1. Role of Non-Invasive Imaging Modalities in Portal Vein Thrombosis
1.1. Color-Doppler Ultrasonography (CDUS) and Contrast-Enhanced Ultrasound (CEUS)
Color-Doppler Ultrasonography (CDUS) is the first-line diagnostic approach when portal vein thrombosis (PVT) is suspected. It is non-invasive, low cost, bedside, repeatable, and does not expose the patient to radiation. The diagnostic sensitivity and specificity for CDUS range from 66% to 100% depending on the expertise of the individual examiner and the extent of PVT
[1]. CDUS is commonly used in the initial diagnosis and follow-up of patients with suspected or known PVT as it readily depicts occlusion of the portal vein and the presence of cavernous transformation with a sensitivity and specificity of approximately 95%
[2].
Compared to further imaging modalities, CDUS has shown to be useful for an accurate assessment of the portal venous system. Extrahepatic visceral vessels leading to portal cavernoma as well as collaterals caused by portal hypertension can be detected in a dynamic mode and with good tissue–vessel discrimination
[3].
The ultrasonographic diagnosis of PVT relies on the presence of echogenic material obstructing the lumen of the vessel and the complete or partial absence of flow in the portal vein at color flow Doppler imaging
[4]. As the chronic thrombus ages and calcifies, CDUS may show areas of increased echogenicity. In addition, the collateral veins from cavernous transformation around the segments of PVT display a serpiginous mixed-color pattern on CDUS and are anechoic on gray-scale imaging
[5][6]. The appearance of the portal cavernoma has been described as a “spongelike mass”, a distinctive tangle of tortuous vessels in the porta hepatis characterized by hepatopetal flow with loss of normal portal phasicity
[7]. However, discriminating cavernous collaterals from the portal vein in children with PVT can be very difficult.
One study
[8] showed that CDUS can play an important role in the diagnosis of PVT by evaluating indices of arterial resistance at the hepatic and splenic levels. PVT produces a buffer response that involves vasodilatation of the hepatic artery to maintain adequate hepatic perfusion. In the acute and subacute phases, this phenomenon can be documented with CDUS as decreased hepatic artery resistance. Moreover, PVT may be associated with increased arterial resistance indices at the splenic level because of the obstruction to splenic outflow
[8].
CDUS is useful in assessing the patency of the intrahepatic portal veins as well as the splenic and superior mesenteric veins, which may also be occluded in PVT, but is less reliable in determining the extension of the thrombus to the mesenteric circulation
[2][9].
In portal biliopathy CDUS provides additional information regarding the presence of gallbladder varices, observed in about 35% of patients. Gallbladder varices appear as tortuous, dilated vessels in or around the wall of the gallbladder or in the gallbladder fossa. Moreover, ultrasound can detect the presence of cholelithiasis caused by biliary obstruction secondary to portal biliopathy: gallstones are characteristically echogenic and demonstrate posterior acoustic shadowing regardless of the gallstone composition
[10].
Limitations of CDUS include the patient’s body habitus and intestinal gas interfering with the examination, specifically when evaluating PVT extension into the mesenteric and splenic veins. Additionally, ultrasound is operator-dependent, requiring an experienced sonographer
[11].
Contrast-enhanced ultrasound (CEUS) has been proposed to overcome the limitations of CDUS by improving visualization of portal vein vasculature, especially in post-LT follow-up
[12]. The European Federation of Societies for Ultrasound in Medicine and Biology (EFSUMB) published an update in 2012 which recommended CEUS for the confirmation of occlusion of the portal vein
[13].
CEUS typically shows the absence of portal vein enhancement within the thrombosed segment. Distal to the thrombosed segment, there may be enhancement of the intrahepatic portal vein branches, due to a phenomenon called “arterial backflow”. This is secondary to the physiological connections between the hepatic artery and portal vein branches at the levels of sinusoids, vasa vasorum of the portal vein, or peribiliary vascular plexuses
[14].
Despite the advantages of CEUS examination such as the non-nephrotoxicity of the ultrasound contrast agents (unlike the contrast agents used in CT and MR), the wide availability, the lesser expensiveness than other imaging methods, CEUS is still not commonly used in post-LT work-up examinations and literature on its use for the evaluation of PVT is scarce. This is probably because CEUS presents the same limitation of general Ultrasound, in particular poor standardization, and patient’s condition/operator’s skillness dependence
[12].
1.2. CT and Magnetic Resonance (MR)
CT and MR can be used to confirm the presence of chronic PVT. The thrombosed vessel may be obliterated, or it may appear attenuated with the incorporation of a clot into the wall. In the chronic form, the thrombosed vessel can be hypodense, containing linear areas of calcification within the thrombus. In addition, CT and MR can depict features of portal hypertension, including ascites, splenomegaly, and portosystemic shunts
[5][15].
Advantages of CT and MR over CDUS include the possibility of detecting the wide spectrum of PVT features, such as bowel ischemia, septic foci, intraabdominal malignancies, and higher sensitivity in the detection of thrombosis in the splenic vein and superior mesenteric vein (SMV).
1.2.1. CT
CT provides a wider field of view and is less operator dependent than CDUS. Many of the findings are easily identified on portal venous phase images, though multiphase imaging with non-contrast, arterial, portal venous, and potentially delayed phases may optimize the information obtained from the examination
[2].
A CT scan in the pre-contrast phase can show hyperattenuating material in the portal vein. After contrast injection, lack of luminal enhancement increased portal vein caliber, increased arterial hepatic enhancement, and decreased portal hepatic enhancement are typical findings of PVT
[16]. Transient hepatic attenuation differences related to altered hepatic perfusion can also be demonstrated on CT with geographic areas of increased attenuation at the periphery of the liver on arterial phase imaging. These transient attenuation differences are related to decreased portal venous perfusion and compensatory increased arterial perfusion from the periphery of the liver
[2][17].
In the chronic form, the thrombosed vessel can be hypodense, containing linear calcifications, obliterated or shrunk, with cavernous transformation
[5][15]. In candidates for LT, calcifications in the portal vein should be actively investigated, because they indicate a more fragile vessel and can hinder surgical anastomoses creation
[15]. Splenomegaly and splenic artery aneurysms are also relevant findings readily depicted by CT.
CT provides a better assessment of thrombus extension within the portal venous system than CDUS, because of its higher sensitivity and accuracy than CDUS for identifying thrombosis of the SMV and splenic vein
[18]. If the SMV is involved by thrombosis, thinning of the intestinal wall or lack of mucosal enhancement of a thickened intestinal wall after contrast injection are further pieces of evidence for intestinal infarction
[16].
Enlargement of pre-existing veins in the porta hepatis and around biliary structures is seen as early as a few days after the onset of acute PVT. This is particularly conspicuous in the gallbladder wall, which is thickened and enhanced after contrast injection, and should be carefully distinguished from acute cholecystitis
[16][19][20].
Disadvantages of CT include the use of ionizing radiation and potentially nephrotoxic contrast material which are particularly relevant in the pediatric population and in chronically affected patients, respectively. Moreover, CT provides a morphological examination of the abdominal anatomy, not giving information about the dynamic blood flow of the portal venous system, unlike CDUS examination
[11]. Thus, CDUS and CT can be considered complementary.
1.2.2. MR
MR is less operator dependent than CDUS, though more so than CT. Similar to CT, MR readily depicts typical findings of PVT including the extent of portal vein occlusion, potential occlusion of the superior mesenteric and splenic veins, and the extent of portosystemic collaterals and secondary changes in the liver. In the evaluation of portal cavernoma cholangiopathy, magnetic resonance cholangiopancreatography (MRCP) better depicts the changes in the biliary system than CT—avoiding ionizing radiation and providing high-quality images of the bile ducts and vessels—and replaces the diagnostic role of percutaneous transhepatic cholangiography and ERCP, reducing the risks of bleeding and acute pancreatitis associated with the latter
[2]. One typical finding is the ‘‘wavy’’ appearance of the biliary ducts related to extrinsic compression by the collateral vessels, which was described in 87.5% of patients in one small retrospective study
[17][21]. Other typical MR findings of the biliary system include areas of stenosis and dilatation, angulation of the common bile duct at the level of the superior pancreatic head (with an angle of ≤145 degrees considered significant)
[22], and wall thickening of the gallbladder and bile ducts. Wall thickening may be related to intramural varices, as previously described, or ischemic fibrosis
[17]. Epicholedochal collateral vessels may appear as punctate foci of intramural enhancement within the ducts on postcontrast MR images
[22]. Bile duct and gallbladder stones are also well depicted on MRCP images. MR can also show other potential complications, including cholangitis and hepatic abscesses
[17].
