Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Oluwatoyin Oladayo Oladayo Onipe | -- | 1998 | 2022-11-28 14:35:19 | | | |
| 2 | Conner Chen | Meta information modification | 1998 | 2022-11-29 09:39:46 | | | | |
| 3 | Conner Chen | + 3 word(s) | 2001 | 2022-12-01 01:34:26 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Onipe, O.O.; Ramashia, S.E. Finger Millet Seed Coat. Encyclopedia. Available online: https://encyclopedia.pub/entry/36902 (accessed on 30 May 2026).
Onipe OO, Ramashia SE. Finger Millet Seed Coat. Encyclopedia. Available at: https://encyclopedia.pub/entry/36902. Accessed May 30, 2026.
Onipe, Oluwatoyin Oladayo, Shonisani Eugenia Ramashia. "Finger Millet Seed Coat" Encyclopedia, https://encyclopedia.pub/entry/36902 (accessed May 30, 2026).
Onipe, O.O., & Ramashia, S.E. (2022, November 28). Finger Millet Seed Coat. In Encyclopedia. https://encyclopedia.pub/entry/36902
Onipe, Oluwatoyin Oladayo and Shonisani Eugenia Ramashia. "Finger Millet Seed Coat." Encyclopedia. Web. 28 November, 2022.
Copy Citation
Finger millet (FM) is one of the little millets grown in Asia and Africa. Although still classified as an “orphan crop”, there is an increasing interest in the research of FM seed coat (FMSC), also known as bran. It houses 90% of the seed’s polyphenols and dietary fibre.
finger millet bran
polyphenols
arabinoxylan
dietary fibre
1. Finger Millet Seed Coat (Bran)
The finger millet (FM) seed coat or bran is the by-product of millet grain milling and has a healthy amount of dietary fibre, minerals, and phytochemicals [1][2]. Normally, FM is milled along with its seed coat, where most nutrients, dietary fibre, and bioactive compounds are concentrated, thus offering nutritional and health benefits [3][4]. The FM wholemeal flour has a dark hue, thereby limiting its use in some bakery products owing to the low consumer appeal and the chewy, gritty texture [5]. To obtain a more acceptable refined millet flour with more potential for food formulation, the bran or seed coat is removed, constituting waste products that end up in landfills or are used as animal feed. The FM seed coat (FMSC) can be used to make composite flour for bakery products. The seed coat contains a significant number of tannins which accounts for the astringency associated with FM-based products. FM-rich foods alleviate diabetes, obesity, and related co-morbidities. An FMSC-rich diet has been shown to reduce inflammation, improve plasma lipid profile, reduce oxidative stress, control the expression levels of numerous obesity-related genes, and increase beneficial gut bacteria such as Roseburia, Bifidobacteria and Lactobacillus spp [6][7].
2. Extraction of Finger Millet Seed Coat
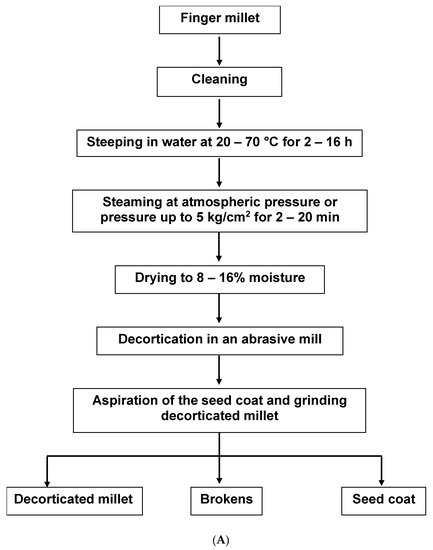
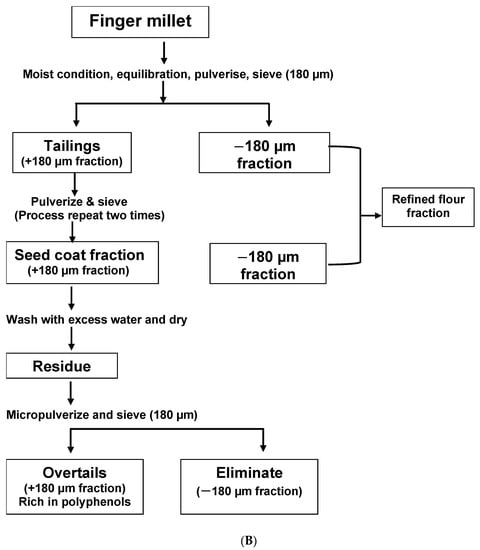
Two main methods of extraction are explained in the literature by Malleshi [8] and Shobana et al. [9]. The aim of the former was to produce decorticated FM grains (Figure 1A). The seed coat (SC) was a by-product of this process. Most cereal grains are easily de-branned, but due to the small size and tight adherence of FM bran to the endosperm, a hydrothermal pre-treatment was needed to extract the SC, leaving an intact endosperm-rich decorticated FM grain [8]. The aim of the method of Shobana et al., on the other hand, was to extract the seed coat directly [9][10]. The process of de-branning starts with tempering the grains with 5–10% w/v water for ten to thirty minutes. The grains are milled and sieved three times to obtain the tailings retained on the sieve. The tailings are washed, dried, and pulverised to obtain the bran (Figure 1B). With FM still cultivated on a minor scale in Africa, the SC is not commercially available like the bran of other cereals such as oat and wheat. An increase in the cultivation and food application of FM will potentially lead to the commercial availability of FMSC in stores.
