
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Dean Liu | -- | 3204 | 2022-11-22 01:35:52 |
Video Upload Options
A spontaneous cerebrospinal fluid leak (sCSF leak) is a cerebrospinal fluid leak – a leak of cerebrospinal fluid that surrounds the brain and spinal cord from the protective dural sac for no apparent reason. The dura mater is the tough, outermost of layer of the meninges, the membranes surrounding the brain and spinal cord. A spontaneous CSF leak, arises idiopathically, and is opposed to traumatically caused CSF leaks. It is one of several types of cerebrospinal fluid leaks caused by one or more holes in the dura. A loss of CSF greater than its rate of production leads to a decreased volume inside the skull known as intracranial hypotension. Any CSF leak is most often characterized by orthostatic headaches, which worsen when standing, and improve when lying down. Other symptoms can include neck pain or stiffness, nausea, vomiting, dizziness, fatigue, and a metallic taste in the mouth. A CT scan can identify the site of a cerebrospinal fluid leakage. Once identified, the leak can often be repaired by an epidural blood patch, an injection of the patient's own blood at the site of the leak, a fibrin glue injection, or surgery. The set of symptoms associated with a sCSF leak is referred to as a spontaneous cerebrospinal fluid leak syndrome (SCSFLS). A sCSF leak is rare, affecting five out of every 100,000 people. On average, the condition develops at age 42, and women are twice as likely to be affected. Some people with a sCSF leak have a chronic leak of cerebrospinal fluid despite repeated patching attempts, leading to long-term disability due to pain. SCSFLS was first described by German neurologist Georg Schaltenbrand in 1938 and by American neurologist Henry Woltman of the Mayo Clinic in the 1950s.
1. Classification
Spontaneous cerebrospinal fluid leaks are classified into two main types: cranial leaks,[1] and spinal leaks.[2] The vast majority of leaks are spinal.[3] Cranial leaks occur in the head, and in some of these cases, CSF can leak from the nose, or from the ear.[4] Spinal leaks occur when one or more holes form in the dura along the spinal cord.[2] Both cranial and spinal spontaneous CSF leaks cause neurological symptoms as well as spontaneous intracranial hypotension (SIH), diminished volume, and pressure of the cranium.[5] While this symptom can be referred to as intracranial hypotension, the intracranial pressure may be normal, with the underlying issue instead being low CSF volume. For this reason, a SCSFL is referred to as CSF hypovolemia as opposed to CSF hypotension.[6][7][8][9]
2. Signs and Symptoms
| Nerve | Function | Symptoms |
|---|---|---|
| optic (2) |
optic nerve crossing |
blurred and or double vision |
| chorda tympani (Branch of 7) |
taste | taste distortion |
| facial (7) |
facial nerve | facial weakness and numbness |
| vestibulocochlear (8) |
hearing, balance |
hearing and balance problems |
| glossopharyngeal (9) |
taste | taste distortion |
Most people who develop a sCSF leak feel the sudden onset of a severe and acute headache.[9][11] This headache is usually made worse by standing and typically becomes prominent throughout the day, with the pain becoming less severe when lying down.[12] Orthostatic headaches can become chronic and disabling to the point of incapacitation.[9][13][14][15] Some patients with a SCSFL will develop headaches that begin in the afternoon. This is known as second-half-of-the-day headache. This may be an initial presentation of a spontaneous CSF leak or appear after treatment such as an epidural patch, and likely indicates a slow CSF leak.[16]
Apart from headache, about 50% of people with a CSFL experience neck pain or stiffness, nausea, and vomiting.[17] Other symptoms include dizziness and vertigo, facial numbness or weakness, unusually blurry or double vision, neuralgia, fatigue, or a metallic taste in the mouth.[9] Leaking CSF can sometimes be felt or observed as a discharge from the nose or ear.[18]
Lack of CSF pressure and volume can allow the brain to sag and descend through the foramen magnum (large opening) of the occipital bone, at the base of the skull. The lower portion of the brain is believed to stretch or impact one or more cranial nerve complexes, thereby causing a variety of sensory symptoms. Nerves that can be affected and their related symptoms are detailed in the table at right.[9][10][15]
2.1. Complications
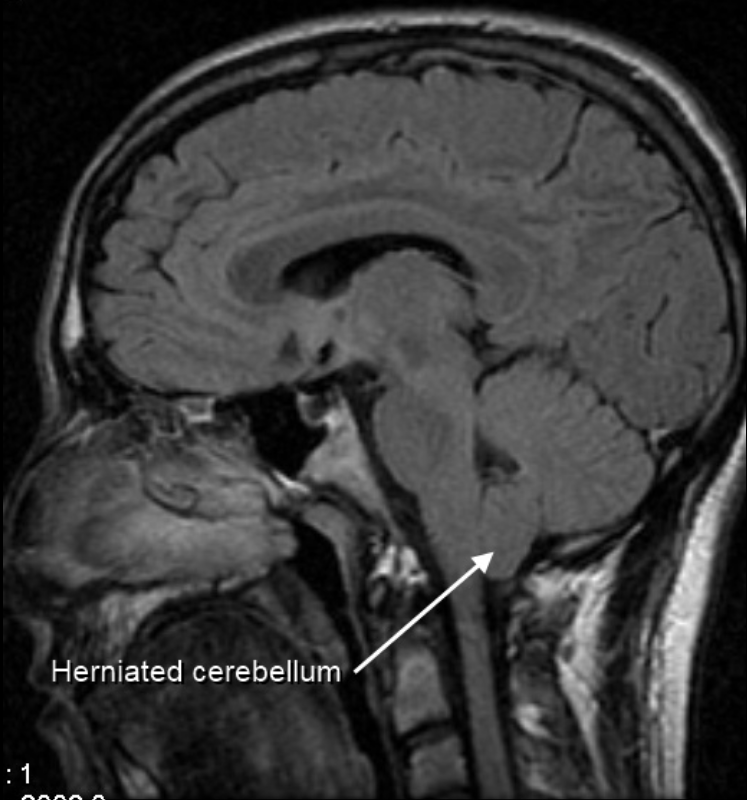
Several complications can occur as a result of SCSFLS including decreased cranial pressure, brain herniation, infection, blood pressure problems, transient paralysis, and coma. The primary and most serious complication of SCSFLS is spontaneous intracranial hypotension, where pressure in the brain is severely decreased.[9][19][20] This complication leads to the hallmark symptom of severe orthostatic headaches.[10][20]
People with cranial CSF leaks, the rarer form, have a 10% risk of developing meningitis per year.[21] If cranial leaks last more than seven days, the chances of developing meningitis are significantly higher.[21] Spinal CSF leaks cannot result in meningitis due to the sterile conditions of the leak site.[10] When a CSF leak occurs at the temporal bone, surgery becomes necessary in order to prevent infection and repair the leak.[22] Orthostatic hypotension is another complication that occurs due to autonomic dysfunction when blood pressure drops significantly.[23] The autonomic dysfunction is caused by compression of the brainstem, which controls breathing and circulation.[23]
