Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Nina Filip | -- | 2889 | 2023-03-07 22:12:49 | | | |
| 2 | Conner Chen | + 8 word(s) | 2897 | 2023-03-09 04:23:00 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Bordeianu, G.; Filip, N.; Cernomaz, A.; Veliceasa, B.; Hurjui, L.L.; Pinzariu, A.C.; Pertea, M.; Clim, A.; Marinca, M.V.; Serban, I.L. Nanotechnologies and Lung Cancer Therapy. Encyclopedia. Available online: https://encyclopedia.pub/entry/41948 (accessed on 07 February 2026).
Bordeianu G, Filip N, Cernomaz A, Veliceasa B, Hurjui LL, Pinzariu AC, et al. Nanotechnologies and Lung Cancer Therapy. Encyclopedia. Available at: https://encyclopedia.pub/entry/41948. Accessed February 07, 2026.
Bordeianu, Gabriela, Nina Filip, Andrei Cernomaz, Bogdan Veliceasa, Loredana Liliana Hurjui, Alin Constantin Pinzariu, Mihaela Pertea, Andreea Clim, Mihai Vasile Marinca, Ionela Lacramioara Serban. "Nanotechnologies and Lung Cancer Therapy" Encyclopedia, https://encyclopedia.pub/entry/41948 (accessed February 07, 2026).
Bordeianu, G., Filip, N., Cernomaz, A., Veliceasa, B., Hurjui, L.L., Pinzariu, A.C., Pertea, M., Clim, A., Marinca, M.V., & Serban, I.L. (2023, March 07). Nanotechnologies and Lung Cancer Therapy. In Encyclopedia. https://encyclopedia.pub/entry/41948
Bordeianu, Gabriela, et al. "Nanotechnologies and Lung Cancer Therapy." Encyclopedia. Web. 07 March, 2023.
Copy Citation
Lung cancer remains a major public health problem both in terms of incidence and specific mortality despite developments in terms of prevention, such as smoking reduction policies and clinical management advances. Better lung cancer prognosis could be achieved by early and accurate diagnosis and improved therapeutic interventions. Nanotechnology is a dynamic and fast-developing field; various medical applications have been developed and deployed, and more exist as proofs of concepts or experimental models.
nanomedicine
lung cancer
drugs
1. Introduction
The birth of the term nanotechnology is anecdotally linked to the American physicist Richard Feynman in the early 1960s; currently, this is an umbrella term for technologies dealing with structures between 1 and 100 nanometers [1]. Nanotechnology has established a foothold in the medical space; it is employed in various branches spanning from diagnosis to treatment. Atheroma plaque healing, regenerating damaged nerves, and targeting tumor tissues are only a few examples of the practical applications of nanotechnology [2].
Recent years have brought significant improvements concerning survival and quality of life for various hematological and solid malignant disease patients; lung cancer lags behind despite new emerging therapies [3]. Various strategies have been devised to improve lung cancer outcomes; early diagnosis and better therapeutic options are focus points. Early diagnosis enables radical therapeutic procedures such as surgical resection or curative intent radiotherapy while maintaining quality of life; however, in the case of lung cancer, late onset of clinical signs, lack of reliable biomarkers, and imaging-related limitations make such an approach difficult [4].
Lung cancer therapeutic protocols are usually chosen considering the histology of the tumor and the extension and mainly combine surgery, radiation therapy, and chemotherapy. For non-resectable lung cancer, the prognosis is linked to oncological treatment efficacy, which is generally limited by tolerability and toxic side effects [5]; immuno- and targeted therapy are recent additions gaining ground and leading towards personalized medicine [6] but are adequate only in a minority of cases.
Nanotechnologies could alleviate some conventional therapy drawbacks and improve efficacy; tailoring drug pharmacokinetics (by facilitating intra- and intercellular traffic or navigating the tumor micro-environment), targeting various cellular lines, and modulating the immune response are only a few possibilities [7].
Nanoparticles is an umbrella term encompassing a plethora of chemically different structures potentially useful in both early diagnosis and better therapy.
2. Nanotechnologies and Lung Cancer Therapy
Lung cancer remains one of the most frequently diagnosed malignant diseases; despite some progress in prevention, early detection, and advanced therapy, its prognosis is usually severe, and associated mortality remains high. Tobacco smoking was identified as the main risk factor, and some population-level risk mitigation measures have been implemented; other external factors, such as air pollution (environmental and domestic), also have a role [5]. Surgery, radiotherapy, and oncological therapies are the main pillars of lung cancer treatment; early diagnosis and correct staging are paramount to optimize outcomes [6].
Classic oncological management of lung cancer involves chemotherapy, though recent molecular biology developments have brought out new therapeutic methods with increased efficacy and better safety profiles, such as targeted agents and immunotherapy.
Paclitaxel is a chemotherapic agent frequently used in breast, ovary, prostate, and lung cancer protocols. The doublet paclitaxel platinum salt may be considered the mainstay of non-small cell lung cancer therapy. Paclitaxel acts as a tubulin-binding agent, stopping mitosis and promoting cellular death; it has low hydro-solubility, and therefore, an emulsifier vehicle is necessary, usually the solvent oil cremophor ethanol (CrEL). The administration of a CrEL-paclitaxel formula may be followed by potentially lethal adverse events such as hypersensitivity reactions, peripheral neuropathy, or myelosuppression [8][9][10]. Various mitigation strategies are used in clinical settings, such as corticoid and antihistamine premedication and low infusion rates; one potential alternative may be the use of albumin-bound paclitaxel nanoparticles, which seem to have increased plasma life and antitumor activity, at least in murine human tumor xenograft models [11]. The nab-paclitaxel formula (under the trade name Abraxane) was initially FDA-approved in 2004 for metastatic breast cancer; in 2012, it was accepted for the first-line treatment of locally advanced or metastatic non-small cell lung cancer in combination with carboplatin in patients who are not candidates for curative surgery or radiation therapy [12]. Using the nab-paclitaxel form allowed enhanced tumor penetration and cellular uptake [13] (probably by transporter-mediated mechanisms) with a higher clinical response rate and a better safety profile than classic CrEL paclitaxel [14]. There are data supporting the use of nab-paclitaxel as higher effective concentrations can be reached with a shorter infusion time, eliminating the need for premedication used to alleviate the risk of solvent-induced hypersensitivity reactions [15].
