| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Angela Nguyen | + 3008 word(s) | 3008 | 2020-11-27 06:45:03 | | | |
| 2 | Bruce Ren | Meta information modification | 3008 | 2020-12-09 03:13:20 | | |
Video Upload Options
Virtual memory T (TVM) cells are a recently described population of conventional CD8+ T cells that, in spite of their antigen inexperience, express markers of T cell activation. TVM cells exhibit rapid responsiveness to both antigen-specific and innate stimuli in youth but acquire intrinsic antigen-specific response defects in the elderly.
1. Introduction
Traditionally, CD8+ T cells have been considered to exist along a single spectrum; resting naïve CD8+ T (TN) cells, upon recognition of cognate antigen and subsequent activation, differentiate into effector T cells, which contract upon antigen clearance, leaving a conventional memory T (TMEM) cell population. The TMEM cell population is comprised largely of effector memory T cells (TEM), found predominantly in tissues and primed for rapid effector function, and central memory T (TCM) cells, found mainly in lymph nodes and responsible for self-renewal and supplying the pipeline of effector T cells [1]. TMEM cells are quiescent but poised for activation, and present at a relatively high antigen-specific frequency. These features of CD8+ T cell memory underpin their ability to respond rapidly after reencounter with the same antigen and are a hallmark of adaptive immunity. Recently, a novel population(s) of CD8+ T cells has been identified, referred to variously as virtual memory T (TVM) cells, memory phenotype (MP) T cells, antigen-inexperienced memory T (TAIM) cells, and innate memory T (TIM) cells, that exhibit many characteristics of TMEM cells - including cell surface phenotype and rapid responsiveness to both antigen-specific and innate stimuli - despite having not previously encountered specific antigen. Herein, we generally refer to this population of antigen-naïve memory phenotype CD8+ T cells as TVM cells. The interest in TVM cells stems from their ability to exert robust and rapid effector functions never previously attributed to antigen-inexperienced T cells, their responsiveness to both antigen-specific and innate stimuli, their superior survival capacity and their intrinsic dysfunction in elderly mice and humans [2][3][4][5][6]. The following review highlights how TVM cells have blurred the traditional boundaries between TN cells and TMEM cells and have driven the need for a re-evaluation of conventionally accepted TMEM characteristics. In addition, we discuss recent advances in our understanding of TVM cells, including their development as a distinct cell lineage and their biological relevance in protection from infection and cancers[7][8][9][10][11].
2. Conflation of Mouse TVM cells with Conventional TMEM cells
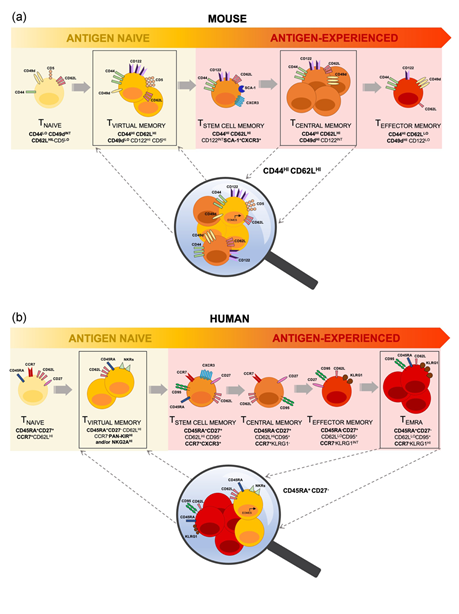
In mice, the phenotype of conventional TMEM cell populations is well established [12][13] with all antigen-experienced memory T cells expressing the definitive activation marker, CD44, and differential expression of the lymph node homing receptor, CD62L, allowing distinction between TCM and TEM subsets [14]. However, the discovery of TVM cells has revealed a substantial overlap in the cell surface phenotype of conventional TCM cells and TVM cells in mice, with both expressing high levels of CD44 and CD62L [15][16]. Consequently, TVM cells have historically fallen into the conventional phenotypic definition of antigen-experienced CD8+ TCM population (Figure 1a). This can be easily overcome by the inclusion of CD49d, an integrin involved in cell trafficking, which is stably upregulated on TMEM cells but, even with advanced age or certain infection models, remains low on TVM cells. TVM cells can also be identified by high level expression of IL-2Rβ/IL-15Rβ (CD122) compared to lower expression on TMEM cells, reflecting TVM cell sensitivity to IL-15 [17]. It has recently been demonstrated that conventionally defined TCM cells are, in both young and aged mice, comprised predominantly (~80%) of TVM cells. Even in mice recently infected with LCMV, which induces a robust CD8+ T cell memory population, over 60% of CD8+ CD44hi CD62Lhi cells (i.e. conventionally defined TCM cells) are TVM cells [18]. Findings regarding the TCM population may therefore be influenced by the inclusion of TVM cells.
Figure 1. The Differentiation Continuum and Defining Phenotype of Steady-State CD8+ T cells. CD8+ TVM cells are a semi-differentiated yet antigenically naïve T cell population, indicating the potential for antigen-independent T cell differentiation and representing a link between antigen naïve (yellow shaded) and antigen-experienced (red shaded) memory T cells. TVM cells have historically been phenotypically included within (a) the TCM cell population in mice, and (b) the TEMRA cell population in humans. Lineage-defining markers (in bold), in conjunction with other additional markers, demarcate TVM cells and reinforce their phenotypic and functional uniqueness.