3. Effect of Grain Pre-Treatments on Nutrient Composition of Finger Millet Seed
The purpose of pre-treatments is to weaken or degrade the cellulosic-phenolic network in plants to release and recover phenolic compounds while also disrupting the structure of the plant cell wall [11]. Bran of other cereals, including wheat [11][12], oat [13], corn [14], rice [15], foxtail millet [16], and pearl millet [17] have been pre-treated to improve their functionality and nutritional profile [18]. However, in the case of FM, the documented pre-treatment attempts have been on the grains, followed by bran extraction analysis. This is mostly due to the size of the seeds, leading to low bran yield. Hithamani and Srinivasan [19] assessed the effect of roasting, sprouting, microwave heating, open-pan boiling and pressure cooking on the phenolic compounds of FM wholemeal. They found out that roasting and sprouting increased the bioaccessibility of FM polyphenols. Krishnan et al. [5] utilised two pre-treatments: malting and hydrothermal treatments and characterised the physicochemical composition. They observed that malting increased protein, soluble and total dietary fibre and reduced the fat, carbohydrate, ash, calcium, phosphorus and phytate content. On the other hand, hydrothermal treatment reduced protein, ash and phosphorus while increasing fat, calcium, and insoluble and total dietary fibre. These nutrient variations are due to pre-treatment differences, especially the germination process in the malted FMSC, which reduced the carbohydrate content [5].
4. Nutritional Composition and Health Benefits of Finger Millet Seed Coat
The SC accounts for the amply higher nutrients in wholemeal flour. The protein of FM is said to be easily digestible, hence its common use as raw material for infant weaning foods [20]. The FMSC contains more proteins (12–13.6%) than wholemeal (8.7%) or refined flour (3.6%). This makes the SC a more nutritional food material. FM proteins are made up of albumins (8%), globulins (15%) and prolamins (35–50%) and contain the highest amount of methionine compared to other cereals [21]. With the high digestibility reported, the SC will find applications in various food products, from bakery to powdered products. The lipid content of FMSC is about 3 g/100 g. The lipids in FM are mostly triglycerides, with oleic, linoleic, and palmitic acids as the main fatty acids. FM lipids have been reported to lower the risk of duodenal ulcers [1]. There is, however, a paucity of research on the fatty acid profile of FMSC lipids. With an increase in the lipid content of hydrothermally treated FMSC, there could be a change in the fatty acid profile. This warrants further studies. FMSC contains minerals such as calcium, iron, zinc, manganese, magnesium, and phosphorus (P). FM is especially rich in calcium and phosphorus, which are more concentrated in the seed coat fraction (711–830 mg/100 g). During germination, there is increased phytase activity leading to the phytate content due to the action of endogenous phytase in the grain, as well as exogenous phytase from yeast and sourdough fermentation, which can both release phytic acid complexed minerals. It is well known that phenolic acids function as antioxidants by giving electrons or hydrogen. Additionally, the oxidation of several food constituents, particularly fatty acids and oils, is inhibited by their stable radical precursors. The FMSC is quite low in carbohydrates (<20%) and high in dietary fibre and phenolic compounds. The hypoglycaemic, nephroprotective, hypocholesterolemic, and anti-cataractogenic properties of FMSC were proved in streptozotocin-induced diabetic rats on a 20% FMSC diet [22].
