Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sabrin R. M. Ibrahim | -- | 3264 | 2022-12-16 14:40:54 | | | |
| 2 | Camila Xu | Meta information modification | 3264 | 2022-12-19 03:19:26 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Khayat, M.T.; Mohammad, K.A.; Omar, A.M.; Mohamed, G.A.; Ibrahim, S.R.M. Biological Activities of Bergamotane Sesquiterpenoids. Encyclopedia. Available online: https://encyclopedia.pub/entry/38896 (accessed on 24 May 2026).
Khayat MT, Mohammad KA, Omar AM, Mohamed GA, Ibrahim SRM. Biological Activities of Bergamotane Sesquiterpenoids. Encyclopedia. Available at: https://encyclopedia.pub/entry/38896. Accessed May 24, 2026.
Khayat, Maan T., Khadijah A. Mohammad, Abdelsattar M. Omar, Gamal A. Mohamed, Sabrin R. M. Ibrahim. "Biological Activities of Bergamotane Sesquiterpenoids" Encyclopedia, https://encyclopedia.pub/entry/38896 (accessed May 24, 2026).
Khayat, M.T., Mohammad, K.A., Omar, A.M., Mohamed, G.A., & Ibrahim, S.R.M. (2022, December 16). Biological Activities of Bergamotane Sesquiterpenoids. In Encyclopedia. https://encyclopedia.pub/entry/38896
Khayat, Maan T., et al. "Biological Activities of Bergamotane Sesquiterpenoids." Encyclopedia. Web. 16 December, 2022.
Copy Citation
Bergamotane sesquiterpenoids are an uncommon class of terpenoids. They possess diverse biological properties, such as plant growth regulation, phototoxic, antimicrobial, anti-HIV, cytotoxic, pancreatic lipase inhibition, antidiabetic, anti-inflammatory, and immunosuppressive traits.
bergamotanes
sesquiterpenoids
marine
fungi
biosynthesis
1. Introduction
Nature has substantially participated in the discovery of drugs for human remedial treatments since the beginning of mankind [1]. The marine environment, with more than 70% of the surface of the Earth, represents the largest ecosystem and is characterized by quite variable physicochemical parameters (e.g., limited light access, low temperature, high pressure, and high salinity) [2]. Among the various marine microbes, fungi are a superabundant and ecologically substantial component of marine microbiota [3]. Fungi are one of nature’s treasures that inhabit various environments on the earth’s surface, including the marine environment [4][5][6][7]. They play a growing relevant role in drug development and biomedicine research, either directly as drugs or indirectly as lead structures for bio-inspired drug synthesis [8][9][10][11][12].
2. Structural Assignment and Stereochemistry Determination
A total of 97 metabolites have been separated from various fungal source extracts using different chromatographic techniques and characterized by NMR, MS, and IR spectral analyses as well as chemical derivatization. The relative configuration of these metabolites was established using NOESY or ROESY spectral analyses. Various studies reported the assigning of their absolute stereochemistry using total synthesis [13][14], Mosher’s method [15], X-ray diffraction, chemical conversion [16][17][18], and ECD analyses [19]. The reported metabolites have been categorized into bi-, tri-, and tetracyclic derivatives.
3. Biological Activities of Bergamotane Sesquiterpenoids
Various reported studies revealed the assessment of bergamotane sesquiterpenoids for diverse bioactivities, including plant growth regulation, phototoxic, antimicrobial, anti-HIV, cytotoxic, pancreatic lipase inhibition, antidiabetic, anti-inflammatory, and immunosuppressive
3.1. Anti-Inflammatory Activity
NO (nitric oxide) is a substantial pro-inflammatory mediator, and its excessive production is accompanied with various inflammatory illnesses; therefore, it possesses a remarkable role for regulating immune responses and inflammation [20]. NO production inhibitors may represent the potential capacity for treating various inflammatory disorders. Thus, further research for fungal metabolites must be conducted to discover novel anti-inflammation agents.
The epigenetic chemical manipulation of Eutypella sp. derived from deep-sea hydrothermal sulfide deposit by co-treatment with SBHA (histonedeacetylase inhibitor, suberohydroxamic acid) and 5-Aza (DNA methyltransferase inhibitor, 5-azacytidine) was shown to activate a biosynthetic sesquiterpene-linked gene cluster [21]. From elicitor-treated cultures EtOAc extract, eutypeterpenes A–F (18–22 and 28) along with xylariterpenoids A (16) and B (17) were purified using SiO2/RP-18/HPLC that were identified by spectral analyses, as well as by using chemical conversion, X-ray diffraction, ECD, and calculated NMR for configuration assignments.
Eutypeterpene A (28) is the first bergamotene sesquiterpene incorporating a dioxolanone moiety. These metabolites were assessed for their NO production inhibitory capacity induced by LPS-(lipopolysaccharide) in RAW 264.7 macrophages [21]. The results indicated thatcompound 18 and 19 (IC50 13.4 and 16.8 μM, respectively) displayed more effectiveness than quercetin (IC50 of 17.0 μM), whereas other metabolites had noticeable potentials (IC50 values ranged from 18.7 to 24.3 μM) with weak cytotoxic capacities (IC50 > 100 μM). A structure–activity study revealed that the analog with a triol unit (18) at the side chain was more effective than compound 16, 17, and 19 with a diol unit, which were more potent than compound 20, 21, and 28 with one hydroxy group. Furthermore, the α,β-unsaturated ketone unit (as in compound 21 and 22) and the OH-linked carbon configuration also affected the activities (16 versus 17) [21] (Figure 1).
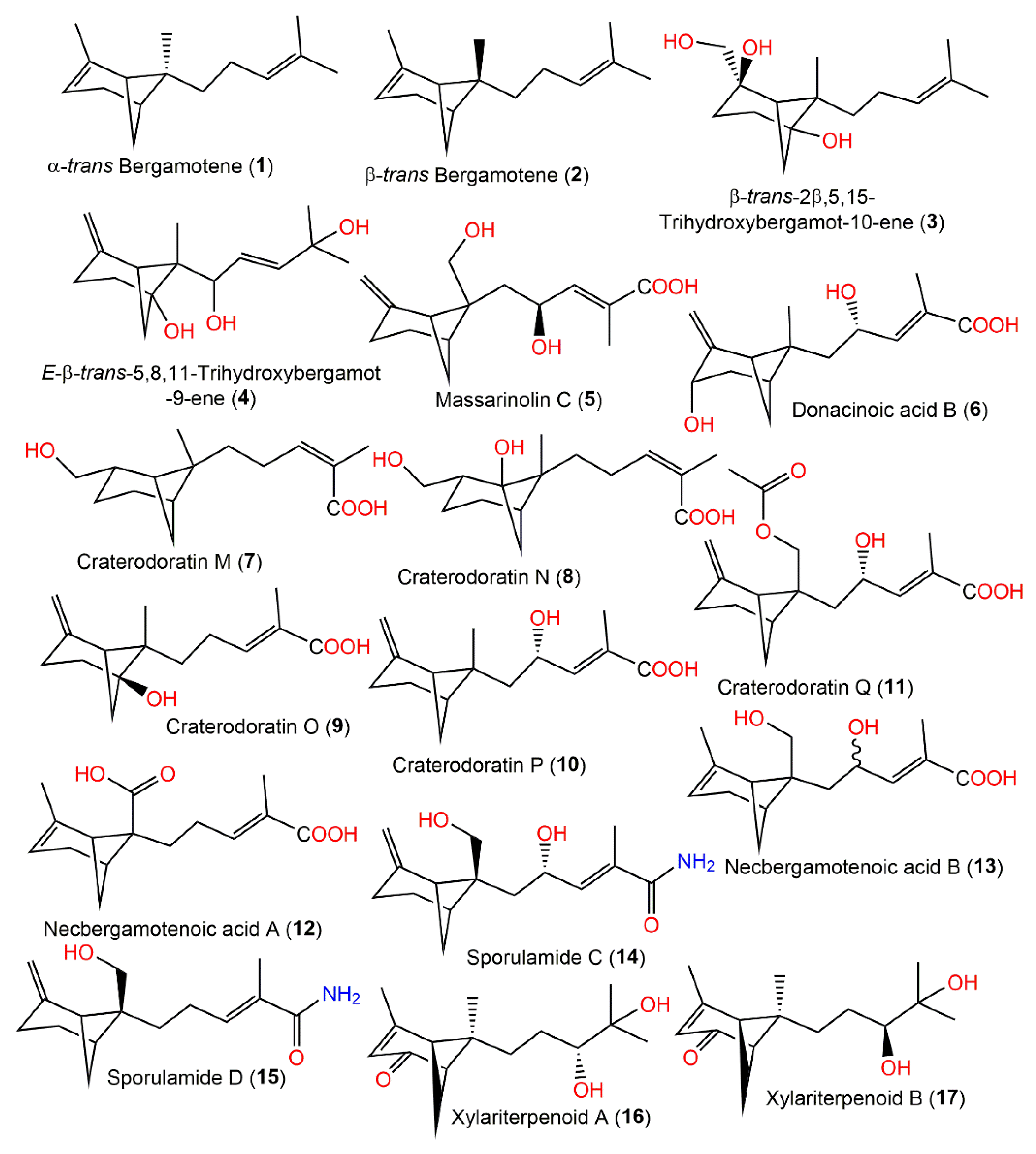
Figure 1. Structures of bicyclic bergamotane sesquiterpenoids (1–17).
Biogenetically, compounds 18–22 are derived from FPP that performs a 1,6-cyclization to produce bisabolane (A). The 4,7-cyclization of A generates bergamotane (B), which further generates 18–22 via diverse oxidation and reduction processes. Additionally, compound 28 is formed from 18 by carbonate incorporation [21] (Scheme 1).
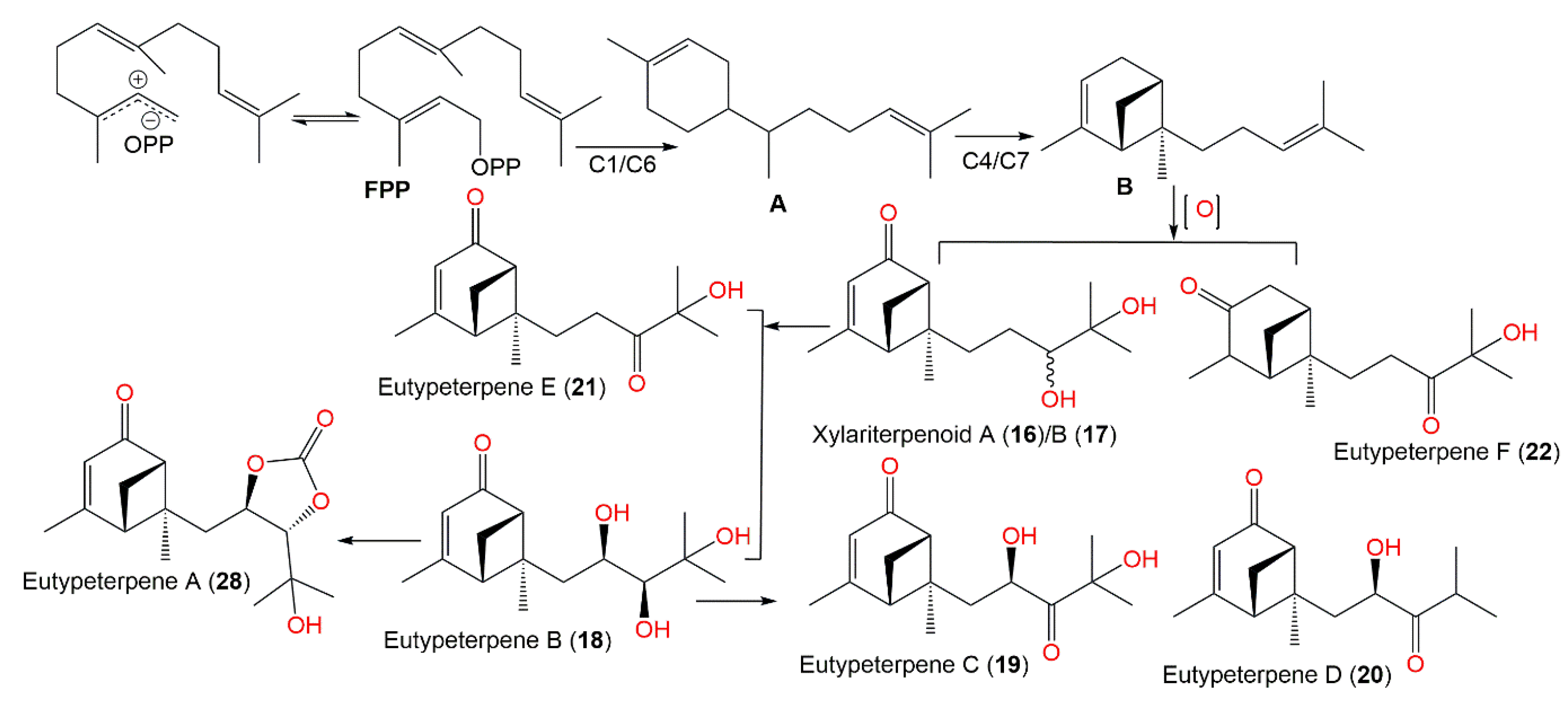
Scheme 1. Biosynthetic pathway of eutypeterpenes A–F (compounds 18–22 and 28) [21].