Inaccurate attribution of characteristics as a consequence of the conflation of TVM and TCM cells is exemplified by our understanding of the reliance of CD8+ TMEM cells on IL-15 for survival. The current paradigm indicates that IL-15 is critical for TMEM cell survival. However, recent studies have shown that in young and aged mice lacking IL-15 there is a complete loss of TVM cells (CD44hi CD49dlo) whilst TMEM (CD44hi CD49dhi) cells are relatively unaffected[19][20]. Similarly, in mice lacking CD122, the generation and maintenance of TMEM cells remains relatively intact whilst TVM cells fail to develop [17] These findings call into question other widely accepted characteristics of TCM cells. Of particular interest is the dependence of TVM and TCM cells on tonic peptide + Major Histocompatibility Complex I molecule (MHCI)-TCR signalling for survival in the periphery. It has long been appreciated that circulating naïve CD8+ T cells require low affinity self-pMHCI:TCR interactions in order to provide tonic signals for survival [21], contrasting memory cells whose survival is independent of MHCI but dependent on homeostatic cytokines such as IL-7 and IL-15 [22]. However, the requirements for survival of TVM cells is contentious. Early adoptive transfer experiments demonstrated the survival of LCMV-specific memory phenotype (MP) cells in β2m-/- hosts [23]. However, transferred MP cells in this experiment were defined only by high expression of CD44. Expanding on this finding, later adoptive transfer experiments demonstrated that MP cells expressing high levels of CD122 (characteristic of TVM cells) were maintained in the periphery of MHC-Ia-/- mice, whilst MP cells expressing low levels of CD122 failed to survive [24]. These CD122lo MP cells expressed a cell surface phenotype reminiscent of recently activated effector CD8+ T cells, including low expression of CD62L. Similarly, inspection of the endogenous population of peripheral CD8+ T cells in MHC-I-/- hosts revealed the majority of these cells exhibited CD44hi CD122hi phenotype reminiscent of TVM cells [25]. Thus, the extent to which conventional TCM cells and TVM cells rely on MHC-I for peripheral survival is yet to be definitively determined.
3. TVM cells are Contained within the Human TEMRA cell Population
While largely characterized in mice, a putative TVM population has also been identified in humans, which displays both functional and phenotypic similarities to mouse TVM cells [26]. These human TVM cells display a differentiated phenotype typically associated with effector memory T cells re-expressing CD45RA (TEMRA) (CD45RA+CD27-), express NK cell receptors (NKRs) such as KIRs and NKG2A, show high expression of Nur77 (indicative of high self-pMHCI affinity), and show high expression of the transcription factor Eomes, with rapid production of IFNγ upon innate-like stimulation. Furthermore, human TVM cells accumulate with age and acquire defects in TCR-mediated proliferation. In addition to these parallels with mouse TVM cells, human TVM cells have been detected in human cord blood and thus their development appears to be independent of antigen exposure [26]. The limited study of human TVM cells can be attributed to the lack of definitive surface markers that distinguish them from TEMRA cells (CD8+CD45RA+CCR7-). Currently, identification of human TVM cells is based on the additional expression of NK cell markers, pan-KIR2D and KIR3DL, and/or NKG2A, which separates TVM cells from the entire CD45RA+ subset of CD8+ T cells. Non-TVM CD45RA+ cells are further subdivided into TN cells (CD27+CD45RA+CD95-), TSCM cells (CD27+CD45RA+CD95+) [27] and TEMRA cells. Owing to the overlap in surface marker expression between TEMRA and TVM cells, and the lack of routine inclusion of defining NKRs, TVM cells are typically included within the TEMRA population [28] (Figure 1b).
Despite their apparent similarities, there are key differences that distinguish TVM cells from TEMRA cells. Firstly, TVM cells are considered to be antigen-inexperienced [29] whilst TEMRA cells are antigen-experienced memory cells, as evidenced by the observation that they can comprise up to 39% of the CD8+ T cells within a given epitope-specific population [30]. Secondly, TVM cells have a higher proliferative capacity than TEMRA cells which are non-proliferative in both young and aged individuals. Thirdly, although a direct comparison between human TVM and TEMRA cell metabolism has not been performed to date, our recent work in mouse models have shown that TVM cells not only have the highest oxygen consumption rate (OCR) of all CD8+ subsets in steady state but that it is further increased with infection and ageing. In addition, our study indicates that there is no difference in basal mitochondrial characteristics, such as mitochondrial mass and number of mitochondria per cell, between TVM cells compared to other CD8+ subsets. In contrast, TEMRA cells have a lower basal OCR and extracellular acidification rate (ECAR), following overnight CD3 stimulation [31], as well as lower basal mitochondrial mass and fewer mitochondria per cell compared to conventional memory subsets [32].
The inclusion of NKRs to separate putative TVM cells from TEMRA cells in humans marks the beginning of the quest to better investigate this distinct cell population. It is clear that this putative TVM population parallels many of the functional characteristics observed in murine TVM cells, emphasising the need to identify unique and definitive markers for future studies. Indeed, a recent single cell transcriptional analysis of human memory T cells has identified novel subsets of stem-like CD8+ memory T cells, highlighting the heterogeneity that has confounded a complete understanding of memory phenotype T cells in humans.
