
Video Upload Options
Synthetic bone substitute materials (BSMs) are becoming the general trend of replacing autologous grafting for bone tissue engineering (BTE) in orthopedic research and clinical practice. As the main component of bone matrix, collagen type I has been playing a critical role in the construction of ideal synthetic BSMs for decades. Significant strides have been made in the field of collagen research, including the exploration of various collagen types, structure and sources, the optimization of preparation techniques, modification technologies, and the manufacture of various collagen-based materials. However, the poor mechanical properties, fast degradation, and lack of osteoconductive activity of collagen-based materials caused inefficient bone replacement and limited their translation into clinical reality. In the area of BTE, so far, attempts focused on the preparation of collagen-based biomimetic BSMs together with other inorganic materials and bioactive substances. By reviewing the approved products on the market, this manuscript updates the latest applications of collagen-based materials in bone regeneration and highlights the potential for further development in the field of BTE over the next ten years.
1. Introduction
2. Collagen in Native Bone Tissues
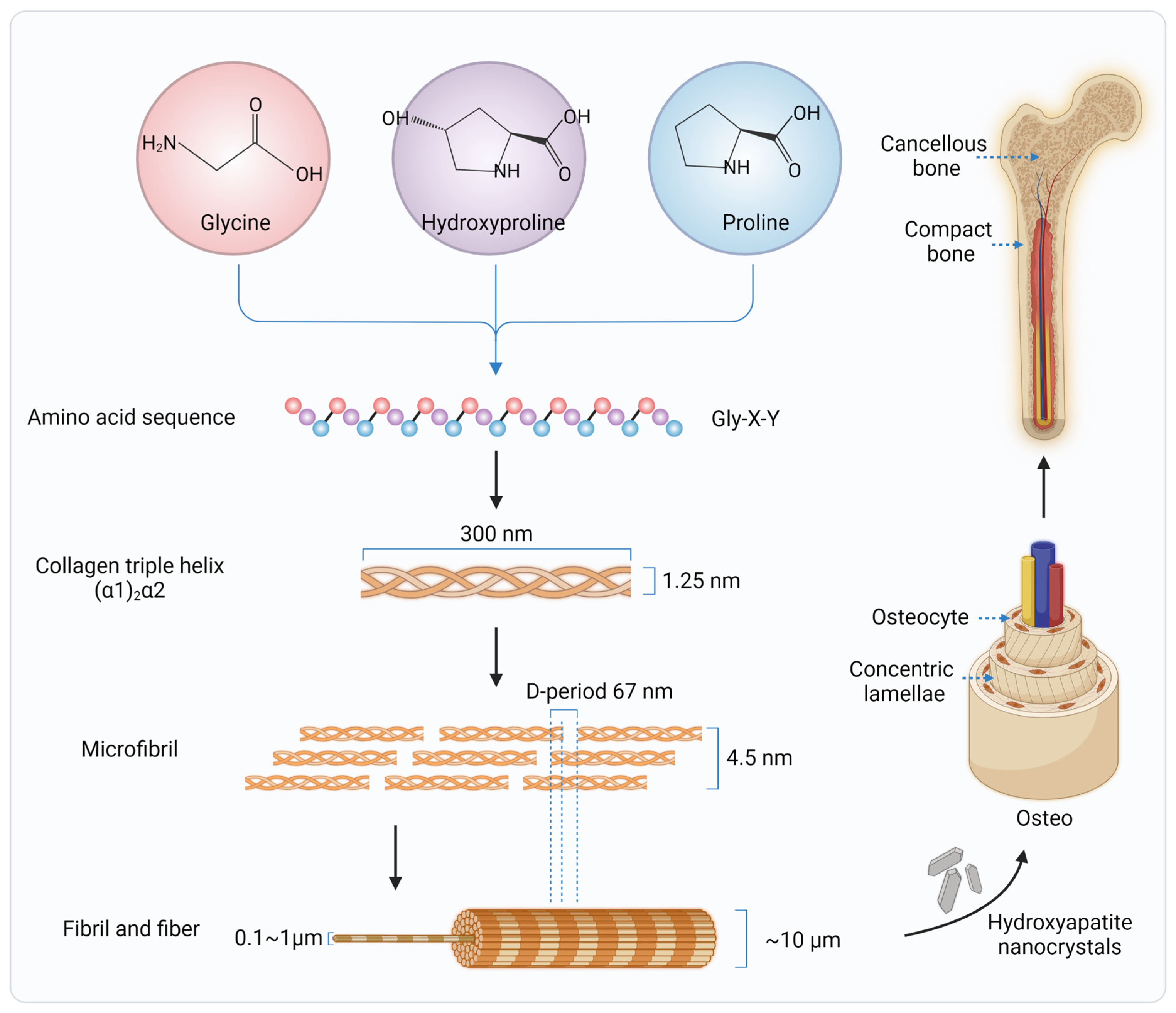
3. Applications of Collagen in Bone Tissue Regeneration and Engineering

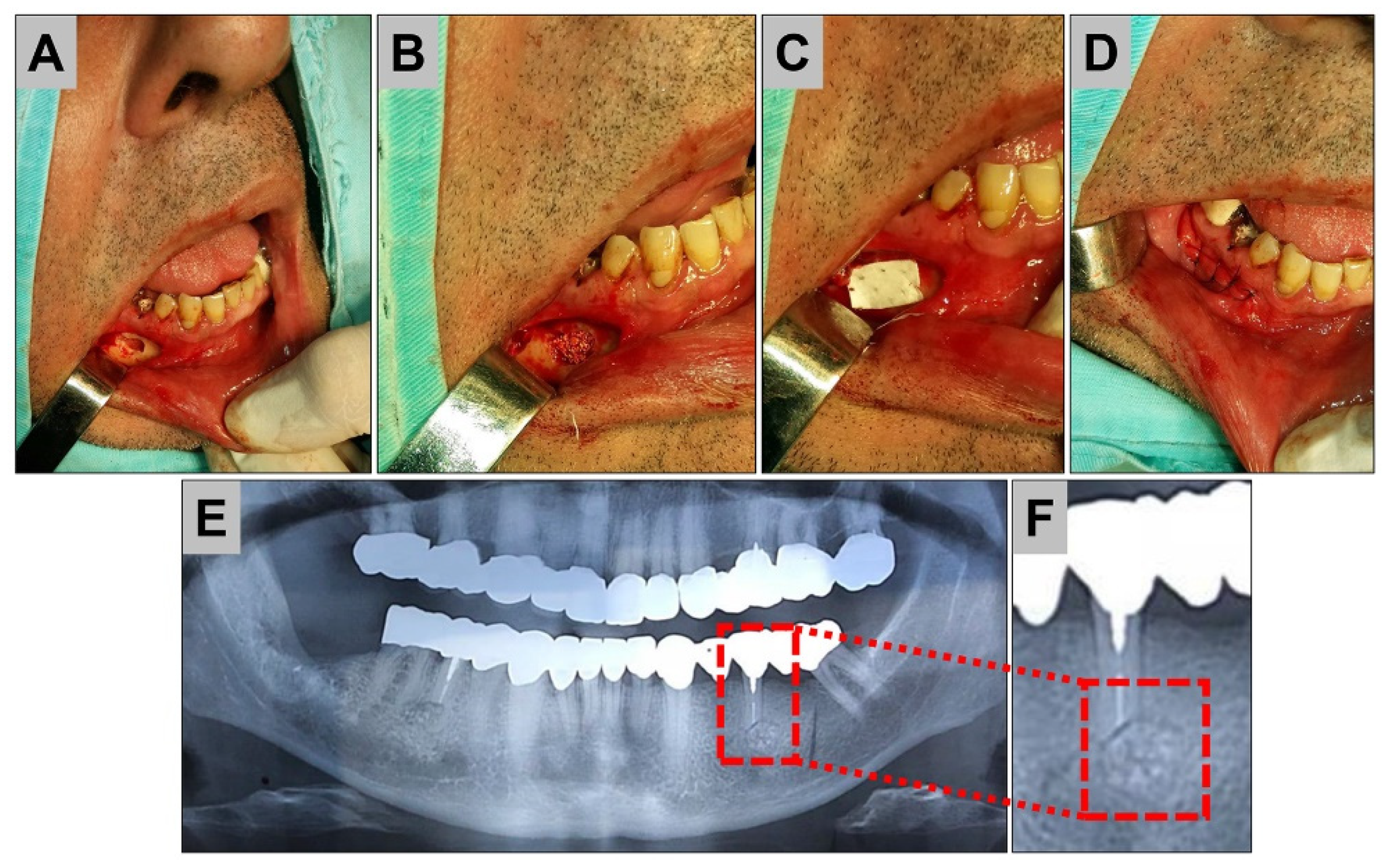
3.1. Collagen Membranes
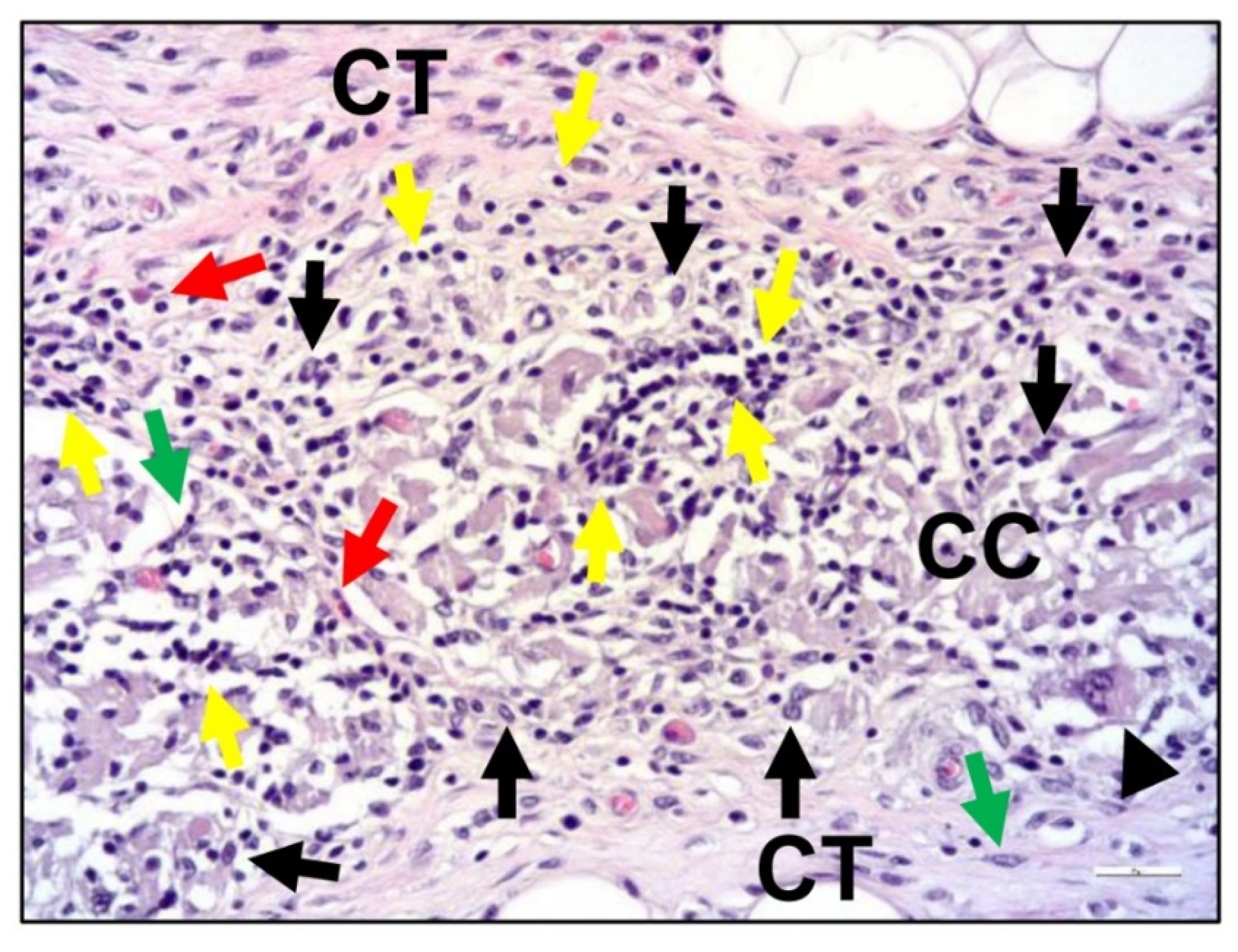
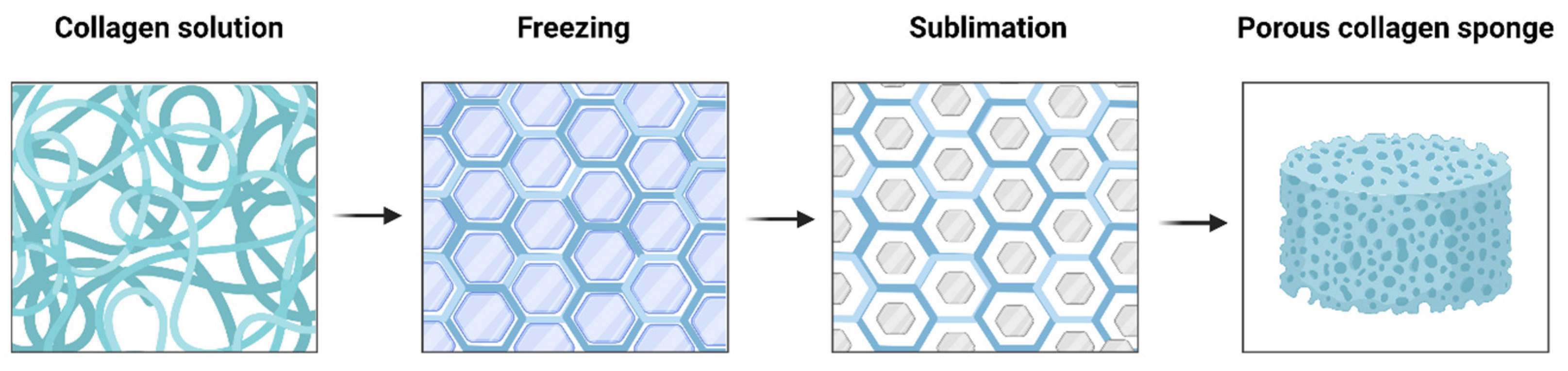
3.2. Collagen Sponges or Matrices
3.3. Collagen Hydrogels
