Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | John Joseph | -- | 1925 | 2022-11-24 21:29:52 | | | |
| 2 | Catherine Yang | Meta information modification | 1925 | 2022-11-25 02:13:22 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Joseph, J. Trained Immunity against Emerging Respiratory Pathogens. Encyclopedia. Available online: https://encyclopedia.pub/entry/36408 (accessed on 21 May 2026).
Joseph J. Trained Immunity against Emerging Respiratory Pathogens. Encyclopedia. Available at: https://encyclopedia.pub/entry/36408. Accessed May 21, 2026.
Joseph, John. "Trained Immunity against Emerging Respiratory Pathogens" Encyclopedia, https://encyclopedia.pub/entry/36408 (accessed May 21, 2026).
Joseph, J. (2022, November 24). Trained Immunity against Emerging Respiratory Pathogens. In Encyclopedia. https://encyclopedia.pub/entry/36408
Joseph, John. "Trained Immunity against Emerging Respiratory Pathogens." Encyclopedia. Web. 24 November, 2022.
Copy Citation
Although parental vaccines offer long-term protection against homologous strains, they rely exclusively on adaptive immune memory to produce neutralizing antibodies that are ineffective against emerging viral variants. Growing evidence highlights the multifaceted functions of trained immunity to elicit a rapid and enhanced innate response against unrelated stimuli or pathogens to subsequent triggers. Training immunity is pertinent in the advent of unprecedented respiratory infections, which are usually limited to the timeline of vaccine development.
trained immunity
innate immune memory
respiratory pathogens
1. Characteristics of Adaptive and Trained Immunity
Classical vaccines mediate long-lasting adaptive immunity from antigen-specific B and T memory cells. Although vaccines are highly effective against the pathogen strain against which it was originally developed, they are considered less efficient against variants. However, certain vaccines such as Bacillus Calmette–Guérin (BCG, an attenuated bacterial vaccine derived from Mycobacterium bovis), influenza, polio, or measles-mumps-rubella (MMR) bestow heterogenous protection by the activation of trained innate immunity [1][2][3][4]. The non-specific recognition of pathogens occurs through the diverse pathogen recognition receptors (PPRs) [5]. The trained immune response is manifested by metabolic reprogramming and epigenetic modifications happening at the transcriptional level compared to the gene recombination and clonal expansion, a hallmark feature of adaptive immunity [6][7]. The onset of innate immune memory upon a secondary challenge (exposure to pathogens or antigens) is faster and typically takes minutes to hours, resulting in elevated non-specific innate response mediated by the secretion of higher levels of proinflammatory cytokines, reactive oxygen species (ROS), and phagocytosis [8].
Chromatin is a repeated unit of nucleosome that comprises 147 bp of DNA wrapped around in octameric histone proteins with a pair of H3 and H4 homodimers and a set of H2A/H2B heterodimers [9]. Methylation and acetylation occur at the N-terminal histone tails induced by enzymes, such as histone acetyltransferase (HAT), histone deacetylase (HDAC), lysine methyltransferase (KMT), and lysine demethylase (KDM). Gene expression is regulated by the extent of histone modification facilitating the accessibility of transcriptional machinery to DNA. A marked transcriptional change occurs with H3 histone as exemplified by HAT-mediated acetylation at lysine 9 (H3K9ac) and lysine 27 (H3K27ac), which are associated with elevated inflammation in innate immune cells [10][11]. On the contrary, HADC enzymes facilitate the removal of an acetyl group from the histone tail. A balance in HAT/HDAC is an essential factor determining the gene expression [12]. Similarly, KMT and KDM enzymes are highly specific to amino acids in the H3 histones. Alterations in the location of a lysine residue in histones determine the functional outcome. A repressed gene is correlated with the methylation of lysine9 (H3K9me) and lysine27(H3K27me) at H3 histone. In striking contrast, a permissive epigenetic modification is characterized by the methylation of lysine4 (H3K4me) and lysine36 (H3K36me) [13]. Moreover, transcriptional activity depends on the degree of methylation, which can be mono, di, or trimethylation on H3 histone.