4. Heterogeneity within the TVM cell Compartment
Broadly, two populations of antigen-inexperienced MP cells have been described – TVM cells and TIM cells. Whilst TVM cells and TIM cells were originally distinguished from one another by the thymic expression of CD49d, their dependence on IL-15 vs IL-4, and their emergence in the periphery versus the thymus, the two populations are indistinguishable once in the periphery [33][34]. It is likely that these cells represent the same population, but their original identification in different mouse strains has resulted in the attribution of distinct characteristics. TIM cells are highly abundant in BALB/c mice due to the ability of unconventional PLZF+ NKT cells in this strain to produce large amounts of IL-4, facilitating TIM differentiation [33][34][35] In contrast, TIM cells are not readily detectable in C57Bl/6 mice, however, genetic alterations in these mice, such as knockout of tyrosine kinases ITK and RLK, increases the number of IL-4 producing PLZF+ NKT cells and, in turn, increases the number of detectable TIM cells within the thymus and periphery [36][37][38][39]. Moreover, while not readily detectable in a WT C57Bl/6 mouse thymus, recent evidence indicates TVM cell differentiation is programmed during thymic development (see below) [40][41][42].
Although peripheral TVM cells in mice are readily identified using cell surface markers such as CD49d, CD44 and CD122, whether or not this population represents a homogenous population of cells or an amalgamation of disparate cell subsets is unclear. Heterogeneity within the TVM population has been suggested by a recent study which used tamoxifen-induced time stamping to analyse TVM cells generated in the neonatal period (day 1) or later in life (day 28) [43]. TVM cells generated early in life (day 1) exhibit a transcriptional profile more akin to a short-lived effector cell (SLEC), as indicated by expression of tbx21, Ifng and gzma genes, compared to those generated later in life (day 28). This heterogeneity translated to differences in functionality, with day 1 TVM cells responding more rapidly to antigen and inflammatory cues linked with increases in effector molecules such as granzyme B and IFNγ. This exaggerated effector response also translated to a greater propensity to adopt a terminally differentiated (KLRG1hi CD62Llo) phenotype 41 days post-infection.
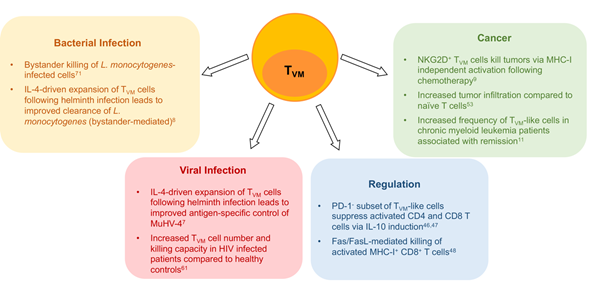
Functional heterogeneity is further observed in a subset of TVM cells that selectively express NK cell markers. NKR expression is a distinguishing feature of mouse TVM cells [44], and a defining characteristic of human TVM cells. Whilst NKR expression on memory CD8+ T cells has conventionally been associated with senescence [45], in TVM cells this subset appears to show heightened functionality, as evidenced by an increased ability to kill MHC-I deficient tumour cells following chemotherapy treatment in both humans and mice(Figure 2).
Figure 2. Virtual memory CD8+ T cells (TVM) participate in various immune responses to pathogens and tumors and may be involved in immune regulation. Boxes indicate the role and possible mechanism of action of TVM cells in different disease or immune contexts.
Adding to evidence of functional heterogeneity within the TVM population, a number of studies have indicated a regulatory role of a subset of CD8+CD44hiCD122hi cells. Early reports have suggested MP CD8+ T cells function similar to that of CD4 Treg cells via IL-10-induced suppression of effector function in activated CD4 and CD8+ cells [46] (Figure 2). Later studies revealed that only the PD-1 negative MP subset displayed regulatory functions [47]. In addition, Akane and colleagues characterised these CD8+CD44hiCD122hi Treg cells and determined they could be further defined from other CD8+ MP cells via a lack of CD49d expression, suggesting they were in fact TVM cells [48]. Taken together, these data suggest both phenotypic and functional heterogeneity within the TVM population.
5. Heightened TCR Reactivity and Cytokine Sensitivity are Key TVM cell Characteristics
TCR reactivity appears to be a key determinant in driving TVM differentiation, phenotype and effector function. Firstly, TVM cells have been shown to express heightened levels of CD5 in mice [49], and Nur77 in humans , which are surrogate markers for TCR signal strength [50][51] and thus are indicative of heightened TCR self-reactivity [52]. In addition, TCR repertoire analyses shows a TCR bias in CD8+ MP cells [53][54], further suggesting the TCR dependence of TVM differentiation. It is likely that this high self-peptide:MHCI reactivity during TVM cell development drives the heightened TVM cell cytokine sensitivity in the periphery, which can, at least in part, be attributed to Eomes expression. Eomes is a Tbox transcription factor which, in CD8+ T cells, shows increased expression following activation [55]. In a study by Miller and colleagues, it was shown that Eomes expression could be upregulated during thymic maturation of CD44hiCD122+ cells, which was attributed to heightened TCR reactivity to self-ligands [53]. Eomes expression has also been shown to bind to the il2rb promoter leading to activation and a subsequent increase in CD122 expression [56]. Thus, the heightened self-peptide MHC reactivity of TVM cells appears to upregulate Eomes expression, which in turn leads to increased CD122 expression, driving TVM cell dependence on, and sensitivity to, IL-15. This is also supported by Gett and colleagues who showed that strong TCR engagement, and subsequent signalling, enhanced survival and responsiveness to IL-15, and other cytokines, through increased expression of cytokine receptors [57].
