Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Siti Hamidah Mohd Setapar | -- | 1926 | 2022-11-17 10:46:40 | | | |
| 2 | Catherine Yang | Meta information modification | 1926 | 2022-11-18 04:35:25 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Mohd-Setapar, S.H.; John, C.P.; Mohd-Nasir, H.; Azim, M.M.; Ahmad, A.; Alshammari, M.B. Nanotechnology in Natural Cosmetics. Encyclopedia. Available online: https://encyclopedia.pub/entry/35095 (accessed on 19 May 2026).
Mohd-Setapar SH, John CP, Mohd-Nasir H, Azim MM, Ahmad A, Alshammari MB. Nanotechnology in Natural Cosmetics. Encyclopedia. Available at: https://encyclopedia.pub/entry/35095. Accessed May 19, 2026.
Mohd-Setapar, Siti Hamidah, Clera Peter John, Hasmida Mohd-Nasir, Muhammad Mohsin Azim, Akil Ahmad, Mohammed B. Alshammari. "Nanotechnology in Natural Cosmetics" Encyclopedia, https://encyclopedia.pub/entry/35095 (accessed May 19, 2026).
Mohd-Setapar, S.H., John, C.P., Mohd-Nasir, H., Azim, M.M., Ahmad, A., & Alshammari, M.B. (2022, November 17). Nanotechnology in Natural Cosmetics. In Encyclopedia. https://encyclopedia.pub/entry/35095
Mohd-Setapar, Siti Hamidah, et al. "Nanotechnology in Natural Cosmetics." Encyclopedia. Web. 17 November, 2022.
Copy Citation
Nanotechnology is a comparatively modern field in the cosmetic industry. Presently, nanotechnology is indeed important as a platform for creating science-based alternatives for advanced therapeutics and cosmetics, resolving antiaging challenges, and enhancing well-being. Nanotechnology is described as an investigation of substances on a molecular and atomic scale. Cosmeceuticals based on nanotechnology offer the benefits of product differentiation, improved bioavailability, and prolonged effects of active ingredients.
nanotechnology-based cosmeceutical
nanoemulsion
micellar nanoparticles
1. Nanomaterials on Skin
Over the last few decades, nanomaterial-based products have been growing and targeting diverse industries, such as cosmetics, pharmaceutics, and dermatology. Nanomaterials are widely used in cosmetics, such as in the preparation of conditioners, hair serums, moisturizing creams, hair-repairing shampoos, skin-whitening creams, and antiwrinkle creams [1]. Cosmeceuticals are cosmetic products that contain naturally active materials and medicinal properties which are transferred onto the skin [2]. These have been used as cosmetics since they have been proven to enhance appearance. Cosmeceutical products provide significant medicinal effects on the skin and are used to treat various symptoms, including acne, dry skin, photoaging, poor appearance, black spots, and hyperpigmentation. Nanocosmeceuticals offer enhanced biocompatibility and stability, as well as a prolonged effect and the potential to improve the payload distribution to the skin. Multiple kinds of nanomaterials, such as niosomes, liposomes, nanostructured lipid carriers (NLC), solid lipid nanoparticles (SLN), gold nanoparticles, nanoemulsions, and polymeric nanoparticles, have been proposed for topical product distribution since they are made from biocompatible materials [3]. Their application in personal care products benefits from the potential to regulate the distribution of cosmeceuticals with the formation of a thin film, along with more precise delivery into the skin. Additionally, skin aging is a complex process that can be induced exogenously and endogenously [4]. However, about 90% of skin aging is activated by UV radiation exposure.