Low CSF volume can cause the cerebellar tonsil position to descend, which can be mistaken for Chiari malformation; however when the CSF leak is repaired the tonsil position often returns to normal (as seen in upright MRI) in this "pseudo-Chiari" condition.[24] A further, albeit rare, complication of CSF leak is transient quadriplegia due to a sudden and significant loss of CSF. This loss results in hindbrain herniation and causes major compression of the upper cervical spinal cord. The quadriplegia dissipates once the patient lies supine.[25] An extremely rare complication of SCSFLS is third nerve palsy, where the ability to move one's eyes becomes difficult and interrupted due to compression of the third cranial nerve.[26]
There are documented cases of reversible frontotemporal dementia and coma.[27] Coma due to a CSF leak has been successfully treated by using blood patches and/or fibrin glue and placing the person in the Trendelenburg position.[28] Empty sella syndrome, a bony structure that surround the pituitary gland, occurs in CSF leak patients.[29][30]
3. Causes
A spontaneous CSF leak is termed so as it has previously been thought to be idiopathic, meaning the cause is unknown.[15][31] The evidence of the last decade suggests however, that these leaks result from either a discogenic pathology, such as microspur, osteophyte or intra-dural disc herniation that pierces the dura like a knife, connective tissue disorder (which can often lead to discogenic pathology), or spinal drainage problems. [32][33]
3.1. Discogenic Causes
The most common cause of an intractable "sCSF" leak is discogenic, either from an intra-dural disc herniation, osteophyte or microspur on the disc or vertebral body. "Recent radiological and microsurgical investigations revealed that a calcified, degenerative bony microspur is often the culprit lesion in cases of intractable CSF leaks. Arising from the level of the intervertebrate disk space, these microspurs pierce the ventral dura and produce a slit-like defect a few millimeters in length. These microspurs and the associated CSF leak have to be localized exactly, and then they are amenable to surgical treatment."[32][33]
3.2. Connective Tissue Theory
Various scientists and physicians have suggested that SCSFLs may be the result of an underlying connective tissue disorder affecting the spinal dura.[9][10][19][34] It may also run in families and be associated with aortic aneurysms and joint hypermobility.[10][35]
Up to two thirds of those affected demonstrate some type of generalized connective tissue disorder.[10][34] Marfan syndrome, Ehlers–Danlos syndrome, and autosomal dominant polycystic kidney disease are the three most common connective tissue disorders associated with SCSFLs.[10] Roughly 20% of patients with a SCSFL exhibit features of Marfan syndrome, including tall stature, hollowed chest (pectus excavatum), joint hypermobility and arched palate. However, no other Marfan syndrome presentations are shown.[10]
3.3. Spinal Drainage Theory
Some studies have proposed that issues with the spinal venous drainage system may cause a CSF leak.[36] According to this theory, dural holes and intracranial hypotension are symptoms caused by low venous pressure in the epidural space. When inferior limb muscles pump blood towards the heart and pressure in the inferior vena cava vein becomes negative, the network of epidural veins is overdrained, causing CSF to be aspirated into the epidural space. True leaks can form at weak points in the spinal meninges. Therefore, the observed CSF hypotension is a result of CSF hypovolemia and reduced epidural venous pressure.[36]
3.4. Other Causes
Cranial CSF leaks result from intracranial hypertension in a vast majority of cases. The increased pressure causes a rupture of the cranial dura mater, leading to CSF leak and intracranial hypotension.[29][37] Patients with a nude nerve root, where the root sleeve is absent, are at increased risk for developing recurrent CSF leaks.[38] Lumbar disc herniation has been reported to cause CSF leaks in at least one case.[39] Degenerative spinal disc diseases cause a disc to pierce the dura mater, leading to a CSF leak.[10]
4. Pathophysiology
Cerebrospinal fluid is produced by the choroid plexus in the ventricles of the brain and contained by the dura and arachnoid layers of the meninges.[9][19][40] The brain floats in CSF, which also transports nutrients to the brain and spinal cord. As holes form in the spinal dura mater, CSF leaks out into the surrounding space. The CSF is then absorbed into the spinal epidural venous plexus or soft tissues around the spine.[10][41] Due to the sterile conditions of the soft tissues around the spine, there is no risk of meningitis.[10]
5. Diagnosis
The primary place of first complaint to a physician is a hospital emergency room.[11][42] Up to 94% of those suffering from SCSFLS are initially misdiagnosed. Incorrect diagnoses include migraines, meningitis, Chiari malformation, and psychiatric disorders. The average time from onset of symptoms until definitive diagnosis is 13 months.[43] A 2007 study found a 0% success rate for proper diagnosis in the emergency department.[42]
Diagnosis of CSF leakage can be done by various imaging techniques, chemical tests of bodily fluid discharged from a head orifice, or clinical examination. The use of CT, MRI, and assays are the most common types of CSF leak instrumental tests. Many CSF leaks do not show up on imaging and chemical assays, thus such diagnostic tools are not definitive to rule out CSF leaks. A clinician may often depend upon patient history and exam to diagnose, for example: discharge of excessive amount of clear fluid from the nose upon bending over, the increase in headache following a Valsalva maneuver or the reduction of headache when the patient takes a prone position are positive indicators.
