
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sirius Huang | -- | 6654 | 2022-11-09 01:37:43 |
Video Upload Options
A COVID‑19 vaccine is a hypothetical vaccine in development against coronavirus disease 2019 (COVID‑19). Although no candidate has completed clinical trials to prove its safety and efficacy, there are multiple efforts in progress to develop one. As of July 2020, there are 24 vaccine candidates planned or undergoing clinical trials, with two beginning Phase III and seven in Phase I-II. Previous attempts to develop a vaccine against the coronavirus diseases SARS and MERS established considerable knowledge about the structure and function of coronaviruses – which accelerated rapid development during early 2020 of varied technology platforms for a COVID‑19 vaccine – but all the previous coronavirus vaccine candidates failed in early-stage clinical trials, with none being advanced to licensing. The Coalition for Epidemic Preparedness Innovations (CEPI) – which is organizing a US$2 billion worldwide fund for rapid investment and development of vaccine candidates – indicated in April that a vaccine may be available under emergency use protocols in less than 12 months or by early 2021. On 4 May 2020, the WHO organized a telethon which received US$8.1 billion in pledges from forty countries to support rapid development of vaccines to prevent COVID‑19 infections. At the same time, the WHO also announced deployment of an international "Solidarity trial" for simultaneous evaluation of several vaccine candidates reaching Phase II-III clinical trials.
1. History
In February 2020, the World Health Organization (WHO) said it did not expect a vaccine against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative virus, to become available in less than 18 months.[1] In July 2020, 218 vaccine candidates were in development.[2][3]
Vaccines have been produced against several animal diseases caused by coronaviruses, including infectious bronchitis virus in birds, canine coronavirus, and feline coronavirus.[4]
Previous projects to develop vaccines for viruses in the family Coronaviridae that affect humans have been aimed at severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS). Vaccines against SARS[5] and MERS[6] have been tested in non-human animal models.
For SARS, as of 2020 there is no cure or protective vaccine that has been shown to be both safe and effective in humans.[7][8] According to research papers published in 2005 and 2006, the identification and development of novel vaccines and medicines to treat SARS was a priority for governments and public health agencies around the world at that time.
There is also no proven vaccine against MERS.[9] When MERS became prevalent, it was believed that existing SARS research may provide a useful template for developing vaccines and therapeutics against a MERS-CoV infection.[7][10] As of March 2020, there was one (DNA based) MERS vaccine which completed Phase I clinical trials in humans,[11] and three others in progress, all of which are viral-vectored vaccines: two adenoviral-vectored (ChAdOx1-MERS, BVRS-GamVac), and one MVA-vectored (MVA-MERS-S).[12]
2. Globally Accelerated Development
After a novel coronavirus pneumonia was detected in December 2019,[13] the genetic sequence of COVID‑19 was published on 11 January 2020, triggering an urgent international response to prepare for the outbreak and hasten development of a preventative vaccine.[14][15][16] The rapidly growing infection rate of COVID‑19 worldwide during early 2020 stimulated international alliances and government efforts to urgently organize resources to make multiple vaccines on shortened timelines,[17] with four vaccine candidates entering human evaluation in March (see the table of clinical trials started in 2020, below).[14][18]
A vaccine for an infectious disease has never before been produced in less than several years, and no vaccine exists for preventing a coronavirus infection.[19] As of April, CEPI estimates that as many as six of the 115 vaccine candidates against COVID‑19 should be chosen by international coalitions for development through Phase II–III trials, and three should be streamlined through regulatory and quality assurance for eventual licensing at a total cost of at least US$2 billion.[14][18][19] Another analysis estimates 10 candidates will need simultaneous initial development, before a select few are chosen for the final path to licensing.[19]
The vaccine effort is being prioritized for speed of rigorous clinical evaluation for safety and efficacy, financing, and planning to manufacture billions of doses, and eventual worldwide deployment and equitable access among developed and undeveloped countries.[18][19] WHO, CEPI, and the Gates Foundation are committing money and organizational resources for the prospect that several vaccines will be needed to prevent continuing COVID‑19 infection.[19] The vaccines will require custom formulation, special packaging, transportation, and storage in all 200 countries with infected citizens.[19][20] The WHO estimates a total cost of US$8 billion to develop a suite of three or more vaccines having different technologies and distribution to prevent COVID‑19 infections worldwide.[17][21][22]
2.1. International Organizations
Organizations have formed international alliances to expedite vaccine development and prepare for distribution, including the WHO which is facilitating collaboration, accelerated research, and international communications on a scale unprecedented in history, beginning in early May by raising US$8.1 billion in pledges.[23] The WHO also implemented an Access to COVID‑19 Tools Accelerator for coordinating global vaccine development.[21] The Coalition for Epidemic Preparedness Innovations (CEPI) is working with international health authorities and vaccine developers to create another US$8 billion fund in a global partnership between public, private, philanthropic, and civil society organizations for accelerated research and clinical testing of eight vaccine candidates, with the 2020-21 goal of supporting three candidates for full development to licensing.[14][18][22] The United Kingdom, Canada, Belgium, Norway, Switzerland, Germany and the Netherlands had already donated US$915 million to CEPI by early May.[23][24] The Bill & Melinda Gates Foundation (Gates Foundation), a private charitable organization dedicated to vaccine research and distribution, is donating US$250 million in support of CEPI for research and public educational support on COVID‑19 vaccines.[15][19][20][25] The Global Research Collaboration for Infectious Disease Preparedness (GLoPID-R) is working closely with the WHO and member states to identify specific funding of research priorities needed for a COVID‑19 vaccine, coordinating among the international funding and research organizations to maintain updated information on vaccine progress and avoid duplicate funding.[26][27] The International Severe Acute Respiratory and Emerging Infection Consortium is organizing and disseminating clinical information on COVID‑19 research to inform public health policy on eventual vaccine distribution.[28]
On 4 June, a virtual summit was coordinated from London, UK among private and government representatives of 52 countries, including 35 heads of state from G7 and G20 nations, to raise US$8.8 billion in support of the Global Alliance for Vaccines and Immunisation (GAVI) to prepare for COVID‑19 vaccinations of 300 million children in under-developed countries through 2025.[19][29][30] Major contributions were $1.6 billion from The Gates Foundation[31] and GB£330 million per year over five years by the UK government (approximately US$2.1 billion in June 2020).[29]
2.2. National Governments
National governments dedicating resources for national or international investments in vaccine research, development, and manufacturing beginning in 2020 included the Canadian government which announced CA$275 million in funding for 96 research vaccine research projects at Canadian companies and universities, with plans to establish a "vaccine bank" of several new vaccines that could be used if another coronavirus outbreak occurs.[32][33] A further investment of CA$1.1 billion was added to support clinical trials in Canada and develop manufacturing and supply chains for vaccines.[27] On 4 May, the Canadian government committed CA$850 million to the WHO's live streaming effort to raise US$8 billion for COVID‑19 vaccines and preparedness.[34] In China, the government is providing low-rate loans to vaccine developers through its central bank, and has enabled land transfers to build production plants.[24] As of June 2020, six of the eleven COVID‑19 vaccine candidates in early-stage human testing were developed by Chinese organizations (table).[20] Three Chinese vaccine companies and research institutes are supported by the government for financing research, conducting clinical trials, and manufacturing the most promising vaccine candidates, while prioritizing rapid evidence of efficacy over safety.[35] On 18 May, China had pledged US$2 billion to support overall efforts by the WHO for programs against COVID‑19.[36]
