EA2174). J. Clin. Oncol. 2021, 39 (Suppl. S15), 4064.
- Smyth, E.; Knödler, M.; Giraut, A.; Mauer, M.; Nilsson, M.; Van Grieken, N.; Wagner, A.D.; Moehler, M.; Lordick, F. VESTIGE: Adjuvant Immunotherapy in Patients with Resected Esophageal, Gastroesophageal Junction and Gastric Cancer Following Preoperative Chemotherapy with High Risk for Recurrence (N+ and/or R1): An Open Label Randomized Controlled Phase-2-Study. Front. Oncol. 2020, 9, 1320.
- Andre, T.; Tougeron, D.; Piessen, G.; De La Fouchardiere, C.; Louvet, C.; Adenis, A.; Jary, M.; Tournigand, C.; Aparicio, T.; Desrame, J.; et al. Neoadjuvant nivolumab plus ipilimumab and adjuvant nivolumab in patients with localized microsatellite instability-high (MSI)/mismatch repair deficient (dMMR) oeso-gastric adenocarcinoma: The GERCOR NEONIPIGA phase II study. J. Clin. Oncol. 2022, 40 (Suppl. S4), 244.
- Raimondi, A.; Palermo, F.; Prisciandaro, M.; Aglietta, M.; Antonuzzo, L.; Aprile, G.; Berardi, R.; Cardellino, G.; De Manzoni, G.; De Vita, F.; et al. TremelImumab and Durvalumab Combination for the Non-OperatIve Management (NOM) of Microsatellite InstabiliTY (MSI)-High Resectable Gastric or Gastroesophageal Junction Cancer: The Multicentre, Single-Arm, Multi-Cohort, Phase II INFINITY Study. Cancers 2021, 13, 2839.
More
Information
Subjects:
Oncology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
594
Revisions:
2 times
(View History)
Update Date:
09 Mar 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