A clinical exam is often used as a means to diagnose CSF leaks. Improved patient response to conservative treatment may further define a positive diagnosis. The lack of clinician awareness of the signs -symptoms and ailments- of a CSF leak is the greatest challenge to proper diagnosis and treatment, in particular: the loss of the orthostatic characteristic of headache and that every chronic CSF leaker will have a unique symptom set that as a whole contributes to the underlying condition, and diagnosis of, a CSF leak.
5.1. CT
Diagnosis of a cerebrospinal fluid leak is performed through a combination of measurement of the CSF pressure and a computed tomography myelogram (CTM) scan of the spinal column for fluid leaks.[10] The opening fluid pressure in the spinal canal is obtained by performing a lumbar puncture, also known as a spinal tap. Once the pressure is measured, a radiocontrast agent is injected into the spinal fluid. The contrast then diffuses out through the dura sac before leaking through dural holes. This allows for a CTM with fluoroscopy to locate and image any sites of dura rupture via contrast seen outside the dura sac in the imagery.[9][18][19]
5.2. MRI
There is disagreement over whether MRI should be the diagnostic tool of choice.[10][17][19] Magnetic resonance imaging is less effective than CT at directly imaging sites of CSF leak. MRI studies may show pachymeningeal enhancement (when the dura mater looks thick and inflamed), sagging of the brain, pituitary enlargement, subdural hygromas, engorgement of cerebral venous sinuses, and other abnormalities.[17] For 20% of patients, MRIs present as completely normal.[17] MRIs performed with the patient seated upright (vs. laying supine) are not better for diagnosing CSF leaks,[44] but are more than twice as effective at diagnosing cerebellar tonsillar ectopia, also known as Chiari malformation.[45] Cerebellar tonsillar ectopia shares many of the same symptoms as CSF leak,[24] but originates either congenitally or from trauma, including whiplash strain to the dura.[45]
An alternate method of locating the site of a CSF leak is to use heavily T2-weighted MR myelography.[10] This has been effective in identifying the sites of a CSF leak without the need for a CT scan, lumbar puncture, and contrast and at locating fluid collections such as CSF pooling.[46] Another highly successful method of locating a CSF leak is intrathecal contrast and MR Myelography.[10]
5.3. Assay
When cranial CSF leak is suspected because of discharge from the nose or ear that is potentially CSF, the fluid can be collected and tested with a beta-2 transferrin assay.[21] This test can positively identify if the fluid is cerebrospinal fluid.[21]
5.4 CSF Analysis
Patients with CSF leaks have been noted to have very low or even negative opening pressures during lumbar puncture. However, patients with confirmed CSF leaks may also demonstrate completely normal opening pressures. In 18–46% of cases, the CSF pressure is measured within the normal range.[10][47][48][49] Analysis of spinal fluid may demonstrate lymphocytic pleocytosis and elevated protein content or xanthochromia. This is hypothesized to be due to increased permeability of dilated meningeal blood vessels and a decrease of CSF flow in the lumbar subarachnoid space.[10]
5.5. Clinical Presentation
The diagnostic criteria for SCSFLS is based on the 2004 International Classification of Headache Disorders, 2nd edn (ICHD-II) (Table 1) (50) criteria. However, the presentation of patients with confirmed diagnosis may be very different from that of the clinical diagnostic criteria and cannot be considered authoritative.[10]
6. Treatment
Initial measures can include rest, caffeine intake (via coffee or intravenous infusion), and hydration.[17] Corticosteroids may provide transient relief for some patients.[17] An abdominal binder, which increases intracranial pressure by compressing the abdomen, can temporarily relieve symptoms for some people.[50]
6.1. Epidural Blood Patch
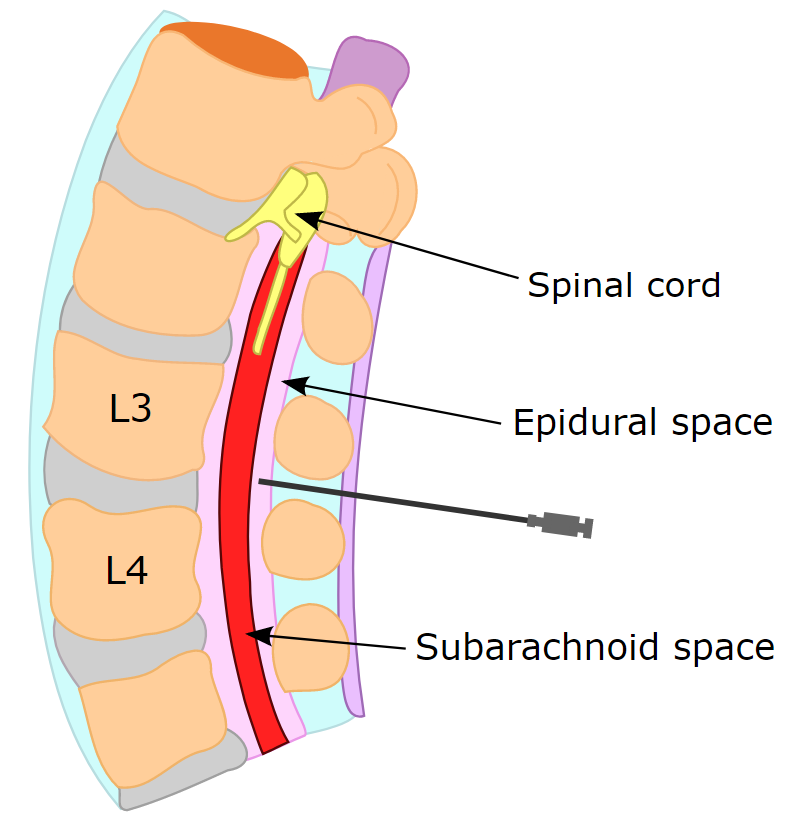
The treatment of choice for this condition is the surgical application of epidural blood patches,[13][51][52] which has a higher success rate than conservative treatments of bed rest and hydration.[53] Through the injection of a person's own blood into the area of the hole in the dura, an epidural blood patch uses blood's clotting factors to clot the sites of holes. The volume of autologous blood and number of patch attempts for patients is highly variable.[13] One-quarter to one-third of SCSFLS patients do not have relief of symptoms from epidural blood patching.[10]
6.2. Fibrin Glue Sealant
If blood patches alone do not succeed in closing the dural tears, placement of percutaneous fibrin glue can be used in place of blood patching, raising the effectiveness of forming a clot and arresting CSF leakage.[2][10][54]
6.3. Surgical Drain Technique