Among European Union countries, France announced a US$4.9 million investment in a COVID‑19 vaccine research consortium via CEPI involving the Institut Pasteur, Themis Bioscience (Vienna, Austria), and the University of Pittsburgh, bringing CEPI's total investment in COVID‑19 vaccine development to US$480 million by May.[37][38] In March, the European Commission made an €80 million investment in CureVac, a German biotechnology company, to develop a mRNA vaccine.[39] The German government announced a separate €300 million investment in CureVac in June.[40] Belgium, Norway, Switzerland, Germany, and the Netherlands have been major contributors to the CEPI effort for COVID‑19 vaccine research in Europe.[24]
In April, the UK government formed a COVID‑19 vaccine taskforce to stimulate British efforts for rapidly developing a vaccine through collaborations of industry, universities, and government agencies across the vaccine development pipeline, including clinical trial placement at UK hospitals, regulations for approval, and eventual manufacturing.[41] The vaccine development initiatives at the University of Oxford and Imperial College of London were financed with GB£44 million in April.[42][43]
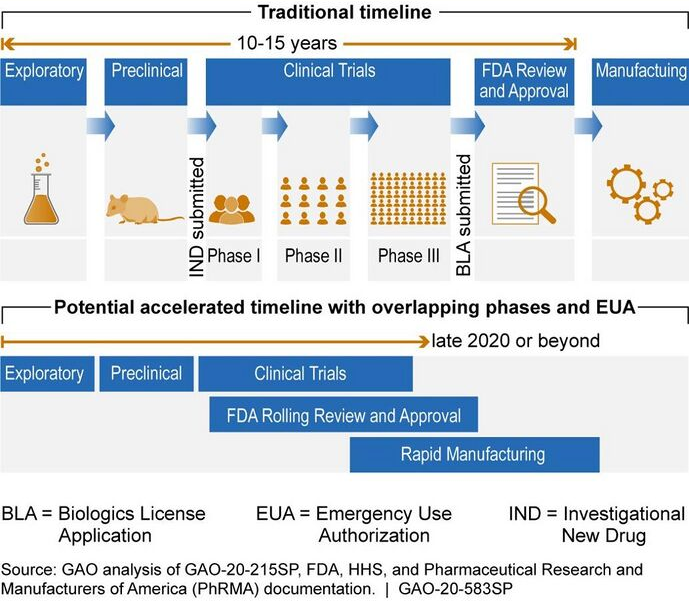
The United States Biomedical Advanced Research and Development Authority (BARDA), a federal agency that funds disease-fighting technology, announced investments of nearly US$1 billion to support American COVID‑19 vaccine development, and preparation for manufacturing the most promising candidates. On 16 April, BARDA made a US$483 million investment in the vaccine developer, Moderna and its partner, Johnson & Johnson.[24][44] BARDA has an additional US$4 billion to spend on vaccine development, and will have roles in other American investment for development of six to eight vaccine candidates to be in clinical studies over 2020-21 by companies such as Sanofi Pasteur and Regeneron.[44][45] On 15 May, the US government announced federal funding for a fast-track program called Operation Warp Speed, which has the goals of placing diverse vaccine candidates in clinical trials by the fall of 2020, and manufacturing 300 million doses of a licensed vaccine by January 2021. The project chief advisor is Moncef Slaoui and its Chief Operating Officer is Army General Gustave Perna.[46][47] In June, the Warp Speed team said it would work with seven companies developing COVID‑19 vaccine candidates: Moderna, Johnson & Johnson, Merck, Pfizer, and the University of Oxford in collaboration with AstraZeneca, as well as two others.[48]
2.3. Partnerships and Competition
WHO Solidarity trial
The WHO has developed a multinational coalition of vaccine scientists defining a Global Target Product Profile (TPP) for COVID‑19, identifying favorable attributes of safe and effective vaccines under two broad categories: "vaccines for the long-term protection of people at higher risk of COVID‑19, such as healthcare workers", and other vaccines to provide rapid-response immunity for new outbreaks.[17] The international TPP team was formed to 1) assess the development of the most promising candidate vaccines; 2) map candidate vaccines and their clinical trial worldwide, publishing a frequently-updated "landscape" of vaccines in development;[49] 3) rapidly evaluate and screen for the most promising candidate vaccines simultaneously before they are tested in humans; and 4) design and coordinate a multiple-site, international randomized controlled trial – the Solidarity trial for vaccines[17][50] – to enable simultaneous evaluation of the benefits and risks of different vaccine candidates under clinical trials in countries where there are high rates of COVID‑19 disease, ensuring fast interpretation and sharing of results around the world.[17] The WHO vaccine coalition will prioritize which vaccines should go into Phase II and III clinical trials, and determine harmonized Phase III protocols for all vaccines achieving the pivotal trial stage.[17]
Adaptive design for the Solidarity trial
A clinical trial design in progress may be modified as an "adaptive design" if accumulating data in the trial provide early insights about positive or negative efficacy of the treatment.[51][52] The WHO Solidarity trial of multiple vaccines in clinical studies during 2020 will apply adaptive design to rapidly alter trial parameters across all study sites as results emerge.[50] Candidate vaccines may be added to the Solidarity trial as they become available if priority criteria are met, while vaccine candidates showing poor evidence of safety or efficacy compared to placebo or other vaccines will be dropped from the international trial.[50]
Adaptive designs within ongoing Phase II-III clinical trials on candidate vaccines may shorten trial durations and use fewer subjects, possibly expediting decisions for early termination or success, avoiding duplication of research efforts, and enhancing coordination of design changes for the Solidarity trial across its international locations.[50][51]
Partnerships, competition, and distribution
Large pharmaceutical companies with experience in making vaccines at scale, including Johnson & Johnson, AstraZeneca, and GlaxoSmithKline (GSK), are forming alliances with biotechnology companies, national governments, and universities to accelerate progression to an effective vaccine.[20][24] To combine financial and manufacturing capabilities for a pandemic adjuvant technology, GSK joined with Sanofi in an uncommon partnership of multinational companies to support accelerated vaccine development.[53]
During a pandemic on the rapid timeline and scale of COVID‑19 infections during 2020, international organizations like the WHO and CEPI, vaccine developers, governments and industry are evaluating distribution of the eventual vaccine(s).[17] Individual countries producing a vaccine may be persuaded to favor the highest bidder for manufacturing or provide first-service to their own country.[15][19][24] Experts emphasize that licensed vaccines should be available and affordable for people at the frontline of healthcare and having the greatest need.[15][19][24] Under their agreement with AstraZeneca, the University of Oxford vaccine development team and UK government agreed that UK citizens would not get preferential access to a new COVID‑19 vaccine developed by the taxpayer-funded university, but rather consented to having a licensed vaccine distributed multinationally in cooperation with the WHO.[42] Several companies plan to initially manufacture a vaccine at low cost, then increase costs for profitability later if annual vaccinations are needed and as countries build stock for future needs.[24]
The WHO and CEPI are developing financial resources and guidelines for global deployment of three or more safe, effective COVID‑19 vaccines, recognizing the need is different across countries and population segments.[14][17][18][50] For example, successful COVID‑19 vaccines would likely be allocated first to healthcare personnel and populations at greatest risk of severe illness and death from COVID‑19 infection, such as the elderly or densely-populated impoverished people.[18][54] Both the WHO and CEPI have discussed concerns that affluent countries should not have priority access to the global supply of eventual COVID‑19 vaccines.[14][18][54]
2.4. Compressed Timelines
Geopolitical issues, safety concerns for vulnerable populations, and manufacturing challenges for producing billions of doses are compressing schedules to shorten the standard vaccine development timeline, in some cases combining clinical trial steps over months, a process typically conducted sequentially over years.[20] As an example, Chinese vaccine developers and the government Chinese Center for Disease Control and Prevention began their efforts in January 2020,[55] and by March were pursuing numerous candidates on short timelines, with the goal to showcase Chinese technology strengths over those of the United States, and to reassure the Chinese people about the quality of vaccines produced in China.[20][56]
| COVID‑19: Vaccine technology platforms July 2020 |
||
|---|---|---|
| Molecular platform | Total number of candidates |
Number of candidates in human trials |
| Non-replicating viral vector | ||
| RNA-based | ||
| Inactivated virus | ||
| DNA-based | ||
| Protein subunit | ||
| Replicating viral vector | ||
| Virus-like particle | ||
| Undefined | ||
| Live attenuated virus | ||
| Replicating bacterial vector | ||
In the haste to provide a vaccine on a rapid timeline for the COVID‑19 pandemic, developers and governments are accepting a high risk of "short-circuiting" the vaccine development process,[24] with one industry executive saying: "The crisis in the world is so big that each of us will have to take maximum risk now to put this disease to a stop".[24] Multiple steps along the entire development path are evaluated, including the level of acceptable toxicity of the vaccine (its safety), targeting vulnerable populations, the need for vaccine efficacy breakthroughs, the duration of vaccination protection, special delivery systems (such as oral or nasal, rather than by injection), dose regimen, stability and storage characteristics, emergency use authorization before formal licensing, optimal manufacturing for scaling to billions of doses, and dissemination of the licensed vaccine.[19][57] If a vaccine fails in development, not providing adequate prevention against the virus – data show that 84-90% of vaccine candidates fail (have "attrition") in Phase III clinical trials[14][58] – the investment by a manufacturer in a vaccine candidate may exceed US$1 billion and end with millions of useless doses.[19][20][24] In the case of COVID‑19 specifically, a vaccine efficacy of 70% may be enough to stop the pandemic, but if it has only 60% efficacy, outbreaks may continue; an efficacy of less than 60% will not provide enough herd immunity to stop the spread of the virus alone.[19]