Another way to improve the chemical stability and solubility of CrEL-paclitaxel made use of liposomes; the cytotoxic effect was similar to classic Taxol, but bioavailability and stability were improved. In 2006, a formulation was approved in China under the trade name LIPUSU [16]. There are data suggesting that higher cellular uptake and better cytotoxicity with a similar safety profile may be attained by altering the lipid components of the liposomes (by adding lysophosphatidylcoline by a simple process) [17].
Other paclitaxel nanoparticles are under scrutiny. Polymeric micellar paclitaxel (pm-Pac) is a CrEL-free structure that was recently tested in phase III trials, showing increased tumor cell penetration and reduced adverse effects in combination with cisplatin, thus potentially becoming a new chemotherapy option for advanced non-small-cell lung cancer patients [18][19]. Polylactic-co-glycolic acid (PLGA) has also been considered as a potential paclitaxel carrier; there are published data supporting higher cytotoxicity, stronger apoptosis signal, weaker migration and invasion for NSCLC cells when using solvent-based paclitaxel as a comparator [20][21].
Doxorubicin is a potentially useful chemotherapeutic agent for many solid tumors [22][23], but its high toxicity and induced resistance may impose limits on its use. Particularly for lung cancers, doxorubicin shows low cellular penetration, low tumor concentrations, and significant toxicity [24][25]. Various nanoparticle–doxorubicin delivery systems have been tested, and some showed better pharmacokinetics and bioavailability and a lower effect on normal cells [20][26].
The dimercaptosuccinic acid terminated poly (amido-amine) (PAMAM) dendrimers conjugated with doxorubicin proved effective in delivering doxorubicin using a glucose moiety as a targeting structure and making use of the increased glucose uptake of tumor cells; furthermore, the dimensions of the conjugates decreased the renal elimination and demonstrated a longer half-life [27].
Doxorubicin-containing PEGylated liposomes are available and widely used in clinical oncology (PLD; CAELYX, Schering-Plough Corp., Kenilworth, NJ, USA/DOXIL, ALZA, Mountain View, CA, USA) as toxic effects (mainly cardiotoxicity and myelosuppression but also vomiting and alopecia) are mitigated compared with conventional doxorubicin [20][28][29].
Molecular cancer targets are products of so-called driver mutations. The most frequent are epidermal growth factor receptor (EGFR), KRAS (Kirsten rat sarcoma virus gene), HER2 (human epidermal growth factor receptor 2 gene), ALK (anaplastic lymphoma kinase), ROS1 (tyrosine-protein kinase ROS gene), cMET (MNNG HOS transforming gene), BRAF (B-Raf gene), RET (rearranged during transfection gene), and NTRK (neurotrophic tyrosine receptor kinase gene) [30].
The best-known driver mutations for NSCLC involve the epidermal growth factor receptor gene (EGFR), being detected in 10–15% of lung adenocarcinoma patients. EGFR is a receptor tyrosine kinase, a member of the ErbB family, and may be physiologically activated by multiple ligands; this interaction may activate various intracellular signaling pathways, such as PI3K/AKT/mTOR, Ras/Raf/MEK/ERK1/2, and the phospholipase C (PLC) cascade—with clear implications regarding cell migration, attachment, angiogenesis, and organogenesis regulation [30][31].
Various activating EGFR mutations have been documented; the in-frame exon 19 deletion and the L858R substitution account for 85% of relevant driver mutations in 85% of NSCLC cases, but there are multiple deletions, insertions, point mutations and duplications reported concerning exons 18–25 [32].
Such mutations may lead to persistent signal pathway activation with decreased apoptosis and cell proliferation and play a role in tumorigenesis; therefore, the EGFR domains became a potential target for novel antitumor agents [33].
The first generation of EGFR tyrosine kinase inhibitors (EGFR-TKI) has a reversible effect on the tyrosine kinase EGFR domain. Various clinical trials have shown improved survival for mutation-harboring patients using standard cytotoxic therapy as a comparator [34][35]. Although the survival rate has improved, the patients acquire resistance to these drugs after 9–14 months [36][37].
EGFR exon 20 T790M deletion, which occurs in 50–60% of NSCLC patients undergoing first-generation EGFR-TKI therapy (such as erlotinib or gefitinib), is the most common mechanism of acquired resistance; the second generation of EGFR-TKI (afatinib and dacomitinib) was developed aiming to circumvent this drawback without noticeable success [38].
The 3rd-generation EGFR-TKI (osimertinib, rocelitinib, olmutinib) proved to be effective in overcoming the resistance induced by the T790M deletion and are currently considered first-line agents in NSCLC protocols for patients with driver mutations [39].
Similar to classic antitumor agents, the idea of boosting the effects of EGFR-TKIs using nanoparticles was investigated. There are data concerning the use of GEF-loaded poly(ε-caprolactone)-poly(ethyleneglycol)-poly(ε-caprolactone) (PCEC)-bearing nanoparticles (GEF-NPs) with improved antitumor effects, prolonged survival time, and less side effects using classic gefitinib as a comparator [40].
Human serum albumin (HAS) is non-immunogenic and has ideal biocompatibility. It is frequently used as a drug vehicle as it improves the solubility of lipophilic drugs. Hyaluronic acid (HA) is a negatively charged polysaccharide that is similarly biocompatible and known to interact with some surface molecules such as CD-44, lymphatic vessel endothelial receptor-1, and receptor for hyaluronan-mediated motility that are frequently overexpressed in malignant cells [41][42]. An erlotinib/hyaluronic acid/human serum albumin complex (ERT-HSA-HA NPs) was developed and tested on tumor cell lines and animal models with promising results, including tumor growth inhibition and lack of recurrence, possibly explained by longer plasma half-life and higher tumor uptake [43][44].