3.4. Collagen-Based Composite Materials
3.4.1. Collagen Integrated with Organic or Inorganic Materials
3.4.2. Collagen-Based Composite Materials Loaded with Growth Factors, Cells, or Drugs
4. Conclusions and Perspectives
Significant strides have been made in the field of collagen (type I) for bone tissue engineering. Advances in exploring various collagen sources, as well as extraction and purification processes, have made collagen preparation available with maximum native structure and minimum immunogenicity/antigenicity. Advances in collagen modifications have offered optimized collagen mechanical properties and biological resistance. For a wide variety of applications in bone grafting, collagens are often modified or combined with other materials to construct various bone substitute materials, such as collagen sponges, hydrogels, nanofibers/microfibers, or nanoparticles/microspheres. Collagen-based composites with bioceramic materials (e.g., HA, TCP, and BGs, etc.), carried with growth factors, peptides, cells, drugs, and genes, have made the multifunctionalized BSMs available with improved osteoconductivity, osteoinductivity, osteointegration, osteogenesis, and vascularization. Although many of them have shown great effects on bone regeneration in in vitro and in vivo studies, only a few of them are currently FDA- or EU- approved for clinical applications. These composites still face challenges regarding complete mechanical properties, biological stability and activity, immune response, regional vascularization, and other safety issues for human bone regeneration, which hinder their