When the thrombus is located at the origin of the portal vein or extended to the splenic vein and/or the SMV, the intra- or peripancreatic cavernoma may form a pseudomass that encases the main bile duct and enhances during the portal phase after contrast injection. In such a case the misleading diagnosis of pancreatic tumor should be carefully avoided. MRCP coupled with MR angiography appears to be of great help in clarifying these differential diagnoses and avoiding the need for biopsy or fine-needle aspiration of a suspected mass, with higher sensitivity than CT
[23].
MR imaging provides better soft tissue resolution and does not involve ionizing radiation; additionally, the paramagnetic contrast agents used in MR imaging have a better safety profile. Similar to CT, a technical issue of MR is its incapability to show dynamic blood flow of the portal venous system in clinically available protocols and its poor spatial resolution, especially in small children
[24]; as previously mentioned for CT, CDUS imaging can be considered complementary. Major disadvantages are also that MR imaging is limited by longer acquisition times (30–60 min) and lower spatial resolution when compared with CT. Moreover, MR is considered quite invasive because it requires deep sedation or general anesthesia in younger children
[11].
2. Role of Invasive Imaging Modalities in Portal Vein Thrombosis
2.1. Retrograde Portography
Retrograde portography is an interventional radiological procedure that provides visualization of the native intrahepatic portal venous system via retrograde opacification through wedge catheterization of the hepatic veins. Through the same technique, wedge hepatic pressure measurement, which reflects portal venous pressure, is possible, though in the case of PVT with cavernous transformation it is not reliable. When wedged hepatic venous pressure is within normal limits and intrasplenic pressure is significantly elevated, a presinusoidal nature of portal hypertension is assumed. Intravariceal pressure closely represents the portal and splenic pressure
[7][25].
In children affected by extrahepatic PVT, the cure of portal hypertension with its associated complications can be achieved by surgical restoration of normal blood flow to the liver
[26]. The preferred technique is the Meso-Rex shunt, which connects the SMV with the left intrahepatic portal branch close to the fetal insertion of the umbilical vein, called the Rex recess. However, a patent intrahepatic portal venous system is an indispensable prerequisite to perform Meso-Rex shunt surgery
[24].
Retrograde portography is the gold standard imaging method to assess the intrahepatic portal venous system with high resolution: if patency of the native intrahepatic portal system is demonstrated, the surgical Meso-Rex bypass can be performed; vice versa, unnecessary operative explorations are avoided. A surgical spleno-renal (Warren) portosystemic shunt or a radiological transjugular intrahepatic portosystemic shunt (TIPS) can be proposed whenever intrahepatic vascular anatomy is compromised
[27]. The choice between the two different approaches is based on other findings such as the presence of a prevalent cavernous vessel to land with the TIPS, the distance between the splenic and the left renal vein to make a surgical shunt, and local expertise/resource availability.
Conventional non-invasive imaging (CDUS, CT, MR) is not always reliable in assessing the patency of the native intrahepatic portal venous system and/or the Rex segment of the left portal vein in patients with cavernoma. At CDUS examination differentiating intrahepatic portal venous branches from venous collaterals in patients with the impaired portal venous flow can be challenging. Similar problems are faced by MR. One study compared MR and retrograde portography for the assessment of the Rex recess in children with extrahepatic PVT before Meso-Rex shunt surgery, demonstrating that only retrograde portography was reliable
[24] (
Figure 1).
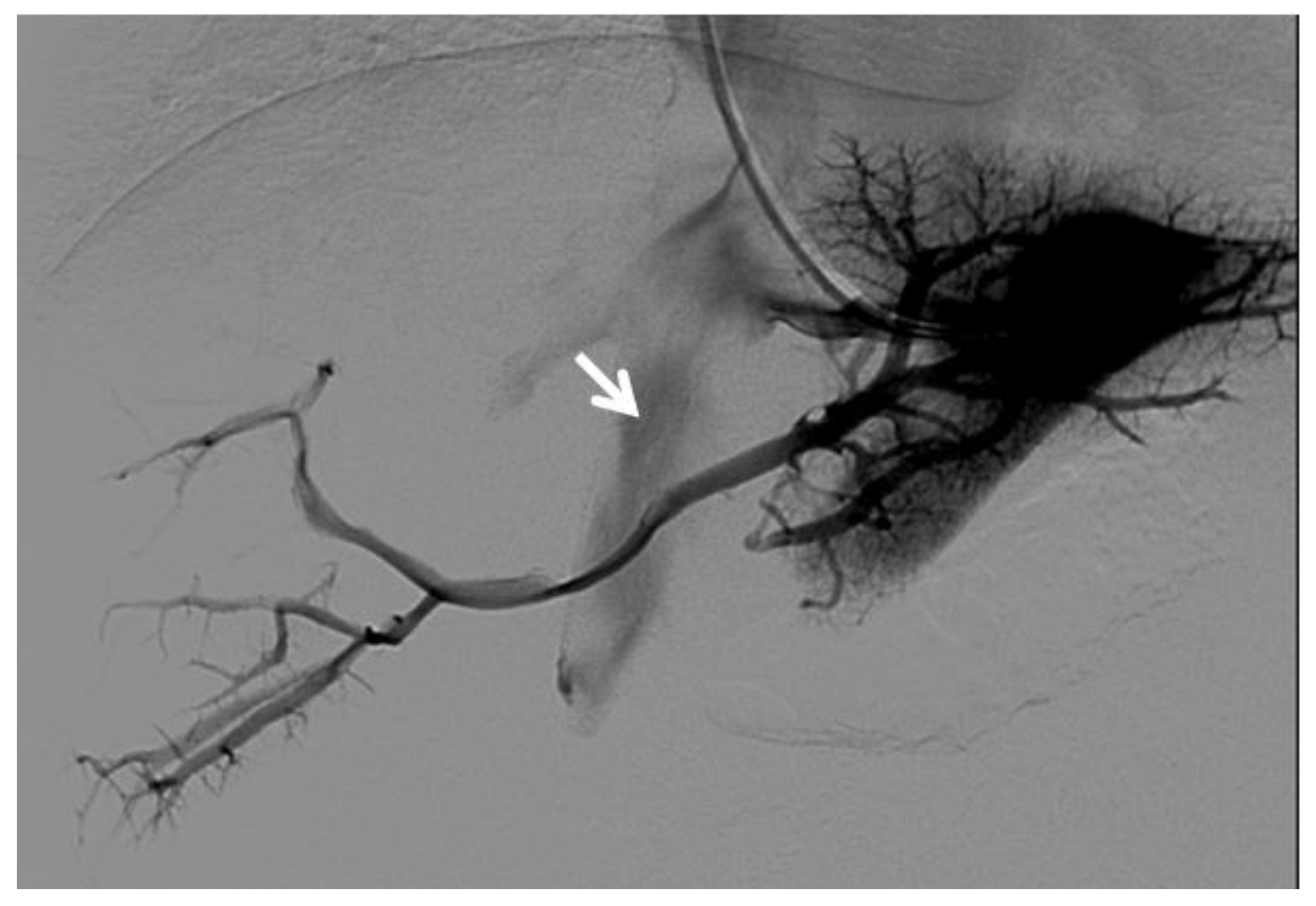
Figure 1. Right oblique anterior (30° cranial) view of a transjugular retrograde portography performed in a 9-year-old female with extrahepatic portal vein cavernoma due to a neonatal umbilical vein catheterization and portal vein thrombosis, who complained of symptoms of portal hypertension. She was a candidate for Meso-Rex bypass surgery. The image shows retrograde filling of the intrahepatic portal vessels with the complete visualization of the Rex segment (arrow) after wedged left hepatic venography. Meso-Rex bypass was then surgically performed.
Major concerns about transjugular retrograde portography in pediatric patients involve invasiveness, with radiation exposure and iodinated contrast agent administration
[24].
Postoperative imaging assessment of the Meso-Rex shunt is treated in a dedicated paragraph.
2.2. Percutaneous Direct Portography
Percutaneous direct portography allows a fine assessment of the portal vein anatomy and PVT degree and extension; a functional and dynamic evaluation of the portal flow; and visualization of retrograde flow towards spontaneous portosystemic shunts
[28]. The feasibility of recanalization of PVT is difficult to predict with preoperative imaging. Thus, the diagnostic role of percutaneous direct portography is emphasized at the beginning of a percutaneous revascularization procedure
[29]. Endovascular transcatheter interventions are less invasive than surgery for the treatment of post-LT vascular complications, particularly in pediatric patients, and are considered the first-line approach for portal vein complications, with high rates of success shown by several studies
[30][31].