In addition, the primary challenge of immunostimulants after recognizing through PRRs prompts a cascade of intracellular metabolic pathways in immune cells, such as the tricarboxylic acid (TCA) cycle, glycolysis, and fatty acid metabolism. Metabolites involved in these processes, such as acetyl coenzyme A (acetyl-CoA) and fumarate, may also facilitate epigenetic reprogramming of the cells through the activation or inhibition of enzymes [6]. These reprogramming of the molecular events occur at long-lived hematopoietic stem cells (HSC) in the bone marrow (centrally trained immunity) and short-lived circulating immune cells, including monocytes, macrophages, natural killer (NK) cells, etc. (Figure 1). It is comprehended that HSC-derived monocytes differentiate into macrophages with augmented effector functions in peripheral regions. Similarly, NK cells with innate immune memory provide enhanced antipathogenic response by faster degranulation and cytokine release than NKs in the normal state. Respiratory epithelial stem cells also featured characteristic innate immune memory by altering chromatin accessibility upon exposure to the stimulus [14].
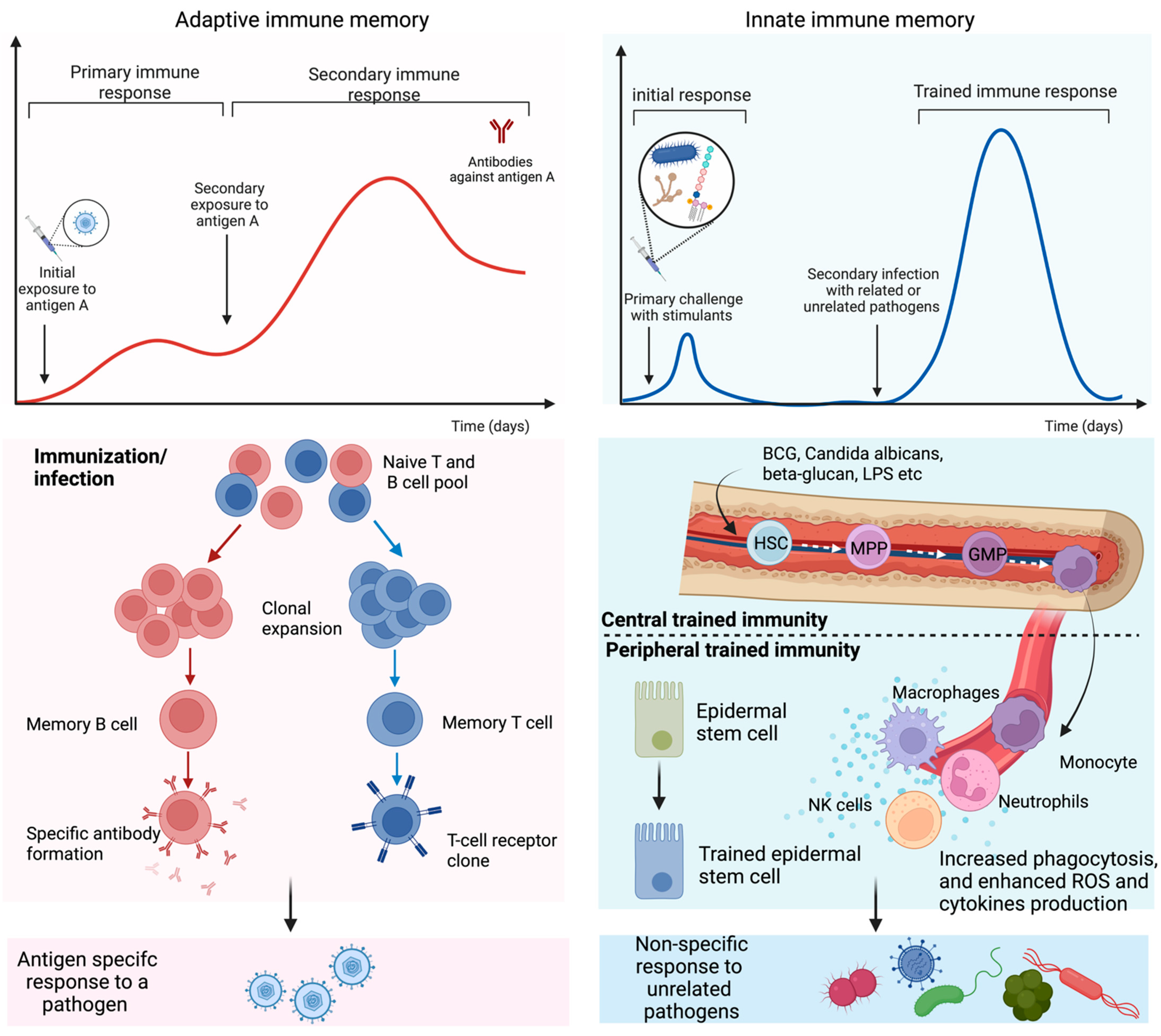
Figure 1. Adaptive and trained immune response. Exposure to antigens or pathogens may trigger innate and adaptive immune responses. In adaptive immunity, an antigen-specific response is attributed to antibody production and long-lived B and T lymphocytes by gene recombination and clonal expansion. Trained immunity is governed by a rapid and heightened innate immune response to certain vaccines and antigens that reprogram the hematopoietic stem cells (HSC) residing in the bone marrow or circulating innate immune cells. HSC undergoes differentiation to bone marrow precursors, multipotent progenitor (MPP), and granulocyte-macrophage progenitor (GMP) and consequently enters peripheral organs as circulating monocytes and tissue macrophages. Epithelial stem cells have also been identified as part of the peripheral trained immunity along with dendritic, NK, and endothelial cells.