Eomes expression in TVM cells has also been shown to be augmented by type I IFN signaling [58]. Indeed, IFNb signalling resulted in an Eomes-dependent increase of both peripheral TVM cells and thymic TIM cells, and TVM cells were significantly diminished in IFNAR-/- mice [58]. Given the observation that tonic type I IFN signalling is received by SP thymocytes as a normal part of T cell development [59], it seems plausible that type I IFN signalling is essential both in the thymus for TVM lineage differentiation at this SP stage [53], as well as in the periphery for the peripheral maintenance of Eomes expression.
Although a characteristic of memory cell subsets in general, TVM cells are particularly sensitive to a range of homeostatic cytokines, such as IL-12, IL-18, IL-4, and IL-7 [60][61][62]. As mentioned, their particularly high sensitivity to IL-15 is likely to be due to increased expression of CD122 , which increases further with age, and leads to a downstream increase in STAT5 phosphorylation following stimulation with IL-15, compared to TMEM cells [63][64][65]. Although the selective impact of cytokines on TVM cells may correspond in part to changes in cytokine receptor expression, age-related changes in TVM cell frequency and function may also be explained by an increase in the levels of these cytokines with aging. For example, there is evidence for elevated IL-15, IL-6, IL-18 and TNF cytokine levels with advanced age, as part of the ‘inflammaging’ process [66][67][68][69]. Outside of IL-15, TVM cells from both mice and humans can be directly activated by other cytokines. Previous in vitro studies of TVM cells have shown that IFNγ production in these cells can be driven by IL-12 and IL-18 stimulation and result in an antigen-independent acquisition of cytotoxic capacity.
The role of cytokines in mediating the expansion and effector function of TVM cells is further reinforced in vivo in the context of infection. Baez and colleagues demonstrated that mice infected with Trypanosoma cruzi showed enhanced expansion of CD44hi CD8+ T cells, owing to increased levels of thymic IL-15 and IL-4, which ultimately promoted antigen-independent proliferation and subsequent protection from parasitemia by this population [70] (Figure 2). Furthermore, the ability of CD44hiNKG2D+ CD8+ T cells to directly kill Listeria monocytogenes-infected target cells occurred independently of strong TCR signalling, but was instead NKG2D-dependent and promoted by direct cytokine exposure (IL-12, IL-15 and IL-18) [71] (Figure 2). This innate-like response was required for effective bacterial clearance during the acute stages of infection [71]. In the context of helminth infections, studies have shown that the robust IL-4 production following infection of B6 or BALB/c mice drives antigen-independent TVM cell expansion, which in turn offered significant protection following subsequent viral or bacterial infections, via either innate or antigen-specific mechanisms [70].
Given the heightened cytokine sensitivity of TVM cells, it will be of interest to determine whether changes in the cytokine milieu associated with infections over a life course, or that occur as a natural part of the aging process (‘inflammaging’), are responsible for changes in TVM cell number and function with age. In this way, it seems possible that the same cytokine responsiveness that may impart the rapid responses and semi-differentiated phenotype in TVM cells from young mice and humans, might also be responsible for the acquisition of a senescent phenotype in advanced age.
References
- Sallusto, F.; Lenig, D.; Förster, R.; Lipp, M.; Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 1999, 401, 708–712, doi:10.1038/44385.
- Quinn, K.M.; Fox, A.; Harland, K.L.; Russ, B.E.; Li, J.; Nguyen, T.H.O.; Loh, L.; Olshanksy, M.; Naeem, H.; Tsyganov, K.; et al. Age-Related Decline in Primary CD8+ T Cell Responses Is Associated with the Development of Senescence in Virtual Memory CD8+ T Cells. Cell Reports 2018, 23, 3512–3524, doi:10.1016/j.celrep.2018.05.057.
- Quinn, K.M.; Hussain, T.; Kraus, F.; Formosa, L.E.; Lam, W.K.; Dagley, M.J.; Saunders, E.C.; Assmus, L.M.; Wynne-Jones, E.; Loh, L.; et al. Metabolic characteristics of CD8+ T cell subsets in young and aged individuals are not predictive of functionality. Nat Commun 2020, 11, 2857, doi:10.1038/s41467-020-16633-7.
- Decman, V.; Laidlaw, B.J.; Doering, T.A.; Leng, J.; Ertl, H.C.J.; Goldstein, D.R.; Wherry, E.J. Defective CD8 T Cell Responses in Aged Mice Are Due to Quantitative and Qualitative Changes in Virus-Specific Precursors. J.I. 2012, 188, 1933–1941, doi:10.4049/jimmunol.1101098.
- Haluszczak, C.; Akue, A.D.; Hamilton, S.E.; Johnson, L.D.S.; Pujanauski, L.; Teodorovic, L.; Jameson, S.C.; Kedl, R.M. The antigen-specific CD8+ T cell repertoire in unimmunized mice includes memory phenotype cells bearing markers of homeostatic expansion. J Exp Med 2009, 206, 435–448, doi:10.1084/jem.20081829.
- Lee, J.-Y.; Hamilton, S.E.; Akue, A.D.; Hogquist, K.A.; Jameson, S.C. Virtual memory CD8 T cells display unique functional properties. Proceedings of the National Academy of Sciences 2013, 110, 13498–13503, doi:10.1073/pnas.1307572110.
- Rolot, M.; Dougall, A.M.; Chetty, A.; Javaux, J.; Chen, T.; Xiao, X.; Machiels, B.; Selkirk, M.E.; Maizels, R.M.; Hokke, C.; et al. Helminth-induced IL-4 expands bystander memory CD8 + T cells for early control of viral infection. Nature Communications 2018, 9, 4516, doi:10.1038/s41467-018-06978-5.