4.1. Dietary Fibre
The SC of FM contains more percentage of insoluble fibre (38.4–51%) than soluble fibre (1.2–6.4%). Dietary fibre is divided into several categories, including pectin, cellulose, lignin, and arabinoxylan. So far, the latter has gained the most research attention, as seen in the subsequent section.
Arabinoxylan
Arabinoxylan (AX) is a non-starch polysaccharide made of branched heteroglycans with a side chain composed of pentose sugars, arabinose, and xylose [21]. Recently, AX has gained popularity due to its health-promoting effects in treating diabetes and colon cancer. They are effective natural immunomodulators and prebiotics and are functional food ingredients [23]. The FM-AX has been successfully extracted using alkaline solutions and water. Water-extractable xylan extracted by Prashanth & Muralikrishna [21] revealed that the xylan was heat stable (up to 200 °C) and amorphous. Given that xylose was the predominant sugar found, the potential of the water-soluble xylan from FM seed coat for producing useful ingredients such as xylitol may be further explored [21]. On the other hand, insoluble AX (primarily made up of arabinose and xylose) was extracted from FMSC using saturated barium hydroxide and 1M potassium hydroxide. The AX had average molecular weights ranging from 40 to 1028 kDa [20][24]. According to recent research, a nutraceutical made from AX extracted from finger millet may alleviate obesity from a high-fat diet [25].
4.2. Polyphenols
Polyphenols are bioactive compounds found in plants with health-promoting properties when consumed [26]. Polyphenols impart anti-ageing, antioxidant and anti-hyperglycemic effects on humans when consumed [27]. Additionally, polyphenols stop the production of advanced glycation end products and restrict glucose absorption. The total polyphenol content of native FMSC ranged from 2356–11,200 mg/100 g [9][28], while the tannins and flavonoids were 2305 and 209 mg/100 g, respectively. The polyphenol content of native FMSC was higher than the wholemeal of FM (2300 mg/100 g) [23]. Conventionally, polyphenols are extracted from FM using the heat-reflux method—this entails refluxing the food material in acidified methanol. Balasubramaniam et al. [29] compared three polyphenol extraction methods: conventional heat-reflux, ultrasonication, and enzyme treatment (xylanase) plus ultrasonication (EU). They observed that enzymatic plus ultrasonic treatment enhanced phenolic yield by 2.3 times compared to the conventional method. However, yield with ultrasonication alone was on par with the conventional method. Total flavonoids increased by 1.4 times and 1.3 times, respectively, in ultrasonicated and EU extracts. Tannin concentrations rose dramatically (1.1-fold in ultrasonicated and 1.2-fold in EU extracts). The phenolic acid composition of FMSC, as identified in high-performance liquid chromatography and direct infusion electrospray ionisation mass spectrometry, includes proto-catechuic acid, naringenin, gallic acid, p-hydroxybenzoic acid, luteolin glycoside, ferulic acid, apigenin, syringic acid, p-coumaric acid, and trans-cinnamic acid, protocatechuic acid, gentistic acid, catechin gallates, epicatechin, caffeic acid, trans-cinnamic acid, kaempferol, and phloroglucinol [9][26][30].
5. Food Applications of Finger Millet Seed Coat
5.1. Food Packaging
The environmental impact of plastic pollution is one of the problems that came with increased industrialisation. This has led to the demand for more sustainable and biodegradable packaging [31][32]. FMSC was recently used as a polysaccharide base for developing green food packaging material made from chitosan and squid gelatine [31]. The thickness of the film with 1% FMSC extracts increased by 65% compared with the film without bran extract. This increase is due to reduced space among the polymer chains in the film. Similarly, film opacity increased from 18% in the control sample to 68% in chitosan film with 1% FMSC. Thereby limiting the amount of light passing through the packaging. Meanwhile, a concomitant decrease in light transmittance (a result of increased thickness), water vapour permeability (62.5%) and absorption (due to polyphenol interaction in the film) was observed. The films with FMSC extract had higher antibacterial, antifungal and antioxidant activity than those without FMSC extract [31].
