
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sirius Huang | -- | 1601 | 2022-11-10 01:36:37 |
Video Upload Options
A cigarette filter is a component of a cigarette, along with cigarette paper, capsules and adhesives. It does not make cigarettes less unhealthy. The filter may be made from cellulose acetate fibre, paper or activated charcoal (either as a cavity filter or embedded into the cellulose acetate). Macroporous phenol-formaldehyde resins and asbestos have also been used in cigarette filters. The acetate and paper modify the particulate smoke phase by particle retention (filtration), and finely divided carbon modifies the gaseous phase (adsorption). Filters can reduce "tar" and nicotine smoke yields up to 50%, with a greater removal rate for other classes of compounds (e.g., phenols), but are ineffective in filtering toxins such as carbon monoxide. Most of these measured reductions occur only when the cigarette is smoked on a smoking machine; when a human smokes them, deliveries remain similar with or without a filter. Most factory-made cigarettes are equipped with a filter; those who roll their own can buy them from a tobacconist. The near-universal adoption of filters on cigarettes has not reduced harms to smokers; for instance, lung cancer rates have not declined. Filling a given length of cigarette with filter is cheaper than filling it with tobacco.
1. History
In 1925, Hungarian inventor Boris Aivaz patented the process of making a cigarette filter from crepe paper.[1]
From 1935, a United Kingdom company began to develop a machine that made cigarettes incorporating the tipped filter. It was considered a specialty item until 1954, when manufacturers introduced the machine more broadly, following a spate of speculative announcements from doctors and researchers concerning a possible link between lung diseases and smoking. Since filtered cigarettes were considered "safer", by the 1960s, they dominated the market. Production of filter cigarettes rose from 0.5 percent in 1950 to 87.7 percent by 1975.[2]
Between the 1930s and the 1950s, most cigarettes were 70 mm (~2 3/4 in) long. The modern cigarette market includes mainly filter cigarettes that are 80 mm (in boxes; ~3 1/8 in), 85 mm (in softpack; ~3 3/8 in), 100 mm (~3 15/16 in), or even 120 mm (~4 3/4 in) long.[3]
Cigarettes filters were originally made of cork and used to prevent tobacco flakes from getting on the smokers' tongue. Many are still patterned to look like cork.[4]
2. Manufacture
— Claude Teague, the inventor of the colour-changing filter[5]

Cellulose acetate is made by esterifying bleached cotton or wood pulp with acetic acid. Of the three cellulose hydroxy groups available for esterification, between two and three are esterified by controlling the amount of acid (degree of substitution (DS) 2.35-2.55). The ester is spun into fibers and formed into bundles called filter tow. Flavors (menthol), sweeteners, softeners (triacetin), flame retardants (sodium tungstate), breakable capsules releasing flavors on demand, and additives colouring the tobacco smoke may be added to cigarette filters.[6][7] The five largest manufactures of filter tow are Hoechst-Celanese and Eastman Chemicals in the United States, Rhodia Acetow in Germany, Daicel and Mitsubishi Rayon in Japan.
Starch glues or emulsion-based adhesives are used for gluing cigarette seams. Hot-melt and emulsion-based adhesives are used for filter seams. Emulsion-based adhesives are used for bonding the filters to the cigarettes.[8] The tip paper may be coated with polyvinyl alcohol.[9]
3. Health Risks
In the 1970s epidemiologic evidence relative to tobacco-related cancers and data for coronary heart disease indicated a reduced risk among filter smokers for these diseases.[10] Between 1970 and 1980 some studies showed a 20-50% reduction in risk of lung cancer for long-term smokers of filtered cigarettes as compared to smokers of non-filtered cigarettes (IARC, 1986) but later studies indicated a similar risk for lung cancer in smokers of filtered and non-filtered cigarettes.[11]