Several studies reported the reprogramming of myeloid cells following exposure to microbes such as fungi and viruses [15][16][17]. Growing evidence of literature indicates the immune memory of dendritic cells (DCs) upon exposure to the fungus, Cryptococcus neoformans in the murine model [18][19][20]. On a subsequent challenge with the same pathogen, epigenetic modifications were observed to trigger the robust production of interferon γ (IFNγ) and Th1 cytokines. This phenomenon was impeded upon treating the animals with histone methyltransferase inhibitors. In addition, herpes infection enhanced the protection against Yersinia pestis via IFNγ and activation of circulating macrophages [21][22]. Another study revealed the characteristic memory similar to adaptive immunity in NK cells when mice were exposed to mouse or human cytomegalovirus [23][24][25].
Similarly, vaccines and vaccine adjuvants have been associated with triggering innate immune memory against early pathogen invasion. For instance, exposure of β-glucan, a polysaccharide of the fungal cell wall, or C. albicans to macrophages or monocytes elicited a profound response in subsequent challenges to related or PAMPs or microorganisms, including parasites and viruses [26]. Key inducers, including IL-1β and GM-CSF (granulocyte-macrophage colony-stimulating factor), were necessary for β-glucan-mediated trained immunity [27][28]. Intriguingly, Toll-like receptors or NOD-like receptors agonists were effective against Toxoplasma and E. coli, respectively [29][30][31]. Likewise, compelling evidence of BCG vaccines unveiled a profound effect on preventing infection without involving adaptive immune cells [3][32]. The nonspecific innate memory induced by BCG was mediated by type II interferon. BCG also induced heterogeneous protection resulting in low parasitemia and viremia in the clinical trial with malarial infection and yellow fever, respectively [33][34]. Most importantly, prophylaxis was not exclusively against tuberculosis (TB) but against several respiratory infections, as detailed in the upcoming section.
Contrary to trained immunity, ‘tolerance’ is characterized by a change in the programming of the immune cells, when subjected to persistent exposure to stimulants, leading to a slower response and gene transcription [27]. This change in the adaptive programming state of the cells is decisive on factors such as the duration (long or short) of stimulation and the dose (low or high) of stimulants [27].
2. Cross-Protection of Trained Immunity against Respiratory Infections
The recent decades witnessed the increased incidence of epidemics and pandemics caused by respiratory tract infections imposing a significant socio-economic burden [35][36]. A growing body of evidence suggests the therapeutic efficacy of trained immunity to confer prophylaxis against respiratory infections caused by bacteria and viruses (Table 1). Harnessing trained immunity could be leveraged as an effective prophylactic tool to bolster the innate immune system to defend against pathogens.
The emergent variants of the influenza virus led to pandemics and seasonal epidemics worldwide, affecting millions of lives. In a randomized pilot study, the administration of BCG vaccines before intramuscular influenza vaccination in healthy volunteers augmented the production of antibody response against the H1N1pdm09 influenza virus [37]. The hemagglutination-inhibiting antibody titer was remarkably higher in the BCG vaccinated cohort than in the placebo group. In another study, intranasal delivery of BCG in murine models enhanced efferocytosis by alveolar phagocytes and maintained lung homeostasis against a lethal mouse-adapted influenza virus A/Puerto Rico/8/34 (PR8) (H1N1) strain [38]. Subcutaneous immunization of BCG failed to confer protection from PR8 infections, and 100% of the animals succumbed after 15 days post-infection. This study connotes the relevance of the administration route of BCG to function as an inducer of trained innate immunity. Besides BCG, other inducers also presented evidence to ponder the potential of trained immunity against the influenza virus. Extensive clinical trials on BCG-induced trained immunity for suppressing disease severity and pathogenesis of SARS-CoV-2 will proffer convincing evidence to bridge the gap for early prophylaxis until an antigen-specific vaccine becomes available. The non-targeted protection against acute lung infections caused by respiratory syncytial viruses in newborns further complements the ubiquitous induction of trained immunity by BCG vaccines [39].