- Lin, J.; Mohrs, K.; Szaba, F.; Kummer, L.; Leadbetter, E.; Mohrs, M. Virtual memory CD8 T cells expanded by helminth infection confer broad protection against bacterial infection. Mucosal Immunol 2019, 12, 258–264, doi:10.1038/s41385-018-0100-x.
- Wang, X.; Waschke, B.C.; Woolaver, R.A.; Chen, S.M.Y.; Chen, Z.; Wang, J.H. MHC class I-independent activation of virtual memory CD8 T cells induced by chemotherapeutic agent-treated cancer cells. Cell Mol Immunol 2020, doi:10.1038/s41423-020-0463-2.
- Rufer, N.; Brümmendorf, T.H.; Kolvraa, S.; Bischoff, C.; Christensen, K.; Wadsworth, L.; Schulzer, M.; Lansdorp, P.M. Telomere Fluorescence Measurements in Granulocytes and T Lymphocyte Subsets Point to a High Turnover of Hematopoietic Stem Cells and Memory T Cells in Early Childhood. The Journal of Experimental Medicine 1999, 190, 157–168, doi:10.1084/jem.190.2.157.
- Cayssials, E.; Jacomet, F.; Piccirilli, N.; Lefèvre, L.; Roy, L.; Guilhot, F.; Chomel, J.-C.; Leleu, X.; Gombert, J.-M.; Herbelin, A.; et al. Sustained treatment-free remission in chronic myeloid leukaemia is associated with an increased frequency of innate CD8(+) T-cells. British Journal of Haematology 2019, 186, 54–59, doi:https://doi.org/10.1111/bjh.15858.
- Kaech, S.M.; Ahmed, R. Memory CD8+ T cell differentiation: initial antigen encounter triggers a developmental program in naïve cells. Nat Immunol 2001, 2, 415–422, doi:10.1038/87720.
- Wherry, E.J.; Ahmed, R. Memory CD8 T-Cell Differentiation during Viral Infection. Journal of Virology 2004, 78, 5535–5545, doi:10.1128/JVI.78.11.5535-5545.2004.
- Harty, J.T.; Badovinac, V.P. Shaping and reshaping CD8 + T-cell memory. Nature Reviews Immunology 2008, 8, 107–119, doi:10.1038/nri2251.
- Kieper, W.C.; Jameson, S.C. Homeostatic expansion and phenotypic conversion of naïve T cells in response to self peptide/MHC ligands. PNAS 1999, 96, 13306–13311, doi:10.1073/pnas.96.23.13306.
- Goldrath, A.W.; Bogatzki, L.Y.; Bevan, M.J. Naive T cells transiently acquire a memory-like phenotype during homeostasis-driven proliferation. J. Exp. Med. 2000, 192, 557–564, doi:10.1084/jem.192.4.557.
- Sosinowski, T.; White, J.T.; Cross, E.W.; Haluszczak, C.; Marrack, P.; Gapin, L.; Kedl, R.M. CD8α + Dendritic Cell Trans Presentation of IL-15 to Naive CD8 + T Cells Produces Antigen-Inexperienced T Cells in the Periphery with Memory Phenotype and Function. J.I. 2013, 190, 1936–1947, doi:10.4049/jimmunol.1203149.
- Chiu, B.-C.; Martin, B.E.; Stolberg, V.R.; Chensue, S.W. Cutting Edge: Central Memory CD8 T Cells in Aged Mice Are Virtual Memory Cells. J.I. 2013, 191, 5793–5796, doi:10.4049/jimmunol.1302509.
- Judge, A.D.; Zhang, X.; Fujii, H.; Surh, C.D.; Sprent, J. Interleukin 15 Controls both Proliferation and Survival of a Subset of Memory-Phenotype CD8+ T Cells. Journal of Experimental Medicine 2002, 196, 935–946, doi:10.1084/jem.20020772.
- White, J.T.; Cross, E.W.; Burchill, M.A.; Danhorn, T.; McCarter, M.D.; Rosen, H.R.; O’Connor, B.; Kedl, R.M. Virtual memory T cells develop and mediate bystander protective immunity in an IL-15-dependent manner. Nat Commun 2016, 7, 11291–11291, doi:10.1038/ncomms11291.
- Jameson, S.C. Maintaining the norm: T-cell homeostasis. Nature Reviews Immunology 2002, 2, 547–556, doi:10.1038/nri853.
- Surh, C.D.; Sprent, J. Homeostasis of Naive and Memory T Cells. Immunity 2008, 29, 848–862, doi:10.1016/j.immuni.2008.11.002.
- Murali-Krishna, K.; Lau, L.L.; Sambhara, S.; Lemonnier, F.; Altman, J.; Ahmed, R. Persistence of Memory CD8 T Cells in MHC Class I-Deficient Mice. Science 1999, 286, 1377–1381, doi:10.1126/science.286.5443.1377.
- Boyman, O.; Cho, J.-H.; Tan, J.T.; Surh, C.D.; Sprent, J. A major histocompatibility complex class I–dependent subset of memory phenotype CD8+ cells. J Exp Med 2006, 203, 1817–1825, doi:10.1084/jem.20052495.
- Kurepa, Z.; Su, J.; Forman, J. Memory Phenotype of CD8+ T Cells in MHC Class Ia-Deficient Mice. The Journal of Immunology 2003, 170, 5414–5420, doi:10.4049/jimmunol.170.11.5414.
