Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Guangbin Zhou | -- | 2050 | 2022-12-08 10:42:48 | | | |
| 2 | Sirius Huang | Meta information modification | 2050 | 2022-12-09 01:54:29 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Cao, B.; Qin, J.; Pan, B.; Qazi, I.H.; Ye, J.; Fang, Y.; Zhou, G. Antioxidant Mechanisms in Oocytes. Encyclopedia. Available online: https://encyclopedia.pub/entry/38303 (accessed on 25 May 2026).
Cao B, Qin J, Pan B, Qazi IH, Ye J, Fang Y, et al. Antioxidant Mechanisms in Oocytes. Encyclopedia. Available at: https://encyclopedia.pub/entry/38303. Accessed May 25, 2026.
Cao, Beijia, Jianpeng Qin, Bo Pan, Izhar Hyder Qazi, Jiangfeng Ye, Yi Fang, Guangbin Zhou. "Antioxidant Mechanisms in Oocytes" Encyclopedia, https://encyclopedia.pub/entry/38303 (accessed May 25, 2026).
Cao, B., Qin, J., Pan, B., Qazi, I.H., Ye, J., Fang, Y., & Zhou, G. (2022, December 08). Antioxidant Mechanisms in Oocytes. In Encyclopedia. https://encyclopedia.pub/entry/38303
Cao, Beijia, et al. "Antioxidant Mechanisms in Oocytes." Encyclopedia. Web. 08 December, 2022.
Copy Citation
Although oocytes can mitigate oxidative stress via intrinsic antioxidant systems, the formation of ribonucleoprotein granules, mitophagy, and the cryopreservation-inflicted oxidative damage cannot be completely eliminated. Therefore, exogenous antioxidants such as melatonin and resveratrol are widely used in oocyte cryopreservation to reduce oxidative damage through direct or indirect scavenging of reactive oxygen species (ROS).
antioxidants
cryopreservation
mitophagy
oocyte
oxidative stress
ROS
1. Antioxidant Mechanisms in Oocytes
1.1. Ribonucleoprotein Particles Can Reduce Oxidative Damage and Protect Cells
Mammalian ribonucleoprotein particles (RNPs) are granules composed of RNA and protein, including stress granules (SGs) and processing bodies (PBs) [1]. SGs are membrane-less dense messenger ribonucleoprotein (mRNP) particles formed in the cytoplasm, containing polyadenylated mRNAs forced to suspend translation due to the stress [2], small ribosomal subunits (40S), translation initiation factor PABP, and RNA binding protein TIA/1R. Similarly, several non-RNA-binding proteins such as signal transduction proteins are also contained in SGs [3]; however, RNAs whose translation is induced by stress are not included in the SGs [4].
Under physiological conditions, PBs contain a large number of mRNAs, protein complexes related to 5′-3′ mRNA degradation such as XRN1 and DCP1 [5], and proteins related to RNA translation inhibition such as GW182 [6]. PBs become larger and more numerous when they are subjected to stress [7][8][9], whereas SGs can only be formed by stress induction. It is precisely due to the formation of the SGs and PBs that cells are able to selectively store mRNAs so that they can recover quickly from various stressful environments.
SGs appear to dock with PBs under oxidative stress. Oxidative damage to RNA results in the formation of 8-hydroxyguanine (8-OHG) at the RNA guanine base. Y box-binding protein 1 (YB-1), which specifically recognizes and binds 8-OHG, is present in PBs [10]. YB-1 is transferred from PBs to SGs under certain types of stress. During stress recovery, the SGs can act as a “sorting” domain for mRNA [10], where mRNA without oxidative damage may be recycled and returned to the polysome to restart protein synthesis. Conversely, oxidized mRNA is transferred to the adjacent PBs for catabolism. Furthermore, SGs are resistant to oxidative damage and their antioxidant activity is regulated by their core components, the GTPase-activating protein SH3 domain-binding protein 1 (G3BP1) and the ubiquitin-specific protease 10 (USP10) [11].
Under physiological conditions, excessive G3BP1 is able to inhibit the antioxidant activity of USP10 to maintain ROS homeostasis. However, upon exposure to external stress such as high concentrations of hydrogen peroxide [12], the inhibition of USP10 by G3BP1 is abrogated and USP10 is induced to phosphorylate by ataxia-telangiectasia-mutated protein (ATM) or activated by ATM-phosphorylated protein, thereby reducing ROS production and apoptosis [11]. Therefore, after cells are subjected to oxidative stress, SGs, on the one hand, dock with P-bodies. On the other hand, as a component of the antioxidant mechanisms, SGs are actively involved in the process of reducing oxidative damage of cells. All these processes relevant to SGs and PBs are depicted in Figure 1.