Percutaneous transluminal angioplasty (PTA) relies on the dilatation of the occluded portal tract with balloon catheters during percutaneous transhepatic portography and it is highly effective for PVT
[32] (
Figure 2). In some cases of late-onset PVT, the fibrosis of the thrombosed segment may be an important determinant of recurrence and PTA may require additional metallic stent placement
[33].
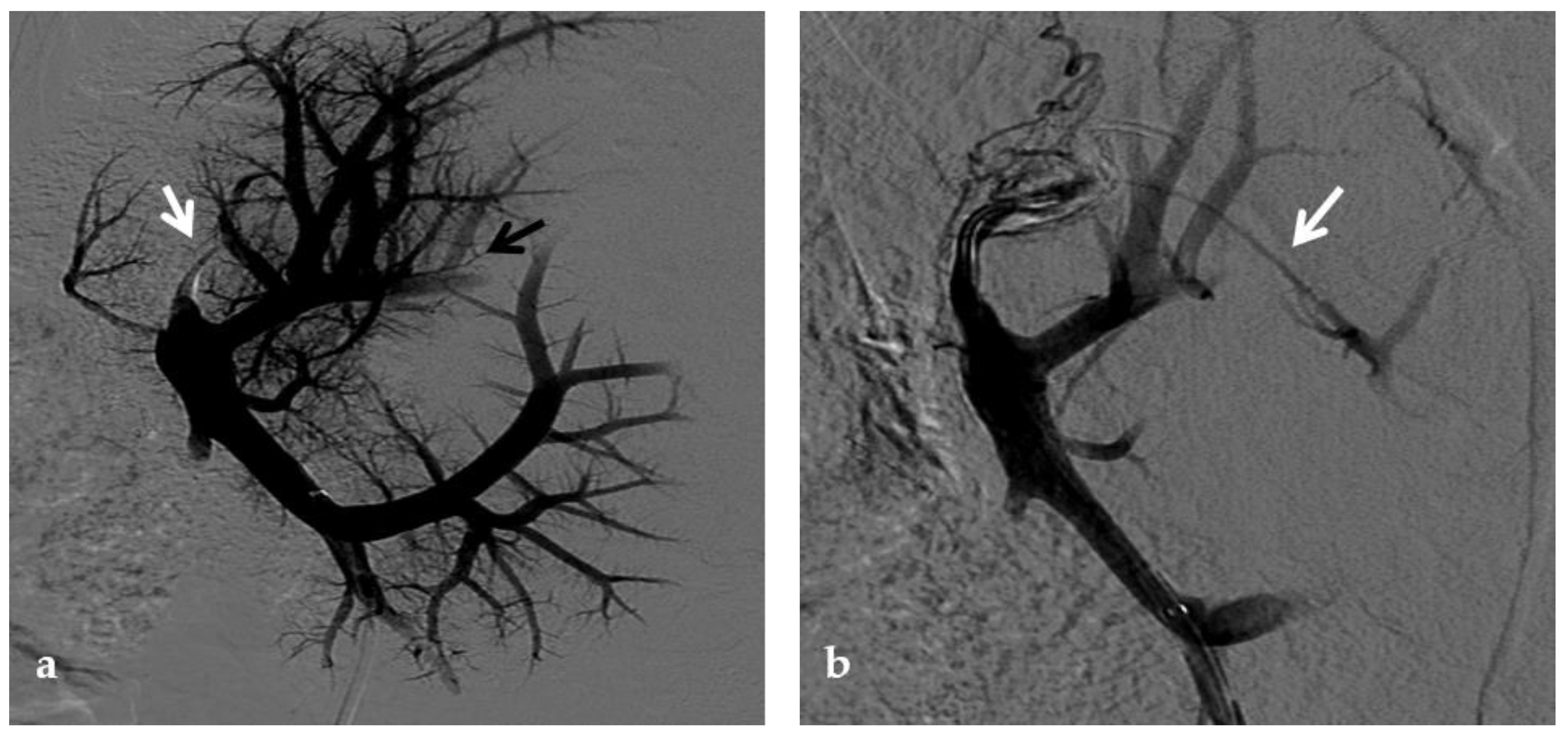
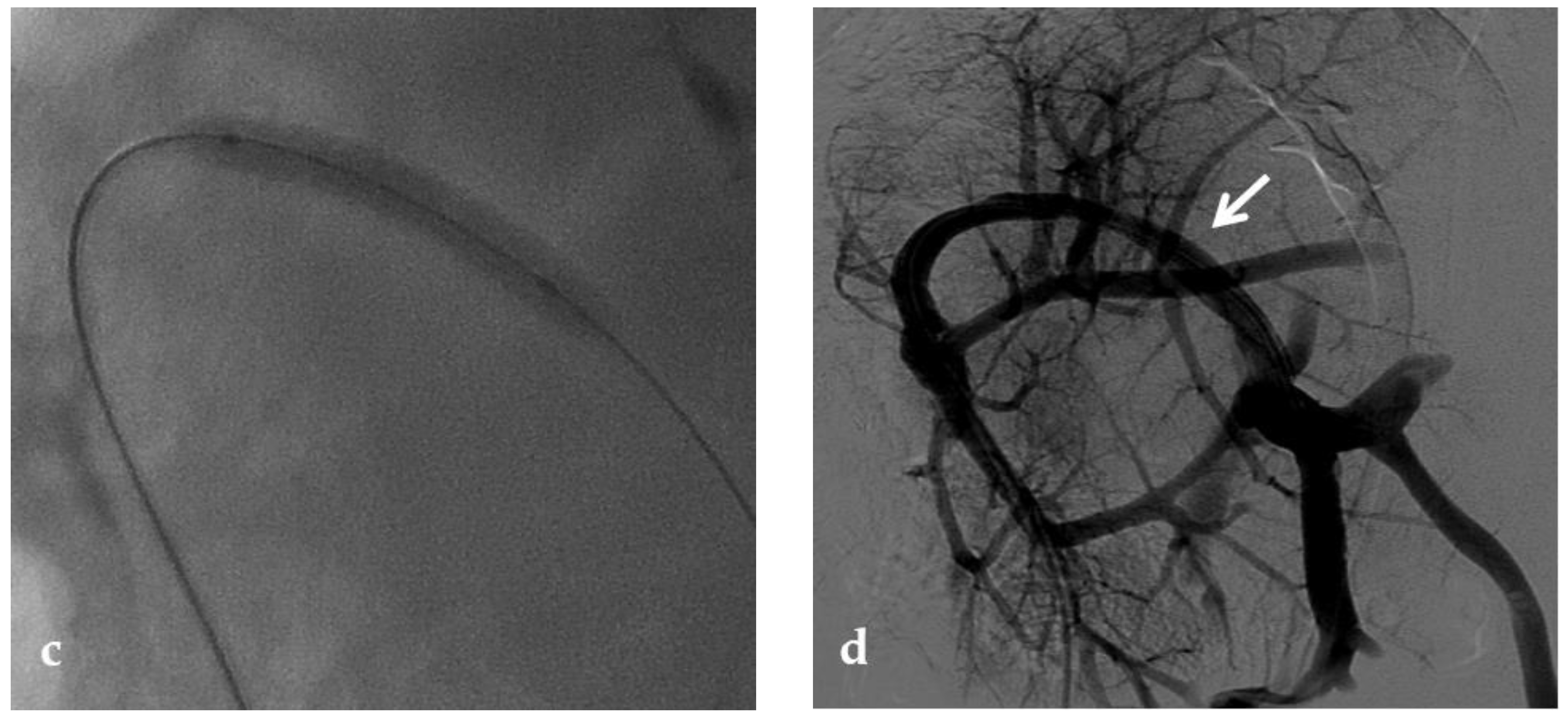
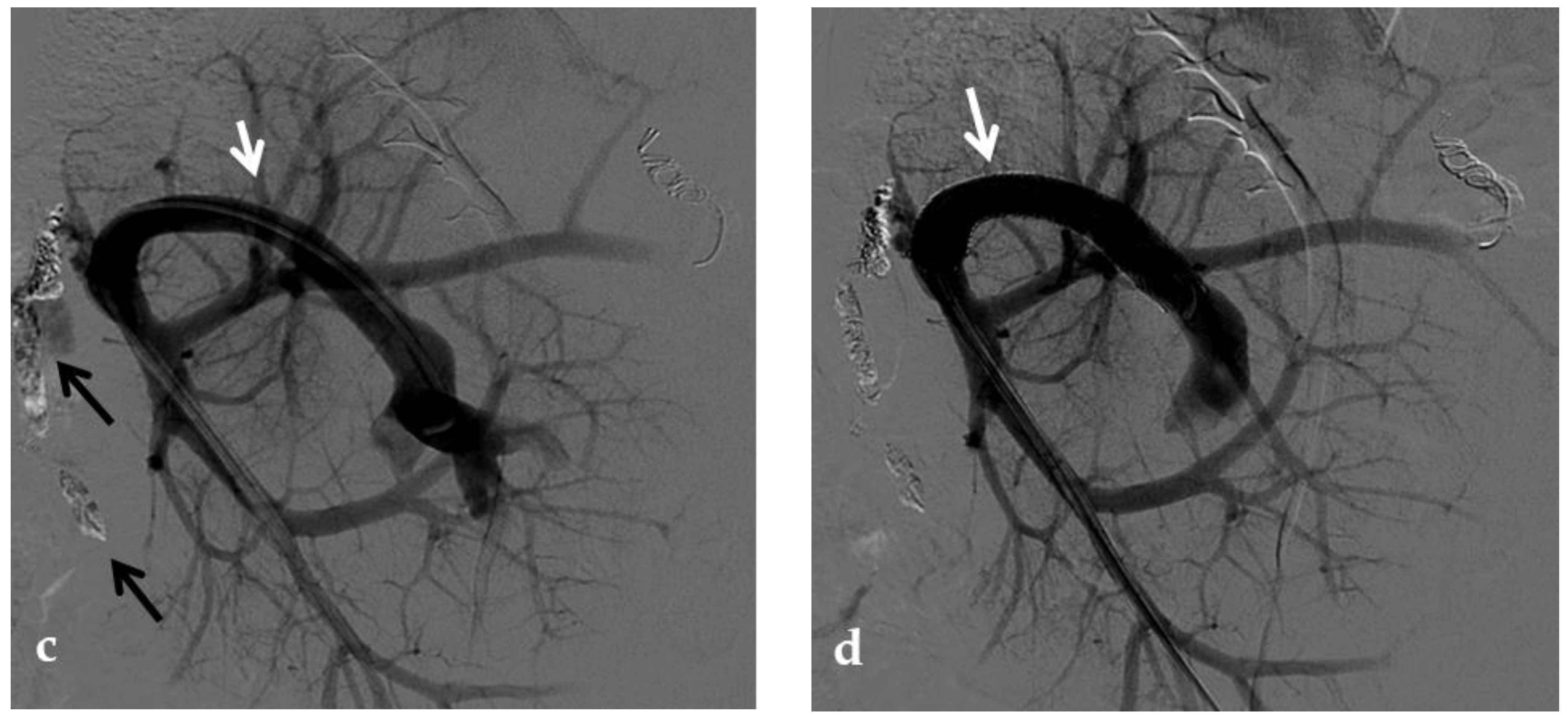
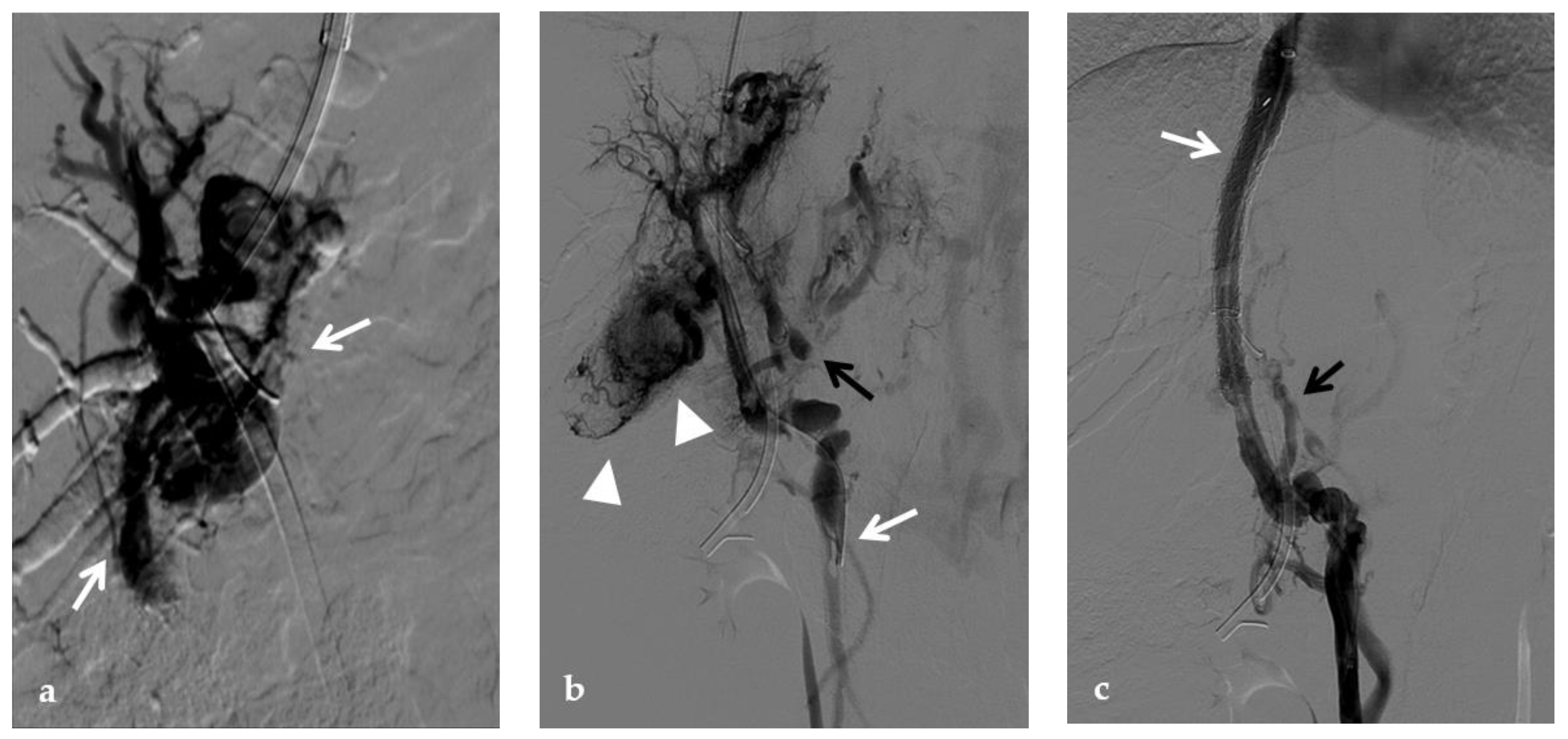
Figure 2. Anteroposterior view of a percutaneous transhepatic portography imaging series performed for chronic complete portal vein thrombosis with cavernous transformation in an 11-year-old boy with a history of gastrointestinal bleeding and portal hypertension 10 years after deceased-donor orthotopic liver transplantation (split II–III) for biliary atresia. (a) Portography image shows a regular representation of the segment 3 portal branch, while the segment 2 portal branch has a peripheral defect of opacification due to flow reversal, resulting from portal cavernoma and extrahepatic capsular feeding from gastric vessels (black arrow). Complete occlusion of the extrahepatic portal vein at the hepatic hilum (white arrow) is demonstrated. (b) Portography image shows the opacification of a very tiny vascular structure (arrow) which represents what remains of the thrombosed extrahepatic portal trunk. (c) Fluoroscopy image shows angioplasty with a non-compliant 8-mm balloon catheter performed on a stiff guidewire which was advanced across the obstructed tract. (d) Control portography shows a re-expanded extrahepatic portal vein (arrow) with normal spleno-portal opacification. Portal vein thrombosis recurred one year after treatment and required repeated angioplasty and metal stenting (shown in the next figure).
No consensus exists regarding the optimal indication for stent placement in PVT. Portal vein stent placement seems to be a safe and effective method for the treatment of post-transplantation PVT in pediatric recipients. The intermediate-term portal vein patency after stent placement is excellent
[34] (
Figure 3). In one study, after recanalization of the segment of portal vein affected by thrombosis through PTA, if an elastic recoil of more than 50% of the normal extrahepatic portal vein, a residual pressure gradient of more than 5 mmHg or a vascular dissection were found, stent placement was performed
[33]. Several other investigators have reported a 100% portal vein patency rate after stent placement with various follow-up times
[35][36]. These patency rates are superior to those previously reported with balloon angioplasty alone (27–50%)
[37].