translations into clinical devices. However, in addition to the continuous development of new tools and technologies for material manufacturing, such as 3D printing and electrospinning, researchers are directing attention to patient-specific collagen-based materials. Rather than relying on the material alone, an integration of functional material manufacture and the innate regeneration potential of patients sheds new light on potential clinical translation within the next decade.
References
- Ghelich, P.; Kazemzadeh-Narbat, M.; Najafabadi, A.H.; Samandari, M.; Memić, A.; Tamayol, A. (Bio)manufactured Solutions for Treatment of Bone Defects with Emphasis on US-FDA Regulatory Science Perspective. Adv. NanoBiomed Res. 2022, 2, 2100073.
- Sakkas, A.; Wilde, F.; Heufelder, M.; Winter, K.; Schramm, A. Autogenous bone grafts in oral implantology-is it still a “gold standard”? A consecutive review of 279 patients with 456 clinical procedures. Int. J. Implant. Dent. 2017, 3, 23.
- Miron, R.J.; Sculean, A.; Shuang, Y.; Bosshardt, D.D.; Gruber, R.; Buser, D.; Chandad, F.; Zhang, Y. Osteoinductive potential of a novel biphasic calcium phosphate bone graft in comparison with autographs, xenografts, and DFDBA. Clin. Oral Implant. Res. 2016, 27, 668–675.
- Li, Y.; Liu, Y.; Li, R.; Bai, H.; Zhu, Z.; Zhu, L.; Zhu, C.; Che, Z.; Liu, H.; Wang, J.; et al. Collagen-based biomaterials for bone tissue engineering. Mater. Des. 2021, 210, 110049.
- Guo, L.; Liang, Z.; Yang, L.; Du, W.; Yu, T.; Tang, H.; Li, C.; Qiu, H. The role of natural polymers in bone tissue engineering. J. Control Release 2021, 338, 571–582.