In extreme cases of intractable CSF leak, a surgical lumbar drain has been used.[55][56][57] This procedure is believed to decrease spinal CSF volume while increasing intracranial CSF pressure and volume.[55] This procedure restores normal intracranial CSF volume and pressure while promoting the healing of dural tears by lowering the pressure and volume in the dura.[55][57] This procedure has led to positive results leading to relief of symptoms for up to one year.[55][56]
6.4. Neurosurgical Repair
For patients that do not respond to either epidural blood patching or fibrin glue, neurosurgery is available to directly repair leaking meningeal diverticula. The areas of dura leak can be tied together in a process called ligation and then a metal clip can be placed in order to hold the ligation closed.[10] Alternatively, a small compress called a pledget can be placed over the dura leak and then sealed with gel foam and fibrin glue.[10] Primary suturing is rarely able to repair a CSF leak, and in some patients exploration of the dura may be required to properly locate all sites of CSF leak.[10]
7. Prognosis
Long-term outcomes for people with SCSFLS remain poorly studied.[10] Symptoms may resolve in as little as two weeks, or persist for months.[17] Less commonly, patients may suffer from unremitting symptoms for many years.[9][17][19][23] People with chronic SCSFLS may be disabled and unable to work.[10][14] Recurrent CSF leak at an alternate site after recent repair is common.[58]
8. Epidemiology
A 1994 community-based study indicated that two out of every 100,000 people suffered from SCSFLS, while a 2004 emergency room-based study indicated five per 100,000.[10][19] SCSFLS generally affects the young and middle aged;[55] the average age for onset is 42.3 years, but onset can range from ages 22 to 61.[59] In an 11-year study, women were found to be twice as likely to be affected as men.[60][61]
Studies have shown that SCSFLS runs in families. It is suspected that genetic similarity in families includes weakness in the dura mater which leads to SCSFLS.[10][62] Large scale population-based studies have not yet been conducted.[19] While a majority of SCSFLS cases continue to be undiagnosed or misdiagnosed, an actual increase in occurrence is unlikely.[19]
9. History
Spontaneous CSF leaks have been described by notable physicians and reported in medical journals dating back to the early 1900s.[63][64] German neurologist Georg Schaltenbrand reported in 1938 and 1953 what he termed "aliquorrhea", a condition marked by very low, unobtainable, or even negative CSF pressures. The symptoms included orthostatic headaches and other features that are now recognized as spontaneous intracranial hypotension. A few decades earlier, the same syndrome had been described in French literature as "hypotension of spinal fluid" and "ventricular collapse". In 1940, Henry Woltman of the Mayo Clinic wrote about "headaches associated with decreased intracranial pressure". The full clinical manifestations of intracranial hypotension and CSF leaks were described in several publications reported between the 1960s and early 1990s.[64] Modern reports of spontaneous CSF leak have been reported to medical journals since the late 1980s.[65]
10. Research
Tetracosactide is a corticosteroid that causes the brain to produce additional spinal fluid to replace the volume of the lost CSF and alleviate symptoms, and has been given intravenously to treat CSF leaks.[66][67]
In three small studies of 1-2 patients suffering from recurrent CSF leaks where repeated blood patches failed to form clots and relieve symptoms, the patients received temporary but complete resolution of symptoms with an epidural saline infusion.[68][69] The saline infusion temporarily restores the volume necessary for a patient to avoid SIH until the leak can be repaired properly.[10] Intrathecal saline infusion is used in urgent cases such as intractable pain or decreased consciousness.[10]
The gene TGFBR2 has been implicated in several connective tissue disorders including Marfan syndrome, arterial tortuosity, and thoracic aortic aneurysm. A study of patients with SCSFLS demonstrated no mutations in this gene.[10] Minor features of Marfan syndrome have been found in 20% of CSF leak patients. Abnormal findings of fibrillin-1 have been documented in these CSF leak patients, but only one patient demonstrated a fibrillin-1 defect consistent with Marfan syndrome.[10][70]
References
- Lloyd, K. M.; Delgaudio, J. M.; Hudgins, P. A. (2008). "Imaging of Skull Base Cerebrospinal Fluid Leaks in Adults". Radiology 248 (3): 725–36. doi:10.1148/radiol.2483070362. PMID 18710972. https://dx.doi.org/10.1148%2Fradiol.2483070362
- Gordon, N. (2009). "Spontaneous intracranial hypotension". Developmental Medicine & Child Neurology 51 (12): 932–935. doi:10.1111/j.1469-8749.2009.03514.x. PMID 19909307. https://www.pediatricneurologybriefs.com/jms/article/view/pedneurbriefs-27-10-5.