Another factor affecting the progression of the aging process is lifestyle, such as drinking, smoking, and sleeping, as well as environmental influences (pollution) and malnutrition. Dry texture, the texture of wrinkles, and loss of skin elasticity are the first symptoms of skin aging. The first symptoms of skin aging can be prevented and/or delayed nowadays. For the topical delivery of active ingredients, conventional topical formulations (suspensions, solutions, gels, aerosols, powder, and emulsion) are acceptable [4]. However, all these formulations have certain restrictions and can impair the treatment’s safety and/or efficacy. To overcome these constraints, a variety of nanomaterials for active-ingredient delivery have been developed. The continuous development of skin products with active ingredients utilizing nanomaterials in health and cosmetic industries creates new possibilities with favorable impacts on society and industries [5]. To promote product innovation by creating nanoproducts, the loading of active pharmaceutical ingredients (APIs) through nano-sized drug delivery systems is currently being exploited. The efficacy of nanoproducts utilized for API delivery into the skin, such as nanocosmeceuticals and nanopharmaceuticals, has previously been demonstrated, as numerous skincare products are already on the market.
The primary challenge in delivering APIs through the skin is overcoming the many physiological layers with different polarities. It is well recognized that skin defends the body from xenobiotics, which can include not just pathogens and hazardous toxins but also drugs, while somehow ensuring that physiological fluids are expelled for homeostasis. Because skin penetration reduces API absorption, the bioavailability of APIs administered topically is lesser than that of intravenous and oral routes. Numerous kinds of nanoparticles have been suggested to resolve the difficulties of API skin administration. Furthermore, Otlatici et al. [4] claim that various APIs may be utilized in cosmetic products to prevent, delay, and, if feasible, cure skin aging. Figure 1 summarizes an illustration of nanomaterials used for loading numerous APIs and their application purposes [5].
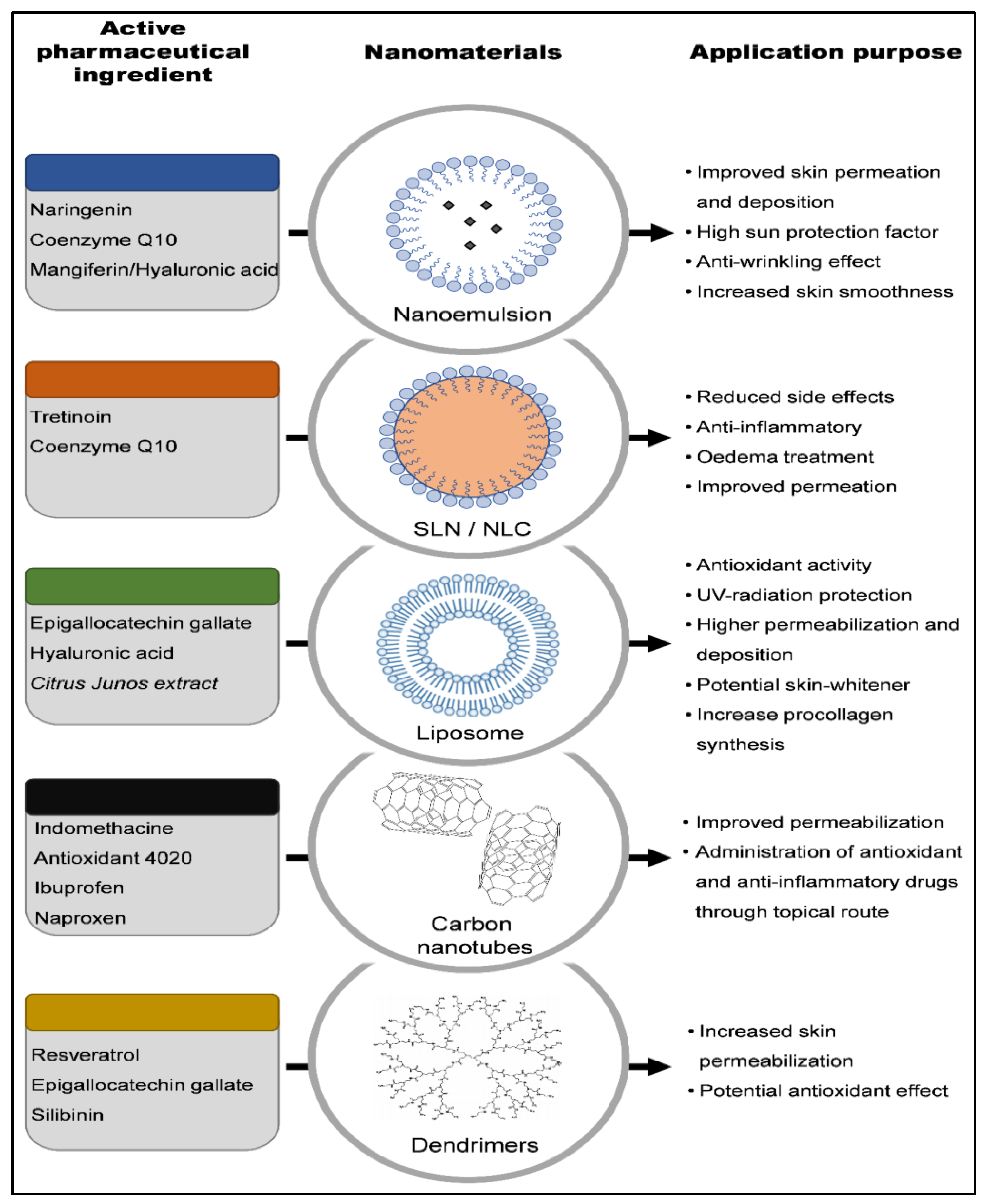
Figure 1. Illustration of nanomaterials that have been utilized to encapsulate a variety of active pharmaceutical ingredients, along with their applications [5]. SLN: Solid lipid nanoparticles; NLC: nanostructured lipid carriers.
2. Formation of Nanoemulsions