Complex associations were also tested. Doxorubicin and icotinib (proven more effective than erlotinib and apatinib) were encapsulated using cationic amphipathic starch and hyaluronic acid. The resulting NPs were tested using lung cancer lines and murine models and were shown to accumulate in tumor cells with a smaller effect on normal cells [45].
Both afatinib and dacomitinib (a second-generation, irreversible EGFR-TKI, FDA-approved) have low solubility, which translates to low pulmonary tissue bioavailability; such a drawback might be circumvented by a direct administration route by using a system of poly-(lactic-co-glycolic-acid) nanoparticles (PLGA NPs) developed for inhalation for pulmonary lesions [46].
Osimertinib is the first FDA-approved third-generation EGFR-TKI; current therapeutic protocols allow its use for both NSCLC patients with activating EGFR mutation and patients with T790M resistance mutation cancers with encouraging results, though still limited by acquired resistance. Among the strategies laid out to overcome osimertinib resistance, the use of complex nanoparticles might play a role; a combination of osimertinib and selumetinib (a MEK inhibitor with limited NSCLC effects) conjugated with PEG using a reactive oxygen species-responsive linker had encouraging in vitro and murine model effects. The PEG-selumetinib complex acted as a micelle carrier for osimertinib and delivered the drug payload in high reactive oxygen species activity zones such as tumor cells; such an approach may combine the benefits of both targeting tumor cells and preventing acquired resistance [47].
The association of nanotechnologies and EGFR-TKIs is not limited to lung cancer therapy. A creative combination of erlotinib and superparamagnetic iron oxide core particles was found to exhibit affinity towards EGFR overexpressing cells. Such an approach may enable magnetic resonance imaging (MRI)-based detection of EGFR mutated tumors; this would be valuable as MRI techniques are generally of little use for lung imaging despite some obvious advantages, such as no ionizing radiation exposure [48].
The anaplastic lymphoma kinase (ALK) gene is located on chromosome 2 and en-codes a transmembrane tyrosine kinase that normally has a low expression in small intestine, nervous system, and testicular cells in adults. Still, ALK gene rearrangement was reported in some NSCLCs; its prevalence is between 3% to 7% in adenocarcinoma cases; many current diagnostic protocols include routine ALK testing for relevant histology samples [49][50].
The c-ros oncogene 1 (ROS1) codes a tyrosine receptor kinase belonging to the insulin receptor family; some rearrangements have been reported particularly in adenocarcinoma cases occurring in young, never smoking patients (Asian descent may also play a role). Multiple ROS1 mutations have been reported (with various signaling pathways involved). Their global prevalence is estimated between 1 and 3% of lung adenocarcinomas [51].
Crizotinib is a tyrosine kinase inhibitor active on ALK, MET and ROS1 available in oral form; its effectivity is limited by various mechanisms such as mutations in the ALK kinase domain, the increased number of ALK fusion genes, and central nervous system progression stemming from low penetration of the blood–brain barrier [52][53].
Polymeric nanoparticles based on polylactide-tocopheryl polyethylene glycol 1000 succinates (PLA-TPGS) may be used to encapsulate crizotinib with better cellular uptake and increased biological effect [54].
Similarly, poly (ethylene glycol)–poly(ε-caprolactones)–poly (ethylene glycol) (PEG–PCL–PEG, PECE) structures have been used as delivery systems for both sorafenib and crizotinib (SORA-CRIZ-NPs), improving their hydrosolubility and reducing their toxic effects [55].
Alectinib was approved in 2015 and is included in current therapeutic protocols for ALK-positive NSCLC cases with resistance or progression under crizotinib therapy [56][57]. Various side effects such as anemia, increased aminotransferase activity, hyperbilirubinemia, and hyperglycemia affect most users [58].
One modern oncological approach to lung cancer is the relatively new check-point immunotherapy; this method makes use of immunoglobulins to prevent the interaction between the programmed death ligand-1 (PD-L1) and its receptor (cluster of differentiation 274 (CD274) or PD-1) the underlying mechanism being T-cell cytotoxic mediated [59][60][61]. Silencing PD-L1 and PD-1 on tumor-infiltrating lymphocytes by deploying siRNA on a lipid-coated calcium phosphate carrier proved to be an effective approach in a breast cancer model, suggesting a way to improve immunotherapy outcomes [61].
There are some completed clinical trials (Table 1) investigating various nanotechnology therapeutic applications in the field of lung cancer. There is considerable variability in terms of investigative products and efficacy endpoints.
Table 1. Completed clinical trials indexed on clinicaltrials.gov relevant to nanoparticle-augmented lung cancer therapy.