- Mokri, B. (2013). "Spontaneous low pressure, low CSF volume headaches: spontaneous CSF leaks". Headache: The Journal of Head and Face Pain 53 (7): 1034–1053. doi:10.1111/head.12149. PMID 23808630. https://dx.doi.org/10.1111%2Fhead.12149
- Tam, EK; Gilbert, AL (November 2019). "Spontaneous cerebrospinal fluid leak and idiopathic intracranial hypertension.". Current Opinion in Ophthalmology 30 (6): 467–471. doi:10.1097/ICU.0000000000000603. PMID 31449087. https://dx.doi.org/10.1097%2FICU.0000000000000603
- Maher, CO; Meyer; Mokri (2000). "Surgical treatment of spontaneous spinal cerebrospinal fluid leaks". Neurosurgical Focus 9 (1): e7. doi:10.3171/foc.2000.9.1.7. PMID 16859268. https://dx.doi.org/10.3171%2Ffoc.2000.9.1.7
- Greenberg, Mark (2006). Handbook of neurosurgery. New York, NY: Thieme Medical Publishers. p. 178. ISBN 978-0-86577-909-9. https://books.google.com/books?id=ExHcxxufG8sC&pg=PA178&dq=Spontaneous+intracranial+hypotension&cd=1#v=onepage. Retrieved 18 December 2009.
- Neil R. Miller; William Fletcher Hoyt (2005). Walsh and Hoyt's clinical neuro-ophthalmology. Lippincott Williams & Wilkins. pp. 1303–. ISBN 978-0-7817-4811-7. https://books.google.com/books?id=ATTlVWi3mvwC&pg=PA1303. Retrieved 8 November 2010.
- Mokri, B. (1999). "Spontaneous cerebrospinal fluid leaks: from intracranial hypotension to cerebrospinal fluid hypovolemia--evolution of a concept". Mayo Clinic Proceedings 74 (11): 1113–1123. doi:10.4065/74.11.1113. PMID 10560599. https://dx.doi.org/10.4065%2F74.11.1113
- Schievink, WI (2000). "Spontaneous spinal cerebrospinal fluid leaks: a review". Neurosurgical Focus 9 (1): 1–9. doi:10.3171/foc.2000.9.1.8. PMID 16859269. https://dx.doi.org/10.3171%2Ffoc.2000.9.1.8
- Schievink, W. I. (2008). "Spontaneous spinal cerebrospinal fluid leaks". Cephalalgia : An International Journal of Headache 28 (12): 1345–1356. doi:10.1111/j.1468-2982.2008.01776.x. PMID 19037970. https://dx.doi.org/10.1111%2Fj.1468-2982.2008.01776.x
- Vaidhyanath, R.; Kenningham, R.; Khan, A.; Messios, N. (2007). "Spontaneous intracranial hypotension: a cause of severe acute headache". Emergency Medicine Journal 24 (10): 739–741. doi:10.1136/emj.2007.048694. PMID 17901290. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2658456
- Schievink, W.; Palestrant, D.; Maya, M.; Rappard, G. (2009). "Spontaneous spinal cerebrospinal fluid leak as a cause of coma after craniotomy for clipping of an unruptured intracranial aneurysm". Journal of Neurosurgery 110 (3): 521–524. doi:10.3171/2008.9.JNS08670. PMID 19012477. https://dx.doi.org/10.3171%2F2008.9.JNS08670
- Mehta, B.; Tarshis, J. (2009). "Repeated large-volume epidural blood patches for the treatment of spontaneous intracranial hypotension". Canadian Journal of Anesthesia 56 (8): 609–13. doi:10.1007/s12630-009-9121-y. PMID 19495908. https://dx.doi.org/10.1007%2Fs12630-009-9121-y
- Mea, E.; Chiapparini, L.; Savoiardo, M.; Franzini, A.; Bussone, G.; Leone, M. (2009). "Clinical features and outcomes in spontaneous intracranial hypotension: a survey of 90 consecutive patients". Neurological Sciences 30 (S1): S11–S13. doi:10.1007/s10072-009-0060-8. PMID 19415418. https://dx.doi.org/10.1007%2Fs10072-009-0060-8
- Victor, Maurice; Ropper, Allan H.; Adams, Raymond Delacy; Brown, Robert F. (2005). Adams and Victor's principles of neurology. New York: McGraw-Hill Medical Pub. Division. pp. 541–543. ISBN 978-0-07-141620-7. https://archive.org/details/adamsvictorsprin00ropp_284.
- Leep Hunderfund, A. N.; Mokri, B. (2011). "Second-half-of-the-day headache as a manifestation of spontaneous CSF leak". Journal of Neurology 259 (2): 306–10. doi:10.1007/s00415-011-6181-z. PMID 21811806. https://dx.doi.org/10.1007%2Fs00415-011-6181-z
- Spears, R. C. (2014). "Low-pressure/spinal fluid leak headache". Current Pain and Headache Reports 18 (6): 1–6. doi:10.1007/s11916-014-0425-4. PMID 24760494. https://dx.doi.org/10.1007%2Fs11916-014-0425-4
- Hofmann, E.; Behr, R.; Schwager, K. (2009). "Imaging of cerebrospinal fluid leaks". Klinische Neuroradiologie 19 (2): 111–121. doi:10.1007/s00062-009-9008-x. PMID 19636501. https://dx.doi.org/10.1007%2Fs00062-009-9008-x
- Schievink, W. I. (2006). "Spontaneous Spinal Cerebrospinal Fluid Leaks and Intracranial Hypotension". Journal of the American Medical Association 295 (19): 2286–96. doi:10.1001/jama.295.19.2286. PMID 16705110. https://dx.doi.org/10.1001%2Fjama.295.19.2286
- Mokri, B (2001). "Spontaneous intracranial hypotension". Current Pain and Headache Reports 5 (3): 284–91. doi:10.1007/s11916-001-0045-7. PMID 11309218. https://dx.doi.org/10.1007%2Fs11916-001-0045-7
- Abuabara, A (2007). "Cerebrospinal fluid rhinorrhoea: diagnosis and management". Medicina Oral, Patologia Oral y Cirugia Bucal 12 (5): E397–400. PMID 17767107. http://www.ncbi.nlm.nih.gov/pubmed/17767107
- Stenzel, M.; Preuss, S.; Orloff, L.; Jecker, P.; Mann, W. (2005). "Cerebrospinal Fluid Leaks of Temporal Bone Origin: Etiology and Management". ORL; Journal for Oto-Rhino-Laryngology and Its Related Specialties 67 (1): 51–5. doi:10.1159/000084306. PMID 15753623. https://dx.doi.org/10.1159%2F000084306
- Schwedt, TJ; Dodick, DW (2007). "Spontaneous intracranial hypotension". Current Pain and Headache Reports 11 (1): 56–61. doi:10.1007/s11916-007-0023-9. PMID 17214923. https://dx.doi.org/10.1007%2Fs11916-007-0023-9
- "Spontaneous Spinal Cerebrospinal Fluid Leaks: Diagnosis". http://www.medscape.com/viewarticle/405623_2.