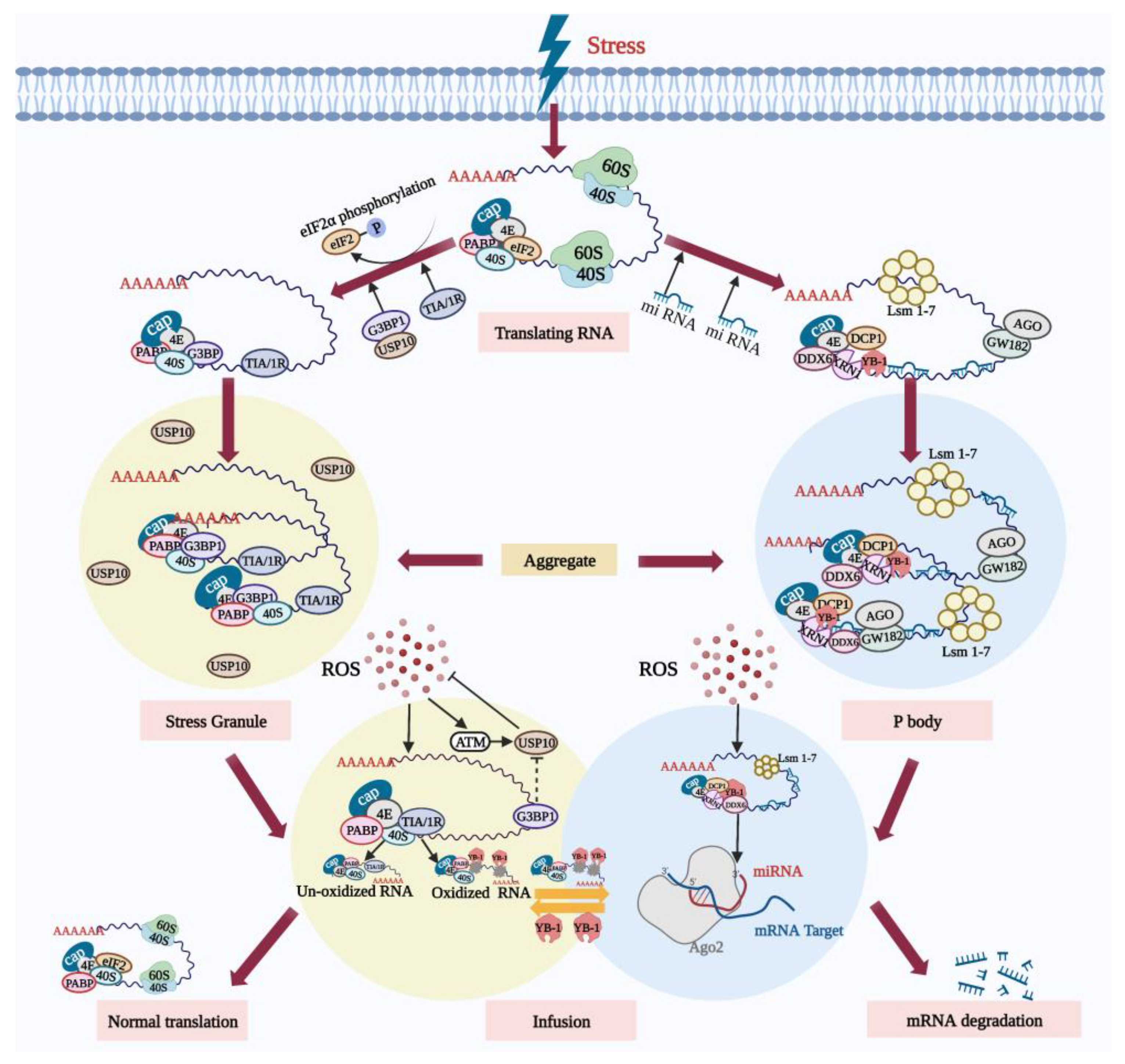
Figure 1. SGs and PBs actively participate in the degradation of oxidized RNA during oxidative stress. Following oxidative stress, the translation of some mRNA in cells is inhibited. On the one hand, the stress-induced phosphorylation of eIF2α inhibits translation initiation, and assembly of the stalled 48S initiation complex promotes the formation and aggregation of SGs. A large amount of RNA that has paused translation is sequestered in the SGs. SGs’ core component G3BP1 relieves the inhibition of USP10. ROS activates ATM and promotes the phosphorylation of USP10, triggering the antioxidant response. On the other hand, the stress-induced PBs mediate the RNA degradation. SGs and PBs dock, and RNA without oxidative damage can be re-translated under stress conditions. However, oxidized RNA is recognized by the YB-1 protein and transferred to PBs for degradation. Created with BioRender.com (accessed on 19 October 2022).
1.2. Mitophagy Can Reduce Oocyte Apoptosis Induced by Oxidative Stress
Mitophagy is a special autophagic process that selectively removes excessive or damaged mitochondria (see Figure 2). MMP loss heralds the onset of mitophagy and induces accumulation of PTEN-inducible kinase1 (PINK1) in the mitochondrial outer membrane, which regulates Parkin recruitment into the damaged mitochondria [13]. Damaged mitochondria can be selectively phagocytosed by autophagic vesicles to form double-membrane autophagosomes and then fuse into lysosomes to be degraded by hydrolytic enzymes [14]. Parkin is an E3 ubiquitous ligase that promotes mitochondrial autophagy in damaged mitochondria. Mitophagy plays an important role in regulating intracellular mitochondrial number and maintaining mitochondrial function.