- Jacomet, F.; Cayssials, E.; Basbous, S.; Levescot, A.; Piccirilli, N.; Desmier, D.; Robin, A.; Barra, A.; Giraud, C.; Guilhot, F.; et al. Evidence for eomesodermin-expressing innate-like CD8(+) KIR/NKG2A(+) T cells in human adults and cord blood samples. Eur. J. Immunol. 2015, 45, 1926–1933, doi:10.1002/eji.201545539.
- Galletti, G.; De Simone, G.; Mazza, E.M.C.; Puccio, S.; Mezzanotte, C.; Bi, T.M.; Davydov, A.N.; Metsger, M.; Scamardella, E.; Alvisi, G.; et al. Two subsets of stem-like CD8 + memory T cell progenitors with distinct fate commitments in humans. Nature Immunology 2020, 1–11, doi:10.1038/s41590-020-0791-5.
- Geginat, J.; Lanzavecchia, A.; Sallusto, F. Proliferation and differentiation potential of human CD8+ memory T-cell subsets in response to antigen or homeostatic cytokines. Blood 2003, 101, 4260–4266, doi:10.1182/blood-2002-11-3577.
- White, J.T.; Cross, E.W.; Kedl, R.M. Antigen-inexperienced memory CD8+ T cells: where they come from and why we need them. Nat Rev Immunol 2017, 17, 391–400, doi:10.1038/nri.2017.34.
- Appay, V.; Dunbar, P.R.; Callan, M.; Klenerman, P.; Gillespie, G.M.A.; Papagno, L.; Ogg, G.S.; King, A.; Lechner, F.; Spina, C.A.; et al. Memory CD8 + T cells vary in differentiation phenotype in different persistent virus infections. Nature Medicine 2002, 8, 379–385, doi:10.1038/nm0402-379.
- Henson, S.M.; Lanna, A.; Riddell, N.E.; Franzese, O.; Macaulay, R.; Griffiths, S.J.; Puleston, D.J.; Watson, A.S.; Simon, A.K.; Tooze, S.A.; et al. p38 signaling inhibits mTORC1-independent autophagy in senescent human CD8+ T cells. J. Clin. Invest. 2014, 124, 4004–4016, doi:10.1172/JCI75051.
- Zinser, M.E.; Highton, A.J.; Kurioka, A.; Kronsteiner, B.; Hagel, J.; Leng, T.; Marchi, E.; Phetsouphanh, C.; Willberg, C.B.; Dunachie, S.J.; et al. Human MAIT cells show metabolic quiescence with rapid glucose-dependent upregulation of granzyme B upon stimulation. Immunology & Cell Biology 2018, 96, 666–674, doi:10.1111/imcb.12020.
- Tripathi, P.; Morris, S.C.; Perkins, C.; Sholl, A.; Finkelman, F.D.; Hildeman, D.A. IL-4 and IL-15 promotion of virtual memory CD8+ T cells is determined by genetic background. European Journal of Immunology 2016, 46, 2333–2339, doi:10.1002/eji.201646404.
- Lee, Y.J.; Holzapfel, K.L.; Zhu, J.; Jameson, S.C.; Hogquist, K.A. Steady-state production of IL-4 modulates immunity in mouse strains and is determined by lineage diversity of iNKT cells. Nat. Immunol. 2013, 14, 1146–1154, doi:10.1038/ni.2731.
- Lai, D.; Zhu, J.; Wang, T.; Hu-Li, J.; Terabe, M.; Berzofsky, J.A.; Clayberger, C.; Krensky, A.M. KLF13 sustains thymic memory-like CD8+ T cells in BALB/c mice by regulating IL-4–generating invariant natural killer T cells. J Exp Med 2011, 208, 1093–1103, doi:10.1084/jem.20101527.
- Jameson, S.C.; Lee, Y.J.; Hogquist, K.A. Innate Memory T cells. Adv Immunol 2015, 126, 173–213, doi:10.1016/bs.ai.2014.12.001.
- Atherly, L.O.; Lucas, J.A.; Felices, M.; Yin, C.C.; Reiner, S.L.; Berg, L.J. The Tec family tyrosine kinases Itk and Rlk regulate the development of conventional CD8+ T cells. Immunity 2006, 25, 79–91, doi:10.1016/j.immuni.2006.05.012.
- Broussard, C.; Fleischacker, C.; Fleischecker, C.; Horai, R.; Chetana, M.; Venegas, A.M.; Sharp, L.L.; Hedrick, S.M.; Fowlkes, B.J.; Schwartzberg, P.L. Altered development of CD8+ T cell lineages in mice deficient for the Tec kinases Itk and Rlk. Immunity 2006, 25, 93–104, doi:10.1016/j.immuni.2006.05.011.
- Weinreich, M.A.; Odumade, O.A.; Jameson, S.C.; Hogquist, K.A. T cells expressing the transcription factor PLZF regulate the development of memory-like CD8+ T cells. Nat. Immunol. 2010, 11, 709–716, doi:10.1038/ni.1898.
- Hirose, S.; Touma, M.; Go, R.; Katsuragi, Y.; Sakuraba, Y.; Gondo, Y.; Abe, M.; Sakimura, K.; Mishima, Y.; Kominami, R. Bcl11b prevents the intrathymic development of innate CD8 T cells in a cell intrinsic manner. Int Immunol 2015, 27, 205–215, doi:10.1093/intimm/dxu104.