The deep-sea-isolated Graphostroma sp. MCCC3A00421 associated with the Atlantic Ocean hydrothermal sulfide deposits biosynthesized new bergamotane sesquiterpenoids: (10S)-xylariterpenoid A (23), (10R)-xylariterpenoid B (24), xylariterpenoid E (25), xylariterpenoid F (26), and xylariterpenoid G (27), which were purified using SiO2/OSD/Sephadex LH-20/RP-18 CC and preparative TLC. They were characterized by extensive spectral data, and their absolute configuration was established by ECD, Cu-Ka-single-crystal X-ray diffraction, and modified Mosher’s method analyses. Compound 25 is trinor-bergamotane. Compounds 23, 26, and 27 revealed moderate inhibition potentials (IC50s of 86, 85, and 85 μM, respectively) of NO production in LPS-stimulated RAW264.7 macrophages compared with aminoguanidine (IC50 of 23 μM). It was noted that bergamotane moiety’s 10S configuration obviously boosted the activity as in compound 23 (10S, IC50 of 85 μM) versus compound 24 (10R, of IC50 230 μM) (Figure 2) [16].
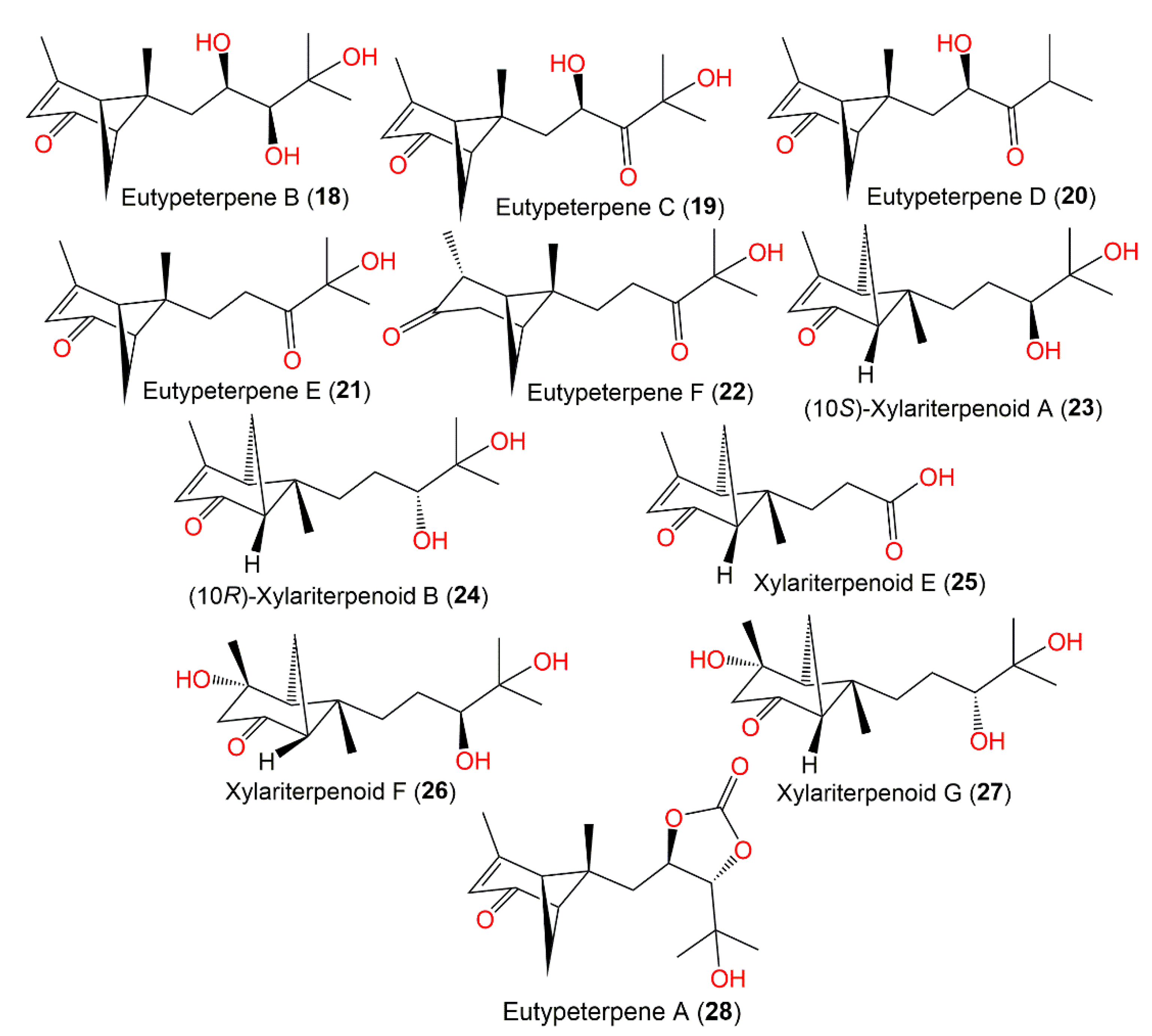
Figure 2. Structures of bicyclic bergamotane sesquiterpenoids (compounds 18–28).
3.2. Phytotoxic Activity
Prehelminthosporol (38) and dihydroprehelminthosporol (34) along with compounds 35–37, 39, 40, and 43 were separated by SiO2, flash CC, and preparative TLC from the EtOAc extract of the Bipolaris species, which is a Sorghum halepnse (Johnson grass) pathogen (Figure 3). These metabolites were assessed for their phytotoxic potential towards Sorghum bicolor (Sorghum) and Sorghum balepense (Johnson grass) in leaf spot assays [22][23]. Compounds 34, 38, and 39 produced similar lesions to those caused by the fungus in the field. The lesions appeared as a reddish-brown area (0.3–0.5 cm diameter) surrounded by a black circle with an outer chlorotic zone. Compounds 34 and 38 (concentration of 25 μg/5 μL) had comparable toxic effectiveness, while compound 38 maintained its effect at a lower concentration of 2.5 μg/5 μL; meanwhile, the other compounds were non-toxic [22][23]. Victoxinine was also toxic to cereals in the order of oats > rye and barley > wheat > sorghum in a root inhibition assay [23]. The phytotoxic influence of compounds 34 and 38–40 versus sorghum, corn, bent-grass, sickle-pod, and morning glory was also assessed in leaf spot assays. Moreover, victoxinine caused a water-soaked translucent appearance with defined irregular necrotic edges. It is worth mentioning that 3-deoxyanthocyanidins are sorghum stress response metabolites (phytoalexins), which were accountable for the red wound response. Compounds 34, 38, and 39 were elicitors of a very strong reddening compared with the wounding-produced reddening, but compound 40 did not elicit a sorghum phytoalexin response. In bent grass and corn, compounds 34 and 38–40 produced a light-brown area limited by a chlorotic region, whereas in sickle pod and morning glory, they showed necrotic lesions that extended at high concentrations beyond the under-drop area. It is noteworthy that compound 38 was the most toxic compound versus all tested plants except for the morning glory [23].
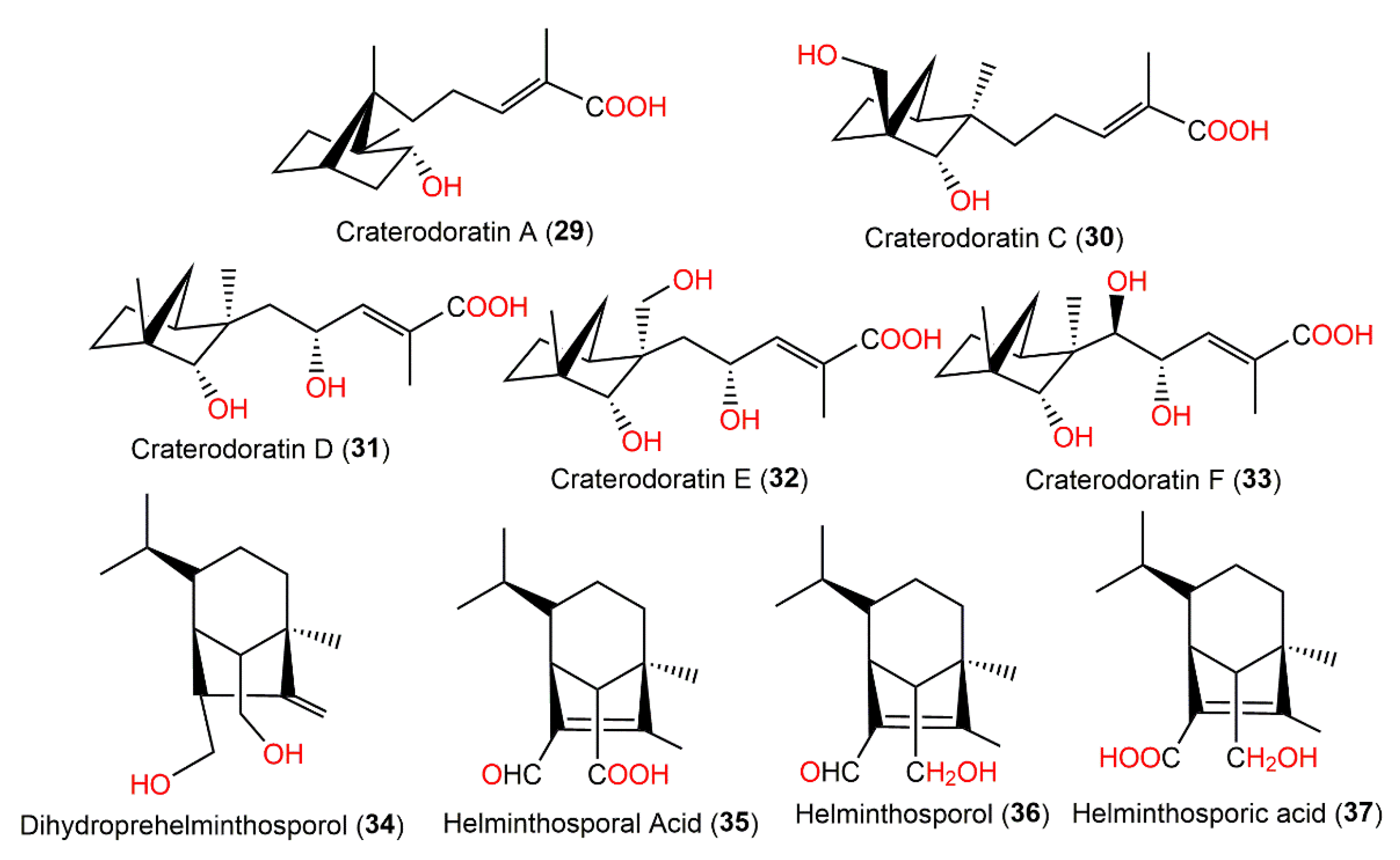
Figure 3. Structures of the bicyclic bergamotane sesquiterpenoids (compounds 29–37).
Helminthosporium victoriae, the causative agent of oats Victoria blight disease yielded phytotoxins, victoxinine (40) and victoxinine α-glycerophosphate (41), which were separated from its diethyl ether extract using Sephadex LH-20 and SiO2 CC and detected on the TLC plate by giving a blue color with 5% vanillin:H2SO4 [24] (Figure 4). The existence of α-glycerophosphate moiety was established by coupling between the phosphorous and carbon. Compound 40 completely prohibited the root growth of toxin-susceptible and toxin-resistant oats (concentration of 2.5 × 10−4 M); it was ≈ 7500 times more toxic for susceptible plants on a weight basis, while its toxicity for resistant plants was nearly similar, suggesting a role of the victoxinine moiety on the toxicity [24][25][26]. On the other side, compound 41 (concentration of 100 µg/mL) demonstrated little or no growth inhibition effectiveness on either susceptible or resistant oats [24].
