
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yoshimi Niwano | -- | 6439 | 2023-01-20 03:17:53 | | | |
| 2 | Yoshimi Niwano | -3082 word(s) | 3357 | 2023-01-25 23:15:29 | | | | |
| 3 | Yoshimi Niwano | -1905 word(s) | 1452 | 2023-01-25 23:18:37 | | | | |
| 4 | Rita Xu | -26 word(s) | 1426 | 2023-01-28 04:23:55 | | |
Video Upload Options
Proanthocyanidins (PACs), which are oligomers or polymers of flavan-3ols with potent antioxidative activity, are well known to exert a variety of beneficial health effects.
1. Introduction
Proanthocyanidins (PACs), also known as condensed tannins, are substances that produce red anthocyanidin pigments when decomposed by acid and are oligomers or polymers of flavan-3-ols, such as epicatechin and catechin. They are widely distributed in fruits, grains, and leaves [1][2][3][4][5], especially in cocoa, black soybeans, cinnamon, apples, and grape seeds [6]. In addition, grape seed PACs have an average degree of polymerization (DP) between 2 and 17 [1]. In in vivo studies, PACs alleviated severe acute pancreatitis in mice via their anti-inflammatory properties [7], exerted anti-obesity and anti-diabetic activity in a type 2 diabetes model of KKAy mice [8] and anti-obesity activity in a mouse model of high-fat-diet-induced obesity [9], and showed neuroprotective activity in zebrafish and rat models of Parkinson’s disease [10][11]. Researchers previously demonstrated that orally administered grape seed PACs prevented bone loss in the lumbar vertebrae and femur in ovariectomized (OVX) mice, and they ameliorated the healing of defects created on the calvaria and osseointegration of a tibial implant in OVX rats, likely by counteracting the accelerated osteoclastogenic activity induced by estrogen deficiency [12]. To attain a better understanding of such health-beneficial activities, pharmacokinetic analysis is imperative. However, there is a paucity of evidence related to the structural complexity of PACs.
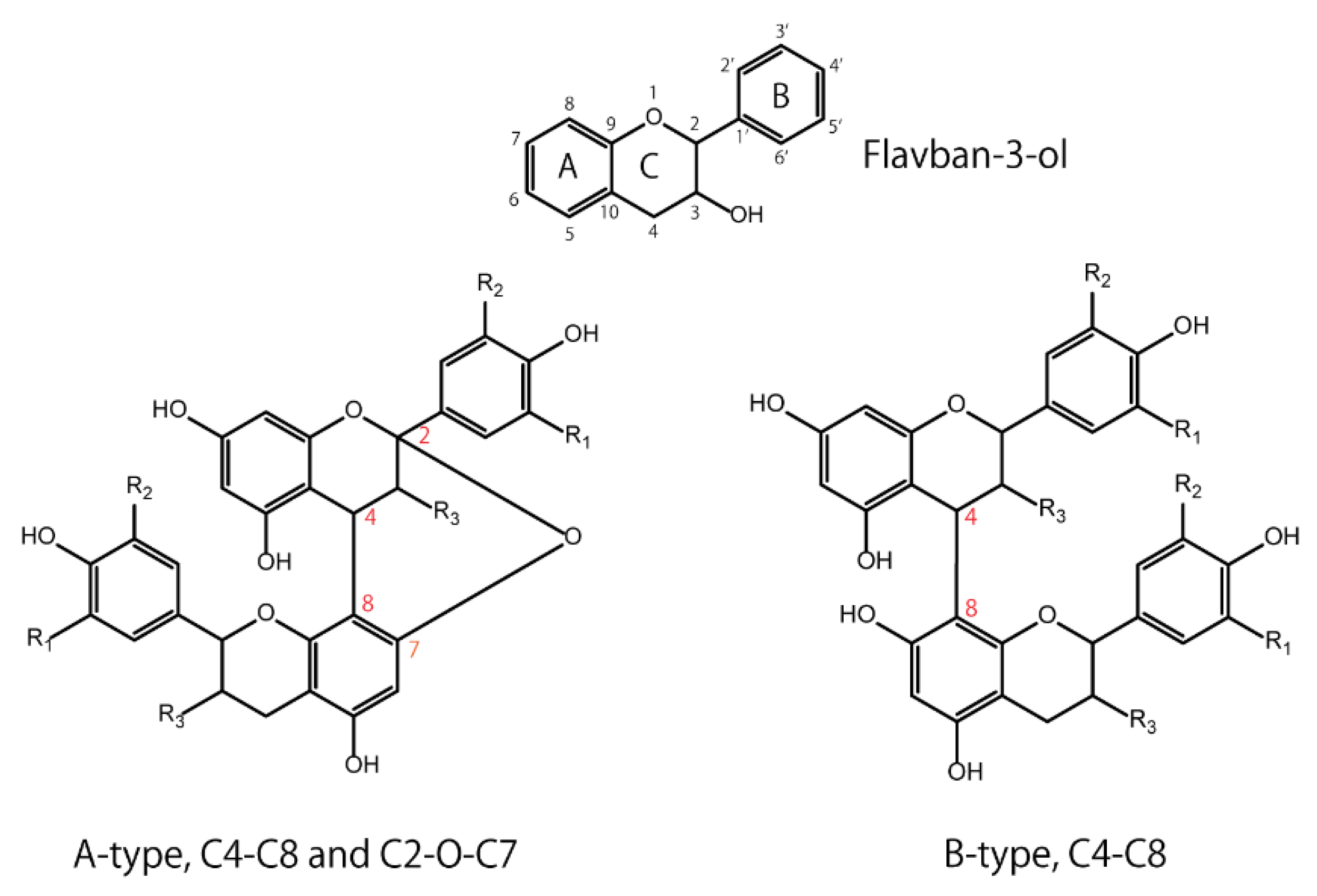
2. Basic Structures of Proanthocyanidins (PACs)
Flavan-3-ols have a basic structure consisting of A, B, and C rings, in which 3, 5, 7, 3′, or 4′ is hydroxylated. For example, the 3-hydroxylated group has two conformations: the 2,3-cis isomer is (−)-epicatechin, and the 2,3-trans isomer is (+)-catechin. Oligomers are formed by C4–C8 or C2–O–C7 bonds between monomers with these basic structures. The isomers are roughly divided into two groups according to their binding modes—those with C4-C8 or C4-C6 bonds are called B-type, and those with additional C2-O-C7 bonds are called A-type (Figure 1). Naturally occurring B-type PACs are predominant in plants such as cocoa, bayberry, and grapes [13][14][15].
3. Metabolic Fate of Orally Ingested Proanthocyanidins (PACs) Is a Key to Their Health Benefit Effects
3.1. Gastric Stability