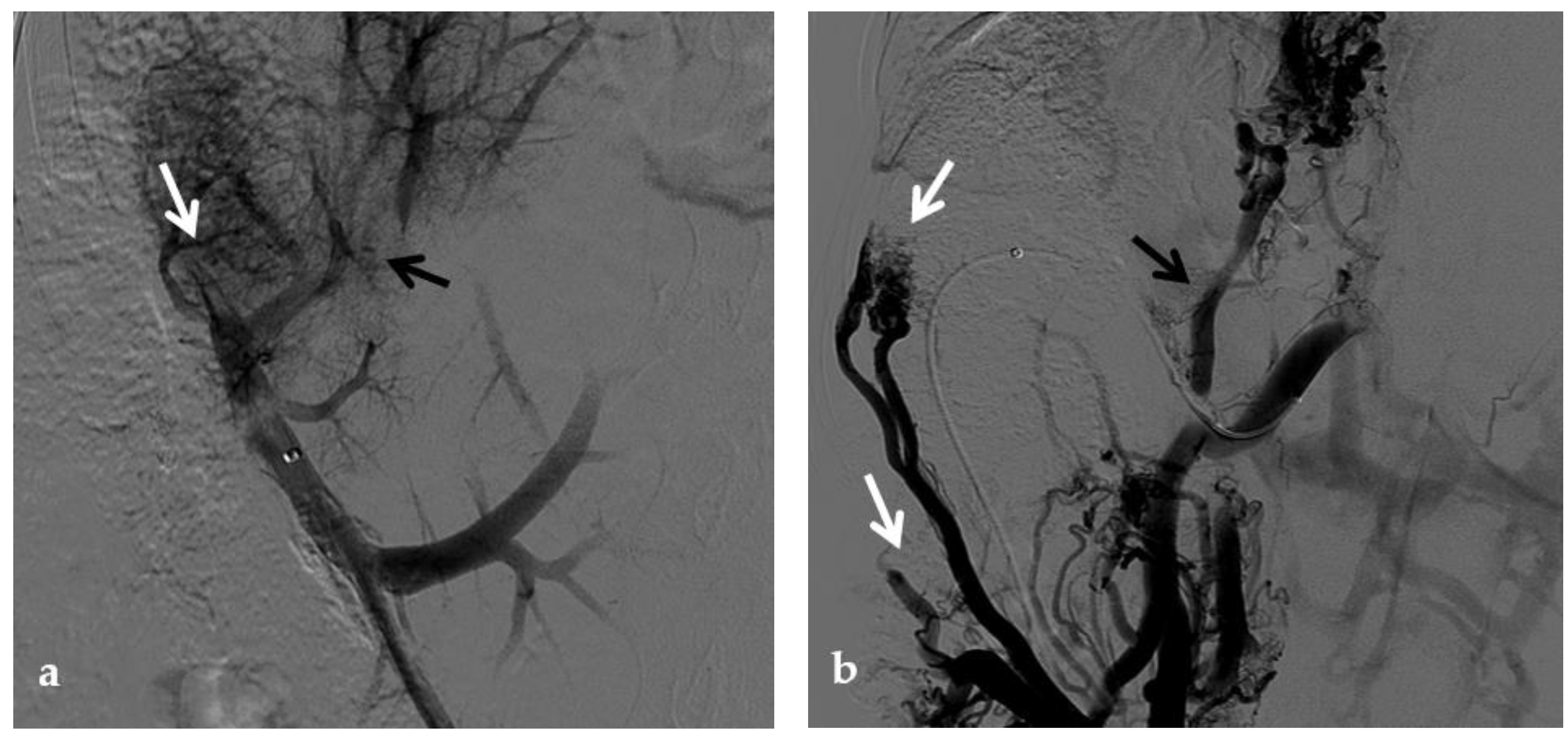
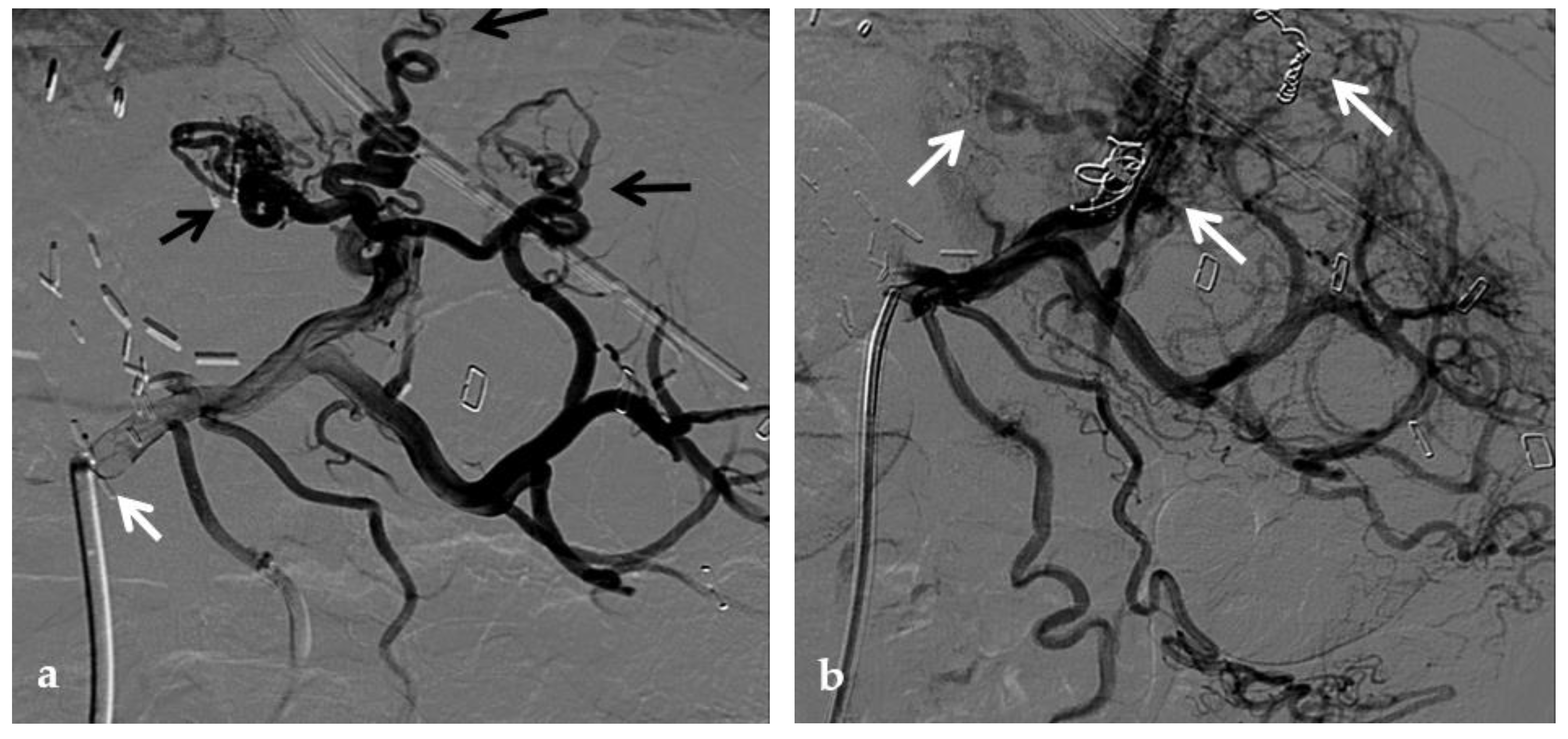
Figure 3. Anteroposterior view of a percutaneous transhepatic portography imaging series in the same patient as in Figure 9. (a) Portography image shows recurrence of complete extrahepatic portal vein thrombosis (white arrow) 1 year after the first angioplasty treatment. Flow reversal in the segment 2 portal branch resulting from portal cavernoma is still evident (black arrow). (b) After transhepatic portal vein recanalization with the angiographic catheter, the splenic portogram shows jejunal cavernoma (white arrows) and gastric (black arrow) varices, due to complete obstruction of the extrahepatic portal trunk. Embolization of varices was performed using a mixture of N-butyl cyanoacrylate and iodized oil and metallic coils. (c) Portography image after angioplasty of the extrahepatic portal vein shows successful endoluminal filling of the portal vein, with a restored but still irregular profile (white arrow). Note subtraction artifacts (black arrows) representing lipiodol and coils in the cavernoma and varices. (d) Final portogram performed after deployment of a 10-mm self-expandable bare metal stent to treat residual stenosis of the extrahepatic portal vein; the main portal vein is now regularly opacified with adequate size and both intrahepatic portal branches present hepatopetal flow.