Intranasal exposure of lipopolysaccharides (LPS) exhibits a noticeable immunogenic activity upon challenge with S. pneumoniae and rewires the metabolism of alveolar macrophages to abrogate the infection [40]. Other adjuvants such as zymosan also orchestrate trained immunity to confer broad-spectrum protection against peritonitis and pneumonia caused by a wide array of pathogens such as S. aureus, E. coli, and Pseudomonas aeruginosa [30]. Together, these research efforts underline the potency of trained immunity inducers to leverage non-specific innate responses against heterologous respiratory pathogens. However, preclinical and clinical studies need to address the longevity of the protection, frequency of administration, and ubiquitous use for all populations, including immunosenescent, immunocompromised, and infants.
Table 1. Cross-reactivity of trained immunity induced by various stimulants against respiratory pathogens.
| Stimulation | Model | Cell Type | Cross Protection | Remarks |
|---|---|---|---|---|
| BCG (i.n) |
Mouse | Monocytes | Influenza A | Intranasal administration of BCG resulted in 100% survival against PR-8 [38] |
| BCG | Ex vivo- Human neutrophils |
Neutrophils | NA | Increased IL-1β, IL-8, ROS production, Phagocytosis [41] |
| BCG (i.m) |
Human and Ex vivo |
NA | Influenza A | BCG modulate innate immune response. Influenza vaccines augment acquired immunity at higher levels [37] |
| BCG (i.d) |
Humans (neonates) | NA | NA | Reduced infectious disease related mortality by 43% [42] |
| BCG | Humans (neonates) | NA | RSV | Reduced risk of acute lower respiratory tract infection [39] |
| BCG (i.d) |
Humans (elderly) |
NA | NA | Reduce disease severity by attenuating matrix metalloproteinases (MMPs), and pro-inflammatory cytokines [43] |
| MV130 (i.n) |
Mouse | NA | SARS-CoV-2 | Improved the survival and elevated B and T lymphocyte response [44] |
| MV130 | Ex vivo and mouse | Dendritic cells | NA | Increased the level of TNF-α, IL-6, IL-1β, and IL-23 [45] |
| MV130 | Ex vivo and mouse | Monocytes | Influenza A | Improved survival [46] |
| β-glucan | Mouse | Monocytes | C. albicans | Enhanced TNF-α and IL-6 production. Improved survival against C. albicans [47] |
| LPS | Ex vivo and human | Monocytes | Influenza A | Enhanced production of TNF-α, IFN-β, and IFN-γ [40] |
i.n—intranasal, i.m—intramuscular, i.d—intradermal.
3. Potential of Trained Immunity to Fight Emerging Pathogens
The recent epidemics and pandemics associated with emerging respiratory viruses have exposed vulnerabilities in tedious, traditional vaccine development and the suboptimal efficacy of vaccines against heterogeneous strains, owing to antigen-specific IgG responses. Although nasal vaccines show elevated levels of mucosal protection compared to systemic vaccines, regulatory hurdles and a lack of appropriate mucosal adjuvants have decelerated their development [48][49]. Despite years of research, FluMist® (Astra Zeneca, Cambridge, UK) remains the only FDA-approved nasal vaccine, which unfortunately showed inconsistent efficacy in heterogeneous patient populations [50]. The entry highlights the pivotal role of inducers of trained immunity, especially BCG, in preventing pathogen invasion, decreased viral load, disease severity, and mortality in preclinical and multicentric clinical trials. Immunodeficiency, genetic predisposition, comorbidities, age, etc. are all attributes that weaken the innate antiviral response and increase exposure to respiratory infection [51]. Moreover, recent studies spotlight the protective role of BCG’s trained immunity in reducing the severity of SARS-CoV-2 infection in the elderly [43][52]. BCG-induced trained immunity also synergistically bolstered cytokine induction of influenza vaccines, and subsequent protection, against respiratory viruses [37]. In these contexts, I envision that trained immunity may represent a complementary prophylactic tool to evoke a timely and robust innate immune response before the availability of antigen-specific vaccines. Thus, inducers of trained immunity may provide a “head start” to fight novel pathogens that attenuate transmission, progression, or severity of infection. For instance, a plethora of evidence on the protective role of BCG resulted in at least 25 clinical trials in the early stage of the COVID-19 pandemic [8].