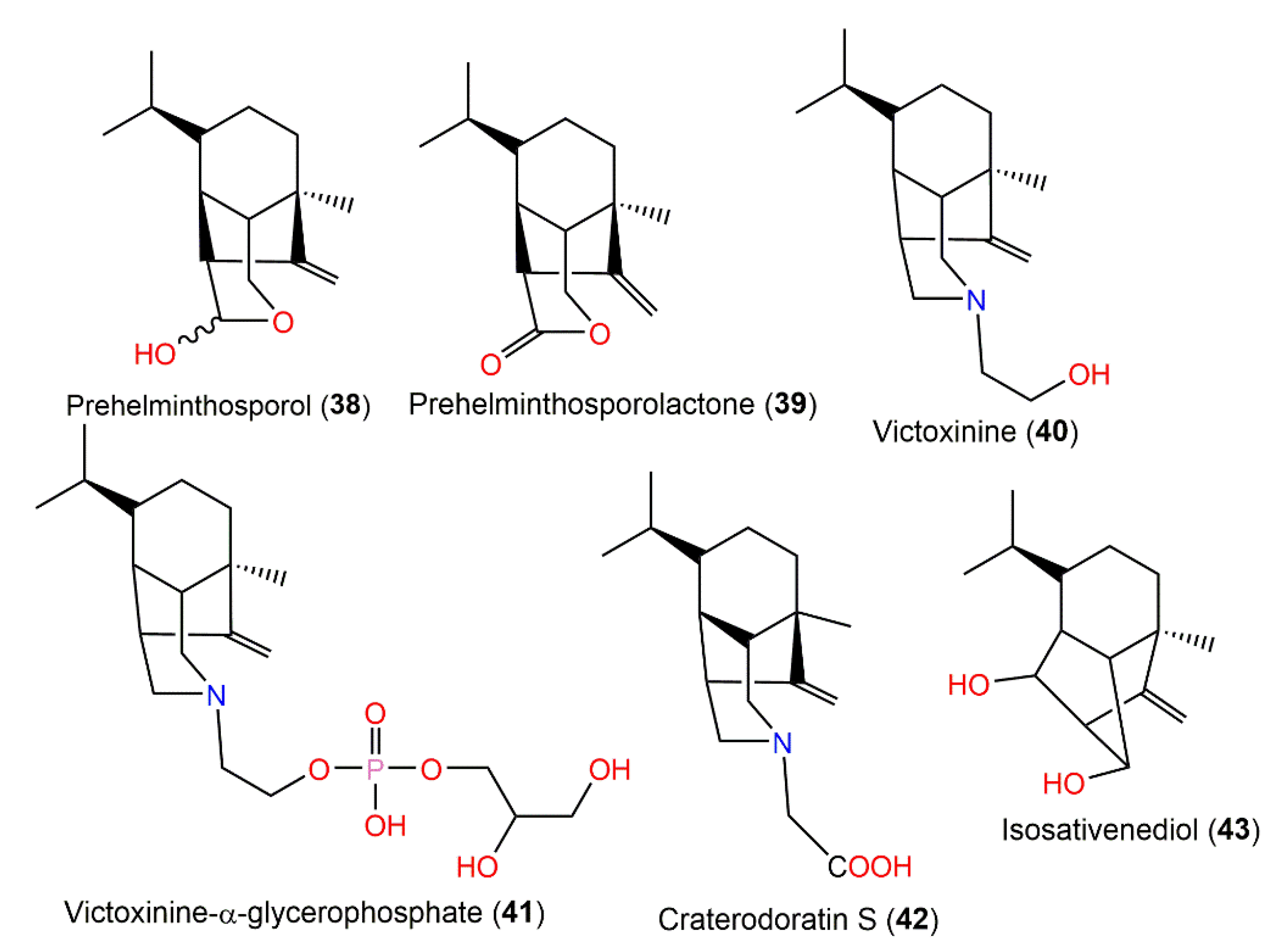
Figure 4. Structures of tricyclic bergamotane sesquiterpenoids (compounds 38–43).
3.3. Anti-HIV Activity
From Paraconiothyrium brasiliense, new tricyclic sesquiterpenoids, brasilamides A–D (45–48) and the formerly reported pinthunamide (44), were separated from the culture’s EtOAc extract utilizing SiO2/Sephadex LH-20 CC and HPLC. Their structures were established using NMR and X-ray analyses (Figure 5). Compounds 45 and 46 are rare metabolites having a 4-oxatricyclo[3.3.1.02,7]nonane moiety with a tetrahydro-2H-pyrone or a tetrahydro-2H-pyran linked with bicyclo[3.1.1]heptane ring at C-5 and C-2, whereas compounds 47 and 48 are analogs of 44, possessing an unprecedented 9-oxatricyclo[4.3.0.04,7]-nonane core.
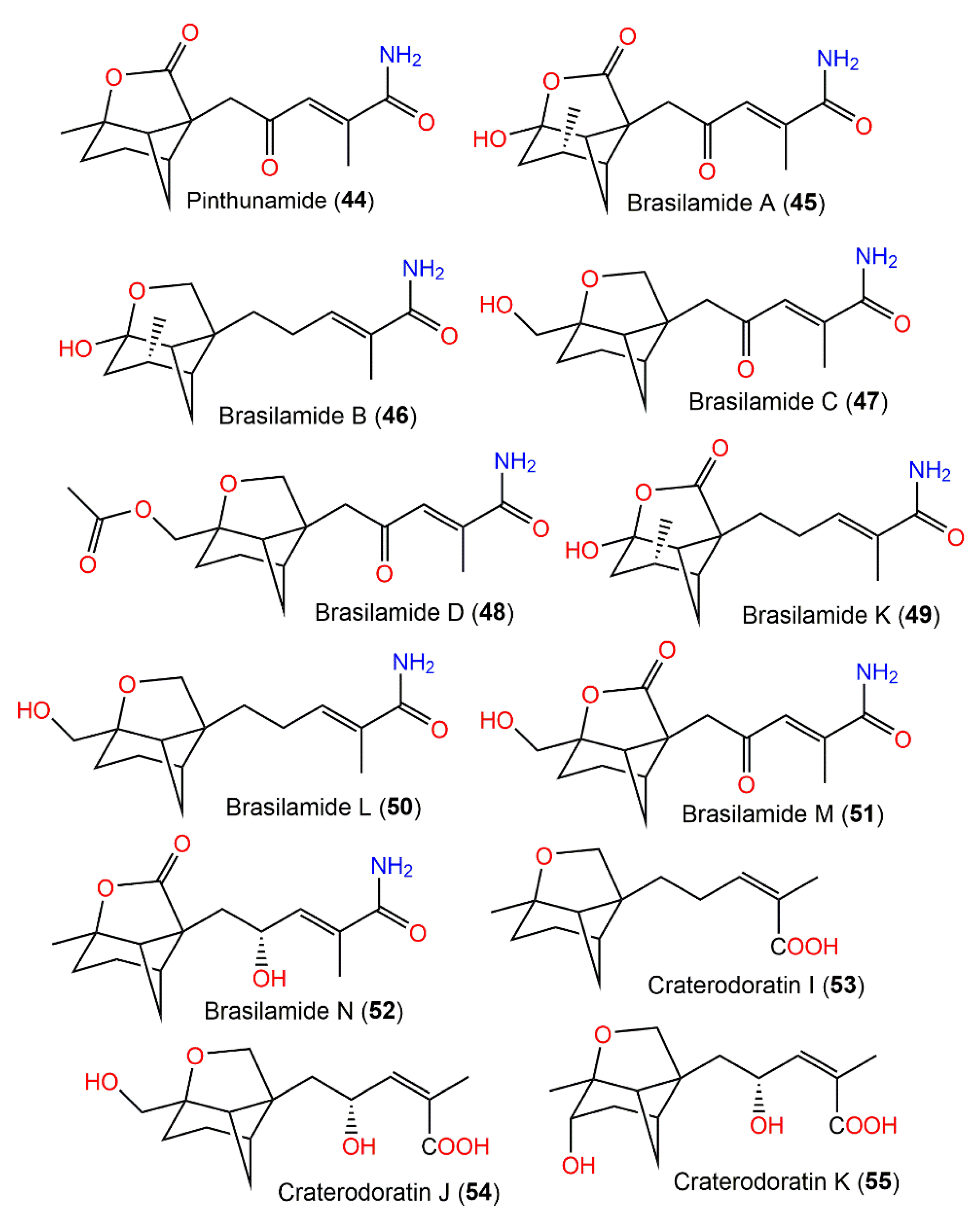
Figure 5. Structures of tricyclic bergamotane sesquiterpenoids (44–55).
The differences of the above-mentioned compounds from 44 were the existence of a tetrahydrofuran moiety connected to the bicycle[3.1.1]heptane unit instead of γ-lactone ring, as well as different C-10 substituents. Compounds 45–48 demonstrated inhibitory effectiveness (EC50s of 108.8, 57.4, and 48.3 µM, respectively) versus HIV-1 replication in C8166 cells compared with indinavir sulfate (EC50 of 8.2 nM) [17]. Biogenetically, they were derived from the mevalonate/trans-cis-farnesol/bisabolene/bergamotane pathway (Scheme 2).
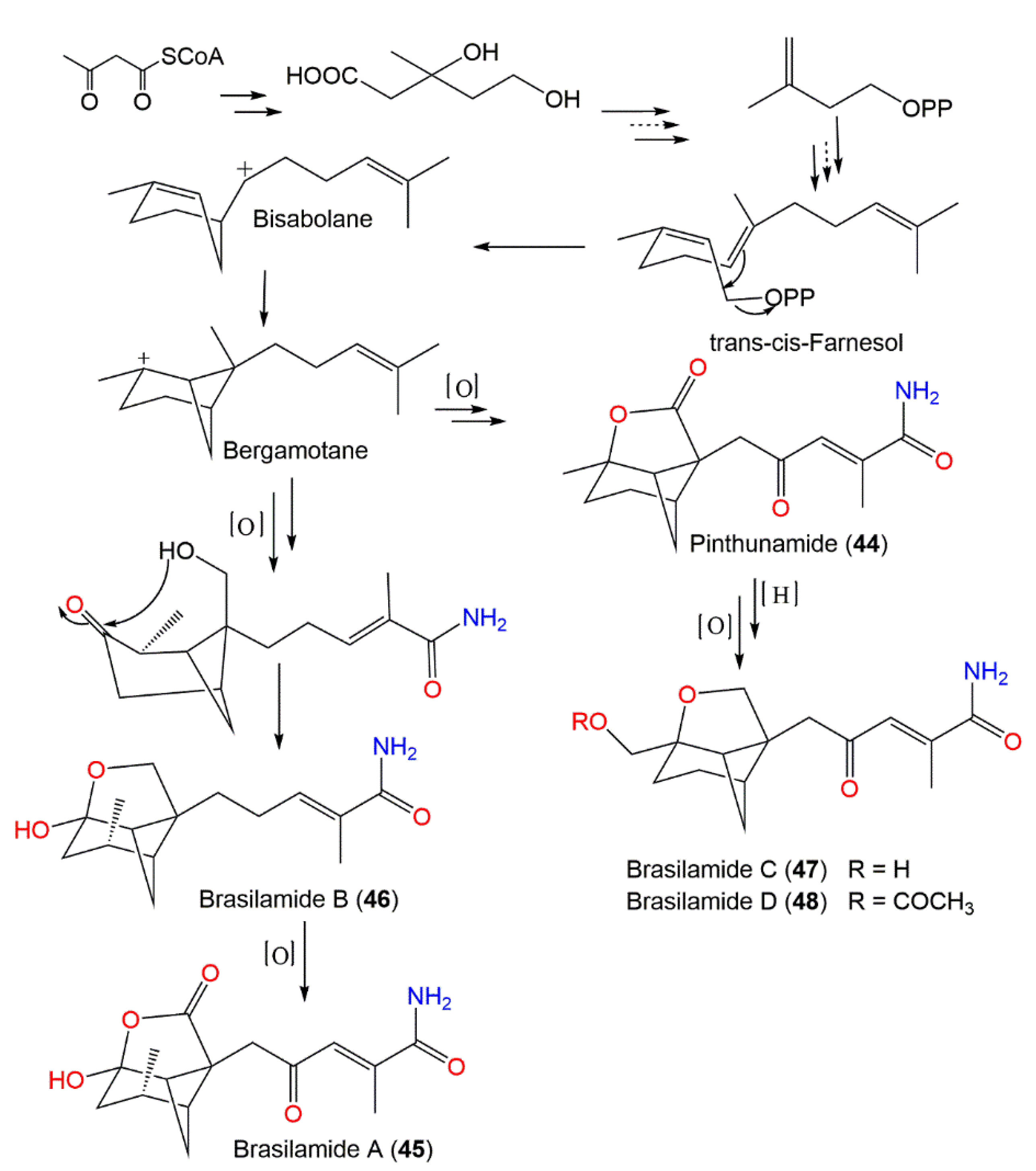
Scheme 2. Biosynthetic pathways of brasilamides A–D (45–48) [17].