Nanoemulsions are colloidal dispersions of water-in-oil or oil-in-water, containing droplets with diameters ranging from a few to 200 nm. An improvement in nanoemulsion-related patent protection activities indicates a growing industrial interest in nanoemulsions [6]. Because of the tiny size of the droplets, they can provide favorable stability and optical, rheological, and ingredient delivery characteristics that are greater than conventional emulsions. Transparency and stability are characteristics of nanoemulsions. Furthermore, their formation increases the water content of the stratum corneum, indicating their efficacy as a moisturizer. Musazzi et al. [7] claim that nanoemulsions may substantially change the permeation profiles of molecules due to their physicochemical characteristics, and specifically, oil/water nanoemulsions greatly enhance the permeation profiles of polar ingredients when compared to conventional emulsions. Compared to conventional emulsions, nanoemulsions act as effective carriers for different bioactive compounds with enhanced properties, such as enhanced bioavailability, physical stability, and high optical consistency [8].
On the other hand, nanoemulsions have a lipophilic interior and are particularly desirable for the transport of hydrophobic substances under aqueous conditions [9]. Compared to nanocapsules, nanoemulsions have a greater ability to penetrate the skin and spread on it. This is possibly due to their lack of polymers and flexibility; this interacts with the stratum corneum. Nanoemulsions encourage water-immiscible active compounds, including lipids, antioxidants, and retinol, to penetrate more intensively into the target tissues and increase their effective concentrations.
Penetration of Nanoemulsion Systems into the Skin
The critical challenge with topical drug delivery is the skin barrier, which prevents the drug from penetrating into the systemic circulation. This barrier can be significantly resolved by nanoemulsion-based topical drug delivery. Sweat ducts, hair follicles, and directly through the stratum corneum are the three most prevalent pathways for drugs to penetrate the skin. In nanoemulsions, small-sized nanoparticles may readily flow across the pores, as illustrated in Figure 2. In addition, the hydrophilic and hydrophobic units make it easier to penetrate both the hydrophobic stratum corneum and the hydrophilic sweat ducts. Nanoemulsion can be used effectively against psoriasis and dermatitis [10]. Positively charged nanoemulsions interact more to penetrate through the negatively charged tissue and thus have been proven to provide greater performance and penetration than negatively charged nanoemulsions [11].
