Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Maria João Rodrigues | -- | 1493 | 2023-02-10 16:19:33 | | | |
| 2 | Jessie Wu | Meta information modification | 1493 | 2023-02-13 06:08:04 | | | | |
| 3 | Jessie Wu | Meta information modification | 1493 | 2023-02-13 06:08:26 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Rodrigues, M.J.; Pereira, C.; Oliveira, M.; Zengin, G.; Custódio, L. Halophyte Plants as Sources of Anthelmintic Agents. Encyclopedia. Available online: https://encyclopedia.pub/entry/41102 (accessed on 10 May 2026).
Rodrigues MJ, Pereira C, Oliveira M, Zengin G, Custódio L. Halophyte Plants as Sources of Anthelmintic Agents. Encyclopedia. Available at: https://encyclopedia.pub/entry/41102. Accessed May 10, 2026.
Rodrigues, Maria João, Catarina Pereira, Marta Oliveira, Gokhan Zengin, Luísa Custódio. "Halophyte Plants as Sources of Anthelmintic Agents" Encyclopedia, https://encyclopedia.pub/entry/41102 (accessed May 10, 2026).
Rodrigues, M.J., Pereira, C., Oliveira, M., Zengin, G., & Custódio, L. (2023, February 10). Halophyte Plants as Sources of Anthelmintic Agents. In Encyclopedia. https://encyclopedia.pub/entry/41102
Rodrigues, Maria João, et al. "Halophyte Plants as Sources of Anthelmintic Agents." Encyclopedia. Web. 10 February, 2023.
Copy Citation
Parasitic diseases, especially those caused by protozoans and helminths, such as malaria, trypanosomiasis, leishmaniasis, Chagas disease, schistosomiasis, onchocerciasis, and lymphatic filariasis, are the cause of millions of morbidities and deaths every year, mainly in tropical regions. Nature has always provided valuable antiparasitic agents, and efforts targeting the identification of antiparasitic drugs from plants have mainly focused on glycophytes. Salt-tolerant plants (halophytes) have attracted the interest of the scientific community due to their medicinal assets, which include antiparasitic properties.
plants
antiparasitic agents
1. Introduction
Human parasitic diseases are predominantly linked to tropical or subtropical areas. However, climate change and the increased mobility of humans and animals trigger vector migration and the upsurge of parasitic infections in developed countries. Parasitic diseases continue to play a major role in human health, particularly infections caused by protozoans and helminths, such as malaria, trypanosomiasis, leishmaniasis, Chagas disease, schistosomiasis, onchocerciasis, lymphatic filariasis, and helminthiases, which are common in tropical regions and cause millions of morbidities and deaths every year [1][2].
For centuries, nature has been a source of medicines for the treatment of a vast array of diseases, with the first records of the ethnomedicinal uses of plants dating back to 2600 BC in Mesopotamia [3]. The easy access to terrestrial plants helps to explain their popularity as a source of bioactive products and innovative drug leads used in the pharmaceutical industry [4]. The importance of plant-based molecules as antiparasitic agents was reinforced by the Nobel Prize laureates in physiology or medicine in 2015, where Youyou Tu was honored for her discovery of artemisinin, a novel drug for the therapy against malaria, which was derived from Artemisia annua L., a plant used in the Chinese traditional medicine [5].
Most research targeting the identification of antiparasitic agents has focused on glycophytes, but salt-tolerant plants (halophytes) have lately aroused the interest of the scientific community due to their multiple medicinal properties, including antiparasitic activities. The harsh habitats where halophytes thrive, such as salt marshes, maritime dunes, and marine cliffs, expose them to extremely variable abiotic conditions, including salinity, light intensity, drought, and temperature [6][7]. This stressful environment contributes to the synthesis and accumulation of bioactive metabolites, including phenolics, alkaloids, and terpenes, conferring to halophyte species important medicinal properties, such as antioxidant, anti-inflammatory, antimicrobial, anti-tumoral, anti-infective, and antiparasitic activities [8][9][10][11][12][13][14][15]. In fact, several halophyte species are used as medicinal (e.g., Mesembryanthemum edule L. (syn. Carpobrotus edulis L.), and/or dietary plants (e.g., Chenopodium quinoa Willd, Salicornia spp.), mainly in rural areas where traditional medicine is the only source of health treatments. Moreover, as the problems associated with the salinization of soils and water bodies and the increasing competition for scarce freshwater resources increase [16][17], recruiting wild halophytes with economic potential is one of the suggested strategies to reduce the damage caused by the salinization of soil and water [18]. While most of the crop species used in traditional agriculture are salt sensitive (glycophytes), having a 10% yield decrease as the soil salinity increases over the 4–8 dS/m range, the growth of several halophytes is stimulated within a salinity range of 15–25 dS/m [18]. Halophytes are, therefore, a real strategy as alternative highly salt-tolerant crops that can cope with adverse saline conditions, to be used in the exploitation of degraded agricultural lands with the irrigation with brackish water for sustainable water management and soil conservation when establishing cost-efficient and environmental-friendly agro-ecosystems.