- Schievink, W. I.; Maya, M. M. (2006). "Quadriplegia and cerebellar hemorrhage in spontaneous intracranial hypotension". Neurology 66 (11): 1777–8. doi:10.1212/01.wnl.0000218210.83855.40. PMID 16769965. https://dx.doi.org/10.1212%2F01.wnl.0000218210.83855.40
- Alonso Cánovas, A; Martínez San Millán, J; Novillo López, ME; Masjuán Vallejo, J (2008). "Third cranial nerve palsy due to intracranial hypotension syndrome". Neurologia (Barcelona, Spain) 23 (7): 462–5. PMID 18726726. http://www.ncbi.nlm.nih.gov/pubmed/18726726
- Sayao, AL; Heran, MK; Chapman, K; Redekop, G; Foti, D (2009). "Intracranial hypotension causing reversible frontotemporal dementia and coma". The Canadian Journal of Neurological Sciences 36 (2): 252–6. doi:10.1017/s0317167100006636. PMID 19378725. https://dx.doi.org/10.1017%2Fs0317167100006636
- Ferrante, E.; Arpino, I.; Citterio, A.; Savino, A. (2009). "Coma resulting from spontaneous intracranial hypotension treated with the epidural blood patch in the Trendelenburg position pre-medicated with acetazolamide". Clinical Neurology and Neurosurgery 111 (8): 699–702. doi:10.1016/j.clineuro.2009.06.001. PMID 19577356. https://dx.doi.org/10.1016%2Fj.clineuro.2009.06.001
- Woodworth, B. A.; Palmer, J. N. (2009). "Spontaneous cerebrospinal fluid leaks". Current Opinion in Otolaryngology & Head and Neck Surgery 17 (1): 59–65. doi:10.1097/MOO.0b013e3283200017. PMID 19225307. https://dx.doi.org/10.1097%2FMOO.0b013e3283200017
- Schievink, W. I.; Moser, F. G.; Pikul, B. K. (2007). "Reversal of coma with an injection of glue". The Lancet 369 (9570): 1402. doi:10.1016/S0140-6736(07)60636-9. PMID 17448827. https://dx.doi.org/10.1016%2FS0140-6736%2807%2960636-9
- Schievink, W. I.; Louy, C. (2007). "Precipitating Factors of Spontaneous Spinal Csf Leaks and Intracranial Hypotension". Neurology 69 (7): 700–702. doi:10.1212/01.wnl.0000267324.68013.8e. PMID 17698794. https://dx.doi.org/10.1212%2F01.wnl.0000267324.68013.8e
- Beck, Jürgen (2018). "Diagnostic challenges and therapeutic possibilities in spontaneous intracranial hypotension". Clincial and Translational Neuroscience 2 (2). doi:10.1177/2514183X18787371. https://dx.doi.org/10.1177%2F2514183X18787371
- Feichter I. (2019). "Intradural non-calcified thoracic disc herniation causing spontaneous intracranial hypotension: a case report". BMC Surg. 19 (66). doi:10.1186/s12893-019-0527-3. https://dx.doi.org/10.1186%2Fs12893-019-0527-3
- Liu, F. -C.; Fuh, J. -L.; Wang, Y. -F.; Wang, S. -J. (2011). "Connective tissue disorders in patients with spontaneous intracranial hypotension". Cephalalgia 31 (6): 691–695. doi:10.1177/0333102410394676. PMID 21220378. https://dx.doi.org/10.1177%2F0333102410394676
- Mokri, B. (2007). "Familial Occurrence of Spontaneous Spinal CSF Leaks: Underlying Connective Tissue Disorder (CME)". Headache: The Journal of Head and Face Pain 48 (1): 146–149. doi:10.1111/j.1526-4610.2007.00979.x. PMID 18184297. https://dx.doi.org/10.1111%2Fj.1526-4610.2007.00979.x
- Franzini, A.; Messina, G.; Nazzi, V.; Mea, E.; Leone, M.; Chiapparini, L.; Broggi, G.; Bussone, G. (2009). "Spontaneous intracranial hypotension syndrome: a novel speculative physiopathological hypothesis and a novel patch method in a series of 28 consecutive patients". Journal of Neurosurgery 112 (2): 300–6. doi:10.3171/2009.6.JNS09415. PMID 19591547. https://dx.doi.org/10.3171%2F2009.6.JNS09415
- Schlosser, RJ; Wilensky, EM; Grady, MS; Bolger, WE (2003). "Elevated intracranial pressures in spontaneous cerebrospinal fluid leaks". American Journal of Rhinology 17 (4): 191–5. doi:10.1177/194589240301700403. PMID 12962187. https://dx.doi.org/10.1177%2F194589240301700403
- Schievink, WI; Jacques, L (2003). "Recurrent spontaneous spinal cerebrospinal fluid leak associated with "nude nerve root" syndrome: case report". Neurosurgery 53 (5): 1216–8; discussion 1218–9. doi:10.1227/01.NEU.0000089483.30857.11. PMID 14580290. https://dx.doi.org/10.1227%2F01.NEU.0000089483.30857.11
- Kim, K. T.; Kim, Y. B. (2010). "Spontaneous Intracranial Hypotension Secondary to Lumbar Disc Herniation". Journal of Korean Neurosurgical Society 47 (1): 48–50. doi:10.3340/jkns.2010.47.1.48. PMID 20157378. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2817515
- Michael Schuenke; Udo Schumacher; Erik Schulte; Edward D. Lamperti; Lawrence M. Ross (2007). Head and neuroanatomy. Thieme. ISBN 978-3-13-142101-2. https://books.google.com/books?id=Y0-Rf_m7xj4C. Retrieved 8 November 2010.