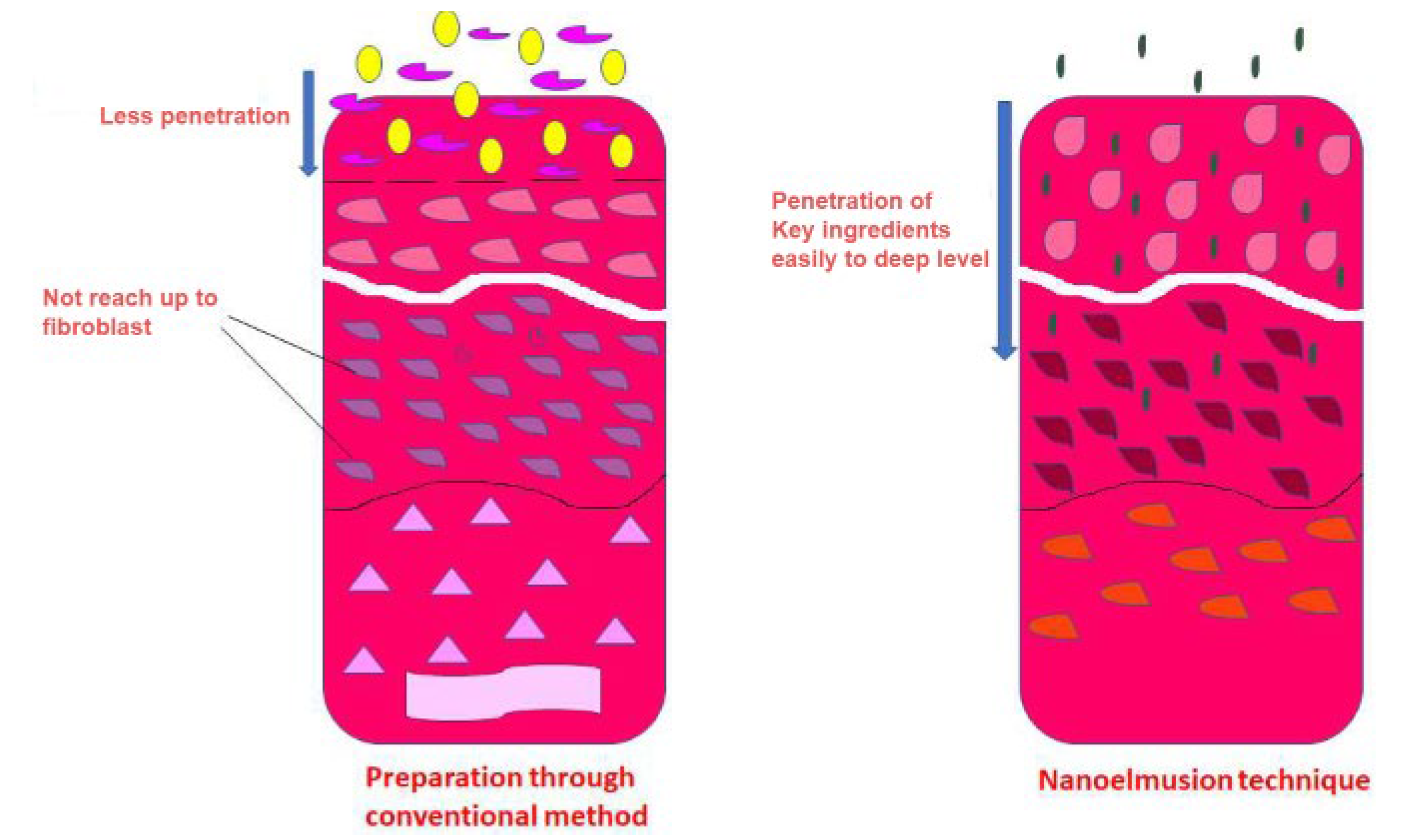
Figure 2. Comparison of nanoemulsions with the conventional transdermal formulations in the case of crossing skin barrier [12].