Reports on the traditional medicinal uses of halophytes comprise 43 families and more than 180 species in the Mediterranean, the Arabian Sea, and Syrian regions [12][19][20]. Some of these halophytes, such as Chenopodium album and Artemisia ramosissima have ethnomedicinal uses for parasitic diseases, including protozoal and helminthic infections [12][18][19], and several scientific reports confirmed their activities by in vitro and in vivo methods and identified their main bioactive constituents.
2. In Vitro Activities and Bioactive Constituents
Most of the reports on the anthelmintic activity of halophytes were performed only in vitro, and only less than half included a chemical characterization of its major constituents. Twelve species from 9 families have been described with in vitro anthelmintic properties, and those reports are included in Table 1.
Oliveira and colleagues [21] have screened 80% acetone extracts from 8 halophyte species for their anthelmintic capacity using the Larval Ensheathment Inhibition Assay (LEIA) and Egg Hatching Inhibition Assay (EHIA) for T. colubriformis. P. lentiscus, L. monopetalum, C. mariscus, and H. italicum picardi were the most active in both GINs and life stages of the nematodes [21]. In particular, C. mariscus aerial parts collected during summer were more active against T. colubriformis (EC50 = 77.8 µg/mL) [22]. Moreover, inflorescences presented the highest anthelmintic activity against T. colubriformis [EC50 (LEIA) = 78.6 µg/mL; IC50 (EHIA) = 848.2 µg/mL]. Flavan-3-ols, proanthocyanidins, luteolin, and glycosylated flavonoids are the main active components [22]. Some authors have also tested some halophyte extracts against S. mansoni, which is a helminth species and the causal agent of schistosomiasis in humans. Methanol extracts of C. ambrosioides reduced S. mansoni cercariae infectivity [23], as well as essential oils from F. vulgare that showed moderate in vitro activity against S. mansoni worms, but with more remarkable effects in egg development, possibly attributable to the two main constituents detected in the essential oil, I-anethole and limonene [24]. A compound isolated from G. inflata, licochalcone A (Figure 1), presented an LC50 of 9 μM towards S. mansoni female and male adult worms [25].
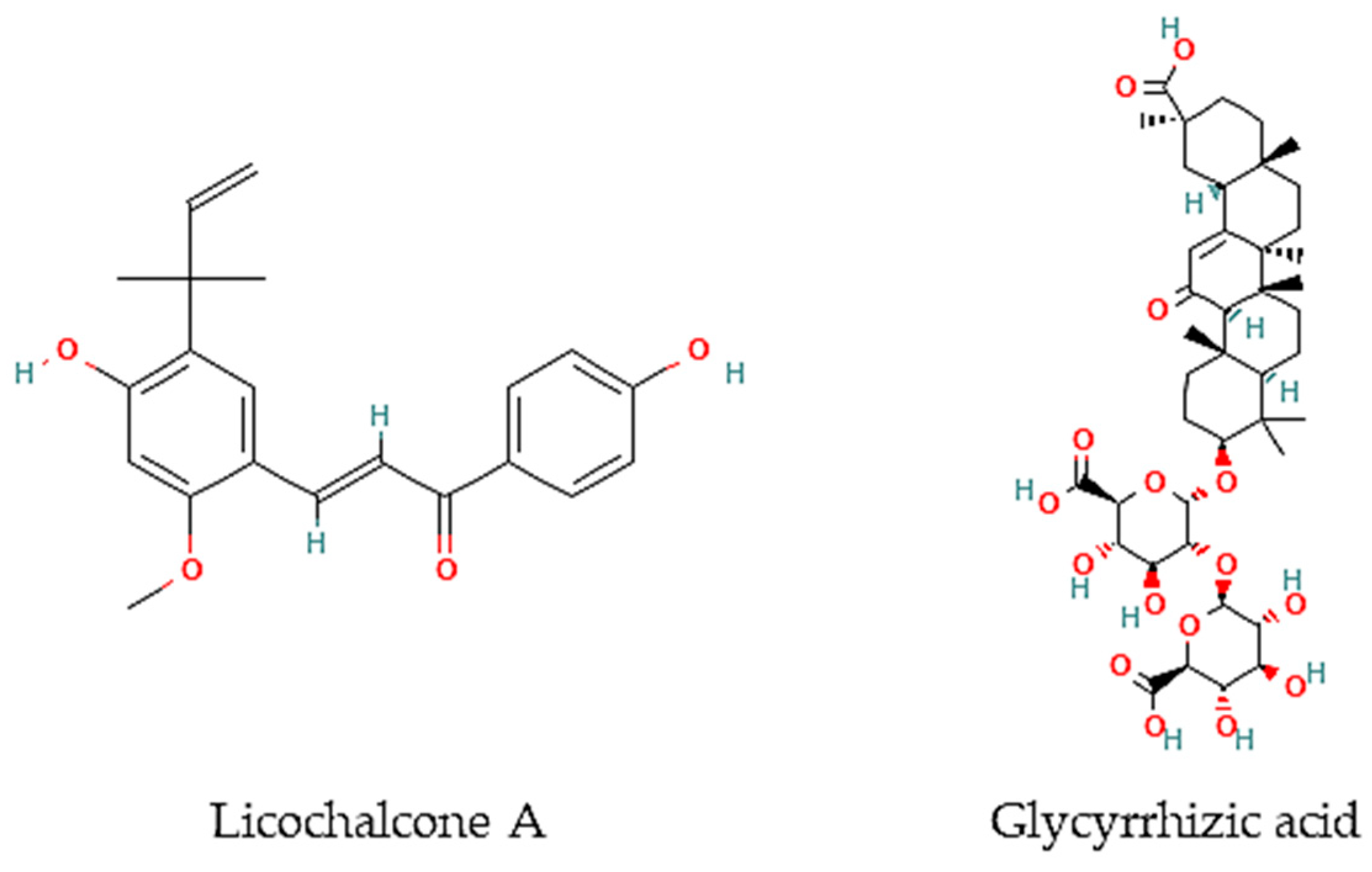
Figure 1. Chemical structures of pure compounds reported with anthelmintic properties.
Besides, other authors have focused on other less frequently tested parasite species. For instance, the roots of G. glabra contain glycyrrhizic acid (Figure 2) that is very active in vitro against B. malayi microfilarae (IC50 = 1.20 μM), one of the most important causative agents of human lymphatic filariasis [26].
Table 1. In vitro anthelmintic activity of halophyte species.