Various add-on cigarette filters ("Water Pik", "Venturi", "David Ross") are sold as stop-smoking or tar-reduction devices. The idea is that filters reduce tar-nicotine levels permitting the smoker to be weaned away from cigarettes.[12]
3.1. Light Cigarettes
The tobacco industry has reduced tar and nicotine yields in cigarette smoke since the 1960s. This has been achieved in a variety of ways including use of selected strains of tobacco plant, changes in agricultural and curing procedures, use of reconstituted sheets (reprocessed tobacco leaf wastes), incorporation of tobacco stalks, reduction of the amount of tobacco needed to fill a cigarette by expanding it (like puffed wheat) to increase its "filling power", and by the use of filters and high-porosity wrapping papers. However, just as a drinker tends to drink a larger volume of beer than of wine or spirits, so many smokers tend to modify their smoking pattern inversely according to the strength of the cigarette being smoked. In contrast to the standardized puffing of the smoking machines on which the tar and nicotine yields are based, when a smoker switches to a low-tar, low nicotine cigarette, they smoke more cigarettes, take more puffs and inhale more deeply. Conversely, when smoking a high-tar, high-nicotine cigarette there is a tendency to smoke and inhale less.[13]

In spite of the changes in cigarette design and manufacturing over the last 50 years, the use of filters and "light" cigarettes neither decreased the nicotine intake per cigarette, nor lowered the incidence of lung cancers (NCI, 2001; IARC 83, 2004; U.S. Surgeon General, 2004).[14] The shift over the years from higher- to lower-yield cigarettes may explain the change in the pathology of lung cancer. That is, the percentage of lung cancers that are adenocarcinomas has increased, while the percentage of squamous cell cancers has decreased. The change in tumor type is believed to reflect the higher nitrosamine delivery of lower-yield cigarettes and the increased depth or volume of inhalation of lower-yield cigarettes to compensate for lower level concentrations of nicotine in the smoke.[15]
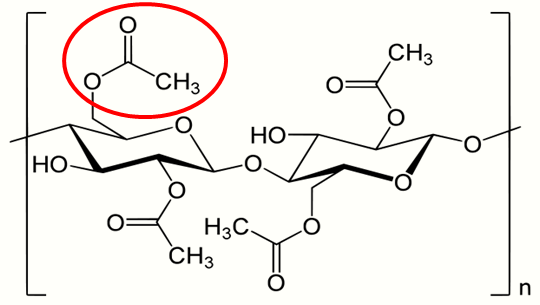
4. Safety
Cellulose acetate is non-toxic, odorless, tasteless, and weakly flammable. It is resistant to weak acids and is largely stable to mineral and fatty oils as well as petroleum. It is biodegradable and the raw material is a renewable natural polymer expected to find application for other uses in the future.[16] Smoked cigarette butts contain 5–7 mg (~ 0.08-0.11 gr) nicotine (about 25% of the total cigarette nicotine content), children ingesting >2 whole cigarettes, 6 cigarette butts or a total of 0.5 mg/kg (~ 0,0035 gr/lbs) of nicotine should be admitted to a hospital.[17] Cellulose acetate is hydrophilic and retains the water-soluble smoke constituents, of which many are irritating (acids, alkali, aldehydes, and phenols), while letting through the lipophilic aromatic compounds.
Smoked cigarette butts and cigarette tobacco are toxic to water organisms such as the marine topsmelt (Atherinops affinis) and the freshwater fathead minnow (Pimephales promelas).[18]
Atmospheric moisture, gastric acid, light, and enzymes hydrolyze cellulose acetate to acetic acid and cellulose. Cellulose may be further hydrolyzed to cellobiose or glucose in an acidic medium, and eventually form valuable humus. Humans cannot digest cellulose and excrete the fibers in feces, because, unlike ruminant animals, rabbits, rodents, termites, and some bacteria and fungi, they lack cellulolytic enzymes such as cellulase. Like wood, paper and cotton, cigarette filters in soil rot slowly over months to years.