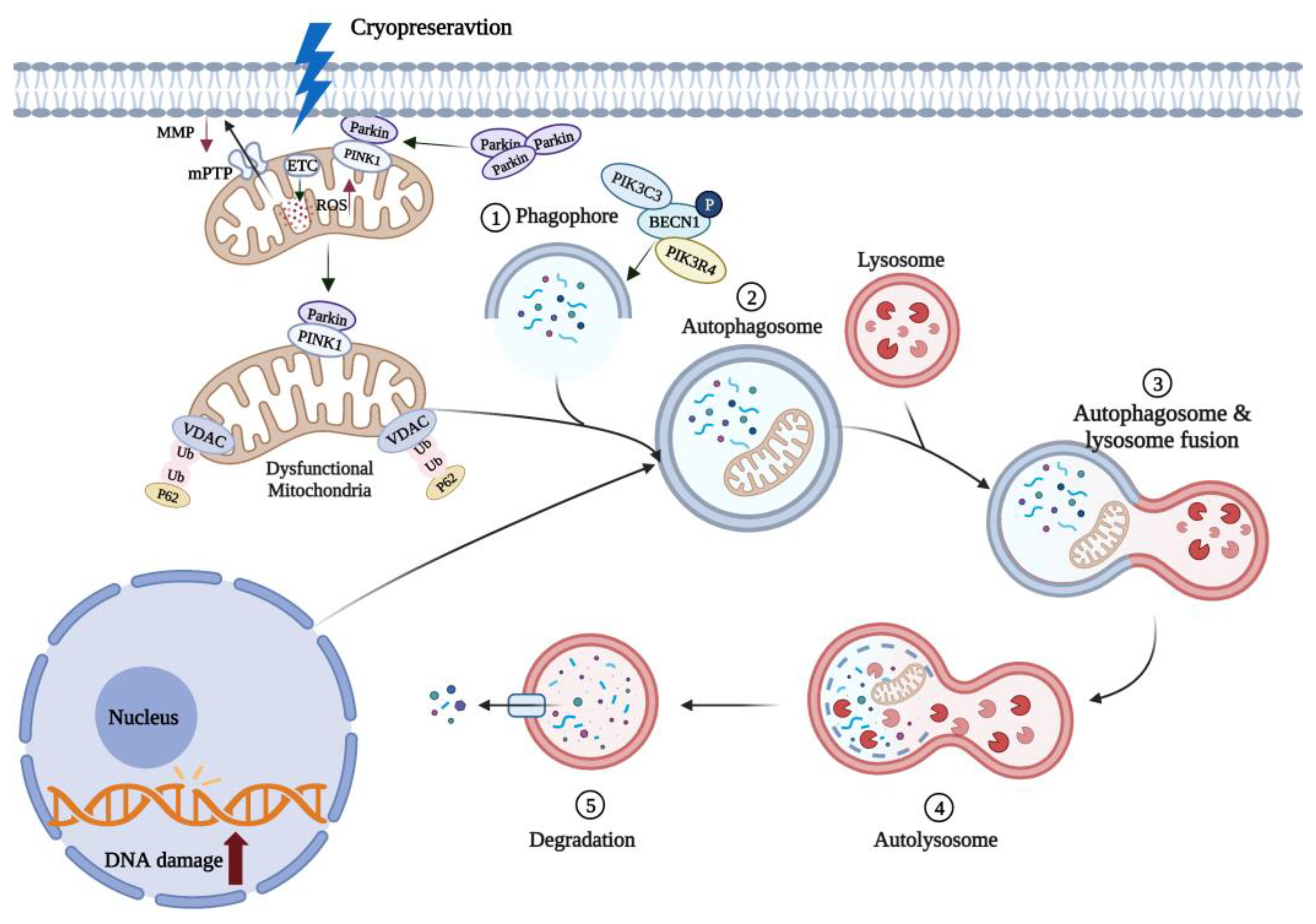
Figure 2. Mitophagy can remove oxidized mitochondria in oocytes. Cryopreservation causes oxidative stress in oocytes and rapid depolarization of MMP, which promotes Parkin entry into damaged mitochondria. Parkin induces polyubiquitination of VDAC in mitochondria, and ubiquitinated VDAC recruits p62 into mitochondria to induce mitophagy. These damaged mitochondria are phagocytosed by autophagic vesicles to form autophagosomes. Mature autophagosomes fuse with lysosomes to form autolysosomes, and the contained mitochondria are subsequently degraded. ↑ and ↓, respectively, indicate a significant increase or decrease. Created with BioRender.com (accessed on 19 October 2022).
It has been experimentally demonstrated that cryopreservation (low-temperatures) can promote mitophagy [15]. The expression level of Beclin1 (BECN1), a key regulator of autophagy in oocytes, was significantly increased following mouse oocyte cryopreservation [15], while the accumulation of P62, a characteristic substrate of autophagy, was decreased [16]. Recently, it has been shown that mitophagy can improve the survival of vitrified-warmed oocytes by eliminating oxidation-damaged mitochondria (see Figure 2). Lately, it has been demonstrated that vitrified-thawed porcine MII oocytes can regulate cell activity through PINK/Parkin-mediated mitophagy [17]. When mitophagy was inhibited with chloroquine (CQ), mitochondrial dysfunction and oxidative damage was increased in vitrified porcine oocytes, reducing the in vitro oocyte developmental potential [18]. Following knockdown of autophagy-related genes in yeast, damaged mitochondria that are not cleared will continue to generate excessive ROS, compromising cell survival [19][20]. It is interesting to note that mitophagy does not always protect the vitrified oocytes, but excessive mitophagy can also lead to a decreased mouse oocyte developmental potential in vitro [21]. This evidence collectively highlights the fact that the mitophagy-mediated protection of vitrified-warmed oocytes is so far limited, therefore, further mechanistic studies are required to improve our understanding of this fascinating phenomenon.