3. Formation of Micelle Nanoparticles
Micelle is a simple structure commonly created by amphibians in aqueous solutions. Typically, the particle size of micelles is in the range of 5–50 nm. They represent a group of associations or amphiphilic colloids that instantaneously form amphiphilic or surface-active agents in certain temperatures and concentrations, of which the molecules consist of two clearly distinct regions with opposite affinities to a given solvent. At low concentrations in an aqueous medium, these amphiphilic molecules exist separately. However, aggregation takes place when their concentration is raised to a specific range. The critical micelle concentration (CMC) is the concentration at which the monomeric amphiphile produces micelles [13].
The aggregation number of a micelle is the number of individual molecules that constitute the micelles. The formation of micelles is driven by the lowering of free energy in the system, resulting in the removal of hydrophobic segments from the aqueous environment and the re-establishment of the hydrogen bond network in water. The formation of Van der Waals attraction between hydrophobic blocks in the center of the produced micelles leads to an increase in energy gain [14]. The hydrophobic regions of amphiphilic molecules create the micelle’s core, while the hydrophilic portions create the micelle’s shell [15]. Micelles, when used as drug carriers in aqueous conditions, solubilize molecules of weakly soluble nonpolar pharmaceuticals within the micelle core, whereas polar molecules are adsorbed in the micelle shell. Drug carrier micelles have several benefits, such as [13]
- i.
-
The use of micelle-forming amphiphiles in drug solubilization leads to an increased bioavailability of sparingly soluble drugs and water solubility;
- ii.
-
Toxicity and other negative consequences are minimized;
- iii.
-
Permeability across physiological barriers is enhanced, and there are promising improvements in drug biodistribution;
- iv.
-
The use of specific amphiphilic substances could also contribute to the characteristic of micelles’ blood having an extended half-life;
- v.
-
Due to their small size, micelles can spontaneously permeate the interstitium with leaky vasculature due to the increased permeability and retention (EPR) effect in bodily compartments;
- vi.
-
The drug in micellar form is adequately shielded from biological surround inactivation and has no adverse side effects on tissues and non-target organs.
Micellar Nanoparticle Formulation
Micellar nanoparticles are formulations based on nanotechnology that achieve high levels of transdermal therapy and can form very fine nanoparticles reaching 10 nm [16]. The formulation provides an excellent and efficient delivery system capable of incorporating a variety of lipophilic components with varying physicochemical characteristics, like a nano-vehicle, to efficiently implement the lipophilic constituent to the skin while shielding the components from effortless decomposition caused by biological reactions within the human body [17]. In comparison to other conventional nanocarriers, such as niosomes, ethosomes, and liposomes, which face challenges such as high production costs, poor bioactive encapsulation performance, and greater particle size, this nanocarrier has emerged as among the most recent nanotechnology-based transdermal drug delivery [18]. Furthermore, one of the efficient micellar formulation properties is the capacity of the nanoemulsion system to be created as a transparent solution, reflecting tiny micelles distributed throughout the aqueous processes. The transparency diagram in Figure 3 represents the tiny micelles distributed throughout the nanoemulsion system. Saberi et al. [19] said that the tiny micelle particle size does not scatter light severely due to its size being smaller than the wavelength of light, which results in the development of a transparent solution.