- Kastner, P.; Chan, S.; Vogel, W.K.; Zhang, L.-J.; Topark-Ngarm, A.; Golonzhka, O.; Jost, B.; Le Gras, S.; Gross, M.K.; Leid, M. Bcl11b represses a mature T-cell gene expression program in immature CD4+CD8+ thymocytes. Eur J Immunol 2010, 40, 2143–2154, doi:10.1002/eji.200940258.
- Hosokawa, H.; Romero-Wolf, M.; Yui, M.A.; Ungerbäck, J.; Quiloan, M.L.G.; Matsumoto, M.; Nakayama, K.I.; Tanaka, T.; Rothenberg, E.V. Bcl11b sets pro-T cell fate by site-specific cofactor recruitment and by repressing Id2 and Zbtb16. Nature Immunology 2018, 19, 1427–1440, doi:10.1038/s41590-018-0238-4.
- Smith, N.L.; Patel, R.K.; Reynaldi, A.; Grenier, J.K.; Wang, J.; Watson, N.B.; Nzingha, K.; Yee Mon, K.J.; Peng, S.A.; Grimson, A.; et al. Developmental Origin Governs CD8+ T Cell Fate Decisions during Infection. Cell 2018, 174, 117-130.e14, doi:10.1016/j.cell.2018.05.029.
- Hussain, T.; Quinn, K.M. Similar but different: virtual memory CD8 T cells as a memory‐like cell population. Immunol Cell Biol 2019, 97, 675–684, doi:10.1111/imcb.12277.
- Pereira, B.I.; De Maeyer, R.P.H.; Covre, L.P.; Nehar-Belaid, D.; Lanna, A.; Ward, S.; Marches, R.; Chambers, E.S.; Gomes, D.C.O.; Riddell, N.E.; et al. Sestrins induce natural killer function in senescent-like CD8 + T cells. Nature Immunology 2020, 21, 684–694, doi:10.1038/s41590-020-0643-3.
- Smith, T.R.F.; Kumar, V. Revival of CD8+ Treg–mediated suppression. Trends in Immunology 2008, 29, 337–342, doi:10.1016/j.it.2008.04.002.
- Dai, H.; Wan, N.; Zhang, S.; Moore, Y.; Wan, F.; Dai, Z. Cutting edge: programmed death-1 defines CD8+CD122+ T cells as regulatory versus memory T cells. J. Immunol. 2010, 185, 803–807, doi:10.4049/jimmunol.1000661.
- Akane, K.; Kojima, S.; Mak, T.W.; Shiku, H.; Suzuki, H. CD8+CD122+CD49dlow regulatory T cells maintain T-cell homeostasis by killing activated T cells via Fas/FasL-mediated cytotoxicity. PNAS 2016, 113, 2460–2465, doi:10.1073/pnas.1525098113.
- Quinn, K.M.; Zaloumis, S.G.; Cukalac, T.; Kan, W.-T.; Sng, X.Y.X.; Mirams, M.; Watson, K.A.; McCaw, J.M.; Doherty, P.C.; Thomas, P.G.; et al. Heightened self-reactivity associated with selective survival, but not expansion, of naïve virus-specific CD8+ T cells in aged mice. PNAS 2016, 113, 1333–1338, doi:10.1073/pnas.1525167113.
- Moran, A.E.; Holzapfel, K.L.; Xing, Y.; Cunningham, N.R.; Maltzman, J.S.; Punt, J.; Hogquist, K.A. T cell receptor signal strength in Treg and iNKT cell development demonstrated by a novel fluorescent reporter mouse. J Exp Med 2011, 208, 1279–1289, doi:10.1084/jem.20110308.
- Ashouri, J.F.; Weiss, A. Endogenous Nur77 Is a Specific Indicator of Antigen Receptor Signaling in Human T and B Cells. The Journal of Immunology 2017, 198, 657–668, doi:10.4049/jimmunol.1601301.
- Azzam, H.S.; Grinberg, A.; Lui, K.; Shen, H.; Shores, E.W.; Love, P.E. CD5 Expression Is Developmentally Regulated By T Cell Receptor (TCR) Signals and TCR Avidity. J Exp Med 1998, 188, 2301–2311, doi:10.1084/jem.188.12.2301.
- Miller, C.H.; Klawon, D.E.J.; Zeng, S.; Lee, V.; Socci, N.D.; Savage, P.A. Eomes identifies thymic precursors of self-specific memory-phenotype CD8 + T cells. Nature Immunology 2020, 21, 567–577, doi:10.1038/s41590-020-0653-1.
- Rudd, B.D.; Venturi, V.; Li, G.; Samadder, P.; Ertelt, J.M.; Way, S.S.; Davenport, M.P.; Nikolich-Zugich, J. Nonrandom attrition of the naive CD8+ T-cell pool with aging governed by T-cell receptor:pMHC interactions. Proceedings of the National Academy of Sciences 2011, 108, 13694–13699, doi:10.1073/pnas.1107594108.
- Pearce, E.L.; Mullen, A.C.; Martins, G.A.; Krawczyk, C.M.; Hutchins, A.S.; Zediak, V.P.; Banica, M.; DiCioccio, C.B.; Gross, D.A.; Mao, C.; et al. Control of Effector CD8+ T Cell Function by the Transcription Factor Eomesodermin. Science 2003, 302, 1041–1043.