2. Oocyte Cryopreservation and Antioxidants
2.1. Endogenous Antioxidants Resist Oxidative Damage Induced by Oocyte Cryopreservation
There are three major enzymatic antioxidants in cells, namely superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPX) [22]. SOD can provide an initial detoxification of O2•−, and its products are then further detoxified to water by CAT or GPX. The upregulation of SOD gene expression levels in mouse and porcine oocytes after vitrification [23][24] facilitates the scavenging of cryopreservation-induced ROS, but the decrease in GSH levels [25] exacerbates the oxidative stress in cells to the extent that 50 IU/mL SOD needs to be added to the freezing solution to scavenge the excessive ROS and to improve fertilization and survival of mouse oocytes [26]. In vitrified-warmed sheep oocytes, it was observed that the CAT activity was significantly higher compared to their control counterparts during in vitro culture [27]. Similarly, the addition of catalase during vitrification of goat ovarian tissue was effective in reducing freezing-induced accumulation of ROS [28].
In addition, glutathione (GSH), cysteine (CYS), and cysteamine (CSH) are key endogenous non-enzymatic antioxidants in oocytes. GSH synthesized in oocytes can regulate the sulfur–oxygen reduction state of cells [29], and promote cytoplasmic maturation. GSH can protect against the oxidative-stress-inflicted damage to the morphology and function of the spindle during in vitro maturation of bovine oocytes [30], and improve the development potential of sheep embryos [31]. Cysteine, a small mercaptan that can enhance the synthesis of GSH, can be added to bovine oocyte maturation medium to reduce oxidative damage, potentially by increasing the level of GSH [32]. Cysteamine can also directly scavenge •OH [33], contributing to maintenance of redox status and high GSH/GSSG ratio in the oocytes. However, after vitrification, the intracellular synthesis of GSH was significantly reduced, and the addition of CYS or CSH also failed to significantly improve the blastocyst formation rate of vitrified-warmed bovine oocytes after IVF [34].
It, therefore, appears that the exogenous antioxidants are beneficial in reducing cryo-oxidative damage, particularly due to the fact that endogenous enzymatic antioxidants alone are unable to mitigate cryopreservation-inflicted ROS.
2.2. Exogenous Non-Enzymatic Antioxidants Protect Oocytes from Oxidative Damage Induced by Cryopreservation
To reduce the cryopreservation-inflicted oxidative damage to oocytes, non-enzymatic antioxidants are usually applied in the vitrification, warming, and/or culture media to mitigate the oxidative stress. Numerous antioxidants including melatonin [35][36][37], resveratrol [38][39][40], L-carnitine [41][42], quercetin [43], vitamin E [44][45], astaxanthin [46], proline [47], and coenzyme Q10 [48] have been demonstrated to have beneficial impact on oocyte maturation and development. The results of the previous studies provide encouraging evidence that the oxidative damage in vitrified-warmed oocytes can be reduced/mitigated, and their in vitro developmental potential can be greatly improved. Table 1 summarized the fascinating results of relevant studies involving different antioxidants.
Table 1. The effects of exogenous non-enzymatic antioxidants on oocyte cryopreservation.
| Components | Concentration | Oocyte Types | Effects | References |
|---|---|---|---|---|
| Melatonin | 10−7 mol/L | Mouse GV-stage oocytes | (↓) ROS levels, spindle damage | [37] |
| 10−9 mol/L | Human MII-stage oocytes | (↑) Blastocyst formation rate, Oocyte maturation rate, MMP, ATP |
[35] | |
| Resveratrol | 1.5 × 10−3 mol/L | Cat GV-stage oocytes | (↑) Oocyte maturation rate, cleavage rate, embryo developmental ratio |
[40] |
| 5 × 10−6 mol/L | Cat COC complexes | (↓) ROS levels (↑) GSH levels |
[39] | |
| 2 × 10−6 mol/L | Porcine MII-stage oocytes | (↓) Cell apoptosis | [38] | |
| Quercetin | 5 × 10−6 mol/L | Mouse GV-stage oocytes | (↑) Oocyte maturation rate, embryo developmental ratio |
[43] |
| Vitamin E | 3 × 10−4 mol/L | Bovine MII-stage oocytes | (↑) Blastocyst formation rate | [44] |
| 10−4 mol/L | Mouse MII-stage oocytes | (↑) Oocyte morphology and ultrastructure |
[45] | |
| Astaxanthin | 2.5 × 10−6 mol/L | Porcine GV-stage oocytes | (↑) GSH levels, lysosomal activity (↓) ROS levels, cathepsin B activity |
[46] |
| Proline | 2 mol/L | Mouse MII-stage oocytes | (↑) Spindle and mitochondrial function | [47] |
| Coenzyme Q10 | 5 × 10−5 mol/L | Bovine COC complexes | (↑) Cell survival after vitrification (↑) Migration of cortical granule |
[48] |
| L-carnitine | 0.8 × 10−3 mol/L | Porcine COC complexes | (↑) SOD1 gene expression | [41] |
| 0.6 g/mL | Mouse COC complexes | (↑) GSH levels | [42] | |
| MitoQ | 2 × 10−8 mol/L | Mouse MII-stage oocytes | (↑) MMP, cell survival | [49] |
↑ and ↓ indicate a significant increase or decrease, respectively.