To investigate the gastric stability of PACs, several in vitro studies using simulated gastric juice were conducted, but the results were controversial. PAC oligomers (trimer to hexamer) purified from cocoa were hydrolyzed to mixtures of epicatechin monomer and dimer [16], apple dimeric PAC B2 was almost completely degraded into (−)-epicatechin [17], and the PAC content in an extract of Hypericum perfoliatum L. significantly decreased by 25% [18]. On the contrary, other studies reported that PACs with a high DP (mean DP ≥ 6) from grape seeds were remarkably stable in the gastric environment and did not degrade into more readily absorbable monomers [19][20], PACs from Acacia mearnsii remained stable during gastric digestion in vitro [21], and the mean DP of PACs isolated from Choerospondias axillaris peel was not affected [22]. A human in vivo study showed that cocoa beverage PACs were stable during gastric transit, with the pH of gastric contents increasing from 1.9 ± 0.2 to 5.4 ± 0.2 after consumption [23]. In summary, PACs are depolymerized to some extent under gastric conditions and then pass into the small intestine.
3.2. Small-Intestinal Stability and Absorption
The first step after gastric digestion is exposure to pancreatic juice in the duodenum. It was reported that slight depolymerization of PACs could be observed in an in vitro small-intestinal model that used pancreatic enzymes and bile salts [20]. A similar in vitro study showed that the mean DP of PACs was slightly decreased, which was possibly due to interactions with digestive enzymes [22]. Collectively, PACs were rather chemically stable with respect to depolymerization during their passage through the simulated duodenal digestion.
3.3. Colonic Stability and Absorption
When PACs reach the colon, they are likely to be affected by gut microbiota. In in vitro fermentation of grape seed extracts that were rich in B-type PACs, the maximum formation of intermediate metabolites, such as valerolactones, valeric acid, several phenolic acids, and gallic acid, was observed at 5–10 h of incubation with fecal microbiota. Subsequently, the incubations (10–48 h) resulted in the appearance of mono- and non-hydroxylated forms of previous metabolites, which was likely due to dehydroxylation reactions [24][25]. These in vitro results were also consistent with those from a human study. When humans consumed a test drink containing PACs with a DP ranging from 2 to 10, γ-valerolactones were mainly detected in the plasma [26], thus rejecting the notion that PACs are broken down into flavanols prior to their absorption. Since two rat studies showed that ingested polymeric PACs were present in the colon as the intact parent compounds [27][28], the parent compounds and γ-valerolactones, as microbiota-generated catabolites of PACs, likely contribute to some health-beneficial effects following oral ingestion of PACs.