One argument against metallic stent deployment is that a metallic stent placed across an extrahepatic portal venous lesion may interfere with future portal venous anastomoses whenever retransplantation becomes necessary
[38].
In the treatment of late-onset PVT with clinical signs of portal hypertension, decreased portal flow due to severe portosystemic shunt may contribute to recurrent PVT itself, and restoration of the portal flow with simultaneous embolization of portosystemic shunts may be important to achieve a sustained patency of the portal vein
[28][33][39].
2.2.1. Percutaneous Transhepatic Portography
The percutaneous transhepatic approach is the traditional method for portal vein catheterization and PVT treatment. The main advantage of direct transhepatic portography is that it allows accurate determination of the thrombus burden and extent, degree of stenosis, as well as direct portal venous pressure measurement which helps in the diagnosis of the cause of PVT
[40].
2.2.2. Percutaneous Transsplenic Portography
Percutaneous transsplenic portography represents a useful alternative access for endovascular portal vein treatments, especially when conventional transhepatic access is challenging or has failed.
A transsplenic access of the portal vein can be performed to achieve orthograde portography and to allow better conditions for recanalization in patients with tight portal vein occlusions and angled stenotic portal vein segments. Especially in young children with occluded, small, and short portal vein branches, this approach may help to achieve optimal conditions for portography and recanalization. The position and anatomy of the portal venous system in its intrahepatic and extrahepatic tracts varies after LT. Thus, recanalization via a transhepatic access can be unsuitable, and the antegrade transsplenic access adds valuable diagnostic information as well as an additional way to cross an unfavorable stenosis. Dilation of the venous splenic branches due to portal hypertension in these patients further increased the suitability of this approach.
The use of ultrasound guidance during puncture is mandatory to reduce the risk of complications
[31][41]. Because of the high vascularity of the spleen, the major technical issue is represented by the risk of bleeding, which can be prevented with tract embolization during introducer sheath removal
[42][43][44][45].
Several investigators reported that portal vein intervention via the transsplenic access is feasible and safe in LT recipients
[42][43][44]. Some studies compared transhepatic and transsplenic access in terms of safety, and demonstrated that both methods are safe, with no difference in the rate of bleeding-related complications
[43][44].
2.2.3. Percutaneous Transmesenteric Portography
The use of a transmesenteric approach has been proposed when the other routes are not adequate to allow PVT treatment
[45][46]. It can be either performed via direct image-guided percutaneous catheterization of the SMV or inferior mesenteric vein
[46] or through SMV cannulation via a mini-laparotomy. This approach is more invasive than the other percutaneous routes, and it is less adopted
[47].
A transmesenteric approach can be used both in early PVT and in chronic PVT when transhepatic and/or transsplenic access fails. Kim et al. and Cheng et al. described the use of the transmesenteric access for portal vein revascularization and stent placement for PVT during transplantation and early PVT
[48][49]. Cavalcante et al. showed that PV revascularization using a transmesenteric approach may be an alternative procedure to reestablish the portal flow to the liver graft in selected cases, being a therapeutic addition to other treatment strategies currently used to treat chronic PVT. It has acceptable technical and clinical success rates, long-term patency, and a low rate of procedure-related complications
[50] (
Figure 4).
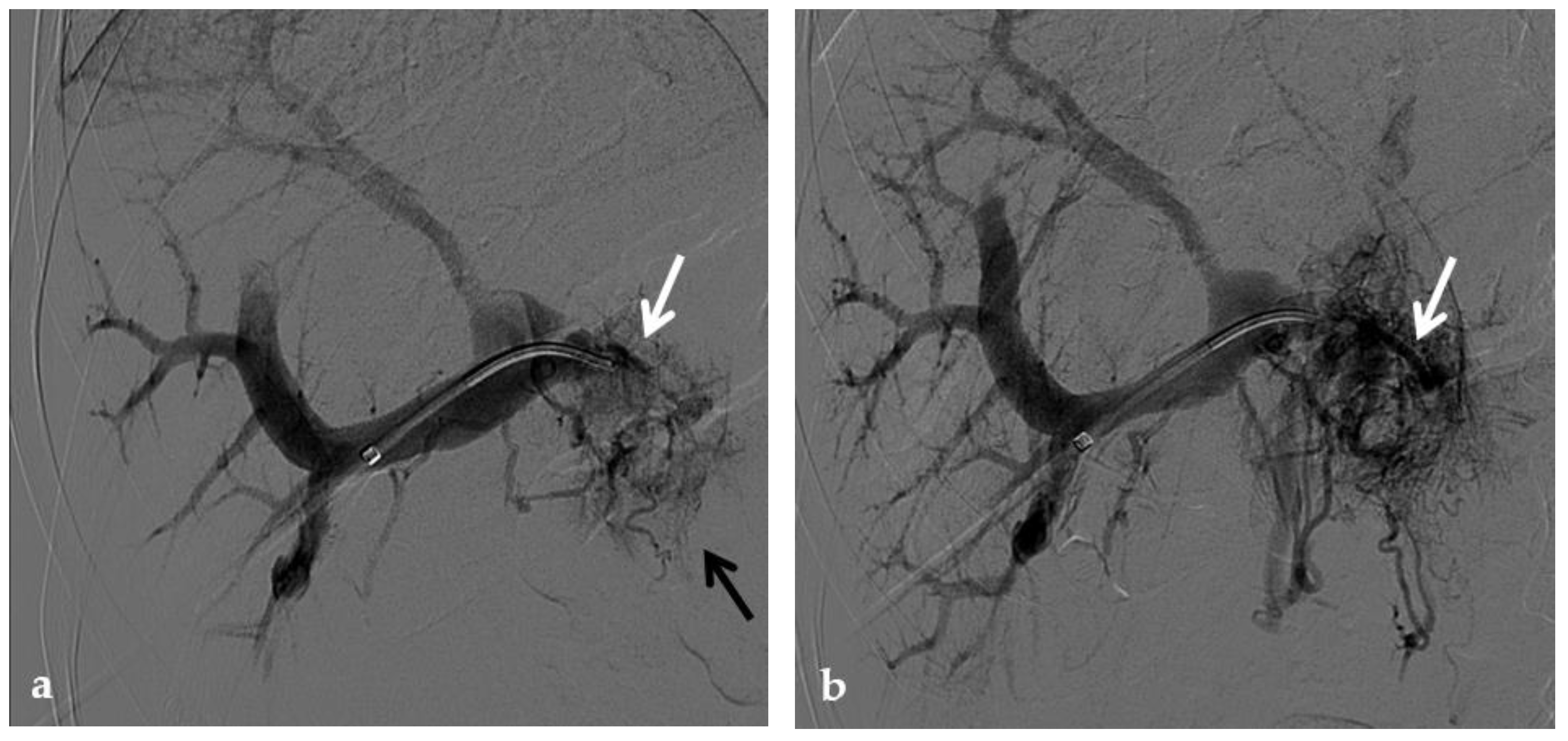
Figure 4. Anteroposterior view of a percutaneous transmesenteric portography imaging series of an acute complete portal vein thrombosis occurred early after pediatric split-liver transplantation in a 1-year-old female with biliary atresia. (a) Superior mesenteric portography performed through a surgically-placed transmesenteric sheath (white arrow) shows total occlusion of the main portal vein and its intrahepatic branches, with opacification of several portosystemic varices (black arrows). (b) Superior mesenteric portography shows effective embolization of the portosystemic shunts to reduce the flow-steal phenomenon. A mixture of N-butyl cyanoacrylate and iodized oil and metallic microcoils (arrows) were used. (c) Portography image shows angioplasty with a 5-mm non-compliant balloon catheter performed on a stiff guidewire advanced through the common portal vein (white arrow). Note the spontaneous remaining opacification caused by the mixture of N-butyl cyanoacrylate and iodized oil (black arrows). (d) Final superior mesenteric portography shows restored opacification of the portal vein anastomosis, the umbilical recess of the portal vein (black arrow) and the intrahepatic segment 2 and segment 3 branches (white arrows), with no contrast filling of the portosystemic shunts.
2.3. Transjugular Intrahepatic Portosystemic Shunt (TIPS)
Although the etiology of PVT in cirrhosis is incompletely understood, reduced portal blood flow velocity is thought to play a central role. Currently, anticoagulation is recommended as the first-line therapy for the management of PVT, though this may put patients with esophageal varices at increased risk for death from variceal hemorrhage.
Traditionally, PVT was considered a relative contraindication for TIPS placement. However, studies have shown that TIPS can facilitate improved blood flow, resulting in clot resorption and recanalization of PVT
[51][52].