| Family/Species | Plant Organ | Extract/Fraction/Compound | Chemical Components | Assay | Results | References |
|---|---|---|---|---|---|---|
| Apiaceae | ||||||
| Foeniculum vulgare Mill. | Fresh leaves | Essential oil | I-anethole and limonene | Schistosoma mansoni adult worms (pairs) and eggs | 50% activity at 100,000 µg/mL (24 and 120 h) | [24] |
| Asteraceae | ||||||
| Helichrysum italicum (Roth) G. Don subsp. picardi (Boiss. & Reut.) Franco |
Aerial parts | 80% acetone extract | Caffeoylquinic and dicaffeoylquinic acids and quercetin glycosides | Trichostrongylus colubriformis | IC50 (LEIA) = 132 µg/mL; IC50 (EHIA) = 3707 µg/mL | [21] |
| Inula crithmoides L. | Aerial parts | 80% acetone extract | ND | Trichostrongylus colubriformis | IC50 (LEIA) = 1031 µg/mL | [21] |
| Cyperaceae | ||||||
| Cladium mariscus L. Pohl | Aerial parts | 80% acetone extract | Proanthocyanins, phenolic acids, and luteolin |
Trichostrongylus colubriformis | IC50 (LEIA) = 77.8 µg/mL; IC50 (EHIA) = 2575 µg/mL | [21] |
| Aerial parts, leaves, and inflorescences collected during spring, summer, autumn, and winter | 80% acetone extract | Flavan-3-ols, proanthocyanidins, luteolin, and glycosylated flavonoids |
Trichostrongylus colubriformis | Summer: EC50 (LEIA) = 77.8 µg/mL; Spring: IC50 (EHIA) = 2275 µg/mL; Leaves: EC50 (LEIA) = 81.1 µg/mL; IC50 (EHIA) = 2289 µg/mL; Inflorescences: EC50 (LEIA) = 78.6 µg/mL; IC50 (EHIA) = 848 µg/mL |
[22] | |
| Convolvulaceae | ||||||
| Calystegia soldanela (L.) R. Br. | Aerial parts | 80% acetone extract | ND | Trichostrongylus colubriformis | IC50 (LEIA) = 2711 µg/mL | [21] |
| Fabaceae | ||||||
| Glycyrrhiza glabra L. | Roots | Glycyrrhizic acid | Brugia malayi microfilarae in vitro | IC50 = 1.20 μM | [26] | |
| Glycyrrhiza inflata Batalin | ND | Licochalcone A | S. mansoni (female and male adult worms) | LC50 = 9 μM | [25] | |
| Medicago marina L. | Aerial parts | 80% acetone extract | ND | Trichostrongylus colubriformis | IC50 (LEIA) = 211 µg/mL | [21] |
| Plantaginaceae | ||||||
| Plantago coronopus L. | Aerial parts | 80% acetone extract | ND | Trichostrongylus colubriformis | IC50 (LEIA) = 212 µg/mL | [21] |
| Plumbaginaceae | ||||||
| Limoniuastrum monopetalum (L.) Boiss. | Aerial parts | 80% acetone extract | Sulphated and/or methylated flavonoids | Trichostrongylus colubriformis | IC50 (LEIA) = 1024 µg/mL; IC50 (EHIA) = 2102 µg/mL | [21] |
| Poaceae | ||||||
| Cynodon dactylon (L.) Pers. | ND | Methanol extract | ND | Hymenolepis diminuta |
40,000 µg/mL: paralysis and mortality at 4.12 h and 5.16 h, respectively | [27] |
| Rubiaceae | ||||||
| Crucianella marítima L. | Aerial parts | 80% acetone extract | ND | Trichostrongylus colubriformis | IC50 (LEIA) = 1024 µg/mL | [27] |
ND: not determined.
3. In Vivo Studies
There is a reduced number of studies focusing on in vivo anthelmintic properties of halophytic species. In fact, to our best knowledge, only three species were tested, mostly using rodents and sheep as models (Table 2).
Pistacia lentiscus is the most studied halophyte species due to its high level of tannins, known to exhibit high anthelmintic activity, as confirmed by in vivo studies against T. colubriformis. Infected goats fed with aerials parts of P. lentiscus presented a 16% reduction in fecal oocyst counts, whereas, in lambs, the reduction ranged between 55.2 and 61.3% [28][29]. In turn, S. mansoni infected mice treated orally with a methanol extract of C. ambrosioides (1250 mg/kg/day) exhibited a 53.7% total worm burden decrease and a 60.3% ova/g tissue in liver reduction, 9 weeks post-infection [30]. Hymenolepis diminuta infected Wistar rats were treated with C. dactylon methanol extract (800 mg/kg) enabling a reduction of 77.6% in eggs and 79% in adult worms counts, respectively [27].