- Laird, N.Z.; Acri, T.M.; Tingle, K.; Salem, A.K. Gene- and RNAi-activated scaffolds for bone tissue engineering: Current progress and future directions. Adv. Drug Deliv. Rev. 2021, 174, 613–627.
- Ballarre, J.; Manjubala, I.; Schreiner, W.H.; Orellano, J.C.; Fratzl, P.; Ceré, S. Improving the osteointegration and bone-implant interface by incorporation of bioactive particles in sol-gel coatings of stainless steel implants. Acta Biomater. 2010, 6, 1601–1609.
- Bellucci, D.; Sola, A.; Cannillo, V. Hydroxyapatite and tricalcium phosphate composites with bioactive glass as second phase: State of the art and current applications. J. Biomed. Mater. Res. Part A 2016, 104, 1030–1056.
- Bharadwaz, A.; Jayasuriya, A.C. Recent trends in the application of widely used natural and synthetic polymer nanocomposites in bone tissue regeneration. Mater. Sci. Eng. C 2020, 110, 110698.
- Wahl, D.; Czernuszka, J. Collagen-hydroxyapatite composites for hard tissue repair. Eur. Cells Mater. 2006, 11, 43–56.
- Gillman, C.E.; Jayasuriya, A.C. FDA-approved bone grafts and bone graft substitute devices in bone regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 130, 112466.
- Chen, X.; Hao, W.; Li, X.; Xiao, Z.; Yao, Y.; Chu, Y.; Farkas, B.; Romano, I.; Brandi, F.; Dai, J. Functional Multichannel Poly(Propylene Fumarate)-Collagen Scaffold with Collagen-Binding Neurotrophic Factor 3 Promotes Neural Regeneration After Transected Spinal Cord Injury. Adv. Heal. Mater. 2018, 7, e1800315.
- Khan, S.N.; Cammisa, F.P., Jr.; Sandhu, H.S.; Diwan, A.D.; Girardi, F.P.; Lane, J.M. The biology of bone grafting. J. Am. Acad. Orthop. Surg. 2005, 13, 77–86.
- Rico-Llanos, G.A.; Borrego-González, S.; Moncayo-Donoso, M.; Becerra, J.; Visser, R. Collagen Type I Biomaterials as Scaffolds for Bone Tissue Engineering. Polymers 2021, 13, 599.
- Ma, C.; Wang, H.; Chi, Y.; Wang, Y.; Jiang, L.; Xu, N.; Wu, Q.; Feng, Q.; Sun, X. Preparation of oriented collagen fiber scaffolds and its application in bone tissue engineering. Appl. Mater. Today 2021, 22, 100902.
- Gurumurthy, B.; Janorkar, A.V. Improvements in mechanical properties of collagen-based scaffolds for tissue engineering. Curr. Opin. Biomed. Eng. 2020, 17, 100253.
- Ferreira, A.M.; Gentile, P.; Chiono, V.; Ciardelli, G. Collagen for bone tissue regeneration. Acta Biomater. 2012, 8, 3191–3200.
- Liu, X.; Zheng, C.; Luo, X.; Wang, X.; Jiang, H. Recent advances of collagen-based biomaterials: Multihierarchical structure, modification and biomedical applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 99, 1509–1522.
- Miller, E.J.; Gay, S. Collagen: An overview. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1982; pp. 3–32.
- Strom, S.C.; Michalopoulos, G. Collagen as a substrate for cell growth and differentiation. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1982; pp. 544–555.
- Wu, S.; Liu, X.; Yeung, K.W.K.; Liu, C.; Yang, X. Biomimetic porous scaffolds for bone tissue engineering. Mater. Sci. Eng. R Rep. 2014, 80, 1–36.
- Canty, E.G.; Kadler, K.E. Procollagen trafficking, processing and fibrillogenesis. J. Cell Sci. 2005, 118, 1341–1353.
- Brodsky, B.; Persikov, A.V. Molecular structure of the collagen triple helix. Adv. Protein Chem. 2005, 70, 301–339.
- Brodsky, B.; Ramshaw, J.A. The collagen triple-helix structure. Matrix Biol. 1997, 15, 545–554.
- Leung, M.K.; Fessler, L.I.; Greenberg, D.B.; Fessler, J.H. Separate amino and carboxyl procollagen peptidases in chick embryo tendon. J. Biol. Chem. 1979, 254, 224–232.
- Bornstein, P. The biosynthesis of collagen. Annu. Rev. Biochem. 1974, 43, 567–603.
- Olsen, B.R.; Hoffmann, H.-P.; Prockop, D.J. Interchain disulfide bonds at the COOH-terminal end of procollagen synthesized by matrix-free cells from chick embryonic tendon and cartilage. Arch. Biochem. Biophys. 1976, 175, 341–350.
- Gallop, P.M.; Blumenfeld, O.O.; Seifter, S. Structure and metabolism of connective tissue proteins. Annu. Rev. Biochem. 1972, 41, 617–672.
- Hodge, A.J.; Schmitt, F.O. The Charge Profile of the Tropocollagen Macromolecule and the Packing Arrangement in Native-Type Collagen Fibrils. Proc. Natl. Acad. Sci. USA 1960, 46, 186–197.
- Kadler, K.; Prockop, D.J. Protein structure and the specific heat of water. Nature 1987, 325, 395.
- Mould, A.P.; Hulmes, D.J.; Holmes, D.F.; Cummings, C.; Sear, C.H.; Chapman, J.A. D-periodic assemblies of type I procollagen. J. Mol. Biol. 1990, 211, 581–594.
- Chao, Y.-H.; Sun, J.-S. Biomechanics of Skeletal Muscle and Tendon. In Frontiers in Orthopaedic Biomechanics; Springer: Berlin/Heidelberg, Germany, 2020; pp. 37–73.
- Parenteau-Bareil, R.; Gauvin, R.; Berthod, F. Collagen-based biomaterials for tissue engineering applications. Materials 2010, 3, 1863–1887.