| Study Type | Description | Primary Outcome | NCT Number | Number of Participants |
|---|---|---|---|---|
| Phase IV | Efficacy and safety of paclitaxel liposome and cisplatin compared with gemcitabine and cisplatin as first-line therapy in advanced squamous non-small-cell lung cancer | Progression-free survival | NCT02996214 | 536 |
| Phase II | ABI-009, human albumin-bound rapamycin, in patients with metastatic, unresectable, low, or intermediate grade neuroendocrine tumors of the lung or gastro-enteropancreatic system who have progressed or been intolerant to everolimus | Disease control rate | NCT03670030 | 5 |
| Phase II | Safety and efficacy of BIND-014 (docetaxel nanoparticles for injectable suspension) as second-line therapy to patients with non-small-cell lung cancer | Objective response rate | NCT01792479 | 64 |
| Phase II | BIND-014 (docetaxel nanoparticles for injectable suspension) as second-line therapy for patients with KRAS positive or squamous cell non-small cell lung cancer | Disease control rate | NCT02283320 | 69 |
| Phase II | Carboplatin and paclitaxel albumin-stabilized nanoparticle formulation together with radiation therapy and erlotinib in treating patients with Stage III NSCLC that cannot be removed by surgery | Overall survival at 12 months | NCT00553462 | 78 |
| Phase II | Paclitaxel albumin-stabilized nanoparticle formulation given together with carboplatin in treating patients with stage IIIB, stage IV, or recurrent NSCLC | Overall response rate | NCT00729612 | 63 |
| Phase I-II | Side effects and optimal dose of ABI-007 (paclitaxel albumin-stabilized nanoparticle formulation) efficacy in treating patients with stage IV NSCLC | Target lesion response (safety, tolerability, antitumor activity) | NCT00077246 | 64 |
| Phase II | CRLX101 (camptothecin (CPT) conjugated to a cyclodextrin-based polymer) vs. best supportive care (BSC) in advanced non-small-cell lung cancer (NSCLC) | Overall survival | NCT01380769 | 157 |
| Phase II | Paclitaxel albumin-stabilized nanoparticle formulation (Abraxane) in treating patients with previously treated advanced non-small-cell lung cancer. | Overall response rate | NCT01620190 | 26 |
| Phase I/II | Safety and antitumor activity of ABI-007 (a unique protein formulation of paclitaxel) in weekly administration in naïve patients with advanced non-small cell lung cancer | Establishing the toxicity | NCT00073723 | 75 |
| Phase I | TargomiRs (targeted minicells containing a microRNA mimic) as 2nd or 3rd line treatment for patients with recurrent malignant pleural mesothelioma and non-small-cell lung cancer. | Establishing maximum tolerated dose and dose-limiting toxicities | NCT02369198 | 27 |
| Phase II | Effectiveness of nab-paclitaxel + carboplatin + MPDL3280A (monoclonal antibody directed against the protein ligand programmed cell death-1 ligand 1 (PD-L1) for treatment of non-small-cell lung carcinoma (NSCLC) | Major pathologic response rate | NCT02716038 | 39 |
| Phase I/II | Combination therapy with NC-6004 (nanoparticle-cisplatin) and gemcitabine in patients with advanced solid tumors or non-small-cell lung, biliary, and bladder cancer | Progression-free survival | NCT02240238 | 209 |
This makes it difficult not only to identify the best therapeutic options but also to identify which way further research should focus. Despite one phase IV trial, the majority of completed trials are phases I and II, which may imply additional data are still required to validate the use of some nanotechnologies in clinical practice.
Current guidelines hold chemotherapy, mutation-targeted therapy, and immunotherapy as standard approaches in lung cancer management, along with surgery and radiotherapy. Nanoparticles may play a role as adjuvants to radiotherapy and topic minimal invasive interventions such as photodynamic therapy. A summary of potential applications is presented in Table 2.
Table 2. Nanoparticle-enabled lung cancer therapeutic procedures.
| Procedure | Nanoparticles | Role | Reference |
|---|---|---|---|
| Photothermal therapy | Gold nanoparticles, Fe3O4, polydopamine | Fluorescent dye, photosensitizer, theragnostic agent | [62][63][64][65] |
| Photodynamic therapy | Quantum dots, photosensitizer nanoparticles (hypocrellin B) | Photosensitizer | [66][67][68][69] |
| Radiation therapy | Gold and platinum-based NPs | Sensitizer | [70][71][72] |
| Gene therapy | Liposomal nucleic acid delivery system (lipofectamine), solid lipid- and polymer-based gene delivery vectors | Nucleic acid delivery systems | [73][74][75] |
| Chemotherapy | Polymers, dendrimers, liposome-based drug delivery systems (various chemotherapeutic agents) | Carriers, targeted carriers | [18][21][27][28][76] |
From a practical point of view some nanoparticles exhibit peculiar properties enabling potential multiple roles at once, both diagnostic and therapeutic (Figure 1). This represents an emerging concept dubbed theranostics. Such an approach is the use of quantum dots as a fluorescent agent able to guide and amplify the biological effect of bronchoscopy-delivered photodynamic therapy, an intervention particularly suited to carcinoma in situ management [66][77]. Similarly, near-infrared emitting QDs were experimentally successfully used to improve the intra-operatory visualization of pulmonary nodules and establish resection limits. The resulting in vivo fluorescence proved to be relatively independent of dimensions and vascularization and allowed improved detection beyond CT data [77][78].
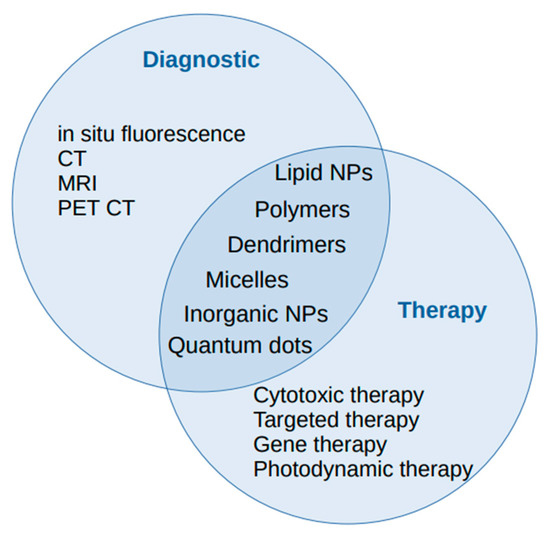
Figure 1. The diagnostic and therapeutic roles of nanoparticles in lung cancer management.
References
- Feynman, R.P. There’s Plenty of Room at the Bottom. Eng. Sci. 1960, 23, 22–36.
- Lin, H.; Datar, R.H. Medical Applications of Nanotechnology. Natl. Med. J. India 2006, 19, 27–32.
- GLOBOCAN—Online Database Providing Global Cancer Statistics Link: Global Cancer Observatory. Available online: https://gco.iarc.fr/ (accessed on 20 December 2022).
- Ramaswamy, A. Lung Cancer Screening: Review and 2021 Update. Curr. Pulmonol. Rep. 2022, 11, 15–28.
- Thai, A.A.; Solomon, B.J.; Sequist, L.V.; Gainor, J.F.; Heist, R.S. Lung Cancer. Lancet 2021, 398, 535–554.