Table 2. In vivo anthelmintic activity of halophyte species.
| Family/Species | Plant Organ | Extract/Fraction/Compound | Chemical Components | Assay | Results | References |
|---|---|---|---|---|---|---|
| Amaranthaceae | ||||||
| Dysphania ambrosioides (L.) Mosyakin & Clemants (syn. Chenopodium ambrosioides L.) | ND | Methanol | ND | S. mansoni infected mice | 1250 mg/kg/day exhibited a 53.7% total worm burden decrease and a 60.3% ova/g tissue in liver reduction | [23][30] |
| Anacardiaceae | ||||||
| Pistacia lentiscus L. | Aerial parts | ND | Tannins | Teladorsagia circumcincta, Trichostrongylus colubriformis, and Chabertia ovina infected goats | Reduced fecal oocyst counts in approx. 16% | [29] |
| ND | Tannins | T. colubriformis infected lambs | Reduction of 55.2–61.3% on faecal egg counts | [28] | ||
| Poaceae | ||||||
| Cynodon dactylon (L.) Pers. | ND | Methanol extract | ND | Hymenolepis diminuta infected Wistar rats |
800 mg/kg: 77.6 and 79% reduction in egg and worms’ reduction, respectively | [27] |
ND: Not determined.
References
- Wink, M. Medicinal plants: A source of anti-parasitic secondary metabolites. Molecules 2012, 17, 12771–12791.
- Pisarski, K. The Global Burden of Disease of Zoonotic Parasitic Diseases: Top 5 Contenders for Priority Consideration. Trop. Med. Infect. Dis. 2019, 4, 44.
- Cragg, G.M.; Newman, D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta 2013, 1830, 3670–3695.
- Mishra, B.B.; Tiwari, V.K. Natural products: An evolving role in future drug discovery. Eur. J. Med. Chem. 2011, 46, 4769–4807.
- The Nobel Prize. Available online: https://www.nobelprize.org/prizes/medicine/2015/tu/facts/ (accessed on 28 December 2022).
- Flowers, T.J.; Colmer, T.D. Salinity tolerance in halophytes. N. Phytol. 2008, 179, 945–963.
- Grigore, M.N.; Villanueva, M.; Boscaiu, M.; Vicente, O. Do halophytes really require salts for their growth and development? An experimental approach. Not. Sci. Biol. 2012, 4, 23–29.
- Menezes-Benavente, L.; Teixeira, F.K.; Kamei, C.L.A.; Margis-Pinheiro, M. Salt stress induces altered expression of genes encoding antioxidant enzymes in seedlings of a Brazilian indica rice (Oryza sativa L.). Plant Sci. 2004, 166, 323–331.
- Amor, N.B.; Jimé Nez, A.B.; Megdiche, W.; Lundqvist, M.; Sevilla, F.; Abdelly, C. Response of antioxidant systems to NaCl stress in the halophyte Cakile maritima. Physiol. Plant. 2006, 126, 446–457.
- Jaleel, C.A.; Manivannan, P.; Whid, A.; Farooq, M.; Al-Juburi, H.J.; Somasundaram, R.; Panneerselvam, R. Drought stress in plants: A review on morphological characteristics and pigments composition. Int. J. Agric. Biol. 2009, 11, 100–105.
- Flowers, T.J.; Galal, H.K.; Bromham, L. Evolution of halophytes: Multiple origins of salt tolerance in land plants. Funct. Plant Biol. 2010, 37, 604–612.
- Ksouri, R.; Ksouri, W.M.; Jallali, I.; Debez, A.; Magné, C.; Hiroko, I.; Abdelly, C. Medicinal halophytes: Potent source of health promoting biomolecules with medical, nutraceutical and food applications. Crit. Rev. Biotechnol. 2012, 32, 289–326.
- Petropoulos, S.A.; Karkanis, A.; Martins, N.; Ferreira, I.C.F.R. Halophytic herbs of the Mediterranean basin: An alternative approach to health. Food Chem. Toxicol. 2018, 114, 155–169.
- Custodio, M.; Moulaert, I.; Asselman, J.; van der Biest, K.; van de Pol, L.; Drouillon, M.; Everaert, G. Prioritizing ecosystem services for marine management through stakeholder engagement. Ocean Coast. Manag. 2022, 225, 106228.
- Ferreira, M.J.; Pinto, D.C.G.A.; Cunha, Â.; Silva, H. Halophytes as medicinal plants against human infectious diseases. Appl. Sci. 2022, 12, 7493.
- Panta, S.; Flowers, T.; Lane, P.; Doyle, R.; Haros, G.; Shabala, S. Halophyte agriculture: Success stories. Environ. Exp. Bot. 2014, 107, 71–83.
- Loconsole, D.; Murillo-Amador, B.; Cristiano, G.; De Lucia, B. Halophyte Common Ice Plants: A future solution to arable land salinization. Sustainability 2019, 11, 6076.