3.4. Immunosuppressive Activity
Immunosuppressants are drugs that prohibit body immunity and are principally utilized in organ transplantation to overcome rejection and in auto-immune illnesses [27]. Currently, many immunosuppressive agents act by prohibiting T-cell proliferation; however, there is no new, safe, and efficient immune-suppressive agent that prohibits B-cell proliferation [28].
Dai et al. separated eighteen bergamotane sesquiterpenoids from the EtOAc extract of Craterellus ordoratus: craterodoratins A–R (7–11, 29–33, 53, 55, 56, 63–65, and 71) and a new victoxinine derivative, craterodoratin S (42), along with the previously isolated 5, 61, 77, and 88 by SiO2/RP-18/Sephadex LH-20/preparative HPLC (Figure 6) [29].
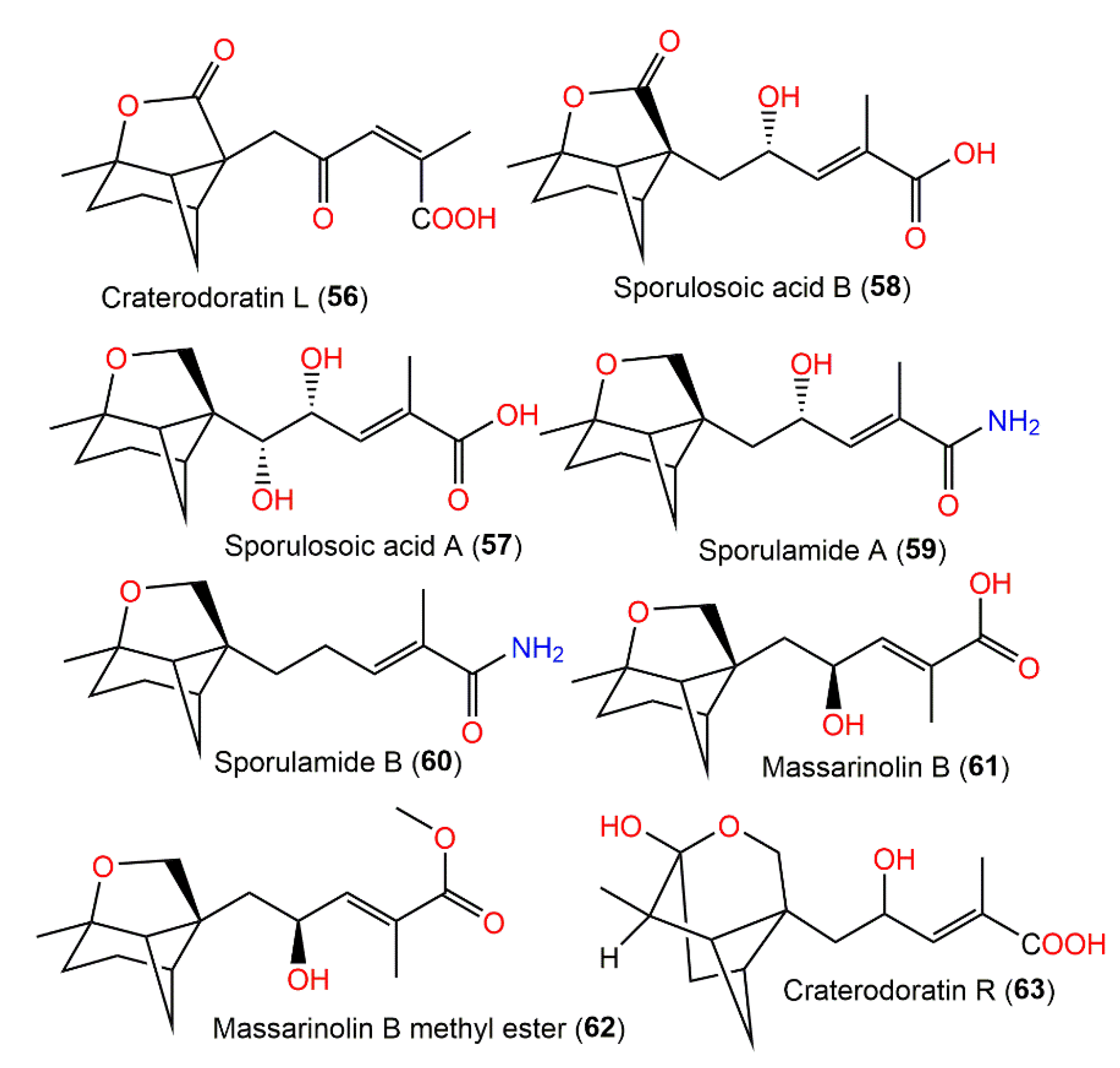
Figure 6. Structures of tricyclic bergamotane sesquiterpenoids (56–63).
Their structures with absolute configurations were established by spectral, X-ray diffraction, and ECD analyses and NMR calculations. Compounds 29 and 71 possess a rare skeleton, where the C-14methyl in 71 showed a further 1,2-migration. On the other hand, compounds 7–11, 53, 55, 56, and 63–65 belong to β-pinene derivatives that produced 30–33 through an alkyl migration (Figure 7). Compounds 7–10, 30, 42, 55, 61, and 88 demonstrated potent inhibitory potential versus LPS-caused B lymphocyte cell proliferation (IC50s ranged from 0.67 to 22.68 μM) in BALB/c mice compared with cyclosporin A (IC50 of 0.47 μM), where compound 61 (IC50 0.67 μM) had the most potent effectiveness. Moreover, compounds 11 and 61 possessed inhibition (IC50s of 31.50 and 0.98 μM, respectively) on T lymphocyte cells proliferation induced by ConA (concanavalin A) compared with cyclosporin A (IC50 0.04 μM). Structurally, it was noted that the α,β-unsaturated-carboxylic acid unit could be the key functional group for the immunosuppressive potential of these metabolites. Furthermore, compounds 61 and 7–10 with a β-pinene main core had a wider range of bioactivities [29].
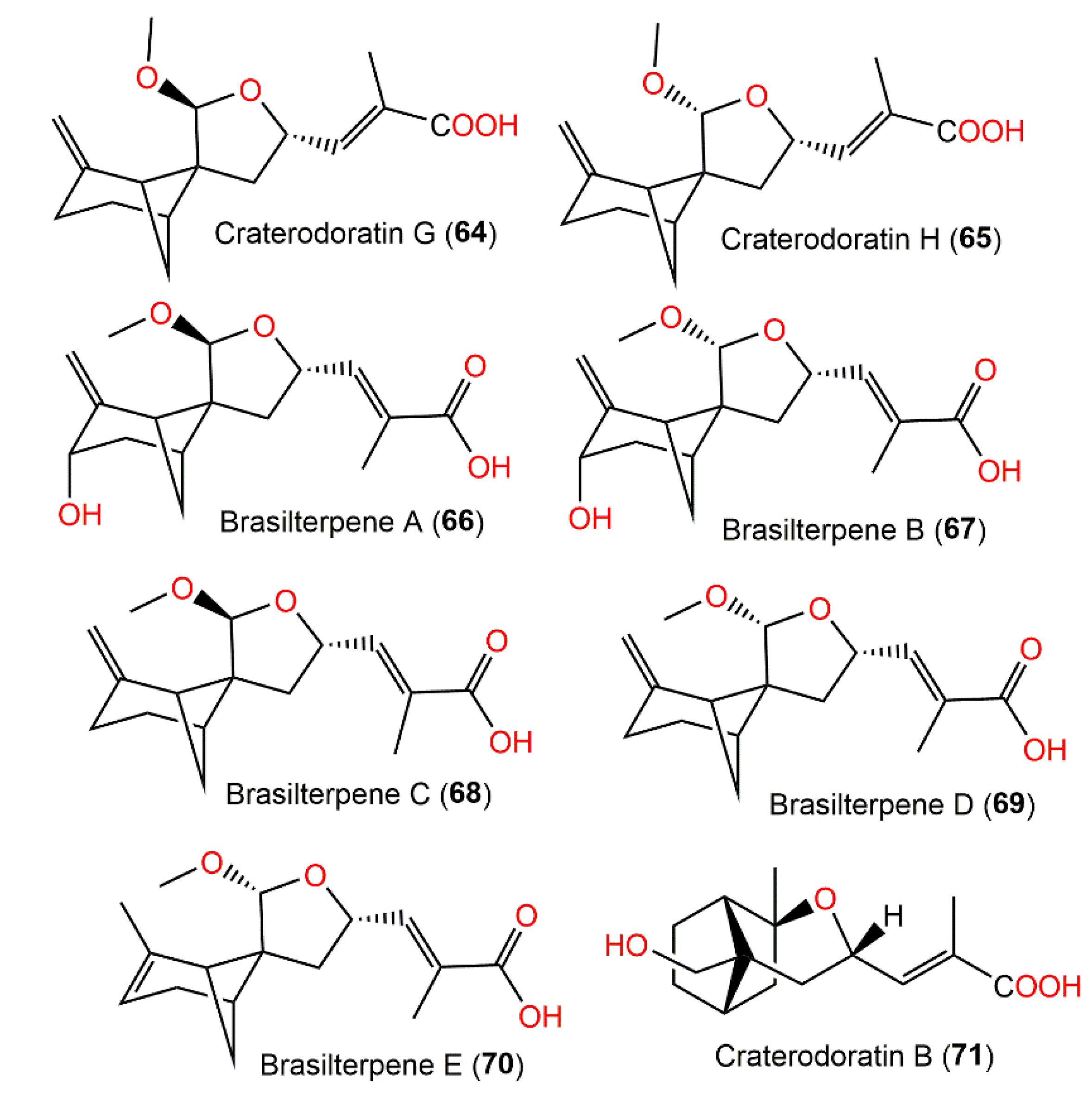
Figure 7. Structures of tricyclic bergamotane sesquiterpenoids (64–71).
3.5. Antimicrobial Activity
From Podospora decipiens, two new tetracyclic sesquiterpenoids, decipienolides A (74) and B (75), were separated from the EtOAc extract by SiO2 CC and HPLC analyses. They were obtained as a mixture of inseparable epimers, having a 3-hydroxy-2,2-dimethylbutyric acid sidechain as elucidated by an NMR analysis (Figure 8). The 74/75 mixture had an antibacterial influence versus B. subtilis (inhibition zone diameter of 9–10 mm, concentration of 200 µg/disk). Neither of them demonstrated capacity versus Ascobolus furfuraceus NRRL6460, Sordaria fimicola NRRL6459, and C. albicans ATCC90029 [30]. Donacinolides A (82) and B (76) (concentration of 50 μg/mL) revealed weak inhibition versus Salmonella enterica subsp. enterica (inhibition rates of 24.3, 23.9, and 26.2%) in the microdilution assay [31]. Furthermore, there were no observed antibacterial activity for purpurolides B (83) and C (84) (concentration of 50 μM) versus E. coli ATCC25922, M. smegmatis mc2155 ATCC70084, S. aureus ATCC25923, and S. epidermidis ATCC12228 [32].