5.2. Natural Antioxidants in Oils-Based Products
There is a rising demand for natural antioxidants from edible plant sources for food preservation and shelf-life extension. High-fat food items such as mayonnaise and salad dressings can be produced with natural antioxidants that are efficient, safe, and health-promoting to satisfy the rising demand for synthetic chemical-free products. The antioxidant capacity of FMSC polyphenols against lipid oxidation in mayonnaise was compared to a synthetic antioxidant (BHT). The study demonstrated that FMSC polyphenols (1.0 mg/g1) are more efficient than BHT at preventing oxidative rancidity in a full-fat spread such as mayonnaise for seven (7) weeks at 4 °C [33]. In addition, the protein and total mineral content of the FMSC-enriched mayonnaise were markedly higher than its synthetic counterpart. Similarly, dose-dependent addition (200–1000 ppm) of FMSC polyphenols inhibited free-radical formation in peanut oil, thereby preventing oxidative rancidity and deterioration in oils during regular and accelerated storage of 7.8 weeks, respectively [29][34]. FMSC polyphenols were significantly effective in reducing primary and secondary oxidation products at 800 and 1000 ppm in oils stored for up to three months.
5.3. Biscuits
Incorporating FM by-products in biscuits may provide natural health benefits, including calcium, iron, and zinc, to consumers. Krishnan et al. [5] studied the physicochemical and functional properties of native, malted and hydrothermally treated FMSC and then substituted them in wheat flour at 10 and 20% to make composite flour for biscuits (labelled as NFMSC, MFMSC and HTFMSC). The biscuits with significant nutrient variation from the control biscuit were 10% NFMSC, 20% MFMSC and 10% HT-FMSC biscuits. The protein content of MFMSC and HTFMSC increased by 8%, and carbohydrates decreased in the three samples by 6.8, 11.3 and 7.5%, respectively. There was a 3.5-, 7.6- and a 5-fold increase in the insoluble dietary fibre of the three biscuits. Dietary fibre, calcium, iron, and zinc of the biscuits made from composite flours increased. The 20% MFMSC biscuit had the highest increase in zinc and polyphenols at 26% and 70%, respectively [5]. The sensory scores showed that native and hydrothermally processed millet, the malted millet seed coat, was the best for making biscuits at 10% and 20%, respectively [5]. The instrumental colour profile of the biscuits was measured using the CIELab colour system, while the texture was assessed by testing the biscuits’ breaking strength using the three-point break technique. The addition of FMSC darkened and hardened the biscuits. Similarly, the average colour sensory score of the biscuits was 7, indicating an above-average likeness. Due to the roughness of the integrated fibre, the score for surface features was slightly lower, at 6.1. The biscuit had a texture score of 6.2, meaning it was less crisp but still maintained an acceptable rating of 7.6 for eating quality [35].
References
- Gull, A.; Ahmad, N.G.; Prasad, K.; Kumar, P. Technological, processing and nutritional approach of finger millet (Eleusine coracana)- a mini-review. J. Food Process Technol. 2016, 7, 2.
- Martins, Z.E.; Pinho, O.; Ferreira, I.M.P.L.V.O. Food industry by-products used as functional ingredients of bakery products. Trends Food Sci. Technol. 2017, 67, 106–128.
- Chandra, D.; Chandra, S.; Sharma, A.K. Review of finger millet (Eleusine coracana (L.) Gaertn): A powerhouse of health-benefiting nutrients. Food Sci. Hum. Wellness 2016, 5, 149–155.
- Saha, S.; Gupta, A.; Singh, S.R.K.; Bharti, N.; Singh, K.P.; Mahajan, V.; Gupta, H.S. Compositional and varietal influence of finger millet flour on rheological properties of dough and quality of biscuit. LWT Food Sci. Technol. 2011, 44, 616–621.
- Krishnan, R.; Dharmaraj, U.; Manohar, R.S.; Malleshi, N.G. Quality characteristics of biscuits prepared from finger millet seed coat-based composite flour. Food Chem. 2011, 129, 499–506.
- Cao, R.; Palanisamy, S.; Ma, N.; Talapphet, N.; Zhang, J.; Wang, C.; You, S. Extraction, structural characterisation, and immunostimulatory activity of soluble non-starch polysaccharides of finger millet. Process Biochem. 2021, 111, 40–50.