As the pandemic expands during 2020, research at universities is obstructed by physical distancing and closing of laboratories.[59][60] Globally, supplies critical to vaccine research and development are increasingly scarce due to international competition or national sequestration.[35] Timelines for conducting clinical research – normally a sequential process requiring years – are being compressed into safety, efficacy, and dosing trials running simultaneously over months, potentially compromising safety assurance.[20][24]
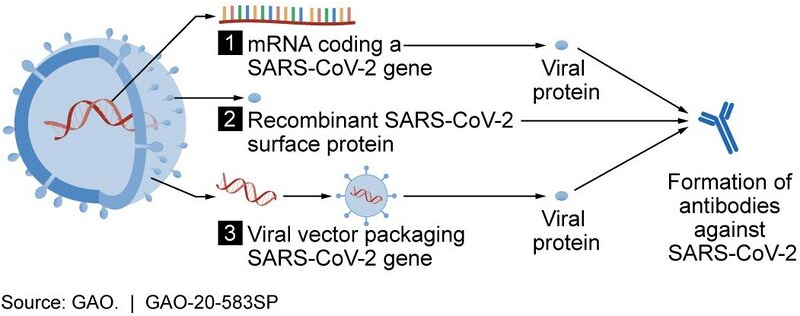
2.5. Technology Platforms
In April, CEPI scientists reported that ten different technology platforms were under research and development during early 2020 to create an effective vaccine against COVID‑19.[14] According to CEPI, the platforms based on DNA or messenger RNA, i.e. DNA vaccines or RNA vaccines, offer considerable promise to alter COVID‑19 antigen functions for strong immune responses, and can be rapidly assessed, refined for long-term stability, and prepared for large-scale production capacity.[14] Other platforms being developed in 2020 focus on non-replicating viral vectors, peptides, recombinant proteins, live attenuated viruses, and inactivated viruses.[14]
In general, the vaccine technologies being developed for COVID‑19 are not like vaccines already in use to prevent influenza, but rather are using "next-generation" strategies for precision on the COVID‑19 infection mechanisms, while hastening development for eventually preventing infection with a new vaccine.[14] Vaccine platforms in development are also designed to address mechanisms for infection susceptibility to COVID‑19 in specific population subgroups, such as the elderly, children, pregnant women, and people with existing weakened immune systems.[14]
3. Vaccine Candidates
CEPI classifies development stages for vaccines as either "exploratory" (planning and designing a candidate, having no evaluation in vivo), "preclinical" (in vivo evaluation with preparation for manufacturing a compound to test in humans), or initiation of Phase I safety studies in healthy people.[14]
Some 205 total vaccine candidates are in early stages of development as either confirmed active projects or in "exploratory" or "preclinical" development, as of mid-July.[2][3]
Phase I trials test primarily for safety and preliminary dosing in a few dozen healthy subjects, while Phase II trials – following success in Phase I – evaluate immunogenicity, dose levels (efficacy based on biomarkers) and adverse effects of the candidate vaccine, typically in hundreds of people.[61][62] A Phase I–II trial consists of preliminary safety and immunogenicity testing, is typically randomized, placebo-controlled, and at multiple sites, while determining more precise, effective doses.[62] Phase III trials typically involve more participants, including a control group, and test effectiveness of the vaccine to prevent the disease (an "interventional" trial), while monitoring for adverse effects at the optimal dose.[61][62]
3.1. Clinical Trials Started in 2020
Phase II-III research is shown in orange; Phase II is in green; Phase I-II is in yellow.
| COVID‑19: candidate vaccines in Phase I–III trials | |||||||
|---|---|---|---|---|---|---|---|
| Vaccine candidate
(developer/sponsor) |
Technology | Phase of trial
(participants) |
Adverse effects | Immune response | Location | Duration | References
and notes |
| AZD1222
(University of Oxford, AstraZeneca) |
adenovirus vector | Phase II–III randomized, interventional (10,260) | ^ spike-specific antibodies at day 28; neutralizing antibodies after a booster dose at day 56 | United Kingdom | May 2020 to August 2021 | [2][49][63] Oxford name: ChAdOx1 nCoV-19; ^ Phase I results, 543 people tested |
|
| Ad5-nCoV
(CanSino Biologics, Institute of Biotechnology of the Academy of Military Medical Sciences) |
recombinant adenovirus type 5 vector | Phase II interventional trial for dosing and side effects (382) | moderate over 7 days: 74% had fever, pain, fatigue[64] | neutralizing antibody and T cell responses[64] | China Canada |
March 2020 to December 2020 | Phase II trial details;[64][65] manufacturing partnership with the National Research Council of Canada and Canadian Center for Vaccinology, Halifax, Nova Scotia[66] |
| mRNA-1273
(Moderna, US National Institute of Allergy and Infectious Diseases, BARDA) |
lipid nanoparticle dispersion containing messenger RNA | Phase II dose-confirmation to evaluate safety, toxicity, and immunogenicity (600) | ^ fever, fatigue, headache, myalgia, and pain at the injection site[67][68] | ^ dose-dependent neutralizing antibody response on two-dose schedule; undetermined durability[67][68] | United States | May 2020 to August 2021 | [2][49][69] ^ preliminary Phase I results, 3 doses, 15 people per group[67] |
| BNT162 (a1, b1, b2, c2)
(BioNTech, Fosun Pharma, Pfizer) |
RNA | Phase I–II of four vaccines, randomized, placebo-controlled, dose-finding, vaccine candidate-selection (7600) | pending Phase I report | pending Phase I report | Germany United States |
April 2020 to May 2021 | [70][71][72][73] |
| CoronaVac
(Sinovac Biotech) |
inactivated SARS-CoV-2 virus | Phase I–II randomized, double-blinded, single-center, placebo-controlled in Xuzhou (744); Phase I–II in Renqiu (422) | pending Phase I report | pending Phase I report | China | April 2020 to December 2020 in Xuzhou; May to July 2020 in Renqiu | [2][49][74][75] |
| INO-4800
(Inovio Pharmaceuticals, CEPI, Korea National Institute of Health, International Vaccine Institute) |
DNA plasmid delivered by electroporation | Phase I–II (40) | pending Phase I report | pending Phase I report | United States South Korea |
April 2020 to November 2020 | South Korean Phase I–II in parallel with Phase I in the US[76][77] |
| unnamed
(Chinese Academy of Medical Sciences) |
inactivated SARS-CoV-2 virus | Phase I–II randomized, double-blinded, single-center, placebo-controlled in Sichuan (942) | China | June 2020 to September 2021 | [2][49][78] | ||
| AG0301-COVID19
(AnGes Inc., AMED) |
DNA plasmid | Phase I–II non-randomized, single-center, two doses (30) | Japan | June 2020 to July 2021 | [3][49][79] | ||
| COVID‑19/aAPC
(Shenzhen Geno-Immune Medical Institute) |
lentiviral vector, pathogen-specific artificial antigen presenting dendritic cells | Phase I (100) | China | March 2020 to 2023 | [2][49][80] | ||
| LV-SMENP-DC
(Shenzhen Geno-Immune Medical Institute) |
lentiviral minigene vaccine, dendritic cells modified with lentiviral vector | Phase I (100) | China | March 2020 to 2023 | [2][49][81] | ||
| unnamed
(Beijing Institute of Biological Products, Wuhan Institute of Biological Products) |
inactivated COVID‑19 virus (vero cells) | Phase I (288) | China | April 2020 to November 2021 | has Phase II design registered for > 1000 participants, including children, not yet recruiting[82][83] | ||
| LNP-nCoVsaRNA
(Medical Research Council Clinical Trials Unit at Imperial College) |
messenger RNA | Phase I randomized trial (105), with dose escalation study (15) and expanded safety study (at least 200) | United Kingdom | June 2020 to July 2021 | [2][49][84] | ||
| NVX-CoV2373
(Novavax) |
SARS-CoV-2 recombinant spike protein nanoparticle with adjuvant | Phase I (131) | Australia | May 2020 to July 2021 | [2][49][85] | ||
| Gam-COVID-Vac Lyo
(Gamaleya Research Institute) |
non-replicating viral vector | Phase I (38) | Russia | June 2020 to August 2020 | [49][86] | ||
| GX-19
(Genexine Consortium, International Vaccine Institute) |
DNA | Phase I (40) | South Korea | June 2020 to June 2022 | [49][87][88] | ||
| SCB-2019
(Clover Biopharm, GlaxoSmithKline) |
spike protein trimeric subunit with GSK adjuvant | Phase I (150) | Australia | June 2020 to March 2021 | [49][89][90] | ||
| COVAX-19
(Vaxine Pty Ltd) |
recombinant protein | Phase I (40) | Australia | June 2020 to July 2021 | [49][91] | ||
| CVnCoV
(CureVac, CEPI) |
messenger RNA | Phase I (168) | Belgium, Germany | June 2020 to August 2021 | [2][49][92] | ||
| unnamed
(Academy of Military Sciences, Walvax Biotech) |
messenger RNA | Phase I (168) | China | June 2020 to December 2021 | [49][93] | ||
| unnamed
(Anhui Zhifei Longcom Biologic Pharmacy Co. Ltd.) |
recombinant protein subunit | Phase I (50) | China | June 2020 to September 2021 | [49][94] | ||
| unnamed
Medicago (Governments of Canada and Quebec) |
plant-derived virus-like particle, recombinant, using GSK adjuvant | Phase I, randomized, dose-ranging (180) | Canada | July 2020 to April 2021 | [49][95][96] VLPs grown in an Australian weed, Nicotiana benthamiana[97] | ||