3.4. Effects on Gut Microbiota
Apart from bacterial transformation, PACs could affect the gut microbiota. It was reported that Lachnospiraceae and Ruminococcus are the major butyrate and propionate producers in human fecal samples [29], and butyrate can modulate oxidative stress in the colonic mucosa of healthy humans [30]. Butyrate was reported to lead to more specific and efficacious therapeutic strategies for the prevention and treatment of different diseases ranging from genetic/metabolic conditions to neurological degenerative disorders [31]. In particular, in a human study, the transfer of intestinal microbiota from lean donors increased insulin sensitivity in individuals with metabolic syndrome along with levels of butyrate-producing intestinal microbiota, suggesting that intestinal microbiota should be developed as therapeutic agents for increasing insulin sensitivity in humans [32]. If PACs have the ability to increase butyrate producers, they may work not only for colonic health, but also for systemic health. More recently, it was reviewed that PACs have a prebiotic and antimicrobial role that favors homeostasis of the intestinal environment, thus reducing the survival of Gram-negative bacteria that produce lipopolysaccharide (LPS) [33]. As LPS triggers the activation of the Toll-like receptor-4 (TLR-4) inflammatory pathway, PACs can minimize endotoxemia.
As for animal studies under pathological conditions, most studies applied high-fat diet (HFD)- or high-fat/high-sucrose diet (HFHSD)-induced metabolic syndrome model animals. PAC-rich grape seed/pomace extract [34][35][36], PAC-rich cranberry extract [37], and apple PACs [38] showed improved symptoms of metabolic syndrome concomitantly with an altered gut microbial environment. Some studies revealed that PACs increase Akkermansia muciniphila [34][36] or Akkermansia at the genus level [38], the former of which is a well-known beneficial gut bacterium that improves host metabolic functions and immune responses [39][40][41][42][43][44][45]. Accounting for 3–5% of the microbial community in healthy individuals, A. muciniphila is a mucinolytic bacterium found in