References
- Chumakov, K.; Avidan, M.S.; Benn, C.S.; Bertozzi, S.M.; Blatt, L.; Chang, A.Y.; Jamison, D.T.; Khader, S.A.; Kottilil, S.; Netea, M.G.; et al. Old Vaccines for New Infections: Exploiting Innate Immunity to Control COVID-19 and Prevent Future Pandemics. Proc. Natl. Acad. Sci. USA 2021, 118, e2101718118.
- Covián, C.; Ríos, M.; Berríos-Rojas, R.V.; Bueno, S.M.; Kalergis, A.M. Induction of Trained Immunity by Recombinant Vaccines. Front. Immunol. 2020, 11, 611946.
- O’Neill, L.A.J.; Netea, M.G. BCG-Induced Trained Immunity: Can It Offer Protection against COVID-19? Nat. Rev. Immunol. 2020, 20, 335–337.
- Debisarun, P.A.; Gössling, K.L.; Bulut, O.; Kilic, G.; Zoodsma, M.; Liu, Z.; Oldenburg, M.; Rüchel, N.; Zhang, B.; Xu, C.-J.; et al. Induction of Trained Immunity by Influenza Vaccination–Impact on COVID-19. PLoS Pathog. 2021, 17, e1009928.
- Medzhitov, R.; Janeway, C.J. Innate Immune Recognition: Mechanisms and Pathways. Immunol. Rev. 2000, 173, 89–97.
- Netea, M.G.; Domínguez-Andrés, J.; Barreiro, L.B.; Chavakis, T.; Divangahi, M.; Fuchs, E.; Joosten, L.A.B.; van der Meer, J.W.M.; Mhlanga, M.M.; Mulder, W.J.M.; et al. Defining Trained Immunity and Its Role in Health and Disease. Nat. Rev. Immunol. 2020, 20, 375–388.
- Kleinnijenhuis, J.; Quintin, J.; Preijers, F.; Joosten, L.A.B.; Ifrim, D.C.; Saeed, S.; Jacobs, C.; van Loenhout, J.; de Jong, D.; Stunnenberg, H.G.; et al. Bacille Calmette-Guerin Induces NOD2-Dependent Nonspecific Protection from Reinfection via Epigenetic Reprogramming of Monocytes. Proc. Natl. Acad. Sci. USA 2012, 109, 17537–17542.
- Kaur, G.; Singh, S.; Nanda, S.; Zafar, M.A.; Malik, J.A.; Arshi, M.U.; Lamba, T.; Agrewala, J.N. Fiction and Facts about BCG Imparting Trained Immunity against COVID-19. Vaccines 2022, 10, 1006.
- Keating, S.T.; El-Osta, A. Epigenetics and Metabolism. Circ. Res. 2015, 116, 715–736.
- Josling, G.A.; Selvarajah, S.A.; Petter, M.; Duffy, M.F. The Role of Bromodomain Proteins in Regulating Gene Expression. Genes 2012, 3, 320–343.
- Eberharter, A.; Becker, P.B. Histone Acetylation: A Switch between Repressive and Permissive Chromatin. Second in Review Series on Chromatin Dynamics. EMBO Rep. 2002, 3, 224–229.
- Hull, E.E.; Montgomery, M.R.; Leyva, K.J. HDAC Inhibitors as Epigenetic Regulators of the Immune System: Impacts on Cancer Therapy and Inflammatory Diseases. Biomed Res. Int. 2016, 2016, 8797206.
- Hyun, K.; Jeon, J.; Park, K.; Kim, J. Writing, Erasing and Reading Histone Lysine Methylations. Exp. Mol. Med. 2017, 49, e324.
- Bigot, J.; Guillot, L.; Guitard, J.; Ruffin, M.; Corvol, H.; Chignard, M.; Hennequin, C.; Balloy, V. Respiratory Epithelial Cells Can Remember Infection: A Proof-of-Concept Study. J. Infect. Dis. 2020, 221, 1000–1005.