5. Litter
Cigarette butts are the most common form of anthropogenic (man-made) litter in the world, as approximately 5.6 trillion cigarettes are smoked every year worldwide.[19] Of those it is estimated that 4.5 trillion cigarette butts become litter every year.[20] The cellulose acetate fibers used as the predominant filter material do not readily biodegrade because of the acetyl groups on the cellulose backbone which in itself can quickly be degraded by various microorganisms employing cellulases.[21] A normal life span of a discarded filter is thought to be up to 15 years.[22]
Many governments have sanctioned stiff penalties for littering of cigarette filters; for example Washington (state) state imposes a penalty of $1,025 for littering cigarette filters.[23] Another option is developing better biodegradable filters. Much of this work relies heavily on the research about the secondary mechanism for photodegradation as stated above. The next option is using cigarette packs with a compartment for discarded cigarette butts, implementing monetary deposits on filters, increasing the availability of butt receptacles, and expanding public education. It may even be possible to ban the sale of filtered cigarettes altogether on the basis of their adverse environmental impact.[19] Recent research has been put into finding ways to utilizes the filter waste, to develop a desired product. One research group in South Korea have developed a simple one-step process that converts the cellulose acetate in discarded cigarette filters into a high-performing material that could be integrated into computers, handheld devices, electrical vehicle and wind turbines to store energy. These materials have demonstrated superior performance as compared to commercially available carbon, graphene and carbon nano tubes. The product is showing high promise as a green alternative for the waste problem.[24] Another group of researchers has proposed adding tablets of food grade acid inside the filters. Once wet enough the tablets will release acid that accelerates degradation to around two weeks (instead of using cellulose triacetate and besides of cigarette smoke being quite acidic).[25] A Dutch startup is training crows to recognize and pick up cigarette butts in exchange for treats.[26]
References
- "The History of Filters". tobaccoasia.com. Archived from the original on August 24, 2003. https://web.archive.org/web/20030824115139/http://www.tobaccoasia.com/news.asp?id=534. Retrieved 2008-05-18.
- Leonard M. Schuman (1977), "Patterns of Smoking Behavior", in Murray E. Jarvik; Joseph W. Cullen; Ellen R. Gritz et al., Research on Smoking Behavior, NIDA Research Monograph, 17, pp. 36–65, http://archives.drugabuse.gov/pdf/monographs/17.pdf
- Lynn T. Kozlowski (1983), "Physical Indicators of Actual Tar and Nicotine Yields of Cigarettes", in John Grabowski; Catherine S. Bell, Measurement in the Analysis and Treatment of Smoking Behavior, NIDA Research Monograph, 48, U.S. Dept. of Health and Human Services, pp. 50–61, http://archives.drugabuse.gov/pdf/monographs/48.pdf
- Kennedy, Pagan (2012-07-06). "Who Made That Cigarette Filter?". The New York Times. ISSN 0362-4331. https://www.nytimes.com/2012/07/08/magazine/who-made-that-cigarette-filter.html. Retrieved 2018-09-04.
- Harris, Bradford (2011-05-01). "The intractable cigarette ‘filter problem’". Tobacco Control 20 (Suppl 1): –10-i16. doi:10.1136/tc.2010.040113. ISSN 0964-4563. PMID 21504917. https://tobaccocontrol.bmj.com/content/20/Suppl_1/i10. Retrieved 2018-09-04.
- Ralf Christoph; Bernd Schmidt; Udo Steinberner; Wolfgang Dilla; Reetta Karinen (2007), "Glycerol", Ullmann's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, pp. 1–16, doi:10.1002/14356007.a12_477.pub2 https://dx.doi.org/10.1002%2F14356007.a12_477.pub2
- Erik Lassner; Wolf-Dieter Schubert; Eberhard Luderitz; Hans Uwe Wolf (2007), "Tungsten, Tungsten Alloys, and Tungsten Compounds", Ullmann's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, pp. 1–37, doi:10.1002/14356007.a27_229 https://dx.doi.org/10.1002%2F14356007.a27_229
- Werner Haller; Hermann Onusseit; Gerhard Gierenz; Werner Gruber; Richard D. Rich; Günter Henke; Lothar Thiele; Horst Hoffmann et al. (2007), "Adhesives", Ullmann's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, pp. 1–70, doi:10.1002/14356007.a01_221 https://dx.doi.org/10.1002%2F14356007.a01_221