- Harley, B.A.; Gibson, L.J. In vivo and in vitro applications of collagen-GAG scaffolds. Chem. Eng. J. 2008, 137, 102–121.
- Ryan, E.J.; Ryan, A.J.; González-Vázquez, A.; Philippart, A.; Ciraldo, F.E.; Hobbs, C.; Nicolosi, V.; Boccaccini, A.R.; Kearney, C.J.; O’Brien, F.J. Collagen scaffolds functionalised with copper-eluting bioactive glass reduce infection and enhance osteogenesis and angiogenesis both in vitro and in vivo. Biomaterials 2019, 197, 405–416.
- Rütsche, D.; Nanni, M.; Rüdisser, S.; Biedermann, T.; Zenobi-Wong, M. Enzymatically Crosslinked Collagen as Versatile Matrix for In Vitro and In Vivo Co-Engineering of Blood and Lymphatic Vasculature. Adv. Mater. 2022. ahead of print.
- Elgali, I.; Omar, O.; Dahlin, C.; Thomsen, P. Guided bone regeneration: Materials and biological mechanisms revisited. Eur. J. Oral Sci. 2017, 125, 315–337.
- Cochran, D.L.; Hermann, J.S.; Schenk, R.K.; Higginbottom, F.L.; Buser, D. Biologic width around titanium implants. A histometric analysis of the implanto-gingival junction around unloaded and loaded nonsubmerged implants in the canine mandible. J. Periodontol. 1997, 68, 186–197.
- Thoma, D.S.; Bienz, S.P.; Figuero, E.; Jung, R.E.; Sanz-Martín, I. Efficacy of lateral bone augmentation performed simultaneously with dental implant placement: A systematic review and meta-analysis. J. Clin. Periodontol. 2019, 46, 257–276.
- Feng, S.; Zhong, Z.; Wang, Y.; Xing, W.; Drioli, E. Progress and perspectives in PTFE membrane: Preparation, modification, and applications. J. Membr. Sci. 2018, 549, 332–349.
- Almazrooa, S.A.; Noonan, V.; Woo, S.-B. Resorbable collagen membranes: Histopathologic features. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2014, 118, 236–240.
- Lee, C.H.; Singla, A.; Lee, Y. Biomedical applications of collagen. Int. J. Pharm. 2001, 221, 1–22.
- Kinoshita, S.; Mera, K.; Ichikawa, H.; Shimasaki, S.; Nagai, M.; Taga, Y.; Iijima, K.; Hattori, S.; Fujiwara, Y.; Shirakawa, J.-I.; et al. Nω-(carboxymethyl) arginine is one of the dominant advanced glycation end products in glycated collagens and mouse tissues. Oxidative Med. Cell. Longev. 2019, 2019, 9073451.
- Bourne, J.W.; Lippell, J.M.; Torzilli, P.A. Glycation cross-linking induced mechanical–enzymatic cleavage of microscale tendon fibers. Matrix Biol. 2014, 34, 179–184.
- Radenković, M.; Alkildani, S.; Stoewe, I.; Bielenstein, J.; Sundag, B.; Bellmann, O.; Jung, O.; Najman, S.; Stojanović, S.; Barbeck, M. Comparative In Vivo Analysis of the Integration Behavior and Immune Response of Collagen-Based Dental Barrier Membranes for Guided Bone Regeneration (GBR). Membranes 2021, 11, 712.
- Rahighi, R.; Panahi, M.; Akhavan, O.; Mansoorianfar, M. Pressure-engineered electrophoretic deposition for gentamicin loading within osteoblast-specific cellulose nanofiber scaffolds. Mater. Chem. Phys. 2021, 272, 125018.
- Liu, Y.; Zhang, Y.; An, Z.; Zhao, H.; Zhang, L.; Cao, Y.; Mansoorianfar, M.; Liu, X.; Pei, R. Slide-ring structure-based double-network hydrogel with enhanced stretchability and toughness for 3D-bio-printing and its potential application as artificial smalldiameter blood vessels. ACS Appl. Bio Mater. 2021, 4, 8597–8606.
- Rider, P.; Kačarević, P.; Elad, A.; Rothamel, D.; Sauer, G.; Bornert, F.; Windisch, P.; Hangyási, D.; Molnar, B.; Hesse, B.; et al. Analysis of a Pure Magnesium Membrane Degradation Process and Its Functionality When Used in a Guided Bone Regeneration Model in Beagle Dogs. Materials 2022, 15, 3106.
- Rider, P.; Kačarević, P.; Elad, A.; Tadic, D.; Rothamel, D.; Sauer, G.; Bornert, F.; Windisch, P.; Hangyási, D.B.; Molnar, B.; et al. Biodegradable magnesium barrier membrane used for guided bone regeneration in dental surgery. Bioact. Mater. 2022, 14, 152–168.
- Chvapil, M. Collagen sponge: Theory and practice of medical applications. J. Biomed. Mater. Res. 1977, 11, 721–741.
- Schoof, H.; Apel, J.; Heschel, I.; Rau, G. Control of pore structure and size in freeze-dried collagen sponges. J. Biomed. Mater. Res. 2001, 58, 352–357.
- Offeddu, G.; Ashworth, J.; Cameron, R.; Oyen, M. Structural determinants of hydration, mechanics and fluid flow in freeze-dried collagen scaffolds. Acta Biomater. 2016, 41, 193–203.
- Zhang, Y.; Wang, C.; Jiang, W.; Zuo, W.; Han, G. Influence of stage cooling method on pore architecture of biomimetic alginate scaffolds. Sci. Rep. 2017, 7, 16150.
- Murphy, C.M.; Haugh, M.G.; O’Brien, F.J. The effect of mean pore size on cell attachment, proliferation and migration in collagen–glycosaminoglycan scaffolds for bone tissue engineering. Biomaterials 2010, 31, 461–466.