- Murtaza, N.; Baboota, R.K.; Jagtap, S.; Singh, D.P.; Khare, P.; Sarma, S.M.; Kondepudi, K.K. Finger millet bran supplementation alleviates obesity-induced oxidative stress, inflammation, and gut microbial derangements in high-fat diet-fed mice. Br. J. Nutri. 2014, 112, 1447–1458.
- Malleshi, N.G. Decorticated Finger Millet (Eleusine coracana) and Process for the Preparation of Decorticated Finger Millet. U.S. Patent 7,029,720, 18 April 2006.
- Shobana, S.; Sreerama, Y.N.; Malleshi, N.G. Composition and enzyme inhibitory properties of finger millet (Eleusine coracana L.) seed coat phenolics: Mode of inhibition of α-glucosidase and pancreatic amylase. Food Chem. 2009, 115, 1268–1273.
- Palaniappan, A.; Yuvaraj, S.S.; Sonaimuthu, S.; Antony, U. Characterization of xylan from rice bran and finger millet seed coat for functional food applications. J. Cereal Sci. 2017, 75, 296–305.
- Onipe, O.O.; Ramashia, S.E.; Jideani, A.I. Wheat bran modifications for enhanced nutrition and functionality in selected food products. Molecules 2021, 26, 3918.
- Zhao, G.; Gao, Q.; Hadiatullah, H.; Zhang, J.; Zhang, A.; Yao, Y. Effect of wheat bran steam explosion pre-treatment on flavours of nonenzymatic browning products. LWT 2021, 135, 110026.
- Alrahmany, R.; Avis, T.J.; Tsopmo, A. Treatment of oat bran with carbohydrases increases soluble phenolic acid content and influences antioxidant and antimicrobial activities. Food Res. Int. 2013, 52, 568–574.
- Gong, L.; Feng, D.; Liu, J.; Yu, Y.; Wang, J. Ionic liquid depolymerize the lignocellulose for the enzymatic extraction of feruloylated oligosaccharide from corn bran. Food Chem. 2022, 15, 100381.
- Wang, S.; Li, X.; Zhu, J.; Liu, H.; Liu, T.; Yu, G.; Shao, M. Covalent interaction between high hydrostatic pressure-pretreated rice bran protein hydrolysates and ferulic acid: Focus on antioxidant activities and emulsifying properties. J. Agric. Food Chem. 2021, 69, 7777–7785.
- Wei, C.; Ge, Y.; Liu, D.; Zhao, S.; Wei, M.; Jiliu, J.; Hu, X.; Quan, Z.; Wu, Y.; Su, Y.; et al. Effects of high-temperature, high-pressure, and ultrasonic treatment on the physicochemical properties and structure of soluble dietary fibres of millet bran. Front. Nutr. 2022, 8, 820715.
- Singh, A.; Eligar, S.M. Bioactive feruloylated xylooligosaccharides derived from pearl millet (Pennisetum glaucum) bran with antiglycation and antioxidant properties. J. Food Measure. Charact. 2021, 15, 5695–5706.
- Ramirez-Gonzalez, B.C.; Castillo-Sanchez, M.I.; Macias-Garbet, R.; Bolaina-Lorenzo, E.D.; Ramos-De-La-Peña, A.M.; Rios-Licea, M.M.; Contreras-Esquivel, J.C. Non-starch polysaccharides extraction from cereal by-products applying pulsed electric fields (PEF) as pre-treatment: An overview. In Natural Food Products and Waste Recovery, 1st ed.; Carvajal-Millan, E., Yaser, A.Z., Haghi, A.K., Eds.; Apple Academic Press: New York, NY, USA, 2021; Volume 1, pp. 71–81.
- Hithamani, G.; Srinivasan, K. Effect of domestic processing on the polyphenol content and bioaccessibility in finger millet (Eleusine coracana) and pearl millet (Pennisetum glaucum). Food Chem. 2014, 164, 55–62.
- Mudau, M.; Ramashia, S.E.; Mashau, M.E.; Silungwe, H. Physicochemical characteristics of bread partially substituted with finger millet flour. Braz. J. Food Technol. 2021, 24, e2020123.