- Gridelli, C.; Rossi, A.; Carbone, D.P.; Guarize, J.; Karachaliou, N.; Mok, T.; Petrella, F.; Spaggiari, L.; Rosell, R. Non-Small-Cell Lung Cancer. Nat. Rev. Dis. Prim. 2015, 1, 15009.
- Mitchell, M.J.; Billingsley, M.M.; Haley, R.M.; Wechsler, M.E.; Peppas, N.A.; Langer, R. Engineering Precision Nanoparticles for Drug Delivery. Nat. Rev. Drug Discov. 2021, 20, 101–124.
- Weiss, R.B.; Donehower, R.C.; Wiernik, P.H.; Ohnuma, T.; Gralla, R.J.; Trump, D.L.; Baker, J.R.; Van Echo, D.A.; Von Hoff, D.D.; Leyland-Jones, B. Hypersensitivity Reactions from Taxol. J. Clin. Oncol. 1990, 8, 1263–1268.
- Irizarry, L.D.; Luu, T.H.; McKoy, J.M.; Samaras, A.T.; Fisher, M.J.; Carias, E.E.; Raisch, D.W.; Calhoun, E.A.; Bennett, C.L. Cremophor EL-Containing Paclitaxel-Induced Anaphylaxis: A Call to Action. Community Oncol. 2009, 6, 132–134.
- Mielke, S.; Sparreboom, A.; Mross, K. Peripheral Neuropathy: A Persisting Challenge in Paclitaxel-Based Regimes. Eur. J. Cancer 2006, 42, 24–30.
- Montana, M.; Ducros, C.; Verhaeghe, P.; Terme, T.; Vanelle, P.; Rathelot, P. Albumin-Bound Paclitaxel: The Benefit of This New Formulation in the Treatment of Various Cancers. J. Chemother. 2011, 23, 59–66.
- Ramalingam, S.; Belani, C.P. Paclitaxel for Non-Small Cell Lung Cancer. Expert Opin. Pharmacother. 2004, 5, 1771–1780.
- Chen, N.; Brachmann, C.; Liu, X.; Pierce, D.W.; Dey, J.; Kerwin, W.S.; Li, Y.; Zhou, S.; Hou, S.; Carleton, M.; et al. Albumin-Bound Nanoparticle (Nab) Paclitaxel Exhibits Enhanced Paclitaxel Tissue Distribution and Tumor Penetration. Cancer Chemother. Pharmacol. 2015, 76, 699–712.
- Gradishar, W.J.; Tjulandin, S.; Davidson, N.; Shaw, H.; Desai, N.; Bhar, P.; Hawkins, M.; O’Shaughnessy, J. Phase III Trial of Nanoparticle Albumin-Bound Paclitaxel Compared with Polyethylated Castor Oil–Based Paclitaxel in Women with Breast Cancer. J. Clin. Oncol. 2005, 23, 7794–7803.
- Gradishar, W.J. Albumin-Bound Paclitaxel: A next-Generation Taxane. Expert Opin. Pharmacother. 2006, 7, 1041–1053.
- Birim, Ö.; Kappetein, A.P.; Stijnen, T.; Bogers, A.J.J.C. Meta-Analysis of Positron Emission Tomographic and Computed Tomographic Imaging in Detecting Mediastinal Lymph Node Metastases in Nonsmall Cell Lung Cancer. Ann. Thorac. Surg. 2005, 79, 375–382.
- Li, L.; Zhan, Q.; Yi, K.; Chen, N.; Li, X.; Yang, S.; Hou, X.; Zhao, J.; Yuan, X.; Kang, C. Engineering Lipusu with Lysophosphatidylcholine for Improved Tumor Cellular Uptake and Anticancer Efficacy. J. Mater. Chem. B 2022, 10, 1833–1842.
- Shi, M.; Gu, A.; Tu, H.; Huang, C.; Wang, H.; Yu, Z.; Wang, X.; Cao, L.; Shu, Y.; Wang, H.; et al. Comparing Nanoparticle Polymeric Micellar Paclitaxel and Solvent-Based Paclitaxel as First-Line Treatment of Advanced Non-Small-Cell Lung Cancer: An Open-Label, Randomized, Multicenter, Phase III Trial. Ann. Oncol. 2021, 32, 85–96.
- Lu, J.; Gu, A.; Wang, W.; Huang, A.; Han, B.; Zhong, H. Polymeric Micellar Paclitaxel (Pm-Pac) Prolonged Overall Survival for NSCLC Patients without Pleural Metastasis. Int. J. Pharm. 2022, 623, 121961.
- Zheng, D.; Wang, J.; Guo, S.; Zhao, Z.; Wang, F. Formulations, Pharmacodynamic and Clinical Studies of Nanoparticles for Lung Cancer Therapy–An Overview. Curr. Drug Metab. 2018, 19, 759–767.
- Zuo, Y.; Shen, W.; Wang, L.; Wang, C.; Pu, J. Study on the Mechanism of Action of Paclitaxel-Loaded Polylactic-Co-Glycolic Acid Nanoparticles in Non-Small-Cell Lung Carcinoma Cells. Comput. Math. Methods Med. 2022, 2022, 1–7.
- Minchinton, A.I.; Tannock, I.F. Drug Penetration in Solid Tumours. Nat. Rev. Cancer 2006, 6, 583–592.
- Panuţa, A.; Radu, I.; Gafton, B.; Ioanid, N.; Terinte, C.; Ferariu, D.; Buna-Arvinte, M.; Scripcariu, D.V.; Scripcariu, V. Multiple versus Unifocal Breast Cancer: Clinicopathological and Immunohistochemical Differences. Romanian J. Morphol. Embryol. Rev. Roum. Morphol. Embryol. 2019, 60, 103–110.
- Primeau, A.J.; Rendon, A.; Hedley, D.; Lilge, L.; Tannock, I.F. The Distribution of the Anticancer Drug Doxorubicin in Relation to Blood Vessels in Solid Tumors. Clin. Cancer Res. 2005, 11, 8782–8788.