- Loh, Q.L.; Choong, C. Three-dimensional scaffolds for tissue engineering applications: Role of porosity and pore size. Tissue Eng. Part B Rev. 2013, 19, 485–502.
- Briquez, P.S.; Tsai, H.-M.; Watkins, E.A.; Hubbell, J.A. Engineered bridge protein with dual affinity for bone morphogenetic protein-2 and collagen enhances bone regeneration for spinal fusion. Sci. Adv. 2021, 7, eabh4302.
- Acri, T.M.; Laird, N.Z.; Jaidev, L.R.; Meyerholz, D.K.; Salem, A.K.; Shin, K. Nonviral gene delivery embedded in biomimetically mineralized matrices for bone tissue engineering. Tissue Eng. Part A 2021, 27, 1074–1083.
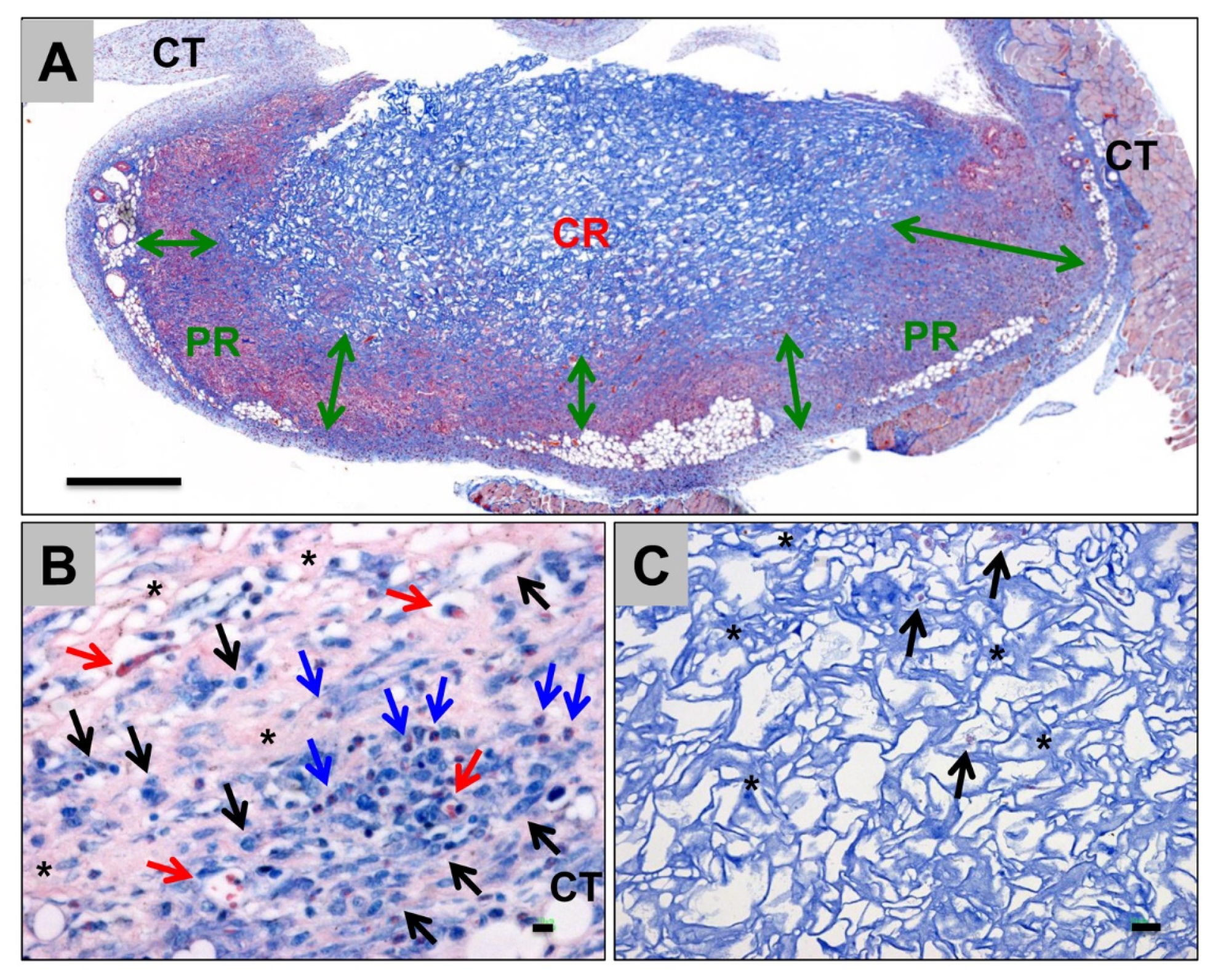
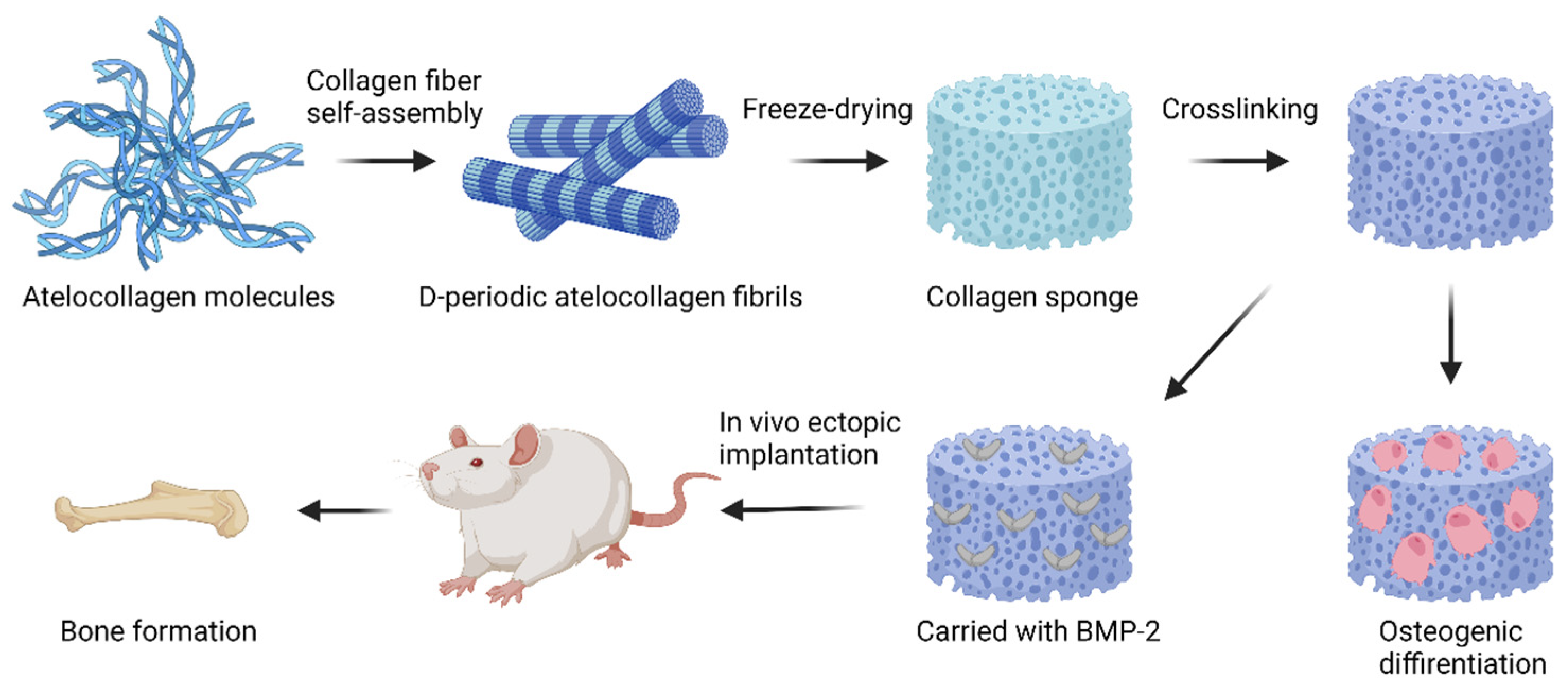
- Borrego-González, S.; Rico-Llanos, G.; Becerra, J.; Díaz-Cuenca, A.; Visser, R. Sponge-like processed D-periodic selfassembled atelocollagen supports bone formation in vivo. Mater. Sci. Eng. C 2021, 120, 111679.
- Cyr, J.A.; Husmann, A.; Best, S.M.; Cameron, R.E. Complex architectural control of ice-templated collagen scaffolds using a predictive model. Acta Biomater. 2022, 153, 260–272.
- Joukhdar, H.; Seifert, A.; Jüngst, T.; Groll, J.; Lord, M.S.; Rnjak-Kovacina, J. Ice templating soft matter: Fundamental principles and fabrication approaches to tailor pore structure and morphology and their biomedical applications. Adv. Mater. 2021, 33, 2100091.
- Xue, X.; Hu, Y.; Deng, Y.; Su, J. Recent advances in design of functional biocompatible hydrogels for bone tissue engineering. Adv. Funct. Mater. 2021, 31, 2009432.
- Nabavi, M.H.; Salehi, M.; Ehterami, A.; Bastami, F.; Semyari, H.; Tehranchi, M.; Semyari, H. A collagen-based hydrogel containing tacrolimus for bone tissue engineering. Drug Deliv. Transl. Res. 2020, 10, 108–121.