- Inamasu, J.; Guiot, B. (2006). "Intracranial hypotension with spinal pathology". The Spine Journal 6 (5): 591–9. doi:10.1016/j.spinee.2005.12.026. PMID 16934734. https://dx.doi.org/10.1016%2Fj.spinee.2005.12.026
- Schievink, W. I.; Maya, M. M.; Moser, F.; Tourje, J.; Torbati, S. (2007). "Frequency of spontaneous intracranial hypotension in the emergency department". The Journal of Headache and Pain 8 (6): 325–328. doi:10.1007/s10194-007-0421-8. PMID 18071632. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=3476164
- Schievink, W. I. (2003). "Misdiagnosis of Spontaneous Intracranial Hypotension". Archives of Neurology 60 (12): 1713–8. doi:10.1001/archneur.60.12.1713. PMID 14676045. https://dx.doi.org/10.1001%2Farchneur.60.12.1713
- Schievink, W. I.; Tourje, J. (2007). "Upright MRI in Spontaneous Spinal Cerebrospinal Fluid Leaks and Intracranial Hypotension". Headache: The Journal of Head and Face Pain 47 (9): 1345–6. doi:10.1111/j.1526-4610.2007.00934.x. PMID 17927653. https://dx.doi.org/10.1111%2Fj.1526-4610.2007.00934.x
- Freeman, MD; Rosa, S; Harshfield, D; Smith, F; Bennett, R; Centeno, C. J.; Kornel, E; Nystrom, A et al. (2010). "A case-control study of cerebellar tonsillar ectopia (Chiari) and head/neck trauma (whiplash)". Brain Injury 24 (7–8): 988–94. doi:10.3109/02699052.2010.490512. PMID 20545453. https://dx.doi.org/10.3109%2F02699052.2010.490512
- Wang, Y. -F.; Lirng, J. -F.; Fuh, J. -L.; Hseu, S. -S.; Wang, S. -J. (2009). "Heavily T2-weighted MR myelography vs CT myelography in spontaneous intracranial hypotension". Neurology 73 (22): 1892–8. doi:10.1212/WNL.0b013e3181c3fd99. PMID 19949036. https://dx.doi.org/10.1212%2FWNL.0b013e3181c3fd99
- Kelley, G (2004). "CSF hypovolemia vs intracranial hypotension in "spontaneous intracranial hypotension syndrome"". Neurology 62 (8): 1453. doi:10.1212/wnl.62.8.1453. PMID 15111706. https://dx.doi.org/10.1212%2Fwnl.62.8.1453
- Canas, N; Medeiros, E; Fonseca, AT; Palma-Mira, F (2004). "CSF volume loss in spontaneous intracranial hypotension". Neurology 63 (1): 186–7. doi:10.1212/01.wnl.0000132964.07982.cc. PMID 15249640. https://dx.doi.org/10.1212%2F01.wnl.0000132964.07982.cc
- Mark S. Greenberg (2006). Handbook of neurosurgery. Thieme. pp. 178–. ISBN 978-3-13-110886-9. https://books.google.com/books?id=ExHcxxufG8sC&pg=PA178. Retrieved 8 November 2010.