the mucus layer of the human gut [46], and it has the potential to restore mucus thickness and intestinal barrier integrity [47][48]. This bacterium also has the ability to decrease the progression of many diseases, such as obesity and type 2 diabetes mellitus [49][50]. As such, A. muciniphila is considered a promising probiotic candidate [44]. At the phylum level, PACs could decrease the ratio of Firmicutes/Bacteroidetes [35][38]. The dominant gut microbial phyla are Firmicutes, Bacteroidetes, Actinobacteria, Proteobacteria, Fusobacteria, and Verrucomicrobia, with the first two phyla being the most common in healthy human individuals [51]. Phylum-level analyses of Firmicutes and Bacteroidetes have shown that they are associated with obesity and that an increased population of Bacteroidetes, as well as a reduced population of Firmicutes, could improve obesity [52][53][54][55][56]. In a human study, the relative proportion of Bacteroidetes was decreased in obese people in comparison with that in lean people, and this proportion increased with weight loss with two types of low-calorie diets [57]. Aside from HFD- or HFHSD-fed animals, PACs normalized the imbalanced Firmicutes/Bacteroidetes ratio observed in OVX mice in a menopause model and prevented OVX animals from having an increased weight [58].
The aforementioned metabolic fate of PACs through the digestive tract and their health-beneficial effects in association with gut microbiota are summarized in Figures 2 and 3, respectively.
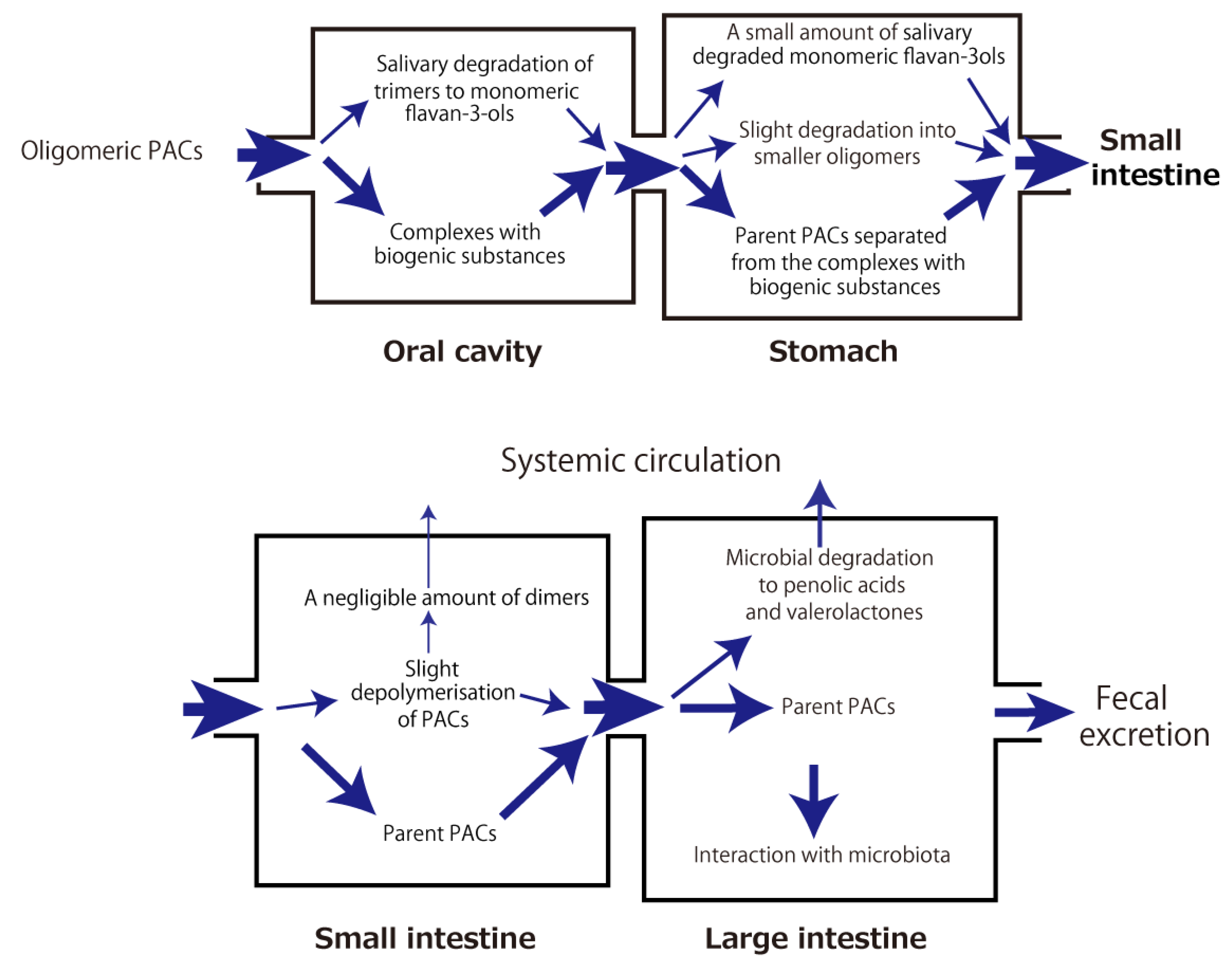
Figure 2. Metabolic fate of orally ingested proanthocyanidins
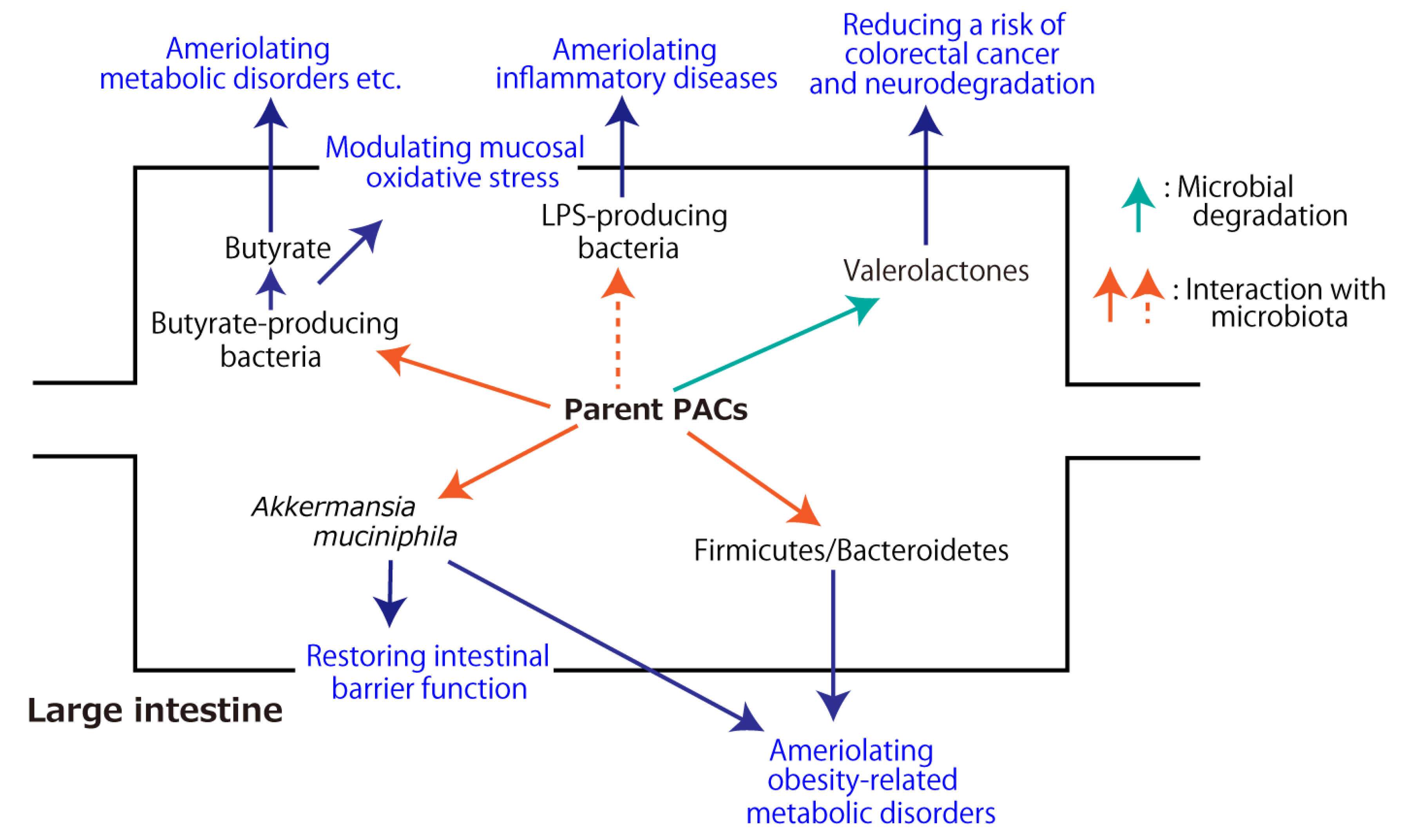
Figure 3. Health-beneficial effects of proanthocyanidins (PACs) associated with gut microbiota. The dashed arrow indicates the inhibitory effect.
4. Summarized concept of health benefit effects of orally ingested proanthocyanidins