- Stienstra, R.; Netea-Maier, R.T.; Riksen, N.P.; Joosten, L.A.B.; Netea, M.G. Specific and Complex Reprogramming of Cellular Metabolism in Myeloid Cells during Innate Immune Responses. Cell Metab. 2017, 26, 142–156.
- Kopf, M.; Nielsen, P.J. Training Myeloid Precursors with Fungi, Bacteria and Chips. Nat. Immunol. 2018, 19, 320–322.
- Foster, S.L.; Hargreaves, D.C.; Medzhitov, R. Gene-Specific Control of Inflammation by TLR-Induced Chromatin Modifications. Nature 2007, 447, 972–978.
- Hole, C.R.; Wager, C.M.L.; Castro-Lopez, N.; Campuzano, A.; Cai, H.; Wozniak, K.L.; Wang, Y.; Wormley, F.L. Induction of Memory-like Dendritic Cell Responses in Vivo. Nat. Commun. 2019, 10, 2955.
- Eastman, A.J.; Xu, J.; Bermik, J.; Potchen, N.; den Dekker, A.; Neal, L.M.; Zhao, G.; Malachowski, A.; Schaller, M.; Kunkel, S.; et al. Epigenetic Stabilization of DC and DC Precursor Classical Activation by TNFα Contributes to Protective T Cell Polarization. Sci. Adv. 2022, 5, eaaw9051.
- Fu, M.S.; Drummond, R.A. The Diverse Roles of Monocytes in Cryptococcosis. J. Fungi 2020, 6, 111.
- Barton, E.S.; White, D.W.; Cathelyn, J.S.; Brett-McClellan, K.A.; Engle, M.; Diamond, M.S.; Miller, V.L.; Virgin, H.W. Herpesvirus Latency Confers Symbiotic Protection from Bacterial Infection. Nature 2007, 447, 326–329.
- Yager, E.J.; Szaba, F.M.; Kummer, L.W.; Lanzer, K.G.; Burkum, C.E.; Smiley, S.T.; Blackman, M.A. Gamma-Herpesvirus-Induced Protection against Bacterial Infection Is Transient. Viral Immunol. 2009, 22, 67–72.
- Grassmann, S.; Pachmayr, L.O.; Leube, J.; Mihatsch, L.; Andrae, I.; Flommersfeld, S.; Oduro, J.; Cicin-Sain, L.; Schiemann, M.; Flossdorf, M.; et al. Distinct Surface Expression of Activating Receptor Ly49H Drives Differential Expansion of NK Cell Clones upon Murine Cytomegalovirus Infection. Immunity 2019, 50, 1391–1400.e4.
- Björkström, N.K.; Lindgren, T.; Stoltz, M.; Fauriat, C.; Braun, M.; Evander, M.; Michaëlsson, J.; Malmberg, K.-J.; Klingström, J.; Ahlm, C.; et al. Rapid Expansion and Long-Term Persistence of Elevated NK Cell Numbers in Humans Infected with Hantavirus. J. Exp. Med. 2011, 208, 13–21.
- Foley, B.; Cooley, S.; Verneris, M.R.; Curtsinger, J.; Luo, X.; Waller, E.K.; Anasetti, C.; Weisdorf, D.; Miller, J.S. Human Cytomegalovirus (CMV)-Induced Memory-like NKG2C+NK Cells Are Transplantable and Expand In Vivo in Response to Recipient CMV Antigen. J. Immunol. 2012, 189, 5082–5088.
- Quintin, J.; Saeed, S.; Martens, J.H.A.; Giamarellos-Bourboulis, E.J.; Ifrim, D.C.; Logie, C.; Jacobs, L.; Jansen, T.; Kullberg, B.-J.; Wijmenga, C.; et al. Candida Albicans Infection Affords Protection against Reinfection via Functional Reprogramming of Monocytes. Cell Host Microbe 2012, 12, 223–232.
- Chavakis, T.; Mitroulis, I.; Hajishengallis, G. Hematopoietic Progenitor Cells as Integrative Hubs for Adaptation to and Fine-Tuning of Inflammation. Nat. Immunol. 2019, 20, 802–811.
- Walachowski, S.; Tabouret, G.; Fabre, M.; Foucras, G. Molecular Analysis of a Short-Term Model of β-Glucans-Trained Immunity Highlights the Accessory Contribution of GM-CSF in Priming Mouse Macrophages Response. Front. Immunol. 2017, 8, 1089.