- Schievink, W. I.; Deline, C. R. (2014). "Headache secondary to intracranial hypotension". Current Pain and Headache Reports 18 (457): 1–9. doi:10.1007/s11916-014-0457-9. PMID 25255993. https://dx.doi.org/10.1007%2Fs11916-014-0457-9
- Peng, PW; Farb (2008). "Spontaneous C1-2 CSF leak treated with high cervical epidural blood patch". The Canadian Journal of Neurological Sciences 35 (1): 102–5. doi:10.1017/s0317167100007654. PMID 18380287. https://dx.doi.org/10.1017%2Fs0317167100007654
- Grimaldi, D.; Mea, E.; Chiapparini, L.; Ciceri, E.; Nappini, S.; Savoiardo, M.; Castelli, M.; Cortelli, P. et al. (2004). "Spontaneous low cerebrospinal pressure: a mini review". Neurological Sciences 25 (S3): S135–S137. doi:10.1007/s10072-004-0272-x. PMID 15549523. https://dx.doi.org/10.1007%2Fs10072-004-0272-x
- Wang, S.; Lirng, J.; Hseu, S.; Chan, K. (2008). "Spontaneous Intracranial Hypotension Treated by Epidural Blood Patches". Acta Anaesthesiologica Taiwanica 46 (3): 129–133. doi:10.1016/S1875-4597(08)60007-7. PMID 18809524. https://dx.doi.org/10.1016%2FS1875-4597%2808%2960007-7
- Schievink, W. I.; Maya, M. M.; Moser, F. M. (2004). "Treatment of spontaneous intracranial hypotension with percutaneous placement of a fibrin sealant". Journal of Neurosurgery 100 (6): 1098–1100. doi:10.3171/jns.2004.100.6.1098. PMID 15200130. https://dx.doi.org/10.3171%2Fjns.2004.100.6.1098
- Schievink, W. I. (2009). "A Novel Technique for Treatment of Intractable Spontaneous Intracranial Hypotension: Lumbar Dural Reduction Surgery". Headache: The Journal of Head and Face Pain 49 (7): 1047–1051. doi:10.1111/j.1526-4610.2009.01450.x. PMID 19473279. https://dx.doi.org/10.1111%2Fj.1526-4610.2009.01450.x
- Kitchel, SH; Eismont, FJ; Green, BA (1989). "Closed subarachnoid drainage for management of cerebrospinal fluid leakage after an operation on the spine". The Journal of Bone and Joint Surgery. American Volume 71 (7): 984–7. doi:10.2106/00004623-198971070-00004. PMID 2760094. https://dx.doi.org/10.2106%2F00004623-198971070-00004
- Roosendaal, C. M.; Coppes, M. H.; Vroomen, P. C. A. J. (2009). "The paradox of intracranial hypotension responding well to CSF drainage". European Journal of Neurology 16 (12): e178–9. doi:10.1111/j.1468-1331.2009.02803.x. PMID 19863649. https://dx.doi.org/10.1111%2Fj.1468-1331.2009.02803.x
- Schievink, W. I.; Maya, M. M.; Riedinger, M. (2003). "Recurrent spontaneous spinal cerebrospinal fluid leaks and intracranial hypotension: a prospective study". Journal of Neurosurgery 99 (5): 840–842. doi:10.3171/jns.2003.99.5.0840. PMID 14609162. https://dx.doi.org/10.3171%2Fjns.2003.99.5.0840
- Schievink, W. I.; Morreale, V. M.; Atkinson, J. L. D.; Meyer, F. B.; Piepgras, D. G.; Ebersold, M. J. (1998). "Surgical treatment of spontaneous spinal cerebrospinal fluid leaks". Journal of Neurosurgery 88 (2): 243–246. doi:10.3171/jns.1998.88.2.0243. PMID 9452231. https://dx.doi.org/10.3171%2Fjns.1998.88.2.0243
- Ferrante, E.; Wetzl, R.; Savino, A.; Citterio, A.; Protti, A. (2004). "Spontaneous cerebrospinal fluid leak syndrome: report of 18 cases". Neurological Sciences. 25 Suppl 3 (S3): S293–S295. doi:10.1007/s10072-004-0315-3. PMID 15549566. https://dx.doi.org/10.1007%2Fs10072-004-0315-3
- Schievink, W.; Maya, M.; Pikul, B.; Louy, C. (2009). "Spontaneous spinal cerebrospinal fluid leaks as the cause of subdural hematomas in elderly patients on anticoagulation". Journal of Neurosurgery 112 (2): 295–299. doi:10.3171/2008.10.JNS08428. PMID 19199465. https://dx.doi.org/10.3171%2F2008.10.JNS08428
- Larrosa, D; Vázquez, J; Mateo, I; Infante, J (2009). "Familial spontaneous intracranial hypotension". Neurologia (Barcelona, Spain) 24 (7): 485–7. PMID 19921558. http://www.ncbi.nlm.nih.gov/pubmed/19921558
- Schaltenbrand, G (1953). "Normal and pathological physiology of the cerebrospinal fluid circulation". Lancet 1 (6765): 805–8. doi:10.1016/S0140-6736(53)91948-5. PMID 13036182. https://dx.doi.org/10.1016%2FS0140-6736%2853%2991948-5
- Mokri, B (2000). "Cerebrospinal fluid volume depletion and its emerging clinical/imaging syndromes". Neurosurgical Focus 9 (1): 1–7. doi:10.3171/foc.2000.9.1.6. PMID 16859267. https://dx.doi.org/10.3171%2Ffoc.2000.9.1.6
- null
- Carter, B.; Pasupuleti, R. (2000). "Use of intravenous cosyntropin in the treatment of postdural puncture headache". Anesthesiology 92 (1): 272–274. doi:10.1097/00000542-200001000-00043. PMID 10638928. https://dx.doi.org/10.1097%2F00000542-200001000-00043
- Cánovas, L; Barros, C; Gómez, A; Castro, M; Castro, A (2002). "Use of intravenous tetracosactin in the treatment of postdural puncture headache: our experience in forty cases". Anesthesia and Analgesia 94 (5): 1369. doi:10.1097/00000539-200205000-00069. PMID 11973227. https://dx.doi.org/10.1097%2F00000539-200205000-00069
- Rouaud, T.; Lallement, F.; Choui, R.; Madigand, M. (2009). "Traitement de l'hypotension spontanée du liquide cérébrospinal par perfusion épidurale de sérum salé isotonique". Revue Neurologique 165 (2): 201–5. doi:10.1016/j.neurol.2008.05.006. PMID 19010507. https://dx.doi.org/10.1016%2Fj.neurol.2008.05.006
- Binder, DK; Dillon, WP; Fishman, RA; Schmidt, MH (2002). "Intrathecal saline infusion in the treatment of obtundation associated with spontaneous intracranial hypotension: technical case report". Neurosurgery 51 (3): 830–6; discussion 836–7. doi:10.1097/00006123-200209000-00045. PMID 12188967. https://dx.doi.org/10.1097%2F00006123-200209000-00045
- Schrijver, I.; Schievink, W. I.; Godfrey, M.; Meyer, F. B.; Francke, U. (2002). "Spontaneous spinal cerebrospinal fluid leaks and minor skeletal features of Marfan syndrome: a microfibrillopathy". Journal of Neurosurgery 96 (3): 483–9. doi:10.3171/jns.2002.96.3.0483. PMID 11883832. https://dx.doi.org/10.3171%2Fjns.2002.96.3.0483