A large portion of orally ingested oligomeric and polymeric PACs reach the colon, where a small portion of them are subjected to microbial degradation into phenolic acids and valerolactones. The rest interact with gut microbiota, resulting in improved microbial diversity, which includes an increased amount of beneficial gut bacteria (e.g., Akkermansia muciniphila), which could ameliorate host metabolic functions, and a lowered ratio of Firmicutes/Bacteroidetes at the phylum level, which could mitigate obesity-related metabolic disorders. Further, PACs have the potential to increase butyrate-producing microbiota and decrease LPS-producing bacteria, leading to the prevention and treatment of different diseases ranging from metabolic conditions to neurological degenerative disorders. These microbial changes could confer some of PACs’ health-beneficial effects.
References
- Prieur, C.; Rigaud, J.; Cheynier, V.; Moutounet, M. Oligomeric and polymeric procyanidins from grape seeds. Phytochemistry 1994, 36, 781–784.
- Bao, J.; Cai, Y.; Sun, M.; Wang, G.; Corke, H. Anthocyanins, flavonols, and free radical scavenging activity of Chinese bay-berry (Myrica rubra) extracts and their color properties and stability. J. Agric. Food Chem. 2005, 53, 2327–2332.
- Spranger, I.; Sun, B.; Mateus, A.M.; Freitas, V.; Ricardo-da-Silva, J.M. Chemical characterization and antioxidant activities of oligomeric and polymeric procyanidin fractions from grape seeds. Food Chem. 2008, 108, 519–532.
- Mkandawire, N.L.; Kaufman, R.C.; Bean, S.R.; Weller, C.L.; Jackson, D.S.; Rose, D.J. Effects of sorghum (Sorghum bicolor (L.) Moench) tannins on α-amylase activity and in vitro digestibility of starch in raw and processed flours. J. Agric Food Chem. 2013, 61, 4448–4454.
- Fu, Y.; Qiao, L.; Cao, Y.; Zhou, X.; Liu, Y.; Ye, X. Structural elucidation and antioxidant activities of proanthocyanidins from Chinese bayberry (Myrica rubra Sieb. et Zucc.) leaves. PLoS ONE 2014, 9, e96162.
- Gu, L.; Kelm, M.A.; Hammerstone, J.F.; Beecher, G.; Holden, J.; Haytowitz, D.; Gebhardt, S.; Prior, R.L. Concentrations of proanthocyanidins in common foods and estimations of normal consumption. J. Nutr. 2004, 134, 613–617.
- Sheng, L.P.; Han, C.Q.; Ling, X.; Guo, X.W.; Lin, R.; Ding, Z. Proanthocyanidins suppress NLRP3 inflammasome and M1 macrophage polarization to alleviate severe acute pancreatitis in mice. J. Biochem. Mol. Toxicol. 2022, e23242.