Recently, it has been shown that mitochondria-targeted antioxidants are more effective compared to the traditional antioxidants. For instance, mitoquinone (MitoQ), which consists of a triphenylphosphate positive cation is covalently attached to the benzoquinone portion of coenzyme Q10 in the respiratory chain complex through a ten-carbon aliphatic chain, can selectively scavenge excessive mitochondrial ROS [50] to maintain ROS homeostasis in mitochondria [51]. MitoQ can also protect the integrity of mitochondrial membranes in vitrified-warmed oocytes. It has been reported that the addition of 2 × 10−8 mol/L MitoQ to the freezing solution significantly enhanced the mitochondrial membrane potential and cell survival in vitrified-warmed mouse MII-stage oocytes [49]. However, single administration of targeted antioxidants or autophagy activators such as rapamycin to reduce oxidative stress has limited effects, so dual-targeted therapies that can simultaneously modulate antioxidant signaling pathways and autophagy are being explored [52].
There is an increasing need to upgrade the existing antioxidants to amplify their antioxidant properties. It is promising that the search for forming efficient remedial strategies involving potent antioxidants is also at the center of attention of reproductive biology researchers and clinicians. For instance, curcumin is not only used in clinical applications for its anti-cancer effects, but also has attracted great attention from researchers around the world for its antioxidant and oxygen-radical-scavenging effects [53][54]. Its chemical structure was modified to obtain a new antioxidant molecule, acetyl zingerone, whose ability to reduce H2O2 and •OH is 17.7 and 39.6 times greater than that of the traditional antioxidant vitamin E, respectively [55]. Not only this but it has a stable structure that can withstand ultraviolet light and maintain its high activity, while vitamin E basically drops to practically zero [55]. Therefore, the exploitation of acetyl zingerone is expected to improve the efficiency of oocyte cryopreservation.
It is important to note that the impact of supplementation of exogenous antioxidants on oocyte development has been shown to be concentration-dependent and varies between species [56][57][58]. Even within the same species, antioxidants can render different effects based on the developmental stages of the oocytes at which they are used [56][57][58]. Intriguingly, there is evidence that, instead of having beneficial effects, high concentrations of antioxidants can also disrupt cell development by interfering with intracellular antioxidant mechanisms and converting oxidative stress into reductive stress [59][60], leading to further damage. There is an evidence that antioxidants can sometimes render more beneficial effects when used in combination or in different chemical forms/presentations than those used alone. For instance, co-encapsulation of melatonin and resveratrol in solid lipid nano-carriers (SLNS) may provide more effective antioxidant synergy and can significantly reduce ROS levels in vitrified-warmed mouse GV-stage oocytes [61]. However, not all antioxidants are more effective when used in combination. This notion is supported by evidence that the single administration of catalase had better clonogenic parameters compared to its combination with alginose [62].
References
- Mittag, T.; Parker, R. Multiple Modes of Protein-Protein Interactions Promote RNP Granule Assembly. J. Mol. Biol. 2018, 430, 4636–4649.
- Anderson, P.; Kedersha, N. RNA granules. J. Cell Biol. 2006, 172, 803–808.
- Cadena Sandoval, M.; Heberle, A.M.; Rehbein, U.; Barile, C.; Ramos Pittol, J.M.; Thedieck, K. mTORC1 Crosstalk with Stress Granules in Aging and Age-Related Diseases. Front. Aging 2021, 2, 761333.
- Kedersha, N.; Anderson, P. Stress granules: Sites of mRNA triage that regulate mRNA stability and translatability. Biochem. Soc. Trans. 2002, 30, 963–969.
- Parker, R.; Sheth, U. P bodies and the control of mRNA translation and degradation. Mol. Cell 2007, 25, 635–646.
- Standart, N.; Minshall, N. Translational control in early development: CPEB, P-bodies and germinal granules. Biochem. Soc. Trans. 2008, 36, 671–676.
- Riggs, C.L.; Kedersha, N.; Ivanov, P.; Anderson, P. Mammalian stress granules and P bodies at a glance. J. Cell Sci. 2020, 133, jcs242487.
- Kedersha, N.; Anderson, P. Mammalian stress granules and processing bodies. Methods Enzymol. 2007, 431, 61–81.
- Kedersha, N.; Stoecklin, G.; Ayodele, M.; Yacono, P.; Lykke-Andersen, J.; Fritzler, M.J.; Scheuner, D.; Kaufman, R.J.; Golan, D.E.; Anderson, P. Stress granules and processing bodies are dynamically linked sites of mRNP remodeling. J. Cell Biol. 2005, 169, 871–884.
- Yang, W.H.; Bloch, D.B. Probing the mRNA processing body using protein macroarrays and “autoantigenomics”. RNA 2007, 13, 704–712.
- Takahashi, M.; Higuchi, M.; Matsuki, H.; Yoshita, M.; Ohsawa, T.; Oie, M.; Fujii, M. Stress granules inhibit apoptosis by reducing reactive oxygen species production. Mol. Cell. Biol. 2013, 33, 815–829.