| COVID‑19: candidate vaccines scheduled for Phase I trials in 2020 |
||
|---|---|---|
| Vaccine candidate
(developer) |
Technology | Start date announced |
| DPX-COVID‑19
(IMV, Inc., |
protein subunit, lipid-based delivery | mid-2020 |
| PittCoVacc
(University of Pittsburgh) |
protein subunit, microneedle arrays | mid-2020 |
| unnamed
(University of Cambridge) |
protein subunit, S protein | mid-2020 |
| LUNAR-COV19
(Arcturus Therapeutics, |
RNA, mRNA | mid-2020 |
| unnamed
(Sanofi Pasteur, GlaxoSmithKline) |
protein subunit, S protein | mid-2020 |
| unnamed
(Cobra Biologics, Karolinska Institute) |
DNA plasmid | mid-2020 |
| unnamed
(Generex Biotech) |
synthetic viral peptides combined with Ii-key immune activation | mid-2020 |
| Ad26
(Janssen (Johnson & Johnson; Beth Israel Deaconess Medical Center) |
non-replicating viral vector[99] | Late July |
| unnamed
(SK Biosciences, Government of Saskatchewan; |
COVID-19 antigen subunits | September |
| CoroFlu
(University of Wisconsin-Madison; |
self-limiting influenza virus | late 2020 |
| unnamed
(Takis; Applied DNA Sciences; Evvivax) |
DNA | late 2020 |
| AdCOVID
(Altimmune; University of Alabama at Birmingham) |
non-replicating viral vector; intranasal | late 2020 |
| unnamed
(Vaxart; Emergent BioSolutions) |
non-replicating viral vector; oral | June-September 2020 |
| unnamed
(VBI Vaccines; National Research Council of Canada) |
pan-coronavirus | No earlier than December 2020 |
3.2. Preclinical Research
In April 2020, the WHO issued a statement representing dozens of vaccine scientists around the world, pledging collaboration to speed development of a vaccine against COVID‑19.[100] The WHO coalition is encouraging international cooperation between organizations developing vaccine candidates, national regulatory and policy agencies, financial contributors, public health associations, and governments, for eventual manufacturing of a successful vaccine in quantities sufficient to supply all affected regions, particularly low-resource countries.[14]
Industry analysis of past vaccine development shows failure rates of 84-90%.[14][58] Because COVID‑19 is a novel virus target with properties still being discovered and requiring innovative vaccine technologies and development strategies, the risks associated with developing a successful vaccine across all steps of preclinical and clinical research are high.[14]
To assess potential for vaccine efficacy, unprecedented computer simulations and new COVID‑19-specific animal models are being developed multinationally during 2020, but these methods remain untested by unknown characteristics of the COVID‑19 virus.[14] Of the confirmed active vaccine candidates, about 70% are being developed by private companies, with the remaining projects under development by academic, government coalitions, and health organizations.[14]
Most of the vaccine developers are small firms or university research teams with little experience in successful vaccine design and limited capacity for advanced clinical trial costs and manufacturing without partnership by multinational pharmaceutical companies.[14] The general geographic distribution of COVID‑19 vaccine development involves organizations in the United States and Canada, together having about 46% of the world's active vaccine research, compared with 36% in Asian countries, including China, and 18% in Europe.[14]
By July, one organization reported that 205 vaccine candidates were in development as either "exploratory/preclinical" projects or in Phase I-III trials in human participants.[2] The table in this section lists emerging vaccine candidates whose Phase I trial starts are scheduled in 2020.
Scheduled Phase I trials in 2020
Many vaccine candidates under design or preclinical development for COVID‑19 will not gain approval for human studies in 2020 due to toxicity, ineffectiveness to induce immune responses or dosing failures in laboratory animals, or because of underfunding.[101][102] The probability of success for an infectious disease vaccine candidate to pass preclinical barriers and reach Phase I of human testing is 41-57%.[101]
Commitment to first-in-human testing of a vaccine candidate represents a substantial capital cost for vaccine developers, estimated to be from US$14 million to US$25 million for a typical Phase I trial program, but possibly as much as US$70 million.[101][103] For comparison, during the Ebola virus epidemic of 2013-16, there were 37 vaccine candidates in urgent development, but only one eventually succeeded as a licensed vaccine, involving a total cost to confirm efficacy in Phase II–III trials of about US$1 billion.[101]
3.3. Non-Specific Vaccines
Some vaccines have non-specific effects beyond the disease they prevent.[104] Two existing vaccines are being tested to determine if either has a protective effect against COVID‑19.
Assertions have been made that COVID‑19 mortality has been lower in countries having routine BCG vaccine administered against tuberculosis,[105][106][107][108] though the World Health Organization (WHO) has said there is no evidence that this vaccine is effective against the COVID‑19 virus.[109]
In March 2020, a randomized trial of BCG vaccine to reduce COVID‑19 illness began in the Netherlands, seeking to recruit 1,000 healthcare workers.[110] A further randomized trial in Australia is seeking to enroll 4,170 healthcare workers.[111][112] A further 700 healthcare workers from Boston and Houston will be recruited in another trial,[113] and 900 healthcare workers in Egypt are to participate in a trial registered by a university in Cairo, Egypt.[114] An additional trial in the Netherlands is testing whether BCG vaccine provides protection for older people, recruiting 1,000 people over 65 years and 600 younger adults.[115] A trial of BCG in 1,000 healthcare workers in Medellín, Colombia was registered on 24 April 2020.[116] Other trials of BCG in healthcare workers were registered in late April – early May: 1,100 participants in Brazil,[117] 1,120 in France,[118] 1,500 in Denmark,[119] and 500 in South Africa.[120] In May 2020, a trial seeking 900 people over 50 in Greece to test BCG vaccine as protection against COVID‑19 was registered.[121]
A randomized placebo-controlled trial to test whether the measles-mumps-rubella vaccine (MMR) can protect healthcare workers from COVID‑19 began with 200 participants during June 2020, in Cairo.[122]
4. Potential Limitations
The rapid development and urgency of producing a vaccine for the COVID‑19 pandemic may increase the risks and failure rate of delivering a safe, effective vaccine.[14][123][124] One study found that between 2006 and 2015, the success rate of obtaining approval from Phase I to successful Phase III trials was 16.2% for vaccines,[58] and CEPI indicates a potential success rate of only 10% for vaccine candidates in 2020 development.[14]
An April 2020 CEPI report stated: "Strong international coordination and cooperation between vaccine developers, regulators, policymakers, funders, public health bodies and governments will be needed to ensure that promising late-stage vaccine candidates can be manufactured in sufficient quantities and equitably supplied to all affected areas, particularly low-resource regions."[14] However, some 10% of the public perceives vaccines as unsafe and unnecessary, refusing vaccination – a global health threat called vaccine hesitancy[125] – which increases the risk of further viral spread that could lead to COVID‑19 outbreaks.[126]
4.1. Biosafety Concern
Early research to assess vaccine efficacy using COVID‑19-specific animal models, such as ACE2-transgenic mice, other laboratory animals, and non-human primates, indicates a need for biosafety-level 3 containment measures for handling live viruses, and international coordination to ensure standardized safety procedures.[14][123]
4.2. Antibody-Dependent Enhancement
Although the quality and quantity of antibody production by a potential vaccine is intended to neutralize the COVID‑19 infection, a vaccine may have unintended effects by causing antibody-dependent disease enhancement (ADE), which is associated with augmentation of virus attachment to its target cells, upregulation of proinflammatory cytokine secretion, and increased organ pathology.[123][127] The vaccine technology platform (for example, viral vector vaccine, spike (S) protein vaccine or protein subunit vaccine), vaccine dose, timing of repeat vaccinations for the possible recurrence of COVID‑19 infection, and elderly age are factors determining the risk and extent of ADE.[123][127] The antibody response to a vaccine is a variable of vaccine technologies in development, including whether the vaccine has precision in its mechanism,[123] and choice of the route for how it is given (intramuscular, intradermal, oral, or nasal).[127][128]
4.3. Causes of Failure