- Prashanth, M.R.S.; Muralikrishna, G. Arabinoxylan from finger millet (Eleusine coracana, v. Indaf 15) bran: Purification and characterisation. Carbohydr. Polym. 2014, 99, 800–807.
- Shobana, S.; Harsha, M.R.; Platel, K.; Srinivasan, K.; Malleshi, N.G. Amelioration of hyperglycaemia and its associated complications by finger millet (Eleusine coracana L.) seed coat matter in streptozotocin-induced diabetic rats. Br. J. Nutr. 2010, 104, 1787–1795.
- Palaniappan, A.; Balasubramaniam, V.G.; Antony, U. Prebiotic potential of xylooligosaccharides derived from finger millet seed coat. Food Biotechnol. 2017, 31, 264–280.
- Kumari, L.P.; Sumathi, S. Effect of consumption of finger millet on hyperglycemia in non-insulin-dependent diabetes mellitus (NIDDM) subjects. Plant Foods Hum. Nutr. 2002, 57, 205–213.
- Sarma, S.M.; Singh, D.P.; Singh, P.; Khare, P.; Mangal, P.; Singh, S.; Kondepudi, K.K. Finger millet arabinoxylan protects mice from high-fat diet-induced lipid derangements, inflammation, endotoxemia and gut-bacterial dysbiosis. Int. J. Biol. Macromol. 2018, 106, 994–1003.
- Viswanath, V.; Urooj, A.; Malleshi, N.G. Evaluation of antioxidant and antimicrobial properties of finger millet polyphenols (Eleusine coracana). Food Chem. 2009, 114, 340–346.
- Kumar, A.; Rani, M.; Kumari, R.; Shah, P.; Mani, S.; Sood, S. Novel prospective on suppression of ageing by the consumption of finger millet. In The Finger Millet Genome; Springer: Berlin/Heidelberg, Germany, 2022; pp. 261–276.
- Soni, B.; Sarita, S. Comparative evaluation of milling bi-products: Finger millet seed coat (FMSC), chickpea husk (CPH) and wheat bran (WB) for their Nutritional, nutraceutical potential and storage quality. Int. J. Basic Appl. Agric. Res. 2014, 12, 104–110.
- Balasubramaniam, V.G.; Ayyappan, P.; Sathvika, S.; Antony, U. Effect of enzyme pre-treatment in the ultrasound-assisted extraction of finger millet polyphenols. J. Food Sci. Technol. 2019, 56, 1583–1594.
- Banerjee, S.; Sanjay, K.R.; Chethan, S.; Malleshi, N.G. Finger millet (Eleusine coracana) polyphenols: Investigation of their antioxidant capacity and antimicrobial activity. Afr. J. Food Sci. 2012, 6, 362–374. Available online: http://ir.cftri.res.in/id/eprint/11195 (accessed on 29 September 2022).
- Xu, M.; Yu, H.; Chen, X.; Yuan, G. Physico-chemical, biological properties of chitosan/gelatin-based films with Finger Millet bran extract. J. Food Measure. Charact. 2022, 16, 2939–2947.
- Sharma, S.; Barkauskaite, S.; Jaiswal, A.K.; Jaiswal, S. Essential oils as additives in active food packaging. Food Chem. 2021, 343, 128403.
- Balasubramaniam, V.G.; Sukumar, S.; Alagarsamy, M.; Velpandi, P.S.D.; Palaniappan, A.; Roy, L.M.; Antony, U. Inhibition of lipid oxidation in mayonnaise by finger millet seed coat polyphenols. J. Am. Oil Chem. Soc. 2022, 99, 79–90.
- Balasubramaniam, V.G.; Sathvika, S.; Ayyappan, P.; Antony, U. Improved oxidative stability of peanut oil through the addition of finger millet (Eleusine coracana) seed coat polyphenols. J. Food Process Eng. 2020, 43, e13194.
- Rajasekhar, M.; Edukondalu, L.; Smith, D.D.; Veeraprasad, G. Development of value-added products from milled (decorticated) finger millet and analysis of cooking quality and sensory evaluation. J. Pharmacogn. Photochem. 2018, 7, 2573–2577.
More
Information
Subjects:
Food Science & Technology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.8K
Revisions:
3 times
(View History)
Update Date:
01 Dec 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No