- Emara, M.M.; Fujimura, K.; Sciaranghella, D.; Ivanova, V.; Ivanov, P.; Anderson, P. Hydrogen peroxide induces stress granule formation independent of eIF2α phosphorylation. Biochem. Biophys. Res. Commun. 2012, 423, 763–769.
- Kim, I.; Rodriguez-Enriquez, S.; Lemasters, J.J. Selective degradation of mitochondria by mitophagy. Arch. Biochem. Biophys. 2007, 462, 245–253.
- Wang, R.; Wang, G. Autophagy in Mitochondrial Quality Control. In Autophagy: Biology and Diseases: Basic Science; Qin, Z.-H., Ed.; Springer: Singapore, 2019; pp. 421–434.
- Bang, S.; Shin, H.; Song, H.; Suh, C.S.; Lim, H.J. Autophagic activation in vitrified-warmed mouse oocytes. Reproduction 2014, 148, 11–19.
- Ito, J.; Shirasuna, K.; Kuwayama, T.; Iwata, H. Resveratrol treatment increases mitochondrial biogenesis and improves viability of porcine germinal-vesicle stage vitrified-warmed oocytes. Cryobiology 2020, 93, 37–43.
- Xu, J.; Sun, L.; Wu, C.; Zhang, S.; Ju, S.; Rui, R.; Zhang, D.; Dai, J. Involvement of PINK1/Parkin-mediated mitophagy in mitochondrial functional disruption under oxidative stress in vitrified porcine oocytes. Theriogenology 2021, 174, 160–168.
- Xu, J.; Zhang, D.; Ju, S.; Sun, L.; Zhang, S.; Wu, C.; Rui, R.; Dai, J. Mitophagy is involved in the mitochondrial dysfunction of vitrified porcine oocytes. Mol. Reprod. Dev. 2021, 88, 427–436.
- Kurihara, Y.; Kanki, T.; Aoki, Y.; Hirota, Y.; Saigusa, T.; Uchiumi, T.; Kang, D. Mitophagy plays an essential role in reducing mitochondrial production of reactive oxygen species and mutation of mitochondrial DNA by maintaining mitochondrial quantity and quality in yeast. J. Biol. Chem. 2012, 287, 3265–3272.
- Yorimitsu, T.; Klionsky, D.J. Autophagy: Molecular machinery for self-eating. Cell Death Differ. 2005, 12 (Suppl. S2), 1542–1552.
- Lee, G.K.; Shin, H.; Lim, H.J. Rapamycin Influences the Efficiency of in vitro Fertilization and Development in the Mouse: A Role for Autophagic Activation. Asian-Australas. J. Anim. Sci. 2016, 29, 1102–1110.
- Wang, L.; Tang, J.; Wang, L.; Tan, F.; Song, H.; Zhou, J.; Li, F. Oxidative stress in oocyte aging and female reproduction. J. Cell. Physiol. 2021, 236, 7966–7983.
- Habibi, A.; Farrokhi, N.; Moreira da Silva, F.; Bettencourt, B.F.; Bruges-Armas, J.; Amidi, F.; Hosseini, A. The effects of vitrification on gene expression in mature mouse oocytes by nested quantitative PCR. J. Assist. Reprod Genet. 2010, 27, 599–604.
- Niu, Y.; Dai, J.; Wu, C.; Chen, Y.; Zhang, S.; Zhang, D. The application of apoptotic inhibitor in apoptotic pathways of MII stage porcine oocytes after vitrification. Reprod. Domest. Anim. = Zuchthyg. 2016, 51, 953–959.
- Somfai, T.; Ozawa, M.; Noguchi, J.; Kaneko, H.; Kuriani Karja, N.W.; Farhudin, M.; Dinnyés, A.; Nagai, T.; Kikuchi, K. Developmental competence of in vitro-fertilized porcine oocytes after in vitro maturation and solid surface vitrification: Effect of cryopreservation on oocyte antioxidative system and cell cycle stage. Cryobiology 2007, 55, 115–126.
- Dinara, S.; Sengoku, K.; Tamate, K.; Horikawa, M.; Ishikawa, M. Effects of supplementation with free radical scavengers on the survival and fertilization rates of mouse cryopreserved oocytes. Hum. Reprod. 2001, 16, 1976–1981.
- Succu, S.; Gadau, S.D.; Serra, E.; Zinellu, A.; Carru, C.; Porcu, C.; Naitana, S.; Berlinguer, F.; Leoni, G.G. A recovery time after warming restores mitochondrial function and improves developmental competence of vitrified ovine oocytes. Theriogenology 2018, 110, 18–26.
- Carvalho, A.A.; Faustino, L.R.; Silva, C.M.; Castro, S.V.; Lobo, C.H.; Santos, F.W.; Santos, R.R.; Campello, C.C.; Bordignon, V.; Figueiredo, J.R.; et al. Catalase addition to vitrification solutions maintains goat ovarian preantral follicles stability. Res. Vet. Sci. 2014, 97, 140–147.
- Hammond, C.L.; Lee, T.K.; Ballatori, N. Novel roles for glutathione in gene expression, cell death, and membrane transport of organic solutes. J. Hepatol. 2001, 34, 946–954.