- Lesniak, M.S.; Upadhyay, U.; Goodwin, R.; Tyler, B.; Brem, H. Local Delivery of Doxorubicin for the Treatment of Malignant Brain Tumors in Rats. Anticancer Res. 2005, 25, 3825–3831.
- Maheswaran, S.; Sequist, L.V.; Nagrath, S.; Ulkus, L.; Brannigan, B.; Collura, C.V.; Inserra, E.; Diederichs, S.; Iafrate, A.J.; Bell, D.W.; et al. Detection of Mutations in EGFR in Circulating Lung-Cancer Cells. N. Engl. J. Med. 2008, 359, 366–377.
- Sztandera, K.; Działak, P.; Marcinkowska, M.; Stańczyk, M.; Gorzkiewicz, M.; Janaszewska, A.; Klajnert-Maculewicz, B. Sugar Modification Enhances Cytotoxic Activity of PAMAM-Doxorubicin Conjugate in Glucose-Deprived MCF-7 Cells–Possible Role of GLUT1 Transporter. Pharm. Res. 2019, 36, 140.
- O’Brien, M.E.R.; Wigler, N.; Inbar, M.; Rosso, R.; Grischke, E.; Santoro, A.; Catane, R.; Kieback, D.G.; Tomczak, P.; Ackland, S.P.; et al. Reduced Cardiotoxicity and Comparable Efficacy in a Phase IIItrial of Pegylated Liposomal Doxorubicin HCl(CAELYXTM/Doxil®) versus Conventional Doxorubicin Forfirst-Line Treatment of Metastatic Breast Cancer. Ann. Oncol. 2004, 15, 440–449.
- Chittasupho, C.; Lirdprapamongkol, K.; Kewsuwan, P.; Sarisuta, N. Targeted Delivery of Doxorubicin to A549 Lung Cancer Cells by CXCR4 Antagonist Conjugated PLGA Nanoparticles. Eur. J. Pharm. Biopharm. 2014, 88, 529–538.
- Inoue, A. Progress in Individualized Treatment for EGFR-Mutated Advanced Non-Small Cell Lung Cancer. Proc. Jpn. Acad. Ser. B 2020, 96, 266–272.
- Alharbi, K.S.; Javed Shaikh, M.A.; Afzal, O.; Alfawaz Altamimi, A.S.; Almalki, W.H.; Alzarea, S.I.; Kazmi, I.; Al-Abbasi, F.A.; Singh, S.K.; Dua, K.; et al. An Overview of Epithelial Growth Factor Receptor (EGFR) Inhibitors in Cancer Therapy. Chem. Biol. Interact. 2022, 366, 110108.
- Harrison, P.T.; Vyse, S.; Huang, P.H. Rare Epidermal Growth Factor Receptor (EGFR) Mutations in Non-Small Cell Lung Cancer. Semin. Cancer Biol. 2020, 61, 167–179.
- Gandhi, J.; Zhang, J.; Xie, Y.; Soh, J.; Shigematsu, H.; Zhang, W.; Yamamoto, H.; Peyton, M.; Girard, L.; Lockwood, W.W.; et al. Alterations in Genes of the EGFR Signaling Pathway and Their Relationship to EGFR Tyrosine Kinase Inhibitor Sensitivity in Lung Cancer Cell Lines. PLoS ONE 2009, 4, e4576.
- Zhou, C.; Wu, Y.-L.; Chen, G.; Feng, J.; Liu, X.-Q.; Wang, C.; Zhang, S.; Wang, J.; Zhou, S.; Ren, S.; et al. Erlotinib versus Chemotherapy as First-Line Treatment for Patients with Advanced EGFR Mutation-Positive Non-Small-Cell Lung Cancer (OPTIMAL, CTONG-0802): A Multicentre, Open-Label, Randomised, Phase 3 Study. Lancet Oncol. 2011, 12, 735–742.
- Mitsudomi, T.; Morita, S.; Yatabe, Y.; Negoro, S.; Okamoto, I.; Tsurutani, J.; Seto, T.; Satouchi, M.; Tada, H.; Hirashima, T.; et al. Gefitinib versus Cisplatin plus Docetaxel in Patients with Non-Small-Cell Lung Cancer Harbouring Mutations of the Epidermal Growth Factor Receptor (WJTOG3405): An Open Label, Randomised Phase 3 Trial. Lancet Oncol. 2010, 11, 121–128.
- Sequist, L.V.; Waltman, B.A.; Dias-Santagata, D.; Digumarthy, S.; Turke, A.B.; Fidias, P.; Bergethon, K.; Shaw, A.T.; Gettinger, S.; Cosper, A.K.; et al. Genotypic and Histological Evolution of Lung Cancers Acquiring Resistance to EGFR Inhibitors. Sci. Transl. Med. 2011, 3, 75ra26.
- Ohashi, K.; Maruvka, Y.E.; Michor, F.; Pao, W. Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitor–Resistant Disease. J. Clin. Oncol. 2013, 31, 1070–1080.
- Wu, S.-G.; Liu, Y.-N.; Tsai, M.-F.; Chang, Y.-L.; Yu, C.-J.; Yang, P.-C.; Yang, J.C.-H.; Wen, Y.-F.; Shih, J.-Y. The Mechanism of Acquired Resistance to Irreversible EGFR Tyrosine Kinase Inhibitor-Afatinib in Lung Adenocarcinoma Patients. Oncotarget 2016, 7, 12404–12413.
- He, J.; Huang, Z.; Han, L.; Gong, Y.; Xie, C. Mechanisms and Management of 3rd-generation EGFR-TKI Resistance in Advanced Non-small Cell Lung Cancer (Review). Int. J. Oncol. 2021, 59, 90.
- Ni, X.L.; Chen, L.X.; Zhang, H.; Yang, B.; Xu, S.; Wu, M.; Liu, J.; Yang, L.L.; Chen, Y.; Fu, S.Z.; et al. In Vitro and In Vivo Antitumor Effect of Gefitinib Nanoparticles on Human Lung Cancer. Drug Deliv. 2017, 24, 1501–1512.