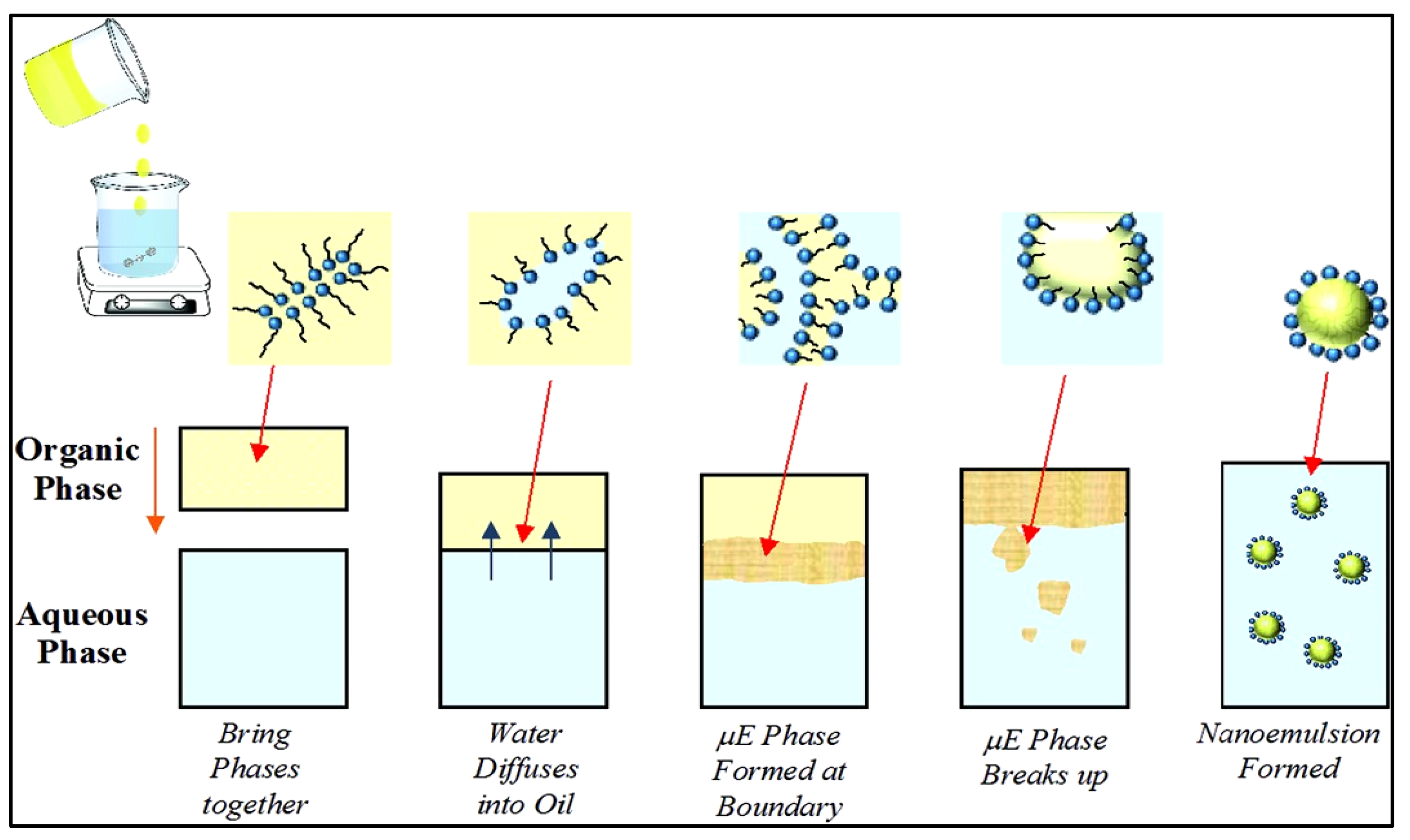
Figure 3. Mechanism of micelle formation in a nanoemulsion system utilizing the spontaneous emulsification method [20].
Surfactants play a critical role in the development of a successful, innovative micellar formulation, which is dependent on the synthesis of fine mean micelle particle diameters and the spontaneous emulsification approach. By easily reducing the interfacial stress between oil and the aqueous phase, the smaller diameters of micelles can be facilitated by surfactants. Furthermore, by dispersing one liquid into another immiscible liquid phase using low- and high-energy approaches, nanoemulsions can generate micellar nanoparticles [20].