- Sánchez-Ramón, S.; Conejero, L.; Netea, M.G.; Sancho, D.; Palomares, Ó.; Subiza, J.L. Trained Immunity-Based Vaccines: A New Paradigm for the Development of Broad-Spectrum Anti-Infectious Formulations. Front. Immunol. 2018, 9, 2936.
- Ciarlo, E.; Heinonen, T.; Théroude, C.; Asgari, F.; Le Roy, D.; Netea, M.G.; Roger, T. Trained Immunity Confers Broad-Spectrum Protection Against Bacterial Infections. J. Infect. Dis. 2020, 222, 1869–1881.
- Ifrim, D.C.; Quintin, J.; Joosten, L.A.B.; Jacobs, C.; Jansen, T.; Jacobs, L.; Gow, N.A.R.; Williams, D.L.; van der Meer, J.W.M.; Netea, M.G. Trained Immunity or Tolerance: Opposing Functional Programs Induced in Human Monocytes after Engagement of Various Pattern Recognition Receptors. Clin. Vaccine Immunol. 2014, 21, 534–545.
- Kanno, A.I.; Boraschi, D.; Leite, L.C.C.; Rodriguez, D. Recombinant BCG Expressing the Subunit 1 of Pertussis Toxin Induces Innate Immune Memory and Confers Protection against Non-Related Pathogens. Vaccines 2022, 10, 234.
- Walk, J.; de Bree, L.C.J.; Graumans, W.; Stoter, R.; van Gemert, G.-J.; van de Vegte-Bolmer, M.; Teelen, K.; Hermsen, C.C.; Arts, R.J.W.; Behet, M.C.; et al. Outcomes of Controlled Human Malaria Infection after BCG Vaccination. Nat. Commun. 2019, 10, 874.
- Arts, R.J.W.; Moorlag, S.J.C.F.M.; Novakovic, B.; Li, Y.; Wang, S.-Y.; Oosting, M.; Kumar, V.; Xavier, R.J.; Wijmenga, C.; Joosten, L.A.B.; et al. BCG Vaccination Protects against Experimental Viral Infection in Humans through the Induction of Cytokines Associated with Trained Immunity. Cell Host Microbe 2018, 23, 89–100.e5.
- Nicola, M.; Alsafi, Z.; Sohrabi, C.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, M.; Agha, R. The Socio-Economic Implications of the Coronavirus Pandemic (COVID-19): A Review. Int. J. Surg. 2020, 78, 185–193.
- Mamelund, S.-E.; Shelley-Egan, C.; Rogeberg, O. The Association between Socioeconomic Status and Pandemic Influenza: Systematic Review and Meta-Analysis. PLoS ONE 2021, 16, e0244346.
- Leentjens, J.; Kox, M.; Stokman, R.; Gerretsen, J.; Diavatopoulos, D.A.; van Crevel, R.; Rimmelzwaan, G.F.; Pickkers, P.; Netea, M.G. BCG Vaccination Enhances the Immunogenicity of Subsequent Influenza Vaccination in Healthy Volunteers: A Randomized, Placebo-Controlled Pilot Study. J. Infect. Dis. 2015, 212, 1930–1938.
- Mukherjee, S.; Subramaniam, R.; Chen, H.; Smith, A.; Keshava, S.; Shams, H. Boosting Efferocytosis in Alveolar Space Using BCG Vaccine to Protect Host against Influenza Pneumonia. PLoS ONE 2017, 12, e0180143.
- Stensballe, L.G.; Nante, E.; Jensen, I.P.; Kofoed, P.-E.; Poulsen, A.; Jensen, H.; Newport, M.; Marchant, A.; Aaby, P. Acute Lower Respiratory Tract Infections and Respiratory Syncytial Virus in Infants in Guinea-Bissau: A Beneficial Effect of BCG Vaccination for Girls Community Based Case-Control Study. Vaccine 2005, 23, 1251–1257.
- Zahalka, S.; Starkl, P.; Watzenboeck, M.L.; Farhat, A.; Radhouani, M.; Deckert, F.; Hladik, A.; Lakovits, K.; Oberndorfer, F.; Lassnig, C.; et al. Trained Immunity of Alveolar Macrophages Requires Metabolic Rewiring and Type 1 Interferon Signaling. Mucosal Immunol. 2022, 15, 896–907.