Vaccine failures result from various factors.[128] Inadequacies of the vaccine itself can occur from incomplete technology efficacy (less than 60% efficacy results in failure to create herd immunity),[19] ineffective vaccination route of administration (muscle vs. skin, oral, or nasal), or failed cold chain distribution or storage.[128] Host-("vaccinee")-related determinants that render a person susceptible to infection, such as genetics, health status (underlying disease, nutrition, pregnancy, sensitivities or allergies), immune competence, age, and economic impact or cultural environment can be primary or secondary factors affecting the severity of infection and response to a vaccine.[128] Elderly (above age 60), allergen-hypersensitive, and obese people have susceptibility to compromised immunogenicity, which prevents or inhibits vaccine effectiveness, possibly requiring separate vaccine technologies for these specific populations or repetitive booster vaccinations to limit virus transmission.[128]
Vaccine developers have to invest resources internationally to find enough participants for Phase II-III clinical trials when the virus has proved to be a "moving target" of changing transmission rate across and within countries, forcing companies to compete for trial participants.[129] As an example in June, the Chinese vaccine developer, Sinovac, formed alliances in Malaysia, Canada, the UK, and Brazil among its plans to recruit trial participants and manufacture enough vaccine doses for a possible Phase III study in Brazil where COVID‑19 transmission was accelerating during June.[129][130] As the COVID‑19 pandemic within China became more isolated and controlled, Chinese vaccine developers sought international relationships to conduct advanced human studies in several countries,[130] creating competition for trial participants with other manufacturers and the international Solidarity trial organized by the WHO.[129] In addition to competition over recruiting participants, clinical trial organizers may encounter people unwilling to be vaccinated due to vaccine hesitancy[126] or disbelieving the science of the vaccine technology and its ability to prevent infection.[131]
Having an insufficient number of skilled team members to administer vaccinations may hinder clinical trials that must overcome risks for trial failure, such as recruiting participants in rural or low-density geographic regions, and variations of age, race, ethnicity, or underlying medical conditions.[129] Definition of vaccine safety, efficacy, and clinical endpoints in a Phase III trial may vary between the trials of different companies, such as defining the degree of side effects, infection or amount of transmission, and whether the vaccine prevents moderate or severe COVID‑19 infection.[129][132][133] Further, there exists a possibility the virus will mutate with altered properties and different transmission or infection mechanisms than those the vaccine technology targets.[134][135]
4.4. Cost
An effective vaccine for coronavirus could save trillions of dollars in global economic impact, according to one expert, and would therefore make any price tag in the billions look small in comparison.[136] It is not yet known if it is even scientifically possible to create a vaccine for this virus, and it is not yet known exactly how much the vaccine development will cost.[15] It is possible that billions of dollars could be invested without success.[19][20][24]
The European Commission organized and held a video conference of world leaders on 4 May 2020, at which US$8 billion was raised for coronavirus vaccine development.[137]
After a vaccine is created, billions of doses will need to be manufactured and distributed worldwide. In April 2020, the Gates Foundation estimated that manufacturing and distribution could cost as much as US$25 billion.[138]
5. Proposed Challenge Studies
Strategies are being considered for fast-tracking the licensing a vaccine against COVID‑19, especially by compressing (to a few months) the usually lengthy duration of Phase II–III trials (typically many years).[139][140][141] Challenge studies have been implemented previously for diseases less deadly than COVID‑19 infection, such as common influenza, typhoid fever, cholera, and malaria. Following preliminary proof of safety and efficacy of a candidate vaccine in laboratory animals and healthy humans, controlled challenge studies might be implemented to bypass typical Phase III research, providing an accelerated path to license a COVID‑19 vaccine.[139][140][142]
A challenge study begins by simultaneously testing a vaccine candidate for immunogenicity and safety in laboratory animals and healthy adult volunteers (100 or fewer), something normally a sequential process using animals first. If the initial tests are promising, the study proceeds by rapidly advancing the effective dose into a large-scale Phase II–III trial in previously-uninfected, low-risk volunteers (such as young adults), who would then be deliberately infected with COVID‑19 for comparison with a placebo control group.[139][140][142] Following the challenge, the volunteers would be monitored closely in clinics with life-saving resources, if needed.[139][140] Volunteering for a vaccine challenge study during the COVID‑19 pandemic is likened to the emergency service of healthcare personnel for COVID‑19-infected people, firefighters, or organ donors.[139]
Although challenge studies are ethically questionable due to the unknown hazards for the volunteers of possible COVID‑19 disease enhancement and whether the vaccine received has long-term safety (among other cautions), challenge studies may be the only option to rapidly produce an effective vaccine that will minimize the projected millions of deaths worldwide from COVID‑19 infection,[139][143] according to some infectious disease experts.[139][140][142] The World Health Organization has developed a guidance document with criteria for conducting COVID‑19 challenge studies in healthy people, including scientific and ethical evaluation, public consultation and coordination, selection and informed consent of the participants, and monitoring by independent experts.[144]
6. Commercialization and Equitable Access
6.1. Commercialization Issues
By June 2020, tens of billions of dollars were invested by corporations, governments, international health organizations, and university research groups to develop dozens of vaccine candidates and prepare for global vaccination programs to immunize against COVID‑19 infection.[15][145][146][147] The corporate investment and need to generate value for public shareholders raised concerns about a "market-based approach" in vaccine development, costly pricing of eventual licensed vaccines, preferred access for distribution first to affluent countries, and sparse or no distribution to where the pandemic is most aggressive, as predicted for densely-populated, impoverished countries unable to afford vaccinations.[15][20][146] The collaboration of the University of Oxford with AstraZeneca (a global pharmaceutical company based in the UK) raised concerns about price and sharing of eventual profits from international vaccine sales, arising from whether the UK government and university as public partners had commercialization rights.[147] AstraZeneca stated that initial pricing of its vaccine would not include a profit margin for the company while the pandemic was still expanding.[147]
In early June, AstraZeneca made deals with CEPI and GAVI to manufacture up to two billion vaccine doses if its Oxford vaccine candidate proves safe and effective.[148] Commercialization of pandemic vaccines is a high-risk business venture, potentially losing billions of dollars in development and pre-market manufacturing costs if the candidate vaccines fail to be safe and effective.[15][20][24][145] The multinational pharmaceutical company, Pfizer, indicated it was not interested in a government partnership, which would be a "third party" slowing progress in Pfizer's vaccine program.[149] Further, there are concerns that rapid-development programs – like the Operation Warp Speed plan of the United States – are choosing vaccine candidates mainly for their manufacturing advantages to shorten the development timeline, rather than for the most promising vaccine technology having safety and efficacy.[149]
6.2. Sovereignty
Favored distribution of vaccines within one or a few select countries, called "vaccine sovereignty", is a criticism of some of the vaccine development partnerships,[146][150] such as for the AstraZeneca-University of Oxford vaccine candidate, concerning whether there may be prioritized distribution first within the UK and to the "highest bidder" – the United States, which made an advance payment of US$1.2 billion to secure 300 million vaccine doses for Americans, even before the AstraZeneca-Oxford vaccine or a Sanofi vaccine is proved safe or effective.[147][151][152] Concerns exist about whether some countries producing vaccines may impose protectionist controls by export or import restrictions that would stockpile a COVID‑19 vaccine for their own population.[145][146][150]
In June, the Serum Institute of India – a major manufacturer of global vaccines – received an advance order from AstraZeneca for 100 million vaccine doses, and indicated that doses manufactured in India may go first to Indian citizens, as required by the Indian government.[153][154] Similar preferential homeland distribution may exist if a vaccine is manufactured in Australia.[155] Amid concerns that vaccines produced in China would be distributed there first, the Chinese government pledged in May that a successful Chinese vaccine would become a "global, public good," inferring enough doses would be manufactured for both national and global distribution.[156]