One study demonstrated that TIPS was effective for the treatment of PVT in patients with cirrhosis. Fifty-seven percent of cases had complete resolution of thrombosis, 30% had reduction in lumen obliteration and/or extension of thrombosis, and only 13% remained unchanged
[52].
Moreover, a systematic review and meta-analysis exploring a total of 18 articles in which patients had a diagnosis of PVT and underwent TIPS for the management of PVT, showed that the use of TIPS may be a safe and feasible option in patients with underlying liver disease for the management of PVT, achieving portal patency in 86% of cases
[53]. This study also showed that the use of TIPS may reduce clot burden with partial recanalization in 84% of patients and complete recanalization in 73% in patients with established PVT.
Acute thrombotic occlusions of the portal system can be managed by mechanical thrombectomy or pharmacologic thrombolysis. Chronic occlusions due to organized or refractory thrombus or fixed venous stenosis are better managed by balloon angioplasty and stent placement. Access into the portal venous system can be established through TIPS creation. A TIPS is also important in the setting of PVT associated with cirrhosis to decompress portal hypertension, improve portal venous flow, and prevent variceal hemorrhage
[54]. The access to the portal system through a TIPS is also advisable in case of abundant perihepatic ascites or coagulopathy that preclude direct portal catheterization through percutaneous liver puncture. In the absence of portal hypertension or other contraindications such as ascites or coagulopathy, portal vein recanalization and mechanical thrombectomy can be also performed through direct transhepatic portal vein catheterization.
In patients with PVT without cirrhosis, the use of TIPS to improve portal flow is still controversial. If on the one hand TIPS may induce acceleration of the portal blood flow preventing the extension of thrombosis into the portal system, on the other hand, it raises concerns about long-term consequences of portal flow shunting on liver function
[55].
Portal cavernoma is regarded as a relative contraindication to TIPS placement due to the high probability of technical failure. There are anecdotal reports of successful TIPS insertion in patients with a portal cavernoma in the absence of cirrhosis
[55]. A study
[56] suggested that TIPS creation by combination with a transhepatic or transsplenic approach is feasible and safe in the presence of portal cavernoma (
Figure 5). However, TIPS was not recommended in the patients with a total fibrotic cord and without a large collateral vein or in those with extensive SMV thrombosis. By definition, TIPS can decrease the portosystemic pressure gradient, thus reducing the incidence of variceal bleeding
[56]. In patients with jaundice or recurrent biliary symptoms due to portal cholangiopathy, although feasible in a limited number of patients, TIPS has been primarily used to address the biliary complications, allowing portal decompression, and staged biliary surgery
[16] (
Figure 6).
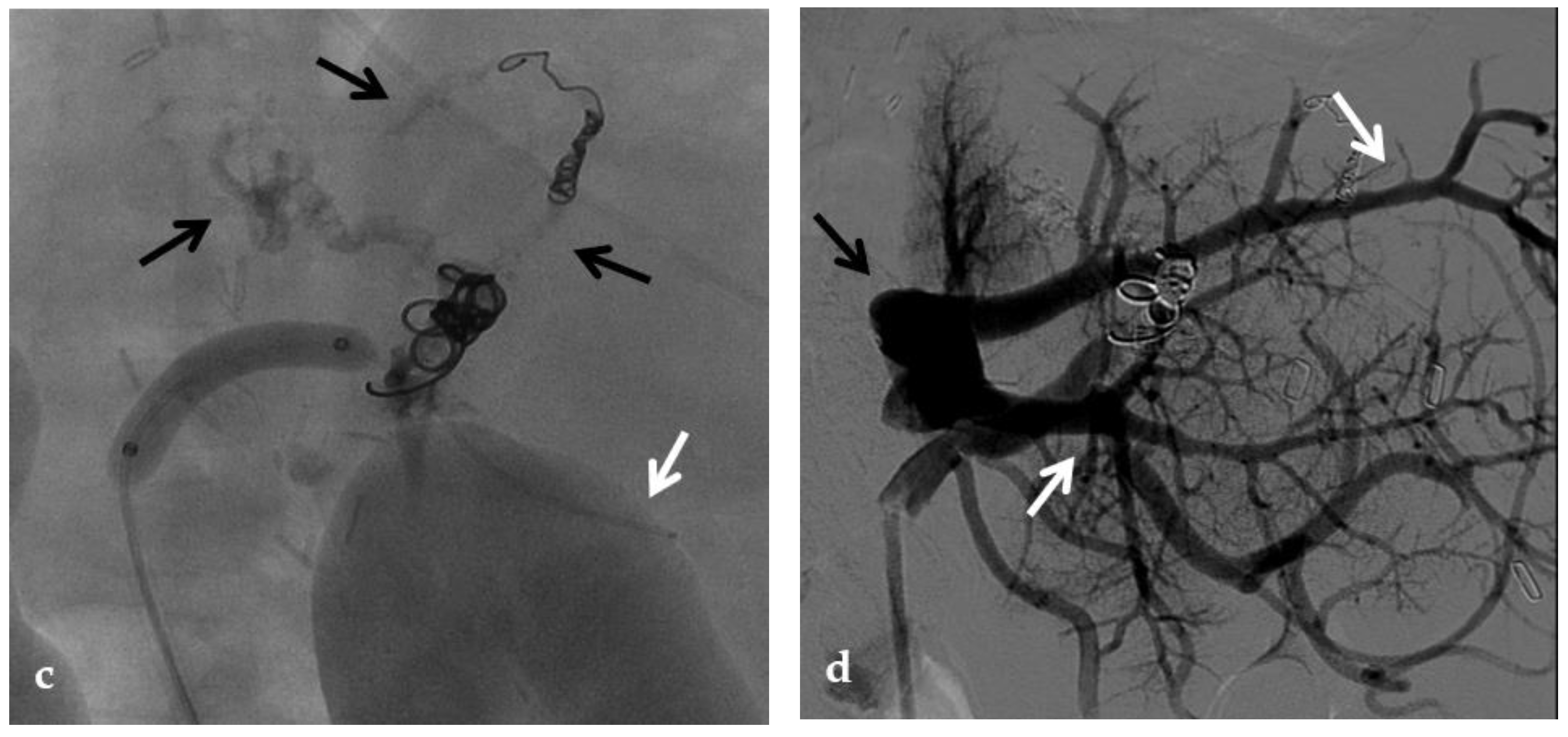
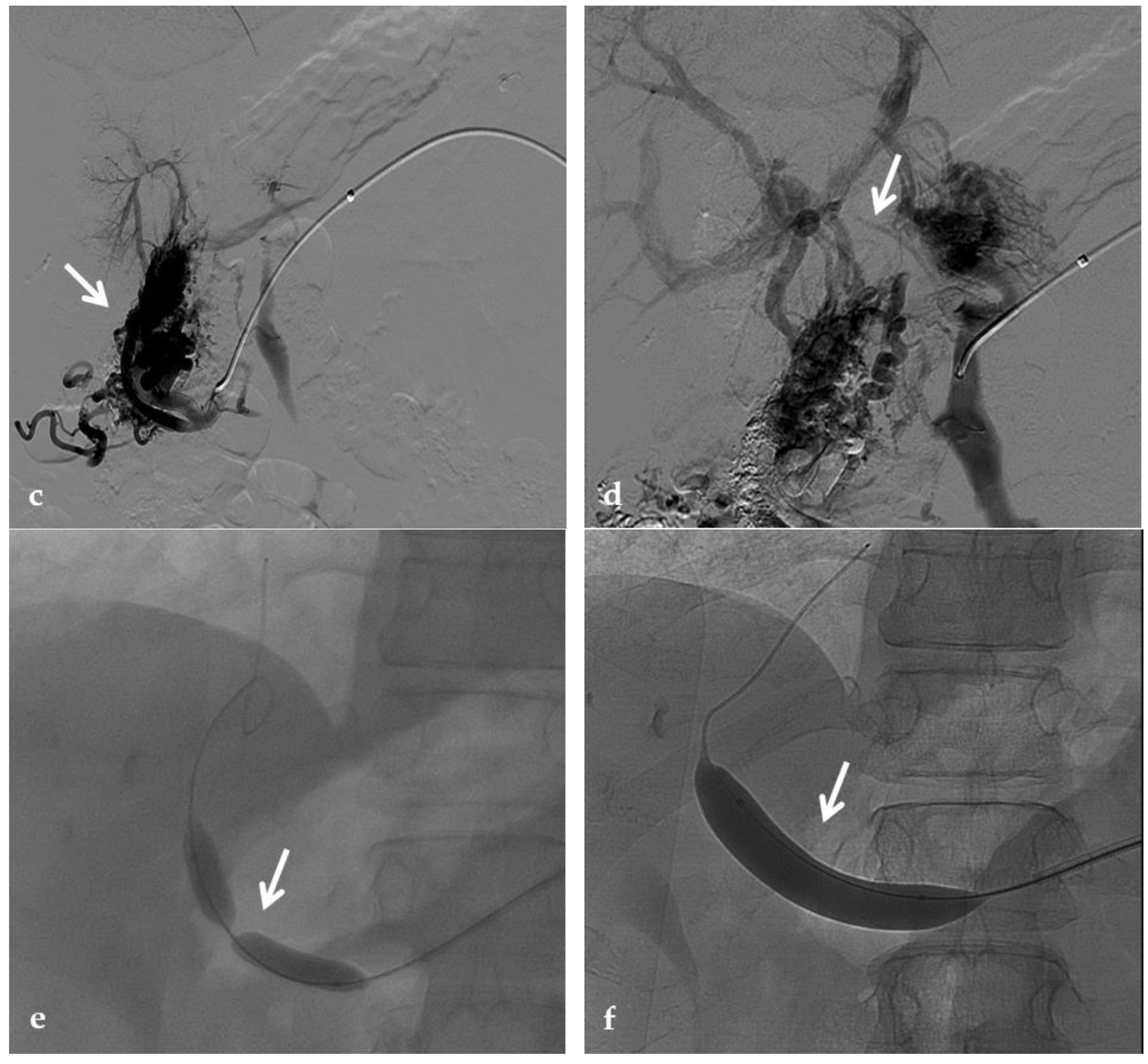