- Moorlag, S.J.C.F.M.; Rodriguez-Rosales, Y.A.; Gillard, J.; Fanucchi, S.; Theunissen, K.; Novakovic, B.; de Bont, C.M.; Negishi, Y.; Fok, E.T.; Kalafati, L.; et al. BCG Vaccination Induces Long-Term Functional Reprogramming of Human Neutrophils. Cell Rep. 2020, 33, 108387.
- de Castro, M.J.; Pardo-Seco, J.; Martinón-Torres, F. Nonspecific (Heterologous) Protection of Neonatal BCG Vaccination Against Hospitalization Due to Respiratory Infection and Sepsis. Clin. Infect. Dis. 2015, 60, 1611–1619.
- Pavan Kumar, N.; Padmapriyadarsini, C.; Rajamanickam, A.; Marinaik, S.B.; Nancy, A.; Padmanaban, S.; Akbar, N.; Murhekar, M.; Babu, S. Effect of BCG Vaccination on Proinflammatory Responses in Elderly Individuals. Sci. Adv. 2022, 7, eabg7181.
- del Fresno, C.; García-Arriaza, J.; Martínez-Cano, S.; Heras-Murillo, I.; Jarit-Cabanillas, A.; Amores-Iniesta, J.; Brandi, P.; Dunphy, G.; Suay-Corredera, C.; Pricolo, M.R.; et al. The Bacterial Mucosal Immunotherapy MV130 Protects Against SARS-CoV-2 Infection and Improves COVID-19 Vaccines Immunogenicity. Front. Immunol. 2021, 12, 748103.
- Cirauqui, C.; Benito-Villalvilla, C.; Sánchez-Ramón, S.; Sirvent, S.; Diez-Rivero, C.M.; Conejero, L.; Brandi, P.; Hernández-Cillero, L.; Ochoa, J.L.; Pérez-Villamil, B.; et al. Human Dendritic Cells Activated with MV130 Induce Th1, Th17 and IL-10 Responses via RIPK2 and MyD88 Signalling Pathways. Eur. J. Immunol. 2018, 48, 180–193.
- Brandi, P.; Conejero, L.; Cueto, F.J.; Martínez-Cano, S.; Dunphy, G.; Gómez, M.J.; Relaño, C.; Saz-Leal, P.; Enamorado, M.; Quintas, A.; et al. Trained Immunity Induction by the Inactivated Mucosal Vaccine MV130 Protects against Experimental Viral Respiratory Infections. Cell Rep. 2022, 38, 110184.
- Saeed, S.; Quintin, J.; Kerstens, H.H.D.; Rao, N.A.; Aghajanirefah, A.; Matarese, F.; Cheng, S.-C.; Ratter, J.; Berentsen, K.; van der Ent, M.A.; et al. Epigenetic Programming of Monocyte-to-Macrophage Differentiation and Trained Innate Immunity. Science 2014, 345, 1251086.
- Lavelle, E.C.; Ward, R.W. Mucosal Vaccines–Fortifying the Frontiers. Nat. Rev. Immunol. 2022, 22, 236–250.
- Yusuf, H.; Kett, V. Current Prospects and Future Challenges for Nasal Vaccine Delivery. Hum. Vaccines Immunother. 2017, 13, 34–45.
- Dibben, O.; Crowe, J.; Cooper, S.; Hill, L.; Schewe, K.E.; Bright, H. Defining the Root Cause of Reduced H1N1 Live Attenuated Influenza Vaccine Effectiveness: Low Viral Fitness Leads to Inter-Strain Competition. Vaccines 2021, 6, 35.
- Jung, S.; Gies, V.; Korganow, A.-S.; Guffroy, A. Primary Immunodeficiencies With Defects in Innate Immunity: Focus on Orofacial Manifestations. Front. Immunol. 2020, 11, 1065.
- Giamarellos-Bourboulis, E.J.; Tsilika, M.; Moorlag, S.; Antonakos, N.; Kotsaki, A.; Domínguez-Andrés, J.; Kyriazopoulou, E.; Gkavogianni, T.; Adami, M.-E.; Damoraki, G.; et al. Activate: Randomized Clinical Trial of BCG Vaccination against Infection in the Elderly. Cell 2020, 183, 315–323.e9.
More
Information
Subjects:
Virology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.6K
Entry Collection:
COVID-19
Revisions:
2 times
(View History)
Update Date:
25 Nov 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