6.3. Equitable Access
As many of the efforts on vaccine candidates have open-ended outcomes, including a high potential for failure during human testing, CEPI, WHO, and charitable vaccine organizations, such as the Gates Foundation and GAVI, raised over US$20 billion during the first half of 2020 to fund vaccine development and preparedness for vaccinations, particularly for children in under-developed countries.[23][29][145][157] CEPI had stated that governments should ensure implementation of a globally-fair allocation system for eventual vaccines, using a coordinated system of manufacturing capacity, financing and purchasing, and indemnification from liability to offset risks taken by vaccine developers.[18] Having been created to monitor fair distribution of infectious disease vaccines to low- and middle-income countries,[150][158] CEPI published guidelines in June for equitable distribution of COVID‑19 vaccines: 1) "prices for vaccines will be set as low as possible for territories that are or may be affected by an outbreak of a disease for which CEPI funding was used to develop a vaccine;" 2) "information, know-how and materials related to vaccine development must be shared with (or transferred to) CEPI" so that it can assume responsibility for vaccine development if a company discontinues expenditures for a promising vaccine candidate; 3) CEPI would have access to, and possible management of, intellectual property rights (i.e., patents) for promising vaccines; 4) "CEPI would receive a share of financial benefits that might accrue from CEPI-sponsored vaccine development, to re-invest in support of its mission to provide global public health benefit"; and 5) data transparency among development partners should maintain the WHO Statement on Public Disclosure of Clinical Trial Results, and encourage publication of results in publicly-available journals.[158] Some vaccine manufacturers opposed parts of these proposals.[146][158]
International groups, such as the Centre for Artistic Activism and Universities Allied for Essential Medicines, advocate for equitable access to licensed COVID‑19 vaccines.[159][160] Scientists have encouraged that the WHO, CEPI, corporations, and governments collaborate to assure evidence-based allocation of eventual COVID‑19 vaccines determined on infection risk,[150][158] particularly urgent vaccinations provided first for healthcare workers, vulnerable populations, and children.[15][145][146]
7. Liability
On 4 February 2020, US Secretary of Health and Human Services Alex Azar published a notice of declaration under the Public Readiness and Emergency Preparedness Act for medical countermeasures against COVID‑19, covering "any vaccine, used to treat, diagnose, cure, prevent, or mitigate COVID‑19, or the transmission of SARS-CoV-2 or a virus mutating therefrom", and stating that the declaration precludes "liability claims alleging negligence by a manufacturer in creating a vaccine, or negligence by a health care provider in prescribing the wrong dose, absent willful misconduct".[161] The declaration is effective in the United States through 1 October 2024.
8. Misinformation
Social media posts have promoted a conspiracy theory that a COVID‑19 vaccine is already available. The patents cited by various social media posts have references to existing patents for genetic sequences and vaccines for other strains such as the SARS coronavirus, but not for COVID‑19.[162][163]
On 21 May 2020, the FDA made public the cease-and-desist notice it had sent to North Coast Biologics, a Seattle-based company that had been selling a purported "nCoV19 spike protein vaccine".[164]
References
- Grenfell, Rob; Drew, Trevor (17 February 2020). "Here's Why It's Taking So Long to Develop a Vaccine for the New Coronavirus". https://www.sciencealert.com/who-says-a-coronavirus-vaccine-is-18-months-away.
- "COVID-19 vaccine development pipeline (Refresh URL to update)". Vaccine Centre, London School of Hygiene and Tropical Medicine. 2020-07-15. https://vac-lshtm.shinyapps.io/ncov_vaccine_landscape/.
- "COVID-19 vaccine tracker (Choose vaccines tab, apply filters to view select data)". Milken Institute. 2020-06-23. https://airtable.com/shrSAi6t5WFwqo3GM/tblEzPQS5fnc0FHYR/viwDBH7b6FjmIBX5x?blocks=hide.
- "Severe acute respiratory syndrome vaccine development: experiences of vaccination against avian infectious bronchitis coronavirus". Avian Pathology 32 (6): 567–82. December 2003. doi:10.1080/03079450310001621198. PMID 14676007. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7154303
- "Effects of a SARS-associated coronavirus vaccine in monkeys". Lancet 362 (9399): 1895–6. December 2003. doi:10.1016/S0140-6736(03)14962-8. PMID 14667748. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7112457
- "Immunogenicity of an adenoviral-based Middle East Respiratory Syndrome coronavirus vaccine in BALB/c mice". Vaccine 32 (45): 5975–82. October 2014. doi:10.1016/j.vaccine.2014.08.058. PMID 25192975. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7115510
- "Development of SARS vaccines and therapeutics is still needed". Future Virology 8 (1): 1–2. January 2013. doi:10.2217/fvl.12.126. PMID 32201503. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7079997
- "SARS (severe acute respiratory syndrome)". National Health Service. 5 March 2020. https://www.nhs.uk/conditions/sars/.
- Shehata, Mahmoud M.; Gomaa, Mokhtar R.; Ali, Mohamed A.; Kayali, Ghazi (20 January 2016). "Middle East respiratory syndrome coronavirus: a comprehensive review". Frontiers of Medicine 10 (2): 120–136. doi:10.1007/s11684-016-0430-6. PMID 26791756. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7089261
- "SARS veterans tackle coronavirus". Nature 490 (7418): 20. October 2012. doi:10.1038/490020a. PMID 23038444. Bibcode: 2012Natur.490...20B. https://dx.doi.org/10.1038%2F490020a
- "Safety and immunogenicity of an anti-Middle East respiratory syndrome coronavirus DNA vaccine: a phase 1, open-label, single-arm, dose-escalation trial". The Lancet. Infectious Diseases 19 (9): 1013–1022. September 2019. doi:10.1016/S1473-3099(19)30266-X. PMID 31351922. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7185789
- "Recent Advances in the Vaccine Development Against Middle East Respiratory Syndrome-Coronavirus". Frontiers in Microbiology 10: 1781. 2019. doi:10.3389/fmicb.2019.01781. PMID 31428074. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=6688523
- "World Health Organization timeline – COVID-19". World Health Organization. 2020-04-27. https://www.who.int/news-room/detail/27-04-2020-who-timeline---covid-19.
- Thanh Le, Tung; Andreadakis, Zacharias; Kumar, Arun; Gómez Román, Raúl; Tollefsen, Stig; Saville, Melanie; Mayhew, Stephen (9 April 2020). "The COVID-19 vaccine development landscape". Nature Reviews Drug Discovery 19 (5): 305–306. doi:10.1038/d41573-020-00073-5. ISSN 1474-1776. PMID 32273591. https://www.nature.com/articles/d41573-020-00073-5. Retrieved 11 April 2020.
- "Responding to Covid-19: A once-in-a-century pandemic?". The New England Journal of Medicine 382 (18): 1677–1679. February 2020. doi:10.1056/nejmp2003762. PMID 32109012. https://dx.doi.org/10.1056%2Fnejmp2003762
- "Covid-19: Navigating the uncharted". The New England Journal of Medicine 382 (13): 1268–1269. March 2020. doi:10.1056/nejme2002387. PMID 32109011. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7121221
- "Update on WHO Solidarity Trial – Accelerating a safe and effective COVID-19 vaccine". World Health Organization. 2020-04-27. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/global-research-on-novel-coronavirus-2019-ncov/solidarity-trial-accelerating-a-safe-and-effective-covid-19-vaccine. "It is vital that we evaluate as many vaccines as possible as we cannot predict how many will turn out to be viable. To increase the chances of success (given the high level of attrition during vaccine development), we must test all candidate vaccines until they fail. [The] WHO is working to ensure that all of them have the chance of being tested at the initial stage of development. The results for the efficacy of each vaccine are expected within three to six months and this evidence, combined with data on safety, will inform decisions about whether it can be used on a wider scale."
- Gavin Yamey, Marco Schäferhoff, Richard Hatchett, Muhammad Pate, Feng Zhao, Kaci Kennedy McDade (2020-05-02). "Ensuring global access to COVID‑19 vaccines". The Lancet 305: 1405–6. https://www.thelancet.com/pdfs/journals/lancet/PIIS0140-6736(20)30763-7.pdf. "CEPI estimates that developing up to three vaccines in the next 12–18 months will require an investment of at least US$2 billion. This estimate includes phase 1 clinical trials of eight vaccine candidates, progression of up to six candidates through phase 2 and 3 trials, completion of regulatory and quality requirements for at least three vaccines, and enhancing global manufacturing capacity for three vaccines.".