Figure 5. Anteroposterior view of a percutaneous transhepatic and transsplenic portography imaging series of a chronic complete extrahepatic portal vein thrombosis with portal cavernous transformation in a 17-year-old boy with a history of gastrointestinal bleeding and portal hypertension after liver transplant for biliary atresia. (a) Percutaneous transhepatic portography shows complete extrahepatic portal vein thrombosis (white arrow), with opacification of an irregular cavernomatous network (black arrow). (b) Opacification of a very tiny fibrotic cord representing the thrombosed portal trunk is seen (arrow). Several attempts to cross the portal vein occlusion failed. (c) Percutaneous transsplenic anterograde portography shows complete extrahepatic portal vein thrombosis and cavernous transformation (arrow) from a different perspective. (d) The tiny residual of the thrombosed main portal trunk (arrow) was easily recognized on the transsplenic superior mesenteric vein portography. (e) Fluoroscopic image shows angioplasty of the main portal vein, performed through a 12-mm non-compliant balloon catheter. The focal notch (arrow) represents the tight anastomotic stenosis that probably led to secondary thrombosis. (f) Fluoroscopic image shows the resolution of the focal notch (arrow) after high-pressure inflation of the non-compliant 12-mm balloon catheter. (g) Control portography shows an expanded main portal vein with improved hepatopetal portal flow, but still with irregular profiles (arrow). (h) Through a hybrid transhepatic and transjugular approach under combined ultrasound and fluoroscopic guidance, the connection between the intrahepatic portal vein and the vena cava was created advancing a 0.018” micro guidewire into the right atrium (arrow). The wire was snared through the right transjugular access providing a through-and-through access for precise transjugular intrahepatic portosystemic shunt (TIPS) placement. The completely image-guided hybrid approach allowed to reduce the attempts of transjugular intrahepatic portal vein puncture. (i) Portography image shows the deployment of two imbricated Viatorr stents to create the TIPS (arrows), with regular intrastent opacification. Despite previous angioplasty, a prestenotic aspect of the main portal vein is seen (black arrow). (j) Portography image shows a self-expandable 9-mm metallic stent placed to cover the main portal vein trunk stenosis (white arrow). Control portography shows regular mesenteric-portal vein opacification with TIPS patency and preserved opacification of intrahepatic portal branches (black arrows); cavernous vessels disappeared.
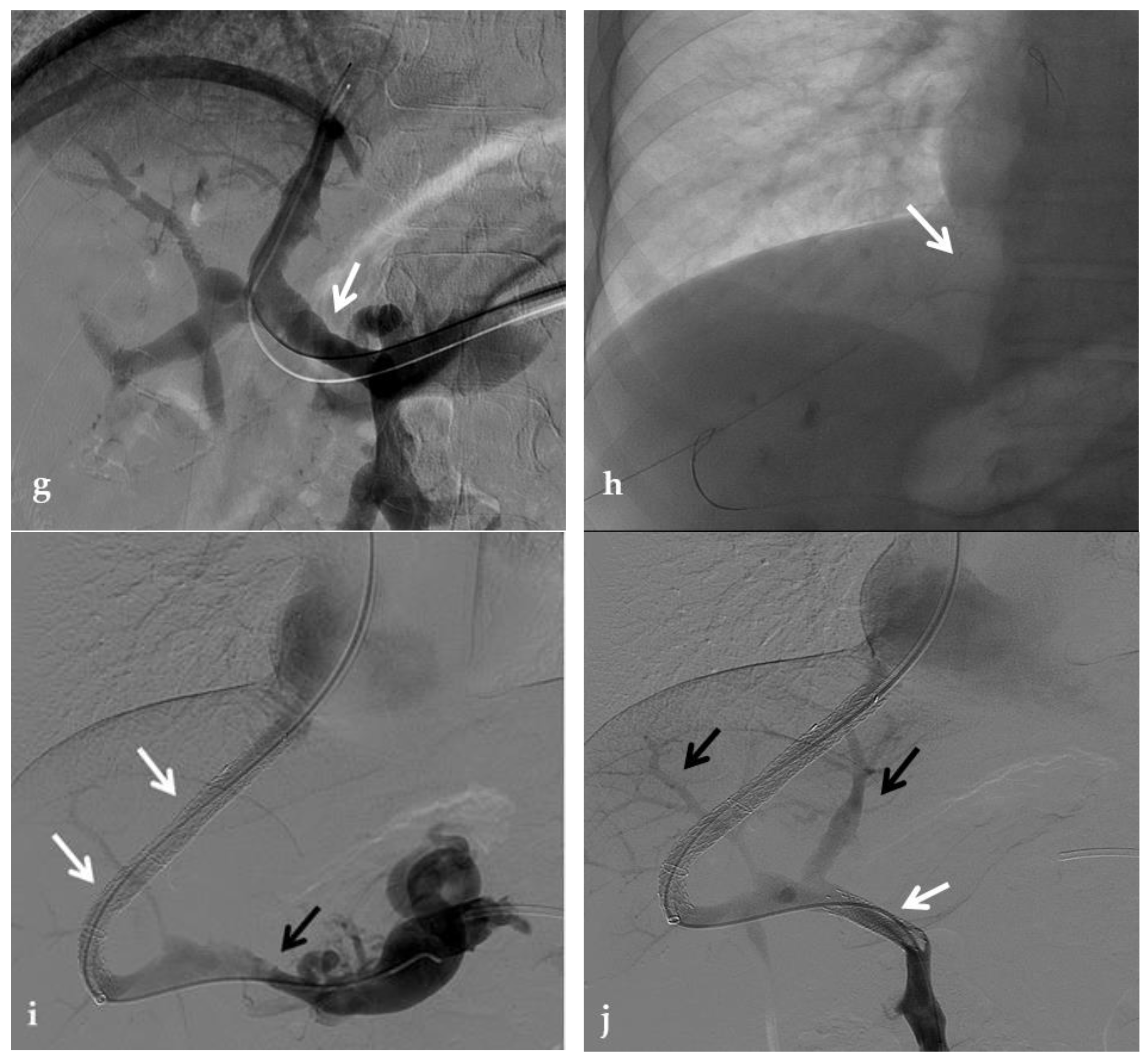
Figure 6. Anteroposterior view of a transjugular intrahepatic portosystemic shunt portography imaging series. (a) Transjugular intrahepatic portal puncture confirms the chronic occlusion of the main portal vein with intrahepatic cavernous transformation (arrows). (b) After transjugular catheterization of the main cavernous vessel portography shows a patent superior mesenteric vein (white arrow) with other minor cavernous vessels at the hepatic hilum (black arrow). Note the aberrant paracholecystic veins forming portoportal collaterals to bypass the obstructed segment of the portal vein (arrowheads). (c) Portography performed after transjugular intrahepatic portosystemic shunt placement shows regular stent opacification (white arrow), with reduced visualization of the cavernoma (black arrow) and disappearance of paracholecystic veins.