- Zeimaran, E.; Pourshahrestani, S.; Fathi, A.; Razak, N.A.B.A.; Kadri, N.A.; Sheikhi, A.; Baino, F. Advances in bioactive glass-containing injectable hydrogel biomaterials for tissue regeneration. Acta Biomater. 2021, 136, 1–36.
- Kadler, K.E.; Hojima, Y.; Prockop, D.J. Assembly of collagen fibrils de novo by cleavage of the type I pC-collagen with procollagen C-proteinase. Assay of critical concentration demonstrates that collagen self-assembly is a classical example of an entropy-driven process. J. Biol. Chem. 1987, 262, 15696–15701.
- Ngo, P.; Ramalingam, P.; Phillips, J.A.; Furuta, G.T. Collagen gel contraction assay. Cell-Cell Interact. 2006, 341, 103–109.
- Gharati, G.; Shirian, S.; Sharifi, S.; Mirzaei, E.; Bakhtirimoghadam, B.; Karimi, I.; Nazari, H. Comparison capacity of collagen hydrogel and collagen/strontium bioglass nanocomposite scaffolds with and without mesenchymal stem cells in regeneration of critical sized bone defect in a rabbit animal model. Biol. Trace Elem. Res. 2022, 200, 3176–3186.
- Yin, B.; Yang, H.; Yang, M. Integrating Soft Hydrogel with Nanostructures Reinforces Stem Cell Adhesion and Differentiation. J. Compos. Sci. 2022, 6, 19.
- Magister, S.; Kolaczko, J.; Sattar, A.; Wetzel, R.J. Clinical parameters and radiographic resorption of a novel magnesium based bone void filler. Injury 2022, 53, 947–952.
- Yang, X.; Wang, X.; Yu, F.; Ma, L.; Pan, X.; Luo, G.; Lin, S.; Mo, X.; He, C.; Wang, H. Hyaluronic acid/EDC/NHS-crosslinked green electrospun silk fibroin nanofibrous scaffolds for tissue engineering. RSC Adv. 2016, 6, 99720–99728.
- Lu, Z.; Liu, S.; Le, Y.; Qin, Z.; He, M.; Xu, F.; Zhu, Y.; Zhao, J.; Mao, C.; Zheng, L. An injectable collagen-genipin-carbon dot hydrogel combined with photodynamic therapy to enhance chondrogenesis. Biomaterials 2019, 218, 119190.