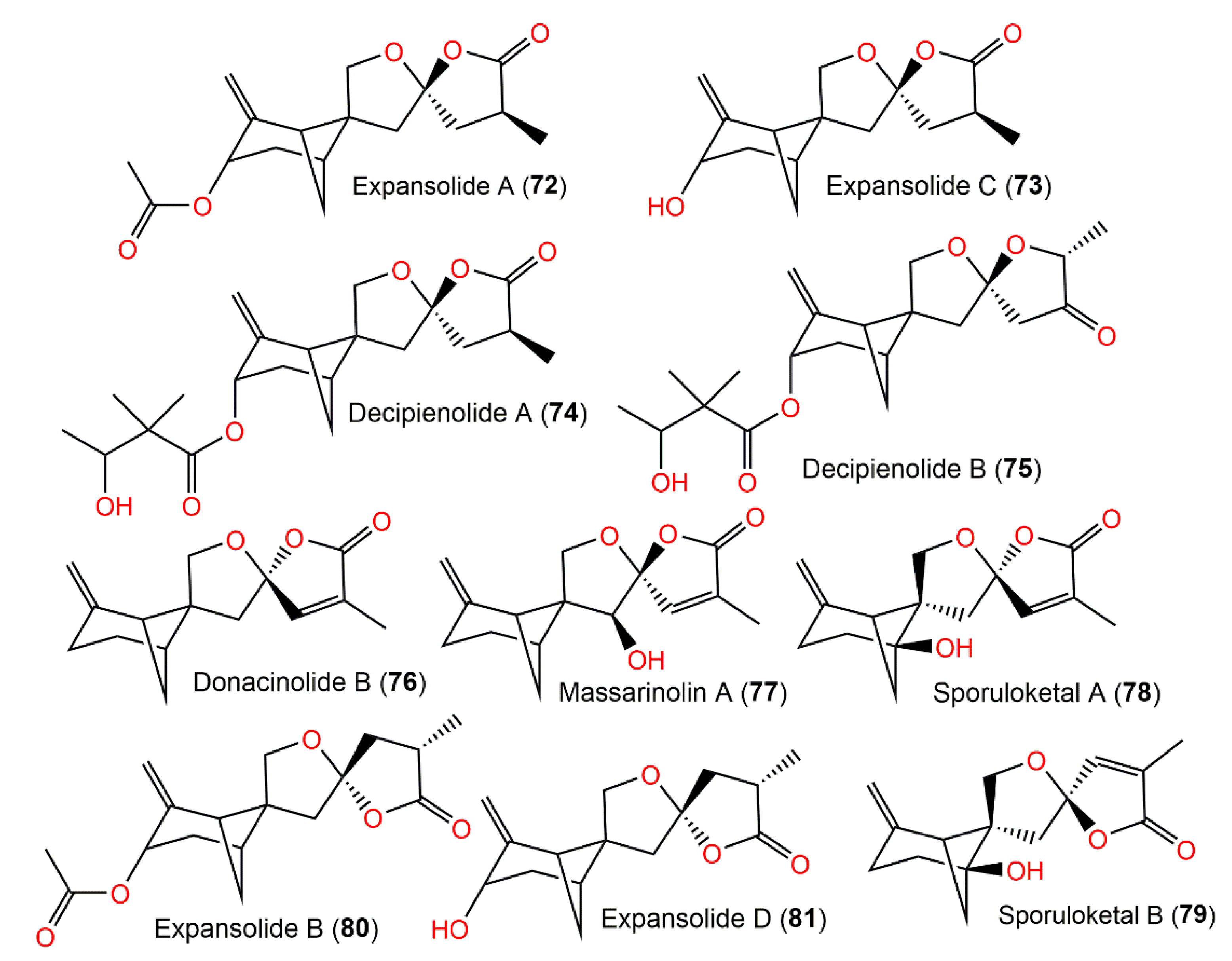
Figure 8. Structures of tetracyclic bergamotane sesquiterpenoids (72–79).
3.6. Pancreatic Lipase Inhibition
Purpurolides B (83) and C (84) are new 6/4/5/5-tetracyclic sesquiterpenoids that were separated from Penicillium purpurogenum IMM003 cultures by SiO2/RP-18/preparative HPLC analysis. The structures and configurations of compounds 83 and 84 were established using spectral and X-ray analyses as well as ECD and GIAO NMR data calculations (Figure 9).
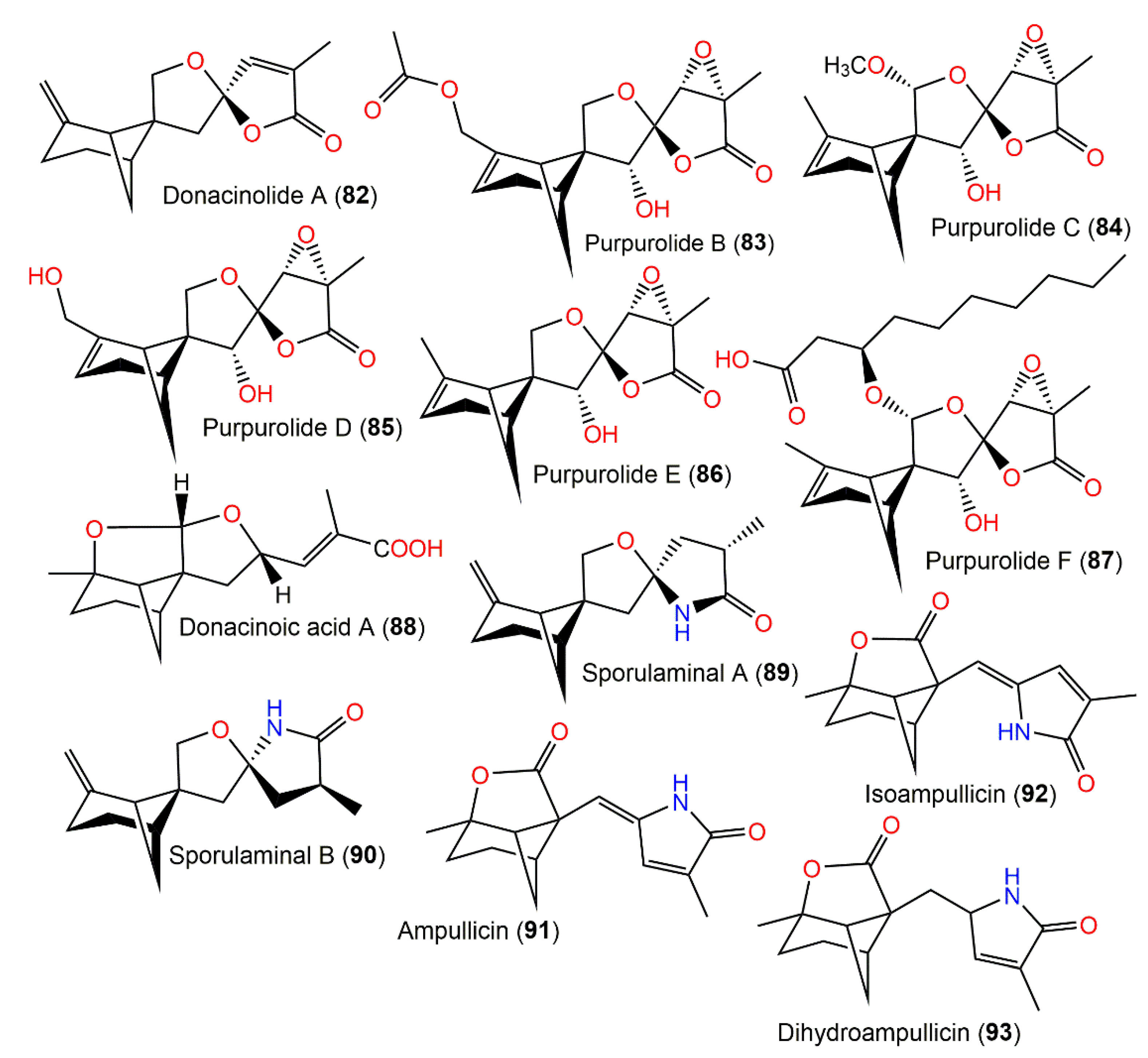
Figure 9. Structures of tetracyclic bergamotane sesquiterpenoids (80–93).
Compounds 83 and 84 demonstrated potent pancreatic lipase inhibition (IC50s of 5.45 and 6.63 μM, respectively), compared with kaempferol (IC50 of 1.50 μM) [32]. These compounds were possibly biosynthesized via numerous the cyclization and enzyme-catalyzed oxidation of FPP (farnesyl pyrophosphate), leading to four- and six-membered rings and the formation of two five-membered heterocyclic rings (Scheme 3) [32].
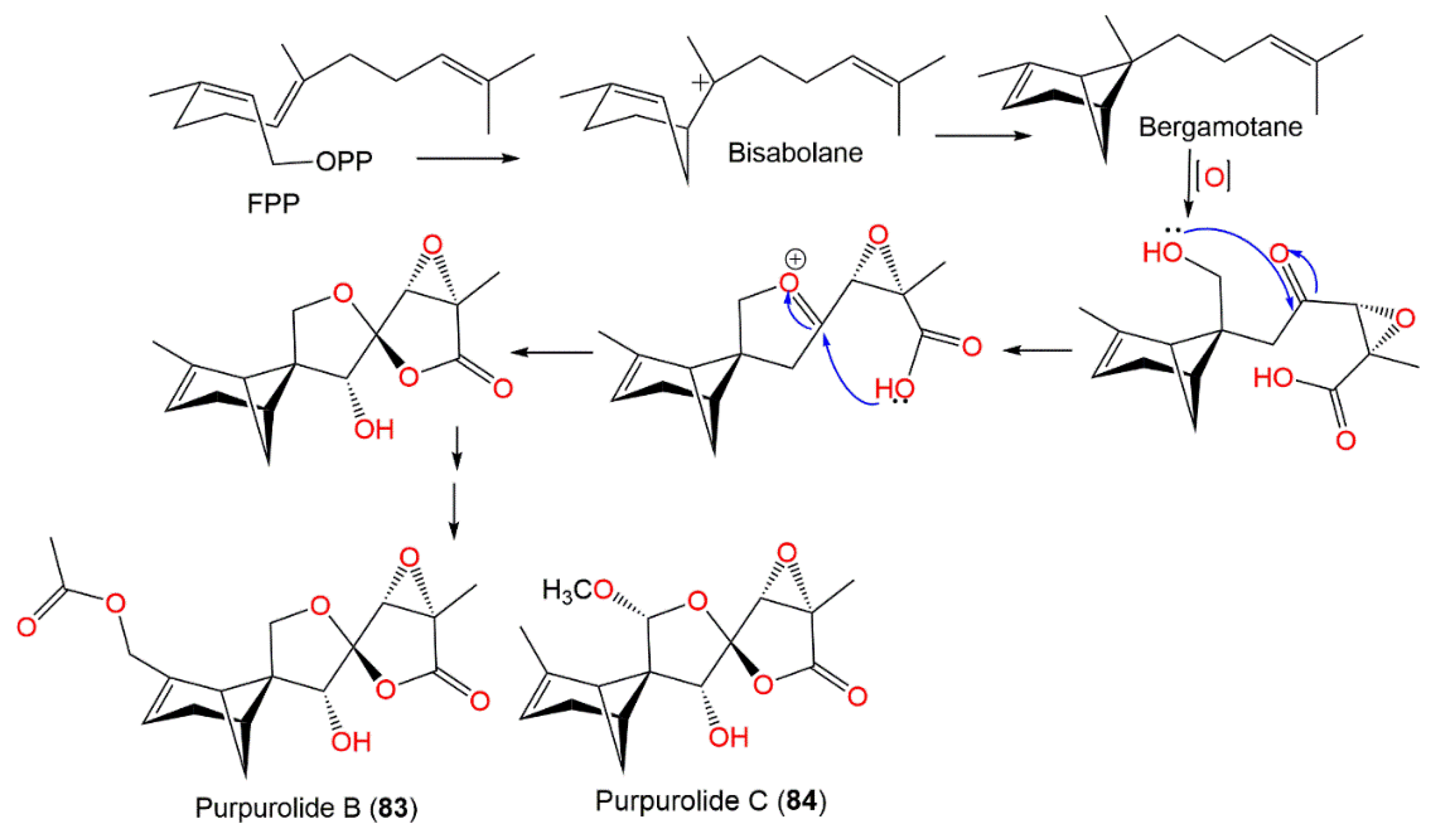
Scheme 3. Biosynthetic pathway of purpurolides B and C (83 and 84) [32].
Xia et al. separated from Penicillium purpurogenum IMM003 purpurolides D–F (85–87), which are new polyoxygenated 6/4/5/5-tetracyclic bergamotanes, using SiO2/Sephadex-LH-20/RP-18 CC and preparative HPLC processing [33]. Their elucidation was accomplished using spectral 13C NMR calculations coupled with DP4+ probability and ECD analyses. Compound 87 had potent pancreatic lipase inhibition potential (IC50 of 1.22 μM) compared with kaempferol (IC50 of 1.50 μM) and orlistat (IC50 of 0.75 μM), whereas compounds 85 and 86 (IC50s of 6.50 and 7.88 μM, respectively) were five or six-fold less powerful than 87, revealing that the C-14 hydroxylated decanoic acid moiety increased the potency [33]. Therefore, polyoxygenated bergamotanes could be viable candidates as pancreatic lipase inhibitors for further clinical development [33].