- Langer, K.; Balthasar, S.; Vogel, V.; Dinauer, N.; von Briesen, H.; Schubert, D. Optimization of the Preparation Process for Human Serum Albumin (HSA) Nanoparticles. Int. J. Pharm. 2003, 257, 169–180.
- Dey, A.; Koli, U.; Dandekar, P.; Jain, R. Investigating Behaviour of Polymers in Nanoparticles of Chitosan Oligosaccharides Coated with Hyaluronic Acid. Polymer 2016, 93, 44–52.
- Pedrosa, S.S.; Pereira, P.; Correia, A.; Gama, F.M. Targetability of Hyaluronic Acid Nanogel to Cancer Cells: In Vitro and In Vivo Studies. Eur. J. Pharm. Sci. 2017, 104, 102–113.
- Shen, Y.; Li, W. HA/HSA Co-Modified Erlotinib–Albumin Nanoparticles for Lung Cancer Treatment. Drug Des. Devel. Ther. 2018, 12, 2285–2292.
- Li, K.; Zhan, W.; Jia, M.; Zhao, Y.; Liu, Y.; Jha, R.K.; Zhou, L. Dual Loading of Nanoparticles with Doxorubicin and Icotinib for the Synergistic Suppression of Non-Small Cell Lung Cancer. Int. J. Med. Sci. 2020, 17, 390–402.
- Elbatanony, R.S.; Parvathaneni, V.; Kulkarni, N.S.; Shukla, S.K.; Chauhan, G.; Kunda, N.K.; Gupta, V. Afatinib-Loaded Inhalable PLGA Nanoparticles for Localized Therapy of Non-Small Cell Lung Cancer (NSCLC)—Development and In-Vitro Efficacy. Drug Deliv. Transl. Res. 2021, 11, 927–943.
- Chen, W.; Yu, D.; Sun, S.-Y.; Li, F. Nanoparticles for Co-Delivery of Osimertinib and Selumetinib to Overcome Osimertinib-Acquired Resistance in Non-Small Cell Lung Cancer. Acta Biomater. 2021, 129, 258–268.
- Ali, A.A.A.; Hsu, F.-T.; Hsieh, C.-L.; Shiau, C.-Y.; Chiang, C.-H.; Wei, Z.-H.; Chen, C.-Y.; Huang, H.-S. Erlotinib-Conjugated Iron Oxide Nanoparticles as a Smart Cancer-Targeted Theranostic Probe for MRI. Sci. Rep. 2016, 6, 36650.
- Iwahara, T.; Fujimoto, J.; Wen, D.; Cupples, R.; Bucay, N.; Arakawa, T.; Mori, S.; Ratzkin, B.; Yamamoto, T. Molecular Characterization of ALK, a Receptor Tyrosine Kinase Expressed Specifically in the Nervous System. Oncogene 1997, 14, 439–449.
- Grande, E.; Bolós, M.-V.; Arriola, E. Targeting Oncogenic ALK: A Promising Strategy for Cancer Treatment. Mol. Cancer Ther. 2011, 10, 569–579.
- Cheng, Y.; Sun, Y.; Wang, L.-Z.; Yu, Y.-C.; Ding, X. Cytoplasmic C-Ros Oncogene 1 Receptor Tyrosine Kinase Expression May Be Associated with the Development of Human Oral Squamous Cell Carcinoma. Oncol. Lett. 2015, 10, 934–940.
- Doebele, R.C.; Pilling, A.B.; Aisner, D.L.; Kutateladze, T.G.; Le, A.T.; Weickhardt, A.J.; Kondo, K.L.; Linderman, D.J.; Heasley, L.E.; Franklin, W.A.; et al. Mechanisms of Resistance to Crizotinib in Patients with ALK Gene Rearranged Non–Small Cell Lung Cancer. Clin. Cancer Res. 2012, 18, 1472–1482.
- Yang, Y.-L.; Xiang, Z.-J.; Yang, J.-H.; Wang, W.-J.; Xiang, R.-L. Effect of Alectinib versus Crizotinib on Progression-Free Survival, Central Nervous System Efficacy and Adverse Events in ALK-Positive Non-Small Cell Lung Cancer: A Systematic Review and Meta-Analysis. Ann. Palliat. Med. 2020, 9, 1782–1796.
- Jiang, Z.-M.; Dai, S.-P.; Xu, Y.-Q.; Li, T.; Xie, J.; Li, C.; Zhang, Z.-H. Crizotinib-Loaded Polymeric Nanoparticles in Lung Cancer Chemotherapy. Med. Oncol. 2015, 32, 193.
- Zhong, T.; Liu, X.; Li, H.; Zhang, J. Co-Delivery of Sorafenib and Crizotinib Encapsulated with Polymeric Nanoparticles for the Treatment of in Vivo Lung Cancer Animal Model. Drug Deliv. 2021, 28, 2108–2118.
- Muller, I.B.; de Langen, A.J.; Giovannetti, E.; Peters, G.J. Anaplastic Lymphoma Kinase Inhibition in Metastatic Non-Small Cell Lung Cancer: Clinical Impact of Alectinib. OncoTargets Ther. 2017, 10, 4535–4541.
- Kodama, T.; Tsukaguchi, T.; Yoshida, M.; Kondoh, O.; Sakamoto, H. Selective ALK Inhibitor Alectinib with Potent Antitumor Activity in Models of Crizotinib Resistance. Cancer Lett. 2014, 351, 215–221.
- Larkins, E.; Blumenthal, G.M.; Chen, H.; He, K.; Agarwal, R.; Gieser, G.; Stephens, O.; Zahalka, E.; Ringgold, K.; Helms, W.; et al. FDA Approval: Alectinib for the Treatment of Metastatic, ALK-Positive Non–Small Cell Lung Cancer Following Crizotinib. Clin. Cancer Res. 2016, 22, 5171–5176.
- Brahmer, J.; Reckamp, K.L.; Baas, P.; Crinò, L.; Eberhardt, W.E.E.; Poddubskaya, E.; Antonia, S.; Pluzanski, A.; Vokes, E.E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 123–135.