In terms of a nanosystem’s capability to transform smaller nanoparticle sizes from 10 to 200 nm, O/W nanoemulsion has outstanding cosmetic-based properties when compared to other emulsions. Furthermore, this enables uniform distribution on the skin, a good aesthetic character, high stability, and improved active delivery properties, surface area, and skin feels [21]. To obtain the maximum efficacy of cosmetics, a certain penetration of the active component integrated with nanotechnology into the skin is required. This is due to the fact that the efficacy of penetration is affected by a variety of characteristics, such as lipophilicity, molecular size, and degree of ionization. An additional critical feature of nanoemulsion-dependent cosmeceuticals is the tiny micellar nanoparticle scale, which has the kinetic stability and strong thermodynamic stability of the cosmetic formulation against sedimentation, flocculation, and Ostwald ripening.
Ostwald ripening plays a critical role in facilitating the instability of O/W nanoemulsions owing to micelle dispersion from small to large droplets through a continuous phase that is known as an aqueous solution [22]. As a result, tiny micelles deform to create significant micelles, jeopardizing the cosmetic formulation’s long-term stability, which can be a major concern in the development of high-quality, as well as effective, cosmetic products [23]. The oil phase plays a crucial role as a component in the creation of micellar nanoparticles in nanoemulsions, where it is essential for the solubilization of the lipophilic active components in cosmetic formulations [24]. Only some types of oil are appropriate for developing cosmetic formulations. The oil phase often contains the lipophilic cosmetic’s bioactive ingredients, such as hydrophobic nutraceuticals, nutrients, vitamins, colors, essential oils, antioxidants or antibacterial agents, and carrier oils. Carrier oils are typically used to promote or enhance the stability of a nanoemulsion system in micellar development [21].
References
- Salvioni, L.; Morelli, L.; Ochoa, E.; Labra, M.; Fiandra, L.; Palugan, L.; Prosperi, D.; Colombo, M. The emerging role of nanotechnology in skincare. Adv. Colloid Interface Sci. 2021, 293, 102437.
- Shah, M.S.; Mirza, U.; Shubeena, S.; Gull, B.; Shafi, M.; Shaheen, S. Nanotechnology: A miniature miracle. J. Entomol. Zool. Stud. 2018, 6, 1984–1988.
- Suter, F.; Schmid, D.; Wandrey, F.; Zülli, F. Heptapeptide-loaded solid lipid nanoparticles for cosmetic anti-aging applications. Eur. J. Pharm. Biopharm. 2016, 108, 304–309.
- Otlatici, G.; Yegen, G.; Gungor, S.; Aksu, B. Overview on nanotechnology based cosmeceuticals to prevent skin aging. Istanb. J. Pharm. 2019, 48, 55–62.
- Souto, E.B.; Fernandes, A.R.; Martins-Gomes, C.; Coutinho, T.E.; Durazzo, A.; Lucarini, M.; Souto, S.B.; Silva, A.M.; Santini, A. Nanomaterials for skin delivery of cosmeceuticals and pharmaceuticals. Appl. Sci. 2020, 10, 1594.
- Miastkowska, M.; Lasoń, E.; Sikora, E.; Wolińska-Kennard, K. Preparation and characterization of water-based nano-perfumes. Nanomaterials 2018, 8, 981.
- Musazzi, U.M.; Marini, V.; Casiraghi, A.; Minghetti, P. Is the European regulatory framework sufficient to assure the safety of citizens using health products containing nanomaterials? Drug Discov. Today 2017, 22, 870–882.
- Maccelli, A.; Vitanza, L.; Imbriano, A.; Fraschetti, C.; Filippi, A.; Goldoni, P.; Maurizi, L.; Ammendolia, M.G.; Crestoni, M.E.; Fornarini, S.; et al. Essential oils: Chemical profiles/phytochemical screening, antimicrobial activity and O/W Nanoemulsion formulations. Pharmaceutics 2019, 12, 7.
- McClements, D.J.; Jafari, S.M. Chapter 1—General aspects of nanoemulsions and their formulation. In Nanoemulsions; Jafari, S.M., McClements, D.J., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 3–20.
- Singh, Y.; Meher, J.G.; Raval, K.; Khan, F.A.; Chaurasia, M.; Jain, N.K.; Chourasia, M.K. Nanoemulsion: Concepts, development and applications in drug delivery. J. Control. Release 2017, 252, 28–49.