- Gates, Bill (2020-04-30). "The vaccine race explained: What you need to know about the COVID-19 vaccine". The Gates Notes. https://www.gatesnotes.com/Health/What-you-need-to-know-about-the-COVID-19-vaccine.
- Sanger, David E.; Kirkpatrick, David D.; Zimmer, Carl; Thomas, Katie; Wee, Sui-Lee (2020-05-02). "With Pressure Growing, Global Race for a Vaccine Intensifies". The New York Times. ISSN 0362-4331. https://www.nytimes.com/2020/05/02/us/politics/vaccines-coronavirus-research.html.
- "Commitment and call to action: Global collaboration to accelerate new COVID-19 health technologies". World Health Organization. 2020-04-24. https://www.who.int/news-room/detail/24-04-2020-commitment-and-call-to-action-global-collaboration-to-accelerate-new-covid-19-health-technologies.
- "Landmark global collaboration launched to defeat COVID-19 pandemic". CEPI. 2020-04-24. https://cepi.net/news_cepi/landmark-global-collaboration-launched-to-defeat-covid-19-pandemic/. "The global nature of a pandemic means that any vaccine or medicine that is successfully developed will be needed immediately all over the world. That means that the challenge we face is not only one of R&D but one of manufacturing at scale, and equitable access."
- Damon Wake (2020-05-04). "World leaders urge cooperation in vaccine hunt, raise $8 billion". Yahoo Finance. https://finance.yahoo.com/news/eu-hosts-virus-telethon-seeking-first-7-5-003500556.html.
- Julie Steenhuysen; Peter Eisler; Allison Martell; Stephanie Nebehay (2020-04-27). "Special Report: Countries, companies risk billions in race for coronavirus vaccine". Reuters. https://www.reuters.com/article/health-coronavirus-vaccine-idUSL2N2CF0JG.
- Hamilton, Isobel Asher (2020-05-01). "Bill Gates thinks there are 8 to 10 promising coronavirus vaccine candidates and one could be ready in as little as 9 months". Business Insider. https://www.businessinsider.com/bill-gates-10-promising-coronavirus-vaccine-candidates-2020-5.
- "GloPID: Novel coronavirus COVID-19". https://www.glopid-r.org/our-work/novel-coronavirus-covid-19.
- "Government of Canada's research response to COVID-19". Government of Canada. 2020-04-23. https://www.canada.ca/en/public-health/services/publications/diseases-conditions/covid-19-government-canada-research-response.html.
- "ISARIC: COVID-19 clinical research resources". ISARIC. 2020-04-27. https://isaric.tghn.org/covid-19-clinical-research-resources/.
- "Global Vaccine Summit 2020: World leaders make historic commitments to provide equal access to vaccines for all". Global Alliance for Vaccines and Immunisation. 2020-06-04. https://www.gavi.org/news/media-room/world-leaders-make-historic-commitments-provide-equal-access-vaccines-all.
- "COVID-19: Gavi steps up response to pandemic". Global Alliance for Vaccines and Immunisation. 2020-04-27. https://www.gavi.org/news/media-room/covid-19-gavi-steps-response-pandemic.
- "Bill & Melinda Gates Foundation pledges US$1.6 billion to Gavi, the Vaccine Alliance, to protect the next generation with lifesaving vaccines". PR Newswire (The Bill & Melinda Gates Foundation). 2020-06-04. https://www.prnewswire.com/news-releases/bill--melinda-gates-foundation-pledges-1-6-billion-to-gavi-the-vaccine-alliance-to-protect-the-next-generation-with-lifesaving-vaccines-301070878.html.
- Abedi, Maham (23 March 2020). "Canada to spend $192M on developing COVID-19 vaccine". Global News. https://globalnews.ca/news/6717883/coronavirus-canada-vaccine-spending/.
- "Government of Canada funds 49 additional COVID-19 research projects – Details of the funded projects". Government of Canada. 23 March 2020. https://www.canada.ca/en/institutes-health-research/news/2020/03/government-of-canada-funds-49-additional-covid-19-research-projects-details-of-the-funded-projects.html.
- Aiello, Rachel (2020-05-04). "'A global challenge': PM Trudeau commits $850 million to global fight against COVID-19". CTV News. https://www.ctvnews.ca/health/coronavirus/a-global-challenge-pm-trudeau-commits-850-million-to-global-fight-against-covid-19-1.4923565.
- Takada, Noriyuki; Satake, Minoru (2020-05-02). "US and China unleash wallets in race for coronavirus vaccine". Nikkei Asian Review. https://asia.nikkei.com/Spotlight/Coronavirus/US-and-China-unleash-wallets-in-race-for-coronavirus-vaccine.
- Yuliya Talmazan, Keir Simmons, Laura Saravia (2020-05-18). "China's Xi announces $2B for coronavirus response as WHO faces calls for investigation". NBC News. https://www.nbcnews.com/news/world/coronavirus-who-faces-global-call-investigation-general-assembly-n1209061.
- "CEPI: Our vaccine and platform portfolio". Coalition for Epidemic Preparedness Innovation (CEPI). 2020-04-30. https://cepi.net/research_dev/our-portfolio/.
- "CEPI collaborates with the Institut Pasteur in a consortium to develop COVID-19 vaccine". Coalition for Epidemic Preparedness Innovations. 19 March 2020. https://cepi.net/news_cepi/cepi-collaborates-with-the-institut-pasteur-in-a-consortium-to-develop-covid-19-vaccine/.
- "Coronavirus: Commission offers financing to innovative vaccines company CureVac". European Commission. 16 March 2020. https://ec.europa.eu/commission/presscorner/detail/en/ip_20_474.
- "Corona-Impfstoff: Bundesregierung beteiligt sich an Impfstoffhersteller CureVac" (in de). Der Spiegel. https://www.spiegel.de/wirtschaft/bundesregierung-beteiligt-sich-an-impfstoffhersteller-curevac-a-2c915a65-5e89-407c-91d0-8fa2200efa00.
- Morriss, Emma (2020-04-22). "Government launches coronavirus vaccine taskforce as human clinical trials start". Pharmafield. https://pharmafield.co.uk/pharma_news/government-launches-coronavirus-vaccine-taskforce-as-human-clinical-trials-start/.
- Gartner, Annelies; Roberts, Lizzie (2020-05-03). "How close are we to a coronavirus vaccine? Latest news on UK trials". The Telegraph. ISSN 0307-1235. https://www.telegraph.co.uk/global-health/science-and-disease/coronavirus-vaccine-uk-trials-news-latest-update-covid-19/.
- "Landmark partnership announced for development of COVID-19 vaccine". University of Oxford. 2020-04-30. http://www.ox.ac.uk/news/2020-04-30-landmark-partnership-announced-development-covid-19-vaccine.
- Kuznia, Robert; Polglase, Katie; Mezzofiore, Gianluca (2020-05-01). "In quest for vaccine, US makes 'big bet' on company with unproven technology". CNN. https://www.cnn.com/2020/05/01/us/coronavirus-moderna-vaccine-invs/index.html.
- Lee, Carol E; Welker, Kristen; Perlmutter-Gumbiner, Elyse (2020-05-01). "Health officials eyeing at least one of 14 potential coronavirus vaccines to fast-track". NBC News. https://www.nbcnews.com/politics/white-house/health-officials-eyeing-least-one-14-potential-coronavirus-vaccines-fast-n1198326.
- Cohen, Jon (2020-05-15). "U.S. 'Warp Speed' vaccine effort comes out of the shadows". Science 368 (6492): 692–693. doi:10.1126/science.368.6492.692. ISSN 0036-8075. PMID 32409451. https://science.sciencemag.org/content/368/6492/692. Retrieved 15 May 2020.
- Justin Sink, Jordan Fabian, Riley Griffin (2020-05-15). "Trump introduces 'Warp Speed' leaders to hasten COVID-19 vaccine". Bloomberg. https://www.bloomberg.com/news/articles/2020-05-15/trump-to-introduce-warp-speed-leaders-after-vaccine-criticism.
- Riley Griffith, Jennifer Jacobs (2020-06-03). "White House Works With Seven Drugmakers in 'Warp Speed' Push". Bloomberg. https://www.bloomberg.com/news/articles/2020-06-03/white-house-working-with-seven-drugmakers-in-warp-speed-push.
- "Draft landscape of COVID 19 candidate vaccines". World Health Organization. 2020-07-21. https://www.who.int/who-documents-detail/draft-landscape-of-covid-19-candidate-vaccines.