- Kashiwada, M.; Nakaishi, S.; Usuda, A.; Miyahara, Y.; Katsumoto, K.; Katsura, K.; Terakado, I.; Jindo, M.; Nakajima, S.; Ogawa, S.; et al. Analysis of anti-obesity and anti-diabetic effects of acacia bark-derived proanthocyanidins in type 2 diabetes model KKAy mice. J. Nat. Med. 2021, 75, 893–906.
- Yu, Y.; Chen, P.; Li, X.; Shen, S.; Li, K. Persimmon proanthocyanidins with different degrees of polymerization possess distinct activities in models of high fat diet induced obesity. Nutrients 2022, 14, 3718.
- Chen, J.; Chen, Y.; Zheng, Y.; Zhao, J.; Yu, H.; Zhu, J.; Li, D. Protective effects and mechanisms of procyanidins on Parkinson’s disease in vivo and in vitro. Molecules 2021, 26, 5558.
- Zhang, Y.; Huang, N.; Chen, M.; Jin, H.; Nie, J.; Shi, J.; Jin, F. Procyanidin protects against 6-hydroxydopamine-induced dopaminergic neuron damage via the regulation of the PI3K/Akt signaling pathway. Biomed. Pharmacother. 2019, 114, 108789.
- Tenkumo, T.; Aobulikasimu, A.; Asou, Y.; Shirato, M.; Shishido, S.; Kanno, T.; Niwano, Y.; Sasaki, K.; Nakamura, K. Pro-anthocyanidin-rich grape seed extract improves bone loss, bone healing, and implant osseointegration in ovariectomized animals. Sci. Rep. 2020, 10, 8812.
- Tomas-Barberan, F.A.; Cienfuegos-Jovellanos, E.; Marín, A.; Muguerza, B.; Gil-Izquierdo, A.; Cerda, B.; Zafrilla, P.; Morillas, J.; Mulero, J.; Ibarra, A.; et al. A new process to develop a cocoa powder with higher flavonoid monomer content and enhanced bioavailability in healthy humans. J. Agric. Food Chem. 2007, 55, 3926–3935.
- Yang, H.; Ye, X.; Liu, D.; Chen, J.; Zhang, J.; Shen, Y.; Yu, D. Characterization of unusual proanthocyanidins in leaves of bayberry (Myrica rubra Sieb. et Zucc.). J. Agric. Food Chem. 2011, 59, 1622–1629.
- Fujimaki, T.; Mori, S.; Horikawa, M.; Fukui, Y. Isolation of proanthocyanidins from red wine, and their inhibitory effects on melanin synthesis in vitro. Food Chem. 2018, 248, 61–69.
- Spencer, J.P.; Chaudry, F.; Pannala, A.S.; Srai, S.K.; Debnam, E.; Rice-Evans, C. Decomposition of cocoa procyanidins in the gastric milieu. Biochem. Biophys. Res. Commun. 2000, 272, 236–241.
- Kahle, K.; Kempf, M.; Schreier, P.; Scheppach, W.; Schrenk, D.; Kautenburger, T.; Hecker, D.; Huemmer, W.; Ackermann, M.; Richling, E.; et al. Intestinal transit and systemic metabolism of apple polyphenols. Eur. J. Nutr. 2011, 50, 507–522.
- Celep, E.; İnan, Y.; Akyüz, S.; Yesilada, E. The bioaccessible phenolic profile and antioxidant potential of Hypericum perfoliatum L. after simulated human digestion. Ind. Crops Prod. 2017, 109, 717–723.
- Fernández, K.; Labra, J. Simulated digestion of proanthocyanidins in grape skin and seed extracts and the effects of digestion on the angiotensin I-converting enzyme (ACE) inhibitory activity. Food Chem. 2013, 139, 196–202.
- Serra, A.; Macià, A.; Romero, M.P.; Valls, J.; Bladé, C.; Arola, L.; Motilva, M.J. Bioavailability of procyanidin dimers and trimers and matrix food effects in in vitro and in vivo models. Br. J. Nutr. 2010, 103, 944–952.
- Chen, X.; Xiong, J.; He, L.; Zhang, Y.; Li, X.; Zhang, L.; Wang, F. Effects of in vitro digestion on the content and biological activity of polyphenols from Acacia mearnsii bark. Molecules 2018, 23, 1824.
- Li, Q.; Chen, J.; Li, T.; Liu, C.; Wang, X.; Dai, T.; McClements, D.J.; Liu, J. Impact of in vitro simulated digestion on the potential health benefits of proanthocyanidins from Choerospondias axillaris peels. Food Res. Int. 2015, 78, 378–387.