- Ventura, Y.; Eshel, A.; Pasternak, D.; Sagi, M. The development of halophyte-based agriculture: Past and present. Ann. Bot. 2015, 115, 529–540.
- Al-Oudat, M.; Qadir, M. The Halophytic Flora of Syria; International Center for Agricultural Research in the Dry Areas (ICARDA): Beirut, Lebanon, 2011.
- Qasim, M.; Abideen, Z.; Adnan, M.Y.; Gulzar, S.; Gul, B.; Rasheed, M.; Khan, M.A. Antioxidant properties, phenolic composition, bioactive compounds and nutritive value of medicinal halophytes commonly used as herbal teas. South Afr. J. Bot. 2017, 110, 240–250.
- Oliveira, M.; Lima, C.S.; Ketavong, S.; Llorent-Martínez, E.J.; Hoste, H.; Custódio, L. Disclosing the bioactive metabolites involved in the In Vitro anthelmintic effects of salt-tolerant plants through a combined approach using PVPP and HPLC-ESI-MSn. Sci. Rep. 2021, 11, 24303.
- Oliveira, M.; Lima, C.S.; Llorent-Martínez, E.J.; Hoste, H.; Custódio, L. Impact of seasonal and organ-related fluctuations on anthelmintic properties and chemical profile of Cladium mariscus (L.) Pohl extracts. Front. Plant Sci. 2022, 13, 934644.
- Kamel, E.G.; El-Emam, M.A.; Mahmoud, S.S.M.; Fouda, F.M.; Bayaumy, F.E. Attenuation of Schistosoma mansoni cercarial infectivity to albino mice by methanol extract of some plant species. Pestic. Biochem. Physiol. 2010, 98, 342–348.
- Wakabayashi, K.A.; de Melo, N.I.; Aguiar, D.P.; de Oliveira, P.F.; Groppo, M.; da Silva Filho, A.A.; Rodrigues, V.; Cunha, W.R.; Tavares, D.C.; Magalhães, L.G.; et al. Anthelmintic effects of the essential oil of fennel (Foeniculum vulgare Mill., Apiaceae) against Schistosoma mansoni. Chem. Biodivers. 2015, 12, 1105–1114.
- Souza, R.L.; Gonçalves, U.O.; Badoco, F.R.; de Souza Galvão, L.; Santos, R.; de Carvalho, P.; de Carvalho, L.; da Silva Filho, A.A.; Veneziani, R.; Rodrigues, V.; et al. Licochalcone A induces morphological and biochemical alterations in Schistosoma mansoni adult worms. Biomed. Pharmacother. 2017, 96, 64–71.
- Kalani, K.; Kushwaha, V.; Verma, R.; Murthy, P.K.; Srivastava, S.K. Glycyrrhetinic acid and its analogs: A new class of antifilarial agents. Bioorg. Med. Chem. Lett. 2013, 23, 2566–2570.
- Yadav, A.K.; Nath, P. Anthelmintic effects and toxicity of Cynodon dactylon (L.) Pers. in rodent models. J. Intercult. Ethnopharmacol. 2017, 6, 407–413.
- Manolaraki, F.; Sotiraki, S.; Stefanakis, A.; Skampardonis, V.; Volanis, M.; Hoste, H. Anthelmintic activity of some Mediterranean browse plants against parasitic nematodes. Parasitology 2010, 137, 685–696.
- Markovics, A.; Cohen, I.; Muklada, H.; Glasser, T.A.; Dvash, L.; Ungar, E.D.; Azaizeh, H.; Landau, S.Y. Consumption of Pistacia lentiscus foliage alleviates coccidiosis in young goats. Vet. Parasitol. 2012, 186, 165–169.
- Kamel, E.G.; El-Emam, M.A.; Mahmoud, S.S.M.; Fouda, F.M.; Bayaumy, F.E. Parasitological and biochemical parameters in Schistosoma mansoni-infected mice treated with methanol extract from the plants Chenopodium ambrosioides, Conyza dioscorides and Sesbania sesban. Parasitol. Int. 2011, 60, 388–392.
More
Information
Subjects:
Parasitology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
861
Revisions:
3 times
(View History)
Update Date:
13 Feb 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