3.7. Antidiabetic Activity
From the deep sea-derived Paraconiothyrium brasiliense HDN15-135 EtOAc extract, new bergamotane sesquiterpenoids, brasilterpenes A-E (66–70), featuring an uncommon 6/4/5-tricyclic ring system, were separated by SiO2/RP-18/Sephadex LH-20/HPLC and assigned by diverse NMR analyses and X-ray diffraction, ECD, and DFT-NMR (density functional theory calculations of nuclear magnetic resonance) data [34]. Their hypoglycemic potential was estimated utilizing β-cell-ablated zebrafish larvae. Compounds 66 and 68 (concentration of 10 μM) remarkably lessened the glucose level down to 449.3 and 420.4 pmol/larva respectively, compared with the β-cell-ablated group (Teton+) (glucose level of 502.8 pmol/larva) and rosiglitazone (glucose level 395.6 pmol/larva) with no toxic influence on zebrafish larvae up to 200 μM. It was found that compounds 66 and 68 notably minimized free blood glucose in vivo in hyperglycemic zebrafish by suppressing gluconeogenesis and improving insulin sensitivity, which revealed that compound 68 had promising antidiabetic potential [34]. The structure–activity study revealed that the activity may be linked to the C-14 S-configuration of compounds 66 and 68, which represent the main structural difference from 67 and 69. The existence of C-3-OH may weaken the influence in 68 versus 66; however, the △2 endocyclic double bond may enhance the potential in 70 versus 69 [34]. Therefore, compound 68 may provide a scaffold for hypoglycemic drug development. Compounds 66–70 are also biosynthesized by the FPP pathway (Scheme 4). The cyclization of FPP via NPP (nerolidyl diphosphate) followed by a bisabol intermediate yields the bergamotane skeleton. These compounds are created by further oxidation, 9-OH-nucleophilic attack, and methylation processes. Because of the nucleophilic attack direction flexibility during the furan ring formation, compounds 66–69 appeared as C14-epimers in pairs [34] (Scheme 4).
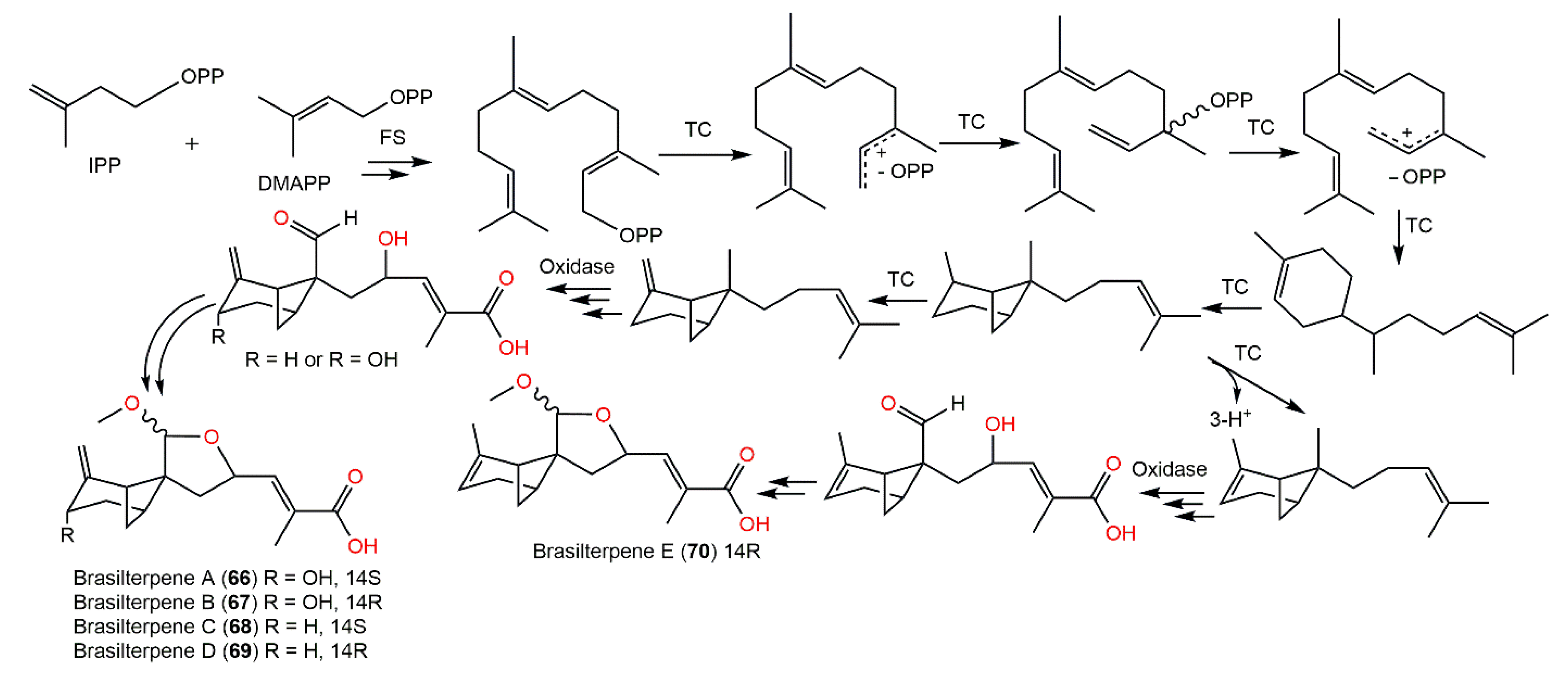
Scheme 4. Biosynthetic pathway of brasilterpenes A-E (66–70) [34]. IPP: isopentenyl diphosphate; FS: farnesyl synthase; NPP: nerolidyl diphosphate; TC: terpenyl cyclase; DMAPP: dimethylallyl diphosphate; FPP: farnesyl diphosphate.
Ying et al. isolated two new derivatives, expansolides C (73) and D (81), in addition to 72 and 80 from the plant pathogen Penicillium expansum ACCC37275 [35]. In an α-glucosidase inhibition assay; the 73/81 epimeric mixture (ratio 2:1) possessed a more powerful effectiveness (IC50 of 0.50 mM) compared with acarbose (IC50 1.90 mM), while the 72/80 epimeric mixture possessed no apparent potential. It was assumed that the acetyl group in compounds 72 and 80 impeded their binding with the α-glucosidase, resulting in loss of activity [35].
3.8. Plant Growth Regulation
Kimura et al. purified the tricyclic amide sesquiterpenoid pinthunamide (44) from the acetone extract of Ampulliferina sp. at pH 2.0 utilizing SiO2 and sephadex LH20 CC processing as well as crystallization from EtOAc extract, which gave positive NH2OH-HCI-FeCl3 and KMnO4 reactions [36]. The compound was assigned by X-ray diffraction and NMR methods. Its plant growth regulation effectiveness was evaluated using a lettuce seedling assay, where it (dose 300 mg/L) produced a 150% root growth acceleration over the control seedlings (100%) while scarcely influencing the hypocotyl elongation at the tested concentrations [36]. Its structure combined a unique configuration of six-, five-, and four-membered rings that was proposed to be biosynthesized via the mevalonate/trans-cis-farnesol/bisabolene/bergamotane pathway (Scheme 5) [36].
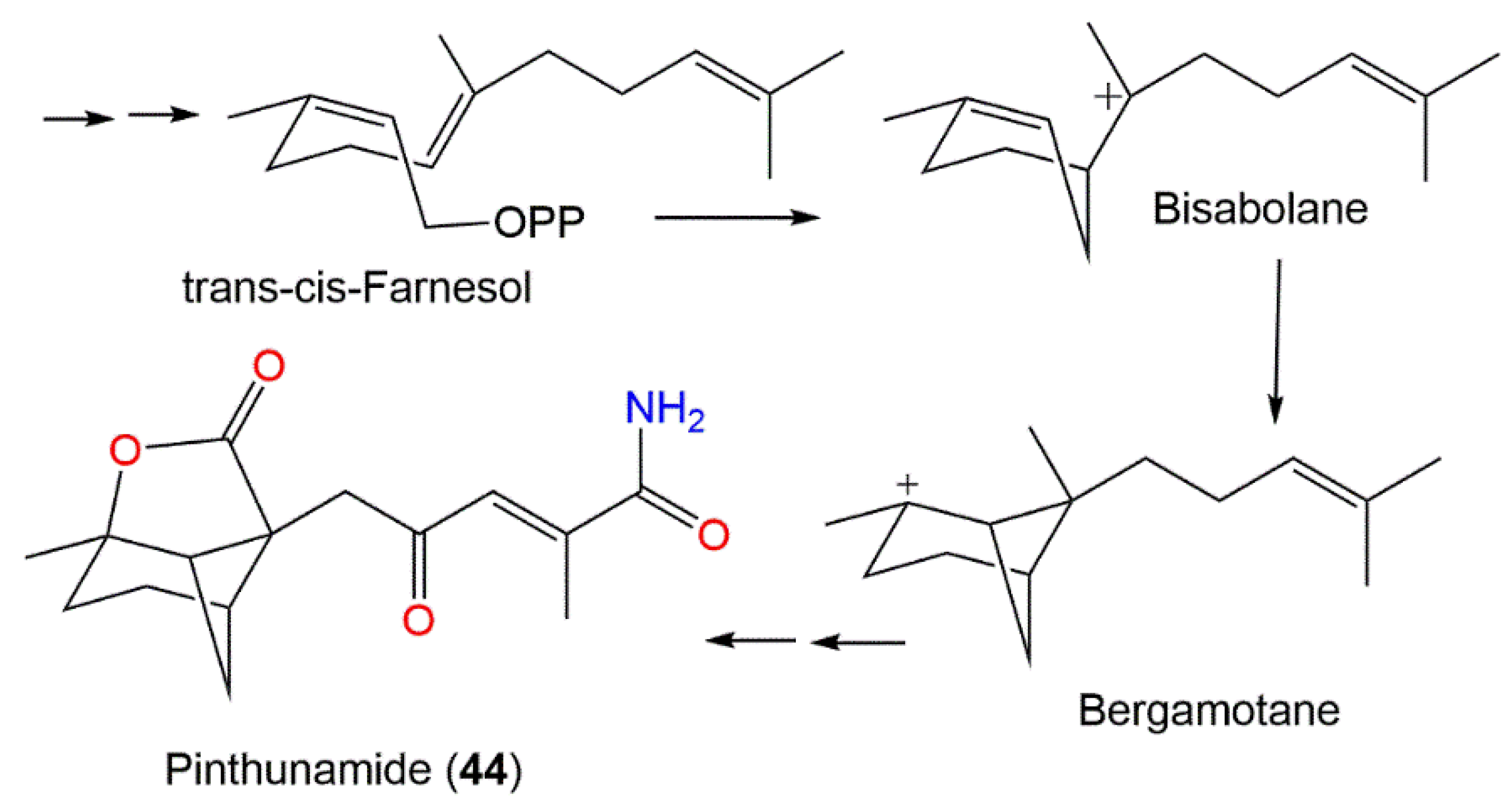
Scheme 5. Biosynthesis pathway of pinthunamide (44) [36].
Furthermore, in 1990, Kimura et al. purified another two new plant growth regulators, ampullicin (91) and isoampullicin (92) from Ampulliferina sp. No. 27 associated with Pinus thunbergii dead tree by SiO2 CC utilizing benzene:acetone as an eluent [37] (Figure 10). They were stereoisomers that had γ-lactam rings. Additionally, they (doses of 300 and 30 mg/L) were shown to promote lettuce seedling root growth by 200% over the control lettuce seedlings (100%) [37]. In 1993, the same group separated a minor metabolite, dihydroampullicin (93), characterized by the absence of the C8-C9 double bond. The compound promoted a 160% growth rate in lettuce seedling roots (dose of 300 mg/L) compared with the control; however, it had no influence on the hypocotyl growth, indicating that the C8-C9-double bond (C8-C9) was substantial in lettuce seedlings` root growth [38]. Bermejo et al. reported the synthesis of (+)−91 and 92 from (R)-(-)-carvone with a 4.5% overall yield using a stereo-selective 18-step sequence application [39]. The EtOAc extract of Aspergillus fumigatus Fresenius separated from leaf litter yielded expansolides A (72) and B (80). They had 2S/4S/6S/7R/9R/11S and 2S/4R/6S/7R/9R/11S, respectively, based on modified Mosher’s method. The compounds noticeably prohibited etiolated wheat coleoptiles growth by 100% and 59% at 10−3 M and 10−4 M solution compared with LOGRAN (commercial herbicide) (%inhibition of 80 and 42%) at the same concentrations [15].