- Rios, L.Y.; Bennett, R.N.; Lazarus, S.A.; Rémésy, C.; Scalbert, A.; Williamson, G. Cocoa procyanidins are stable during gastric transit in humans. Am. J. Clin. Nutr. 2002, 76, 1106–1110.
- Sánchez-Patán, F.; Cueva, C.; Monagas, M.; Walton, C.E.; Gibson, G.R.; Martín-Álvarez, P.J.; Moreno-Arribas, M.V.; Bartolomé, B. Gut microbial catabolism of grape seed flavan-3-ols by human faecal microbiota. Targeted analysis of precursor compounds, intermediate metabolites and end-products. Food Chem. 2012, 131, 337–347.
- Sánchez-Patán, F.; Barroso, E.; van de Wiele, T.; Jiménez-Girón, A.; Martín-Alvarez, P.J.; Moreno-Arribas, M.V.; Martínez-Cuesta, M.C.; Peláez, C.; Requena, T.; Bartolomé, B.; et al. Comparative in vitro fermentations of cranberry and grape seed polyphenols with colonic microbiota. Food Chem. 2015, 183, 273–282.
- Ottaviani, J.I.; Kwik-Uribe, C.; Keen, C.L.; Schroeter, H. Intake of dietary procyanidins does not contribute to the pool of circulating flavanols in humans. Am. J. Clin. Nutr. 2012, 95, 851–858.
- He, J.; Magnuson, B.A.; Giusti, M.M. Analysis of anthocyanins in rat intestinal contents--impact of anthocyanin chemical structure on fecal excretion. J. Agric. Food Chem. 2005, 53, 2859–2866.
- Choy, Y.Y.; Jaggers, G.K.; Oteiza, P.I.; Waterhouse, A.L. Bioavailability of intact proanthocyanidins in the rat colon after ingestion of grape seed extract. J. Agric. Food Chem. 2013, 61, 121–127.
- Reichardt, N.; Duncan, S.H.; Young, P.; Belenguer, A.; McWilliam Leitch, C.; Scott, K.P.; Flint, H.J.; Louis, P. Phylogenetic distribution of three pathways for propionate production within the human gut microbiota. ISME J. 2014, 8, 1323–1335.
- Hamer, H.M.; Jonkers, D.M.; Bast, A.; Vanhoutvin, S.A.; Fischer, M.A.; Kodde, A.; Troost, F.J.; Venema, K.; Brummer, R.J. Butyrate modulates oxidative stress in the colonic mucosa of healthy humans. Clin. Nutr. 2009, 28, 88–93.
- Berni Canani, R.; di Costanzo, M.; Leone, L. The epigenetic effects of butyrate: Potential therapeutic implications for clinical practice. Clin. Epigenetics 2012, 4, 4.
- Vrieze, A.; Van Nood, E.; Holleman, F.; Salojärvi, J.; Kootte, R.S.; Bartelsman, J.F.; Dallinga-Thie, G.M.; Ackermans, M.T.; Serlie, M.J.; Oozeer, R.; et al. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology 2012, 143, 913–916.
- Ferreira, Y.A.M.; Jamar, G.; Estadella, D.; Pisani, L.P. Proanthocyanidins in grape seeds and their role in gut microbiota-white adipose tissue axis. Food Chem. 2023, 404, 134405.
- Roopchand, D.E.; Carmody, R.N.; Kuhn, P.; Moskal, K.; Rojas-Silva, P.; Turnbaugh, P.J.; Raskin, I. Dietary polyphenols promote growth of the gut bacterium Akkermansia muciniphila and attenuate high-fat diet-induced metabolic syndrome. Diabetes 2015, 64, 2847–2858.
- Griffin, L.E.; Witrick, K.A.; Klotz, C.; Dorenkott, M.R.; Goodrich, K.M.; Fundaro, G.; McMillan, R.P.; Hulver, M.W.; Ponder, M.A.; Neilson, A.P.; et al. Alterations to metabolically active bacteria in the mucosa of the small intestine predict anti-obesity and anti-diabetic activities of grape seed extract in mice. Food Funct. 2017, 8, 3510–3522.
- Zhang, L.; Carmody, R.N.; Kalariya, H.M.; Duran, R.M.; Moskal, K.; Poulev, A.; Kuhn, P.; Tveter, K.M.; Turnbaugh, P.J.; Raskin, I.; et al. Grape proanthocyanidin-induced intestinal bloom of Akkermansia muciniphila is dependent on its baseline abundance and precedes activation of host genes related to metabolic health. J. Nutr. Biochem. 2018, 56, 142–151.
- Anhê, F.F.; Roy, D.; Pilon, G.; Dudonné, S.; Matamoros, S.; Varin, T.V.; Garofalo, C.; Moine, Q.; Desjardins, Y.; Levy, E.; et al. A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut 2015, 64, 872–883.