- García-Martínez, T.; Vendrell-Flotats, M.; Martínez-Rodero, I.; Ordóñez-León, E.A.; Álvarez-Rodríguez, M.; López-Béjar, M.; Yeste, M.; Mogas, T. Glutathione Ethyl Ester Protects In Vitro-Maturing Bovine Oocytes against Oxidative Stress Induced by Subsequent Vitrification/Warming. Int. J. Mol. Sci. 2020, 21, 7547.
- de Matos, D.G.; Furnus, C.C. The importance of having high glutathione (GSH) level after bovine in vitro maturation on embryo development: Effect of β-mercaptoethanol, cysteine and cystine. Theriogenology 2000, 53, 761–771.
- de Matos, D.G.; Furnus, C.C.; Moses, D.F.; Baldassarre, H. Effect of cysteamine on glutathione level and developmental capacity of bovine oocyte matured in vitro. Mol. Reprod. Dev. 1995, 42, 432–436.
- Zheng, S.; Newton, G.L.; Gonick, G.; Fahey, R.C.; Ward, J.F. Radioprotection of DNA by thiols: Relationship between the net charge on a thiol and its ability to protect DNA. Radiat. Res. 1988, 114, 11–27.
- Hara, H.; Yamane, I.; Noto, I.; Kagawa, N.; Kuwayama, M.; Hirabayashi, M.; Hochi, S. Microtubule assembly and in vitro development of bovine oocytes with increased intracellular glutathione level prior to vitrification and in vitro fertilization. Zygote 2014, 22, 476–482.
- Zhang, Z.; Mu, Y.; Ding, D.; Zou, W.; Li, X.; Chen, B.; Leung, P.C.; Chang, H.M.; Zhu, Q.; Wang, K.; et al. Melatonin improves the effect of cryopreservation on human oocytes by suppressing oxidative stress and maintaining the permeability of the oolemma. J. Pineal Res. 2021, 70, e12707.
- Yang, J.; Guo, S.; Pan, B.; Qazi, I.H.; Qin, J.; Zang, S.; Han, H.; Meng, Q.; Zhou, G. Melatonin promotes in vitro maturation of vitrified-warmed mouse GV oocytes potentially by modulating MAD2 protein expression of SAC component through MTRs. Cryobiology 2021, 102, 82–91.
- Guo, S.; Yang, J.; Qin, J.; Qazi, I.H.; Pan, B.; Zang, S.; Lv, T.; Deng, S.; Fang, Y.; Zhou, G. Melatonin Promotes In Vitro Maturation of Vitrified-Warmed Mouse Germinal Vesicle Oocytes, Potentially by Reducing Oxidative Stress through the Nrf2 Pathway. Animals 2021, 11, 2324.
- Giaretta, E.; Spinaci, M.; Bucci, D.; Tamanini, C.; Galeati, G. Effects of resveratrol on vitrified porcine oocytes. Oxid. Med. Cell. Longev. 2013, 2013, 920257.
- Piras, A.R.; Ariu, F.; Falchi, L.; Zedda, M.T.; Pau, S.; Schianchi, E.; Paramio, M.; Bogliolo, L. Resveratrol treatment during maturation enhances developmental competence of oocytes after prolonged ovary storage at 4 °C in the domestic cat model. Theriogenology 2020, 144, 152–157.
- Comizzoli, P.; Wildt, D.E.; Pukazhenthi, B.S. In Vitro compaction of germinal vesicle chromatin is beneficial to survival of vitrified cat oocytes. Reprod. Domest. Anim. = Zuchthyg. 2009, 44 (Suppl. S2), 269–274.
- Xu, H.; Jia, C.; Cheng, W.; Zhang, T.; Tao, R.; Ma, Y.; Si, L.; Xu, Y.; Li, J. The Effect of L-Carnitine Additive During In Vitro Maturation on the Vitrification of Pig Oocytes. Cell. Reprogr. 2020, 22, 198–207.
- Zare, Z.; Rezaei, N.; Mohammadi, M. Treatment of mouse cumulus-oocyte complexes with L-carnitine during vitrification and in vitro maturation affects maturation and embryonic developmental rate after parthenogenetic activation. Anat. Histol. Embryol. 2022, 51, 44–50.
- Davoodian, N.; Kadivar, A.; Ahmadi, E.; Nazari, H.; Mehrban, H. Quercetin effect on the efficiency of ovine oocyte vitrification at GV stage. Theriogenology 2021, 174, 53–59.
- Yashiro, I.; Tagiri, M.; Ogawa, H.; Tashima, K.; Takashima, S.; Hara, H.; Hirabayashi, M.; Hochi, S. High revivability of vitrified–warmed bovine mature oocytes after recovery culture with α-tocopherol. Reproduction 2015, 149, 347–355.
- Farzollahi, M.; Tayefi-Nasrabadi, H.; Mohammadnejad, D.; Abedelahi, A. Supplementation of culture media with vitamin E improves mouse antral follicle maturation and embryo development from vitrified ovarian tissue. J. Obstet. Gynaecol. Res. 2016, 42, 526–535.
- Xiang, D.C.; Jia, B.Y.; Fu, X.W.; Guo, J.X.; Hong, Q.H.; Quan, G.B.; Wu, G.Q. Role of astaxanthin as an efficient antioxidant on the in vitro maturation and vitrification of porcine oocytes. Theriogenology 2021, 167, 13–23.