- Amirian, J.; Makkar, P.; Lee, G.H.; Paul, K.; Lee, B.T. Incorporation of alginate-hyaluronic acid microbeads in injectable calcium phosphate cement for improved bone regeneration. Mater. Lett. 2020, 272, 127830.
- Gélébart, P.; Cuenot, S.; Sinquin, C.; Halgand, B.; Sourice, S.; Le Visage, C.; Guicheux, J.; Colliec-Jouault, S.; Zykwinska, A. Microgels based on Infernan, a glycosaminoglycanmimetic bacterial exopolysaccharide, as BMP-2 delivery systems. Carbohydr. Polym. 2022, 284, 119191.
- Glenske, K.; Donkiewicz, P.; Köwitsch, A.; Milosevic-Oljaca, N.; Rider, P.; Rofall, S.; Franke, J.; Jung, O.; Smeets, R.; Schnettler, R.; et al. Applications of Metals for Bone Regeneration. Int. J. Mol. Sci. 2018, 19, 826.
- Kargozar, S.; Mozafari, M.; Ghodrat, S.; Fiume, E.; Baino, F. Copper-containing bioactive glasses and glassceramics: From tissue regeneration to cancer therapeutic strategies. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 121, 111741.
- Kaur, K.; Paiva, S.S.; Caffrey, D.; Cavanagh, B.L.; Murphy, C.M. Injectable chitosan/collagen hydrogels nano-engineered with functionalized single wall carbon nanotubes for minimally invasive applications in bone. Mater. Sci. Eng. C 2021, 128, 112340.
- McKay, W.F.; Peckham, S.M.; Badura, J.M. A comprehensive clinical review of recombinant human bone morphogenetic protein-2 (INFUSE® Bone Graft). Int. Orthop. 2007, 31, 729–734.
- James, A.W.; LaChaud, G.; Shen, J.; Asatrian, G.; Nguyen, V.; Zhang, X.; Ting, K.; Soo, C. A review of the clinical side effects of bone morphogenetic protein-2. Tissue Eng. Part B Rev. 2016, 22, 284–297.
- Oryan, A.; Alidadi, S.; Moshiri, A.; Bigham-Sadegh, A. Bone morphogenetic proteins: A powerful osteoinductive compound with non-negligible side effects and limitations. Biofactors 2014, 40, 459–481.
- Schneider, H.; Sedaghati, B.; Naumann, A.; Hacker, M.C.; Schulz-Siegmund, M. Gene silencing of chordin improves BMP-2 effects on osteogenic differentiation of human adipose tissue-derived stromal cells. Tissue Eng. Part A 2014, 20, 335–345.
- Wong, D.S.H.; Li, J.; Yan, X.; Wang, B.; Li, R.; Zhang, L.; Bian, L. Magnetically Tuning Tether Mobility of Integrin Ligand Regulates Adhesion, Spreading, and Differentiation of Stem Cells. Nano Lett. 2017, 17, 1685–1695.
- Wong, S.H.D.; Yin, B.; Yang, B.; Lin, S.; Li, R.; Feng, Q.; Yang, H.; Zhang, L.; Yang, Z.; Li, G.; et al. Anisotropic Nanoscale Presentation of Cell Adhesion Ligand Enhances the Recruitment of Diverse Integrins in Adhesion Structures and Mechanosensing-Dependent Differentiation of Stem Cells. Adv. Funct. Mater. 2019, 29, 1806822.
- Chamieh, F.; Collignon, A.-M.; Coyac, B.R.; Lesieur, J.; Ribes, S.; Sadoine, J.; Llorens, A.; Nicoletti, A.; Letourneur, D.; Colombier, M.-L.; et al. Accelerated craniofacial bone regeneration through dense collagen gel scaffolds seeded with dental pulp stem cells. Sci. Rep. 2016, 6, 38814.
- Park, H.; Collignon, A.-M.; Lepry, W.C.; Ramirez-GarciaLuna, J.L.; Rosenzweig, D.H.; Chaussain, C.; Nazhat, S.N. Acellular dense collagen-S53P4 bioactive glass hybrid gel scaffolds form more bone than stem cell delivered constructs. Mater. Sci. Eng. C 2021, 120, 111743.
- Raftery, R.M.; Walsh, D.P.; Ferreras, L.B.; Castaño, I.M.; Chen, G.; LeMoine, M.; Osman, G.; Shakesheff, K.M.; Dixon, J.E.; O’Brien, F.J. Highly versatile cell-penetrating peptide loaded scaffold for efficient and localised gene delivery to multiple cell types: From development to application in tissue engineering. Biomaterials 2019, 216, 119277.
- Boda, S.K.; Almoshari, Y.; Wang, H.; Wang, X.; Reinhardt, R.A.; Duan, B.; Wang, D.; Xie, J. Mineralized nanofiber segments coupled with calciumbinding BMP-2 peptides for alveolar bone regeneration. Acta Biomater. 2019, 85, 282–293.
- Egberink, R.O.; Zegelaar, H.M.; El Boujnouni, N.; Versteeg, E.M.M.; Daamen, W.F.; Brock, R. Biomaterial-Mediated Protein Expression Induced by Peptide-mRNA Nanoparticles Embedded in Lyophilized Collagen Scaffolds. Pharmaceutics 2022, 14, 1619.
- Wang, P.; Perche, F.; Midoux, P.; Cabral, S.; Malard, V.; Correia, I.J.; Ei-Hafci, H.; Petite, H.; Logeart-Avramoglou, D.; Pichon, C. In Vivo bone tissue induction by freeze-dried collagennanohydroxyapatite matrix loaded with BMP2/NS1 mRNAs lipopolyplexes. J. Control. Release 2021, 334, 188–200.
- Fan, L.; Körte, F.; Rudt, A.; Jung, O.; Burkhardt, C.; Barbeck, M.; Xiong, X. Encapsulated vaterite-calcite CaCO3 particles loaded with Mg2+ and Cu2+ ions with sustained release promoting osteogenesis and angiogenesis. Front. Bioeng. Biotechnol. 2022, 10, 983988.