- Masumoto, S.; Terao, A.; Yamamoto, Y.; Mukai, T.; Miura, T.; Shoji, T. Non-absorbable apple procyanidins prevent obesity associated with gut microbial and metabolomic changes. Sci. Rep. 2016, 6, 31208.
- Cani, P.D.; de Vos, W.M. Next-generation beneficial microbes: The case of Akkermansia muciniphila. Front. Microbiol. 2017, 8, 1765.
- Routy, B.; le Chatelier, E.; Derosa, L.; Duong, C.P.M.; Alou, M.T.; Daillère, R.; Fluckiger, A.; Messaoudene, M.; Rauber, C.; Roberti, M.P.; et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 2018, 359, 91–97.
- Zhang, T.; Li, Q.; Cheng, L.; Buch, H.; Zhang, F. Akkermansia muciniphila is a promising probiotic. Microb. Biotechnol. 2019, 12, 1109–1125.
- Zhai, Q.; Feng, S.; Arjan, N.; Chen, W. A next generation probiotic, Akkermansia muciniphila. Crit. Rev. Food Sci. Nutr. 2019, 59, 3227–3236.
- Ansaldo, E.; Slayden, L.C.; Ching, K.L.; Koch, M.A.; Wolf, N.K.; Plichta, D.R.; Brown, E.M.; Graham, D.B.; Xavier, R.J.; Moon, J.J.; et al. Akkermansia muciniphila induces intestinal adaptive immune responses during homeostasis. Science 2019, 364, 1179–1184.
- Cheng, D.; Xie, M.Z. A review of a potential and promising probiotic candidate-Akkermansia muciniphila. J. Appl. Microbiol. 2021, 130, 1813–1822.
- Bae, M.; Cassilly, C.D.; Liu, X.; Park, S.M.; Tusi, B.K.; Chen, X.; Kwon, J.; Filipčík, P.; Bolze, A.S.; Liu, Z.; et al. Akkermansia muciniphila phospholipid induces homeostatic immune responses. Nature 2022, 608, 168–173.
- Belzer, C.; de Vos, W.M. Microbes inside--from diversity to function: The case of Akkermansia. ISME J. 2012, 6, 1449–1458.
- Ganesh, B.P.; Klopfleisch, R.; Loh, G.; Blaut, M. Commensal Akkermansia muciniphila exacerbates gut inflammation in Salmonella Typhimurium-infected gnotobiotic mice. PLoS ONE 2013, 8, e74963.
- Zhong, Y.; Teixeira, C.; Marungruang, N.; Sae-Lim, W.; Tareke, E.; Andersson, R.; Fåk, F.; Nyman, M. Barley malt increases hindgut and portal butyric acid, modulates gene expression of gut tight junction proteins and Toll-like receptors in rats fed high-fat diets, but high advanced glycation end-products partially attenuate the effects. Food Funct. 2015, 6, 3165–3176.
- Hansen, C.H.; Krych, L.; Nielsen, D.S.; Vogensen, F.K.; Hansen, L.H.; Sørensen, S.J.; Buschard, K.; Hansen, A.K. Early life treatment with vancomycin propagates Akkermansia muciniphila and reduces diabetes incidence in the NOD mouse. Diabetologia 2012, 55, 2285–2294.
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.M.; Kennedy, S.; et al. Richness of human gut microbiome correlates with metabolic markers. Nature 2013, 500, 541–546.
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the healthy gut microbiota composition? A changing ecosystem across age, environment, diet, and diseases. Microorganisms 2019, 7, 14.
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638.
- Ley, R.E.; Bäckhed, F.; Turnbaugh, P.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075.
- Guo, X.; Xia, X.; Tang, R.; Zhou, J.; Zhao, H.; Wang, K. Development of a real-time PCR method for Firmicutes and Bacteroidetes in faeces and its application to quantify intestinal population of obese and lean pigs. Lett. Appl. Microbiol. 2008, 47, 367–373.
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031.
- Turnbaugh, P.J.; Hamady, M.; Yatsunenko, T.; Cantarel, B.L.; Duncan, A.; Ley, R.E.; Sogin, M.L.; Jones, W.J.; Roe, B.A.; Affourtit, J.P.; et al. A core gut microbiome in obese and lean twins. Nature 2009, 457, 480–484.
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Microbial ecology: Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023.
- Jin, G.; Asou, Y.; Ishiyama, K.; Okawa, A.; Kanno, T.; Niwano, Y. Proanthocyanidin-rich grape seed extract modulates intestinal microbiota in ovariectomized mice. J. Food Sci. 2018, 83, 1149–1152.