- Brahmer, J.R.; Tykodi, S.S.; Chow, L.Q.M.; Hwu, W.-J.; Topalian, S.L.; Hwu, P.; Drake, C.G.; Camacho, L.H.; Kauh, J.; Odunsi, K.; et al. Safety and Activity of Anti–PD-L1 Antibody in Patients with Advanced Cancer. N. Engl. J. Med. 2012, 366, 2455–2465.
- Wu, Y.; Gu, W.; Li, J.; Chen, C.; Xu, Z.P. Silencing PD-1 and PD-L1 with Nanoparticle-Delivered Small Interfering RNA Increases Cytotoxicity of Tumor-Infiltrating Lymphocytes. Nanomedicine 2019, 14, 955–967.
- Vankayala, R.; Lin, C.-C.; Kalluru, P.; Chiang, C.-S.; Hwang, K.C. Gold Nanoshells-Mediated Bimodal Photodynamic and Photothermal Cancer Treatment Using Ultra-Low Doses of near Infra-Red Light. Biomaterials 2014, 35, 5527–5538.
- Jain, S.; Hirst, D.G.; O’Sullivan, J.M. Gold Nanoparticles as Novel Agents for Cancer Therapy. Br. J. Radiol. 2012, 85, 101–113.
- Fang, D.; Jin, H.; Huang, X.; Shi, Y.; Liu, Z.; Ben, S. 3O4 Nanoparticles Inhibit Tumor Growth and Metastasis Through Chemodynamic and Photothermal Therapy in Non-Small Cell Lung Cancer. Front. Chem. 2021, 9, 789934.
- Meng, Z.; Wang, B.; Liu, Y.; Wan, Y.; Liu, Q.; Xu, H.; Liang, R.; Shi, Y.; Tu, P.; Wu, H.; et al. Mitochondria-Targeting Polydopamine-Coated Nanodrugs for Effective Photothermal- and Chemo-Synergistic Therapies against Lung Cancer. Regen. Biomater. 2022, 9, rbac051.
- Juzenas, P.; Chen, W.; Sun, Y.-P.; Coelho, M.A.N.; Generalov, R.; Generalova, N.; Christensen, I.L. Quantum Dots and Nanoparticles for Photodynamic and Radiation Therapies of Cancer. Adv. Drug Deliv. Rev. 2008, 60, 1600–1614.
- Uprety, B.; Abrahamse, H. Semiconductor Quantum Dots for Photodynamic Therapy: Recent Advances. Front. Chem. 2022, 10, 946574.
- He, B.; Jin, H.-Y.; Wang, Y.-W.; Fan, C.-M.; Wang, Y.-F.; Zhang, X.-C.; Liu, J.-X.; Li, R.; Liu, J.-W. Carbon Quantum Dots/Bi4O5Br2 Photocatalyst with Enhanced Photodynamic Therapy: Killing of Lung Cancer (A549) Cells In Vitro. Rare Met. 2022, 41, 132–143.
- Chang, J.-E.; Yoon, I.-S.; Sun, P.-L.; Yi, E.; Jheon, S.; Shim, C.-K. Anticancer Efficacy of Photodynamic Therapy with Hematoporphyrin-Modified, Doxorubicin-Loaded Nanoparticles in Liver Cancer. J. Photochem. Photobiol. B 2014, 140, 49–56.
- Haume, K.; Rosa, S.; Grellet, S.; Śmiałek, M.A.; Butterworth, K.T.; Solov’yov, A.V.; Prise, K.M.; Golding, J.; Mason, N.J. Gold Nanoparticles for Cancer Radiotherapy: A Review. Cancer Nanotechnol. 2016, 7, 8.
- Ngwa, W.; Kumar, R.; Sridhar, S.; Korideck, H.; Zygmanski, P.; Cormack, R.A.; Berbeco, R.; Makrigiorgos, G.M. Targeted Radiotherapy with Gold Nanoparticles: Current Status and Future Perspectives. Nanomedicine 2014, 9, 1063–1082.
- Hao, Y.; Altundal, Y.; Moreau, M.; Sajo, E.; Kumar, R.; Ngwa, W. Potential for Enhancing External Beam Radiotherapy for Lung Cancer Using High-Z Nanoparticles Administered via Inhalation. Phys. Med. Biol. 2015, 60, 7035–7043.
- Kibria, G.; Hatakeyama, H.; Ohga, N.; Hida, K.; Harashima, H. Dual-Ligand Modification of PEGylated Liposomes Shows Better Cell Selectivity and Efficient Gene Delivery. J. Control. Release 2011, 153, 141–148.
- Wasungu, L.; Hoekstra, D. Cationic Lipids, Lipoplexes and Intracellular Delivery of Genes. J. Control. Release 2006, 116, 255–264.
- Nimesh, S. Polyethylenimine as a Promising Vector for Targeted SiRNA Delivery. Curr. Clin. Pharmacol. 2012, 7, 121–130.
- Mittal, P.; Vardhan, H.; Ajmal, G.; Bonde, G.V.; Kapoor, R.; Mittal, A.; Mishra, B. Formulation, Optimization, Hemocompatibility and Pharmacokinetic Evaluation of PLGA Nanoparticles Containing Paclitaxel. Drug Dev. Ind. Pharm. 2019, 45, 365–378.
- Duan, Y.; Shen, C.; Zhang, Y.; Luo, Y. Advanced Diagnostic and Therapeutic Strategies in Nanotechnology for Lung Cancer. Front. Oncol. 2022, 12, 1031000.
- Okusanya, O.T.; Holt, D.; Heitjan, D.; Deshpande, C.; Venegas, O.; Jiang, J.; Judy, R.; DeJesus, E.; Madajewski, B.; Oh, K.; et al. Intraoperative Near-Infrared Imaging Can Identify Pulmonary Nodules. Ann. Thorac. Surg. 2014, 98, 1223–1230.
More
Information
Subjects:
Oncology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
746
Revisions:
2 times
(View History)
Update Date:
09 Mar 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