- Intlekofer, A.M.; Takemoto, N.; Wherry, E.J.; Longworth, S.A.; Northrup, J.T.; Palanivel, V.R.; Mullen, A.C.; Gasink, C.R.; Kaech, S.M.; Miller, J.D.; et al. Effector and memory CD8+ T cell fate coupled by T-bet and eomesodermin. Nat. Immunol. 2005, 6, 1236–1244, doi:10.1038/ni1268.
- Gett, A.V.; Sallusto, F.; Lanzavecchia, A.; Geginat, J. T cell fitness determined by signal strength. Nat Immunol 2003, 4, 355–360, doi:10.1038/ni908.
- Martinet, V.; Tonon, S.; Torres, D.; Azouz, A.; Nguyen, M.; Kohler, A.; Flamand, V.; Mao, C.-A.; Klein, W.H.; Leo, O.; et al. Type I interferons regulate eomesodermin expression and the development of unconventional memory CD8+ T cells. Nat Commun 2015, 6, doi:10.1038/ncomms8089.
- Xing, Y.; Wang, X.; Jameson, S.C.; Hogquist, K.A. Late stages of T cell maturation in the thymus involve NF-κB and tonic type I interferon signaling. Nature Immunology 2016, 17, 565–573, doi:10.1038/ni.3419.
- Akue, A.D.; Lee, J.-Y.; Jameson, S.C. Derivation and maintenance of virtual memory CD8 T cells. J Immunol 2012, 188, 2516–2523, doi:10.4049/jimmunol.1102213.
- Jin, J.-H.; Huang, H.-H.; Zhou, M.-J.; Li, J.; Hu, W.; Huang, L.; Xu, Z.; Tu, B.; Yang, G.; Shi, M.; et al. Virtual memory CD8+ T cells restrain the viral reservoir in HIV-1-infected patients with antiretroviral therapy through derepressing KIR-mediated inhibition. Cellular & Molecular Immunology 2020, 1–9, doi:10.1038/s41423-020-0408-9.
- Tan, J.T.; Ernst, B.; Kieper, W.C.; LeRoy, E.; Sprent, J.; Surh, C.D. Interleukin (IL)-15 and IL-7 jointly regulate homeostatic proliferation of memory phenotype CD8+ cells but are not required for memory phenotype CD4+ cells. The Journal of Experimental Medicine 2002, 195, 1523–1532, doi:10.1084/jem.20020066.
- Berg, R.E.; Crossley, E.; Murray, S.; Forman, J. Memory CD8+ T Cells Provide Innate Immune Protection against Listeria monocytogenes in the Absence of Cognate Antigen. J Exp Med 2003, 198, 1583–1593, doi:10.1084/jem.20031051.
- Liu, K.; Catalfamo, M.; Li, Y.; Henkart, P.A.; Weng, N. IL-15 mimics T cell receptor crosslinking in the induction of cellular proliferation, gene expression, and cytotoxicity in CD8+ memory T cells. PNAS 2002, 99, 6192–6197, doi:10.1073/pnas.092675799.
- Zhang, X.; Sun, S.; Hwang, I.; Tough, D.F.; Sprent, J. Potent and selective stimulation of memory-phenotype CD8+ T cells in vivo by IL-15. Immunity 1998, 8, 591–599, doi:10.1016/s1074-7613(00)80564-6.
- Pangrazzi, L.; Naismith, E.; Meryk, A.; Keller, M.; Jenewein, B.; Trieb, K.; Grubeck-Loebenstein, B. Increased IL-15 Production and Accumulation of Highly Differentiated CD8+ Effector/Memory T Cells in the Bone Marrow of Persons with Cytomegalovirus. Front Immunol 2017, 8, doi:10.3389/fimmu.2017.00715.
- Ershler, W.B.; Keller, E.T. Age-Associated Increased Interleukin-6 Gene Expression, Late-Life Diseases, and Frailty. Annu. Rev. Med. 2000, 51, 245–270, doi:10.1146/annurev.med.51.1.245.
- Ferrucci, L.; Corsi, A.; Lauretani, F.; Bandinelli, S.; Bartali, B.; Taub, D.D.; Guralnik, J.M.; Longo, D.L. The origins of age-related proinflammatory state. Blood 2005, 105, 2294–2299, doi:10.1182/blood-2004-07-2599.
- Bruunsgaard, H.; Skinhøj, P.; Pedersen, A.N.; Schroll, M.; Pedersen, B.K. Ageing, tumour necrosis factor-alpha (TNF-α) and atherosclerosis. Clin Exp Immunol 2000, 121, 255–260, doi:10.1046/j.1365-2249.2000.01281.x.
- Baez, N.S.; Cerbán, F.; Savid-Frontera, C.; Hodge, D.L.; Tosello, J.; Acosta-Rodriguez, E.; Almada, L.; Gruppi, A.; Viano, M.E.; Young, H.A.; et al. Thymic expression of IL-4 and IL-15 after systemic inflammatory or infectious Th1 disease processes induce the acquisition of “innate” characteristics during CD8+ T cell development. PLOS Pathogens 2019, 15, e1007456, doi:10.1371/journal.ppat.1007456.
- Chu, T.; Tyznik, A.J.; Roepke, S.; Berkley, A.M.; Woodward-Davis, A.; Pattacini, L.; Bevan, M.J.; Zehn, D.; Prlic, M. Bystander-activated memory CD8 T cells control early pathogen load in an innate-like, NKG2D-dependent manner. Cell Rep 2013, 3, 701–708, doi:10.1016/j.celrep.2013.02.020.