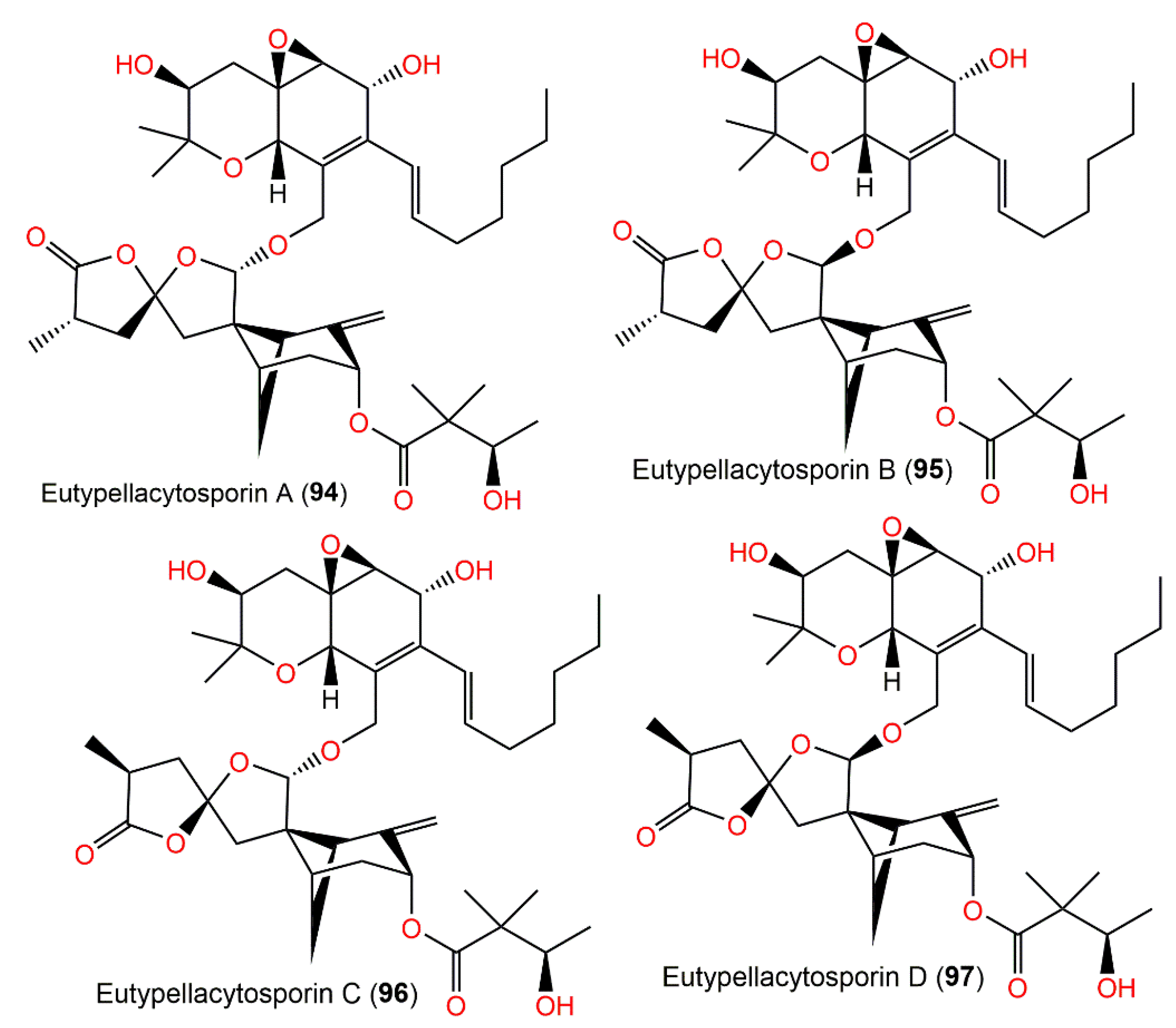
Figure 10. Structures of tetracyclic bergamotane sesquiterpenoids (94–97).
3.9. Cytotoxic Activity
Compounds 3 and 4, which were new β-bergamotane sesquiterpenoids, were separated by SiO2/RP-18/HPLC from the marine-associated Aspergillus fumigatus-YK-7 EtOAc extract. Their antiproliferative effects on the U937 and PC-3 cell lines were measured in vitro in an MTT assay. Compound 4 revealed a weak growth inhibition capacity (IC50 of 84.9 µM) versus the U937 cell line, while 3 had no activity (IC50 > 100 µM) compared with doxorubicin hydrochloride (IC50 of 0.021 µM). On the other sides, both had no effect versus PC-3 cells [40]. Wu et al. reported the separation of two new derivatives, xylariterpenoids A and B (16 and 17), from the EtOAc extract of Xylariaceae fungus by Sephadex LH-20/ODS CC and reversed-phase HPLC processing [41]. Their structures and stereo-configuration were proved utilizing NMR and CD methods. They are C-10 epimers having 2S/6S/7S/10R and 2S/6S/7S/10S configurations, respectively. Unfortunately, they (IC50 > 40 μM) exhibited no cytotoxic potential versus HL-60, MCF-7, SMMC-7721, A-549, and SW480 in an MTT assay [41].
From Paraconiothynium brasiliense Verkley, new bergamotane sesquiterpenoids brasilamides K-N (49–52), featuring 4-oxatricyclo-(3.3.1.02,7)-nonane (as in 49) and 9-oxatricyclo-(4.3.0.04,7)-nonane (as in 50–52) skeletons in addition to the formerly reported brasilamides A and C (45 and 46), were purified from the fungus scale-up fermentation cultures using SiO2/Sephadex LH-20/HPLC processing. They were elucidated via NMR analyses and compound 52’s configuration was assured using modified Mosher’s method. Compound 49 is a 45-hydrogenated analog that has a tetrahydro-2H-pyrone unit linked at C-2 and C-5 to the bicyclo(3.1.1)heptane framework, forming a 4-oxatricyclo-(3.3.1.0 2,7)-nonane skeleton, whereas compounds 50–52 displayed unusual 9-oxatricyclo-(4.3.0.0 4,7)-nonane skeletons. Compounds 50 and 51 are hydrogenated and oxygenated derivatives of 46, respectively, while 52 differed from 46 by having a C-8-carbonyl, C-1-methyl, and C-12 hydroxyl group instead of methylene, oxy-methylene, and ketone carbonyl, respectively. These metabolites (concentration of 50 µM) possessed no potential versus A549, A375, MCF-7, CNE1-LMP1, EC109, MGC, PANC-1, and Hep3B-2 in the MTS assay [42].
Montagnula donacina (edible mushroom) biosynthesized rare tetracyclic bergamotane sesquiterpenoids, donacinolides A (82) and B (76) and donacinoic acids A (88) and B (6), which were separated using SiO2 CC/Sephadex LH-20 CC/HPLC processing and were characterized using spectroscopic data, X-ray diffraction analysis, and computational methods. Compounds 76 and 82 are C9 epimers with a spiroketal moiety having 1S/5S/6S/9R and 1S/5S/6S/9S configurations, respectively, whereas 88 and 6 exhibited α,β-unsaturated carboxylic acid moiety and had 1R/2R/5S/6S/9S/14S and 1R/3S/5R/6R/9S configurations, respectively. These metabolites lacked a marked cytotoxic potential (IC50 > 40 μM) versus HL-60, SW480, A549, SMMC-7721, and MCF-7 [31].
In addition, purpurolides B (83) and C (84) had no cytotoxicity versus M14, HCT-116, U87, A2780, BGC-823, Bel-7402, and A549 [32], whereas compounds 85–87 (concentration of 50 μM) were inactive versus HCT-116, BGC-823, and Bel-7402 cell lines [33].
The chemical investigation of Arctic fungus Eutypella sp. D-1′s EtOAc extract yielded new derivatives, eutypellacytosporins A–D (94–97), which were established by spectroscopic analysis and modified Mosher’s method. Structurally, these metabolites are related to decipienolides and cytosporins. They exhibited (IC50s ranging from 4.9 to 17.1 μM) weak-to-moderate cytotoxic influence versus DU145, SW1990, Huh7, and PANC-1 in the CCK-8 assay, whereas Huh7 and SW1990 cell lines had more sensitivity to 94–97 (IC50s ranging from 4.9 to 8.4 μM). On the other hand, compounds 95 and 97 possessed noticeable potential versus PANC-1 (IC50s of 7.9 and 7.5 μM, respectively) compared with cisplatin (IC50 4.5 μM). The results revealed that the decipienolide moiety was substantial for activity; however, the C-33 configuration did not affect the activity [43]. It was proposed that compounds 94–97 are created from gentisaldehyde precursor with subsequent isoprenyl unit addition, double bond epoxidation, keto group hydrogenation, and an aliphatic chain addition (Scheme 6). The other precursor, the 14-OH of decipienolide A 74 or B 75, is produced from hydroxylation, allylic oxidation, and cyclization of farnesyl diphosphate to give I with a bicycle[3.1.1]heptane. Additionally, (14S)-14-OH-expansolide C, (14R)-14-OH-expansolide C, (14S)-14-OH-expansolide D, and (14R)-14-OH-expansolide D are formed via two steps of reface- and si-face attacks of the OH groups on the ketone and aldehyde groups, respectively. After these steps, compounds 94–97 were produced from the two groups of 14-OH-expansolides C and D through condensation reactions with (S)-3-hydroxy-2,2-dimethylbutanoic acid and cytosporin D, respectively [43].
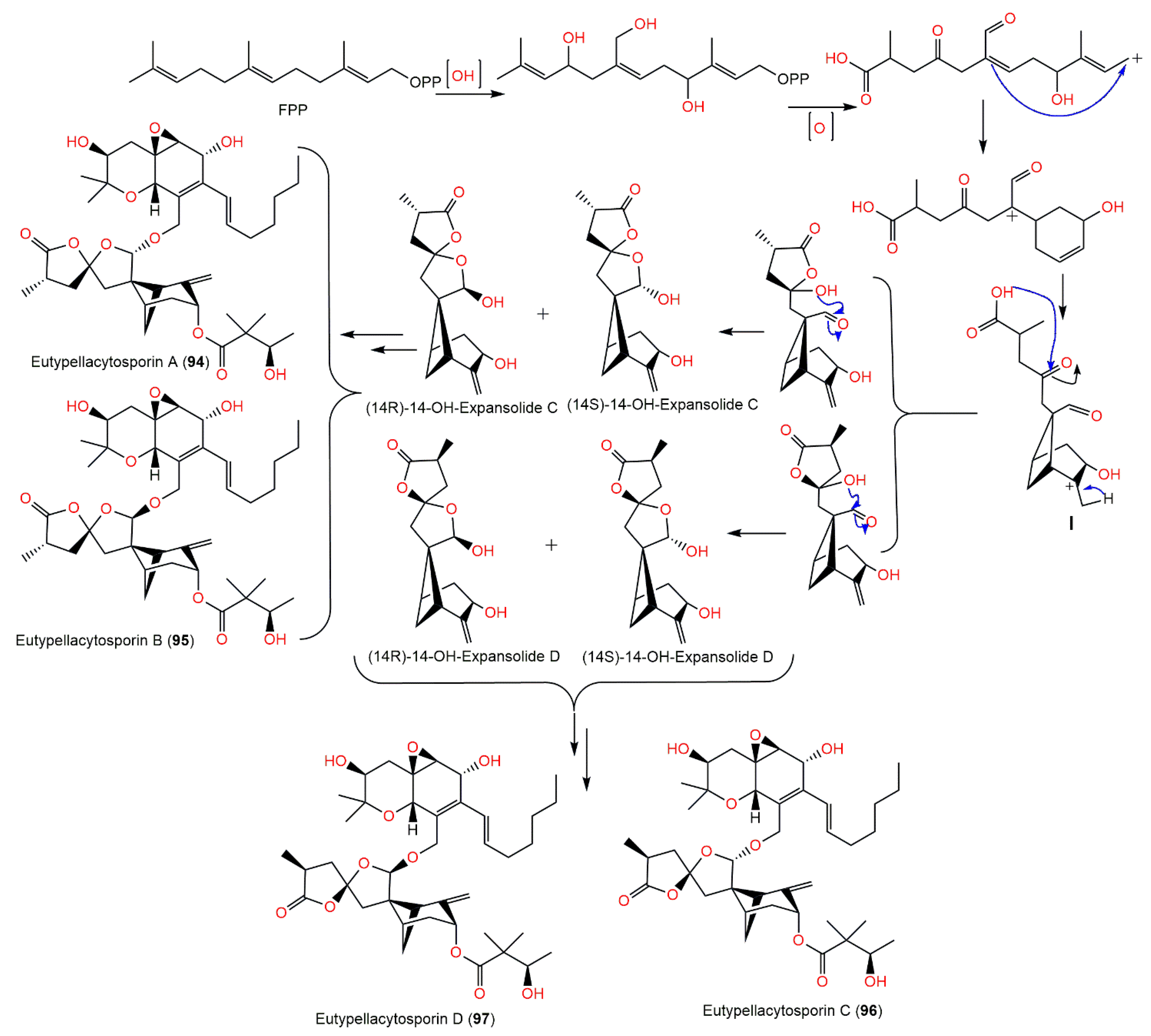
Scheme 6. Biosynthetic pathway of eutypellacytosporins A–D (94–97) [43].