- F. L. Marten (2002), "Vinyl Alcohol Polymers", Kirk-Othmer Encyclopedia of Chemical Technology (5th ed.), Wiley, p. 26, doi:10.1002/0471238961.2209142513011820.a01.pub2 https://dx.doi.org/10.1002%2F0471238961.2209142513011820.a01.pub2
- Ernst L. Wynder (1977), "Interrelationship of Smoking to Other Variables and Preventive Approaches", in Murray E. Jarvik; Joseph W. Cullen; Ellen R. Gritz et al., Research on Smoking Behavior, NIDA Research Monograph, 17, pp. 67–95, http://archives.drugabuse.gov/pdf/monographs/17.pdf
- K. Rothwell (1999), Health effects of interactions between tobacco use and exposure to other agents, Environmental Health Criteria, World Health Organization, http://www.inchem.org/documents/ehc/ehc/ehc211.htm
- Jerome L. Schwartz (1977), "Smoking Cures: Ways to Kick an Unhealthy Habit", in Murray E. Jarvik; Joseph W. Cullen; Ellen R. Gritz et al., Research on Smoking Behavior, NIDA Research Monograph, 17, pp. 308–336, http://archives.drugabuse.gov/pdf/monographs/17.pdf
- Michael A. H. Russell (1977), "Smoking Problems: An Overview", in Murray E. Jarvik; Joseph W. Cullen; Ellen R. Gritz et al., Research on Smoking Behavior, NIDA Research Monograph, 17, pp. 13–34, http://archives.drugabuse.gov/pdf/monographs/17.pdf
- Anthony J. Alberg; Jonathan M. Samet (2010), "Epidemiology of Lung Cancer", in Robert J. Mason; V. Courtney Broaddus; Thomas R. Martin et al., Murray and Nadel's Textbook of Respiratory Medicine, 1 (5th ed.), Saunders, ISBN 978-1-4160-4710-0
- Neal L. Benowitz; Paul G. Brunetta (2010), "Smoking Hazards and Cessation", in Robert J. Mason; V. Courtney Broaddus; Thomas R. Martin et al., Murray and Nadel's Textbook of Respiratory Medicine, 1 (5th ed.), Saunders, ISBN 978-1-4160-4710-0
- Klaus Balser; Lutz Hoppe; Theo Eicher; Martin Wandel; Hans-Joachim Astheimer; Hans Steinmeier (2007), "Cellulose Esters", Ullmann's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, pp. 1–45, doi:10.1002/14356007.a05_419.pub2 https://dx.doi.org/10.1002%2F14356007.a05_419.pub2
- Jerrold B. Leikin; Frank P. Paloucek, eds. (2008), Poisoning and Toxicology Handbook (4th ed.), Informa, pp. 494–496, ISBN 978-1-4200-4479-9
- "Toxicity of cigarette butts, and their chemical components, to marine and freshwater fish". Tobacco Control 20: 25–29. 2011. doi:10.1136/tc.2010.040170. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=3088407
- "Cigarettes butts and the case for an environmental policy on hazardous cigarette waste". International Journal of Environmental Research and Public Health 6: 1691–705. 2009. doi:10.3390/ijerph6051691. https://dx.doi.org/10.3390%2Fijerph6051691
- "The world litters 4.5 trillion cigarette butts a year. Can we stop this?". The Houston Chronicle. http://blog.chron.com/sciguy/2010/05/the-world-litters-4-5-trillion-cigarette-butts-a-year-can-we-stop-this/. Retrieved 2014-09-16.
- Frederic Beaudry (2017). "Are Cigarette Butts Biodegradable?". http://environment.about.com/od/pollution/fl/Are-Cigarette-Butts-Biodegradable.htm.
- Aaron Cantú (2014). "New 'Green' Cigarette Butts Biodegrade Within Days—And Can Even Sprout Into Grass - The company Greenbutts is manufacturing a new filter to address most common litter problem". alternet.org. http://www.alternet.org/environment/new-green-cigarette-butts-biodegrade-within-days-and-can-even-sprout-grass.
- "Accidents, fires: Price of littering goes beyond fines". Washington: State of Washington Department of Ecology. 2004-06-01. http://listserv.wa.gov/cgi-bin/wa?A2=ind0406A&L=ecology-news&F=&S=&P=643.
- "Preparation of energy storage material derived from a used cigarette filter for a supercapacitor electrode". IOP Science 25: 34. 2014. http://iopscience.iop.org/article/10.1088/0957-4484/25/34/345601/meta;jsessionid=6AD229C8CCE8FE4B20613E8171F79CE7.ip-10-40-2-73.
- "No more butts: biodegradable filters a step to boot litter problem". Environmental Health News. 2012-08-14. http://www.environmentalhealthnews.org/ehs/newscience/2012/06/2012-0725-biodegradable-cigarette-filters.
- Cecile Borkhataria; Cheyenne MacDonald (13 October 2017), "The bizarre project to teach CROWS to clean up city streets by collecting cigarette butts in exchange for treats", Daily Mail, http://www.dailymail.co.uk/sciencetech/article-4975664/Startup-teaches-CROWS-collecti-cigarette-butts.html