- Zhang, L.; Xue, X.; Yan, J.; Yan, L.Y.; Jin, X.H.; Zhu, X.H.; He, Z.Z.; Liu, J.; Li, R.; Qiao, J. L-proline: A highly effective cryoprotectant for mouse oocyte vitrification. Sci. Rep. 2016, 6, 26326.
- Ruiz-Conca, M.; Vendrell, M.; Sabés-Alsina, M.; Mogas, T.; Lopez-Bejar, M. Coenzyme Q(10) supplementation during in vitro maturation of bovine oocytes (Bos taurus) helps to preserve oocyte integrity after vitrification. Reprod. Domest. Anim. = Zuchthyg. 2017, 52 (Suppl. S4), 52–54.
- Shirzeyli, M.H.; Eini, F.; Shirzeyli, F.H.; Majd, S.A.; Ghahremani, M.; Joupari, M.D.; Novin, M.G. Assessment of Mitochondrial Function and Developmental Potential of Mouse Oocytes after Mitoquinone Supplementation during Vitrification. J. Am. Assoc. Lab. Anim. Sci. JAALAS 2021, 60, 388–395.
- Fang, L.; Bai, C.; Chen, Y.; Dai, J.; Xiang, Y.; Ji, X.; Huang, C.; Dong, Q. Inhibition of ROS production through mitochondria-targeted antioxidant and mitochondrial uncoupling increases post-thaw sperm viability in yellow catfish. Cryobiology 2014, 69, 386–393.
- Plotnikov, E.Y.; Zorov, D.B. Pros and Cons of Use of Mitochondria-Targeted Antioxidants. Antioxidants 2019, 8, 316.
- Cabello-Verrugio, C.; Ruiz-Ortega, M.; Mosqueira, M.; Simon, F. Oxidative Stress in Disease and Aging: Mechanisms and Therapies. Oxidative Med. Cell. Longev. 2016, 2016, 8786564.
- Meja, K.K.; Rajendrasozhan, S.; Adenuga, D.; Biswas, S.K.; Sundar, I.K.; Spooner, G.; Marwick, J.A.; Chakravarty, P.; Fletcher, D.; Whittaker, P.; et al. Curcumin restores corticosteroid function in monocytes exposed to oxidants by maintaining HDAC2. Am. J. Respir. Cell Mol. Biol. 2008, 39, 312–323.
- Priyadarsini, K.I.; Maity, D.K.; Naik, G.H.; Kumar, M.S.; Unnikrishnan, M.K.; Satav, J.G.; Mohan, H. Role of phenolic O-H and methylene hydrogen on the free radical reactions and antioxidant activity of curcumin. Free. Radic. Biol. Med. 2003, 35, 475–484.
- Chaudhuri, R.K.; Meyer, T.; Premi, S.; Brash, D. Acetyl zingerone: An efficacious multifunctional ingredient for continued protection against ongoing DNA damage in melanocytes after sun exposure ends. Int. J. Cosmet. Sci. 2020, 42, 36–45.
- Zhang, Y.; Wang, T.; Lan, M.; Zang, X.-W.; Li, Y.-L.; Cui, X.-S.; Kim, N.-H.; Sun, S.-C. Melatonin protects oocytes from MEHP exposure-induced meiosis defects in porcine†. Biol. Reprod. 2018, 98, 286–298.
- Li, W.D.; Yu, S.; Luo, S.M.; Shen, W.; Yin, S.; Sun, Q.Y. Melatonin defends mouse oocyte quality from benzoperylene-induced deterioration. J. Cell. Physiol. 2019, 234, 6220–6229.
- An, Q.; Peng, W.; Cheng, Y.; Lu, Z.; Zhou, C.; Zhang, Y.; Su, J. Melatonin supplementation during in vitro maturation of oocyte enhances subsequent development of bovine cloned embryos. J. Cell. Physiol. 2019, 234, 17370–17381.
- Khor, S.P.; Yeow, L.C.; Poobathy, R.; Zakaria, R.; Chew, B.L.; Subramaniam, S. Droplet-vitrification of Aranda Broga Blue orchid: Role of ascorbic acid on the antioxidant system and genetic fidelity assessments via RAPD and SCoT markers. Biotechnol. Rep. 2020, 26, e00448.
- Bisht, S.; Dada, R. Oxidative stress: Major executioner in disease pathology, role in sperm DNA damage and preventive strategies. Front. Biosci. Sch. Ed. 2017, 9, 420–447.
- Aghaz, F.; Vaisi-Raygani, A.; Khazaei, M.; Arkan, E. Enhanced Cryoprotective Effect of Melatonin and Resveratrol by Coencapsulation: Improved In Vitro Development of Vitrified-Warmed Mouse Germinal Vesicle Oocytes. Biopreserv. Biobank. 2021, 19, 184–193.
- Limaye, L.S.; Kale, V.P. Cryopreservation of human hematopoietic cells with membrane stabilizers and bioantioxidants as additives in the conventional freezing medium. J. Hematother. Stem Cell Res. 2001, 10, 709–718.
More
Information
Subjects:
Agriculture, Dairy & Animal Science
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
882
Revisions:
2 times
(View History)
Update Date:
09 Dec 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