References
- Shen, B. A New Golden Age of Natural Products Drug Discovery. Cell 2015, 163, 1297–1300.
- Krabberød, A.K.; Deutschmann, I.M.; Bjorbækmo, M.F.; Balagué, V.; Giner, C.R.; Ferrera, I.; Garcés, E.; Massana, R.; Gasol, J.M.; Logares, R. Long-Term Patterns of an Interconnected Core Marine Microbiota. Environ. Microbiome 2022, 17, 1–24.
- Imhoff, J.F. Natural Products from Marine fungi—Still an Underrepresented Resource. Mar. Drugs 2016, 14, 19.
- Alves, A.; Sousa, E.; Kijjoa, A.; Pinto, M. Marine-Derived Compounds with Potential use as Cosmeceuticals and Nutricosmetics. Molecules 2020, 25, 2536.
- Ibrahim, S.R.; Fadil, S.A.; Fadil, H.A.; Hareeri, R.H.; Alolayan, S.O.; Abdallah, H.M.; Mohamed, G.A. Dactylospongia Elegans—A Promising Drug Source: Metabolites, Bioactivities, Biosynthesis, Synthesis, and Structural-Activity Relationship. Mar. Drugs 2022, 20, 221.
- Ibrahim, S.R.; Fadil, S.A.; Fadil, H.A.; Hareeri, R.H.; Abdallah, H.M.; Mohamed, G.A. Genus Smenospongia: Untapped Treasure of Biometabolites—Biosynthesis, Synthesis, and Bioactivities. Molecules 2022, 27, 5969.
- Mohamed, G.A.; Ibrahim, S.R. Untapped Potential of Marine-Associated Cladosporium Species: An Overview on Secondary Metabolites, Biotechnological Relevance, and Biological Activities. Mar. Drugs 2021, 19, 645.
- Ibrahim, S.R.M.; Mohamed, G.A.; Al Haidari, R.A.; El-Kholy, A.A.; Zayed, M.F.; Khayat, M.T. Biologically Active Fungal Depsidones: Chemistry, Biosynthesis, Structural Characterization, and Bioactivities. Fitoterapia 2018, 129, 317–365.
- Omar, A.M.; Mohamed, G.A.; Ibrahim, S.R.M. Chaetomugilins and Chaetoviridins-Promising Natural Metabolites: Structures, Separation, Characterization, Biosynthesis, Bioactivities, Molecular Docking, and Molecular Dynamics. J. Fungi 2022, 8, 127.
- Ibrahim, S.R.M.; Sirwi, A.; Eid, B.G.; Mohamed, S.G.A.; Mohamed, G.A. Fungal Depsides-Naturally Inspiring Molecules: Biosynthesis, Structural Characterization, and Biological Activities. Metabolites 2021, 11, 683.
- Ibrahim, S.R.M.; Fadil, S.A.; Fadil, H.A.; Eshmawi, B.A.; Mohamed, S.G.A.; Mohamed, G.A. Fungal Naphthalenones; Promising Metabolites for Drug Discovery: Structures, Biosynthesis, Sources, and Pharmacological Potential. Toxins 2022, 14, 154.
- Ameen, F.; AlNadhari, S.; Al-Homaidan, A.A. Marine Microorganisms as an Untapped Source of Bioactive Compounds. Saudi J. Biol. Sci. 2021, 28, 224–231.
- Bermejo, F.A.; Mateos, A.F.; Escribano, A.M.; Lago, R.M.; Burón, L.M.; López, M.R.; González, R.R. Ti (III)-Promoted Cyclizations. Application to the Synthesis of (E)-Endo-Bergamoten-12-Oic Acids. Moth Oviposition Stimulants Isolated from Lycopersicon Hirsutum. Tetrahedron 2006, 62, 8933–8942.
- López, M.R.; Bermejo, F.A. Total Synthesis of ( )-Massarinolin B and ( )-4-Epi-Massarinolin B, Fungal Metabolites from Massarina Tunicata. Tetrahedron 2006, 62, 8095–8102.
- Macías, F.A.; Varela, R.M.; Simonet, A.M.; Cutler, H.G.; Cutler, S.J.; Hill, R.A. Absolute Configuration of Bioactive Expansolides A and B from Aspergillus Fumigatus Fresenius. Tetrahedron Lett. 2003, 44, 941–943.
- Niu, S.; Xie, C.; Zhong, T.; Xu, W.; Luo, Z.; Shao, Z.; Yang, X. Sesquiterpenes from a Deep-Sea-Derived Fungus Graphostroma Sp. MCCC 3A00421. Tetrahedron 2017, 73, 7267–7273.
- Liu, L.; Gao, H.; Chen, X.; Cai, X.; Yang, L.; Guo, L.; Yao, X.; Che, Y. Brasilamides A–D: Sesquiterpenoids from the Plant Endophytic Fungus Paraconiothyrium Brasiliense. Eur. J. Org. Chem. 2010, 3302–3306.
- Zhao, J.; Feng, J.; Tan, Z.; Liu, J.; Zhang, M.; Chen, R.; Xie, K.; Chen, D.; Li, Y.; Chen, X. Bistachybotrysins A-C, Three Phenylspirodrimane Dimers with Cytotoxicity from Stachybotrys Chartarum. Bioorg. Med. Chem. Lett. 2018, 28, 355–359.
- Pei, Y.; Zhang, L.; Wu, X.; Wu, H.; Wang, H.; Wang, Y.; Chen, G. Polyhydroxylated Bergamotane-Type Sesquiterpenoids from Cultures of Paraconiothyrium Sporulosum YK-03 and their Absolute Configurations. Phytochemistry 2022, 194, 113000.
- Zamora, R.; Vodovotz, Y.; Billiar, T.R. Inducible Nitric Oxide Synthase and Inflammatory Diseases. Mol. Med. 2000, 6, 347–373.
- Niu, S.; Liu, D.; Shao, Z.; Liu, J.; Fan, A.; Lin, W. Chemical Epigenetic Manipulation Triggers the Production of Sesquiterpenes from the Deep-Sea Derived Eutypella Fungus. Phytochemistry 2021, 192, 112978.
- Pena-Rodriguez, L.M.; Armingeon, N.A.; Chilton, W.S. Toxins from Weed Pathogens, I. Phytotoxins from a Bipolaris Pathogen of Johnson Grass. J. Nat. Prod. 1988, 51, 821–828.
- Pena-Rodriguez, L.M.; Chilton, W.S. Victoxinine and Prehelminthosporolactone, Two Minor Phytotoxic Metabolites Produced by Bipolaris Sp., a Pathogen of Johnson Grass. J. Nat. Prod. 1989, 52, 899–901.
- Kono, Y.; Takeuchi, S.; Daly, J.M. Isolation and Structure of a New Victoxinine Derivative Produced by Helminthosporium Victoriae. Agric. Biol. Chem. 1983, 47, 2701–2702.
- Pringle, R.B.; Braun, A.C. Constitution of the Toxin of Helminthosporium Victoriae. Nature 1958, 181, 1205–1206.
- Dorn, F.; Arigoni, D. Structure of Victoxinine. J. Chem. Soc. Chem. Commun. 1972, 24, 1342–1343.
- Kovarik, J.M.; Burtin, P. Immunosuppressants in Advanced Clinical Development for Organ Transplantation and Selected Autoimmune Diseases. Expert Opin. Emerg. Drugs 2003, 8, 47–62.
- Klinker, M.W.; Lundy, S.K. Multiple Mechanisms of Immune Suppression by B Lymphocytes. Mol. Med. 2012, 18, 123–137.
- Dai, Q.; Zhang, F.; Li, Z.; He, J.; Feng, T. Immunosuppressive Sesquiterpenoids from the Edible Mushroom Craterellus Odoratus. J. Fungi 2021, 7, 1052.
- Che, Y.; Gloer, J.B.; Koster, B.; Malloch, D. Decipinin A and Decipienolides A and B: New Bioactive Metabolites from the Coprophilous Fungus Podospora Decipiens. J. Nat. Prod. 2002, 65, 916–919.
- Zhao, Z.; Zhao, K.; Chen, H.; Bai, X.; Zhang, L.; Liu, J. Terpenoids from the Mushroom-Associated Fungus Montagnula Donacina. Phytochemistry 2018, 147, 21–29.
- Wang, Y.; Xia, G.; Wang, L.; Ge, G.; Zhang, H.; Zhang, J.; Wu, Y.; Lin, S. Purpurolide A, 5/5/5 Spirocyclic Sesquiterpene Lactone in Nature from the Endophytic Fungus Penicillium Purpurogenum. Org. Lett. 2018, 20, 7341–7344.
- Xia, G.; Wang, L.; Zhang, J.; Wu, Y.; Ge, G.; Wang, Y.; Lin, P.; Lin, S. Three New Polyoxygenated Bergamotanes from the Endophytic Fungus Penicillium Purpurogenum IMM 003 and their Inhibitory Activity Against Pancreatic Lipase. Chin. J. Nat. Med. 2020, 18, 75–80.
- Wang, W.; Shi, Y.; Liu, Y.; Zhang, Y.; Wu, J.; Zhang, G.; Che, Q.; Zhu, T.; Li, M.; Li, D. Brasilterpenes A-E, Bergamotane Sesquiterpenoid Derivatives with Hypoglycemic Activity from the Deep Sea-Derived Fungus Paraconiothyrium Brasiliense HDN15-135. Mar Drugs 2022, 20, 514.
- Ying, Y.; Fang, C.; Yao, F.; Yu, Y.; Shen, Y.; Hou, Z.; Wang, Z.; Zhang, W.; Shan, W.; Zhan, Z. Bergamotane Sesquiterpenes with Alpha-glucosidase Inhibitory Activity from the Plant Pathogenic Fungus Penicillium Expansum. Chem. Biodivers. 2017, 14, e1600184.
- Kimura, Y.; Nakajima, H.; Hamasaki, T.; Sugawara, F.; Parkanyi, L.; Clardy, J. Pinthunamide, a New Tricyclic Sesquiterpene Amide Produced by a Fungus, Ampullifernia Sp. Tetrahedron Lett. 1989, 30, 1267–1270.
- Kimura, Y.; Nakajima, H.; Hamasaki, T.; Matsumoto, T.; Matsuda, Y.; Tsuneda, A. Ampullicin and Isoampullicin, New Metabolites from an Ampulliferina-Like Fungus Sp. no. 27. Agric. Biol. Chem. 1990, 54, 813–814.
- Kimura, Y.; Matsumoto, T.; Nakajima, H.; Hamasaki, T.; Matsuda, Y. Dihydroampullicin, a New Plant Growth Regulators Produced by the Ampulliferina-Like Fungus Sp. no. 27. Biosci. Biotechnol. Biochem. 1993, 57, 687–688.
- Bermejo, F.A.; Rico-Ferreira, R.; Bamidele-Sanni, S.; García-Granda, S. Total Synthesis of (+)-Ampullicin and (+)-Isoampullicin: Two Fungal Metabolites with Growth Regulatory Activity Isolated from Ampulliferina Sp. 27. J. Org. Chem. 2001, 66, 8.
- Wang, Y.; Li, D.; Li, Z.; Sun, Y.; Hua, H.; Liu, T.; Bai, J. Terpenoids from the Marine-Derived Fungus Aspergillus Fumigatus YK-7. Molecules 2016, 21, 31.
- Wu, Z.; Wu, Y.; Chen, G.; Hu, D.; Li, X.; Sun, X.; Guo, L.; Li, Y.; Yao, X.; Gao, H. Xylariterpenoids A–D, Four New Sesquiterpenoids from the Xylariaceae Fungus. RSC Adv. 2014, 4, 54144–54148.
- Guo, Z.; Ren, F.; Che, Y.; Liu, G.; Liu, L. New Bergamotane Sesquiterpenoids from the Plant Endophytic Fungus Paraconiothyrium Brasiliense. Molecules 2015, 20, 14611–14620.
- Zhang, Y.; Yu, H.; Xu, W.; Hu, B.; Guild, A.; Zhang, J.; Lu, X.; Liu, X.; Jiao, B. Eutypellacytosporins A–D, Meroterpenoids from the Arctic Fungus Eutypella Sp. D-1. J. Nat. Prod. 2019, 82, 3089–3095.
More
Information
Subjects:
Biotechnology & Applied Microbiology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
2 times
(View History)
Update Date:
19 Dec 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