- Hagigit, T.; Abdulrazik, M.; Valamanesh, F.; Behar-Cohen, F.; Benita, S. Ocular antisense oligonucleotide delivery by cationic nanoemulsion for improved treatment of ocular neovascularization: An in-vivo study in rats and mice. J. Control. Release 2012, 160, 225–231.
- Sutradhar, K.B.; Amin, M.L. Nanoemulsions: Increasing possibilities in drug delivery. Eur. J. Nanomed. 2013, 5, 97–110.
- Nair, H.A.; Singh Rajawat, G.; Nagarsenker, M.S. Chapter 8-Stimuli-responsive micelles: A nanoplatform for therapeutic and diagnostic applications. In Drug Targeting and Stimuli Sensitive Drug Delivery Systems; Grumezescu, A.M., Ed.; William Andrew Publishing: Norwich, NY, USA, 2018; pp. 303–342.
- Priya, L.B.; Baskaran, R.; Padma, V.V. Chapter 21-Phytonanoconjugates in oral medicine. In Nanostructures for Oral Medicine; Andronescu, E., Grumezescu, A.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 639–668.
- Joseph, M.; Trinh, H.M.; Mitra, A.K. Chapter 7—Peptide and protein-based therapeutic agents*. In Emerging Nanotechnologies for Diagnostics, Drug Delivery and Medical Devices; Mitra, A.K., Cholkar, K., Mandal, A., Eds.; Elsevier: Boston, MA, USA, 2017; pp. 145–167.
- Peng, Y.; Meng, Q.; Zhou, J.; Chen, B.; Xi, J.; Long, P.; Zhang, L.; Hou, R. Nanoemulsion delivery system of tea polyphenols enhanced the bioavailability of catechins in rats. Food. Chem. 2018, 242, 527–532.
- Lee, R.W.; Shenoy, D.B.; Sheel, R. Chapter 2-Micellar nanoparticles: Applications for topical and passive transdermal drug delivery. In Handbook of Non-Invasive Drug Delivery Systems; Kulkarni, V.S., Ed.; William Andrew Publishing: Boston, MA, USA, 2010; pp. 37–58.
- Razavi, H.; Janfaza, S. Ethosome: A nanocarrier for transdermal drug delivery. Arch. Adv. Biosci. 2015, 6, 38–43.
- Saberi, A.H.; Fang, Y.; McClements, D.J. Fabrication of vitamin E-enriched nanoemulsions: Factors affecting particle size using spontaneous emulsification. J. Colloid Interface Sci. 2013, 391, 95–102.
- Komaiko, J.S.; McClements, D.J. Formation of Food-Grade Nanoemulsions using low-energy preparation methods: A review of available methods. Compr. Rev. Food Sci. Food Saf. 2016, 15, 331–352.
- Yukuyama, M.N.; Ghisleni, D.D.M.; Pinto, T.J.A.; Bou-Chacra, N.A. Nanoemulsion: Process selection and application in cosmetics—A review. Int. J. Cosmet. Sci. 2016, 38, 13–24.
- Bilbao-Sáinz, C.; Avena-Bustillos, R.J.; Wood, D.F.; Williams, T.G.; McHugh, T.H. Nanoemulsions prepared by a low-energy emulsification method applied to edible films. J. Agric. Food Chem. 2010, 58, 11932–11938.
- Taylor, P. Ostwald ripening in emulsions. Adv. Colloid Interface Sci. 1998, 75, 107–163.
- Rai, V.K.; Mishra, N.; Yadav, K.S.; Yadav, N.P. Nanoemulsion as pharmaceutical carrier for dermal and transdermal drug delivery: Formulation development, stability issues, basic considerations and applications. J. Control. Release 2018, 270, 203–225.
More
Information
Subjects:
Engineering, Chemical
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.8K
Entry Collection:
Biopharmaceuticals Technology
Revisions:
2 times
(View History)
Update Date:
18 Nov 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