- "An international randomised trial of candidate vaccines against COVID-19: Outline of Solidarity vaccine trial". World Health Organization. 2020-04-09. https://www.who.int/blueprint/priority-diseases/key-action/Outline_CoreProtocol_vaccine_trial_09042020.pdf.
- "Adaptive designs in clinical trials: why use them, and how to run and report them". BMC Medicine 16 (1): 29. February 2018. doi:10.1186/s12916-018-1017-7. PMID 29490655. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=5830330
- "Adaptive designs for clinical trials of drugs and biologics: Guidance for industry". U.S. Food and Drug Administration (FDA). 1 November 2019. https://www.fda.gov/media/78495/download.
- McGrail, Samantha (2020-04-15). "Sanofi, GSK partner to develop adjuvanted COVID-19 vaccine". PharmaNewsIntelligence. https://pharmanewsintel.com/news/sanofi-gsk-partner-to-develop-adjuvanted-covid-19-vaccine.
- "R&D Blueprint: A coordinated global research roadmap – 2019 novel coronavirus". World Health Organization. 2020-03-01. https://www.who.int/blueprint/priority-diseases/key-action/Coronavirus_Roadmap_V9.pdf?ua=1.
- Jeong-ho, Lee; Zheng, William; Zhou, Laura (26 January 2020). "Chinese scientists race to develop vaccine as coronavirus death toll jumps". South China Morning Post. https://www.scmp.com/news/china/society/article/3047676/number-coronavirus-cases-china-doubles-spread-rate-accelerates.
- Wee, Sui-Lee (2020-05-04). "China's coronavirus vaccine drive empowers a troubled industry". The New York Times. ISSN 0362-4331. https://www.nytimes.com/2020/05/04/business/coronavirus-china-vaccine.html.
- Simpson, Shmona; Kaufmann, Michael C.; Glozman, Vitaly; Chakrabarti, Ajoy (May 2020). "Disease X: accelerating the development of medical countermeasures for the next pandemic". The Lancet. Infectious Diseases 20 (5): e108–e115. doi:10.1016/S1473-3099(20)30123-7. ISSN 1474-4457. PMID 32197097. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7158580
- "Clinical Development Success Rates 2006-2015". BIO Industry Analysis. June 2016. https://www.bio.org/sites/default/files/Clinical%20Development%20Success%20Rates%202006-2015%20-%20BIO,%20Biomedtracker,%20Amplion%202016.pdf.
- Tom Blackwell (2020-04-20). "COVID-19 vaccine researchers say pandemic lockdown placing many serious obstacles to their work". National Post. https://nationalpost.com/news/covid-19-vaccine-researchers-say-pandemic-lockdown-placing-many-serious-obstacles-to-their-work.
- Justin Chen (2020-05-04). "Covid-19 has shuttered labs. It could put a generation of researchers at risk". STAT. https://www.statnews.com/2020/05/04/coronavirus-lab-shutdowns-impact-on-scientists-research-delays/.
- "Vaccine Safety – Vaccines". US Department of Health and Human Services. https://www.vaccines.gov/basics/safety.
- "The drug development process". U.S. Food and Drug Administration (FDA). 4 January 2018. https://www.fda.gov/patients/learn-about-drug-and-device-approvals/drug-development-process.
- "Investigating a Vaccine Against COVID-19". https://clinicaltrials.gov/ct2/show/NCT04400838.
- Zhu, Feng-Cai; Guan, Xu-Hua; Li, Yu-Hua; Huang, Jian-Ying; Jiang, Tao; Hou, Li-Hua et al. (July 2020). "Immunogenicity and safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine in healthy adults aged 18 years or older: a randomised, double-blind, placebo-controlled, phase 2 trial" (PDF). Lancet. doi:10.1016/s0140-6736(20)31605-6. ISSN 0140-6736. https://www.thelancet.com/action/showPdf?pii=S0140-6736%2820%2931605-6.
- "A Phase II Clinical Trial to Evaluate the Recombinant Novel Coronavirus Vaccine (Adenovirus Vector)". https://clinicaltrials.gov/ct2/show/NCT04341389.
- "The National Research Council of Canada and CanSino Biologics Inc. announce collaboration to advance vaccine against COVID-19". National Research Council, Government of Canada. 2020-05-12. https://www.canada.ca/en/national-research-council/news/2020/05/the-national-research-council-of-canada-and-cansino-biologics-inc-announce-collaboration-to-advance-vaccine-against-covid-19.html.
- "An mRNA Vaccine against SARS-CoV-2 - Preliminary Report" (PDF). New England Journal of Medicine. July 2020. doi:10.1056/NEJMoa2022483. PMID 32663912. https://www.nejm.org/doi/pdf/10.1056/NEJMoa2022483.
- "An mRNA Vaccine against SARS-CoV-2 - Preliminary Report Supplementary appendix". New England Journal of Medicine. July 2020. doi:10.1056/NEJMoa2022483. PMID 32663912. https://www.nejm.org/doi/suppl/10.1056/NEJMoa2022483/suppl_file/nejmoa2022483_appendix.pdf.
- "Safety and Immunogenicity Study of 2019-nCoV Vaccine (mRNA-1273) for Prophylaxis of SARS-CoV-2 Infection (COVID-19)". 25 February 2020. https://clinicaltrials.gov/ct2/show/NCT04283461.
- "EudraCT Number 2020-001038-36". European Union. https://www.clinicaltrialsregister.eu/ctr-search/trial/2020-001038-36/DE.
- "Mainzer Unternehmen Biontech testet Corona-Impfstoff" (in de). swr.online. SWR. 22 April 2020. https://www.swr.de/swraktuell/rheinland-pfalz/mainz/biontech-impfstoff-coronavirus-100.html.
- "Study to Describe the Safety, Tolerability, Immunogenicity, and Potential Efficacy of RNA Vaccine Candidates Against COVID-19 in Healthy Adults". https://clinicaltrials.gov/ct2/show/NCT04368728.
- Sahin, Ugur; Muik, Alexander; Derhovanessian, Evelyna; Vogler, Isabel; Kranz, Lena M; Vormehr, Mathias et al.. "Concurrent human antibody and TH1 type T-cell responses elicited by a COVID-19 RNA vaccine". medRxiv. doi:10.1101/2020.07.17.20140533v1. https://www.medrxiv.org/content/10.1101/2020.07.17.20140533v1.full.pdf.
- "Safety and Immunogenicity Study of 2019-nCoV Vaccine (Inactivated) for Prophylaxis SARS CoV-2 Infection (COVID-19) (Xuzhou)". https://clinicaltrials.gov/ct2/show/NCT04352608.
- "Safety and Immunogenicity Study of 2019-nCoV Vaccine (Inactivated) for Prophylaxis SARS CoV-2 Infection (COVID-19) (Renqui)". https://clinicaltrials.gov/ct2/show/NCT04383574.
- "IVI, INOVIO, and KNIH to partner with CEPI in a Phase I/II clinical trial of INOVIO's COVID-19 DNA vaccine in South Korea". International Vaccine Institute. 2020-04-16. https://www.ivi.int/ivi-inovio-and-knih-to-partner-with-cepi-in-a-phase-i-ii-clinical-trial-of-inovios-covid-19-dna-vaccine-in-south-korea/.
- "Safety, Tolerability and Immunogenicity of INO-4800 for COVID-19 in Healthy Volunteers". https://clinicaltrials.gov/ct2/show/NCT04336410.
- "Safety and Immunogenicity Study of an Inactivated SARS-CoV-2 Vaccine for Preventing Against COVID-19". https://clinicaltrials.gov/ct2/show/NCT04412538.
- "Study of COVID-19 DNA Vaccine (AG0301-COVID19)". https://clinicaltrials.gov/ct2/show/NCT04463472.
- "Safety and Immunity of Covid-19 aAPC Vaccine". https://clinicaltrials.gov/ct2/show/NCT04299724.
- "Immunity and Safety of Covid-19 Synthetic Minigene Vaccine". https://clinicaltrials.gov/ct2/show/NCT04276896.
- "A randomized, double-blind, placebo parallel-controlled phase I/II clinical trial for inactivated novel coronavirus pneumonia vaccine (vero cells)". Chinese Clinical Trial Registry. 2020-04-11. http://www.chictr.org.cn/showprojen.aspx?proj=52227.
- "Draft landscape of COVID-19 candidate vaccines – 2 June 2020". World Health Organization. 2020-06-02. https://www.who.int/who-documents-detail/draft-landscape-of-covid-19-candidate-vaccines.
- "ISRCTN17072692: Clinical trial to assess the safety of a coronavirus vaccine in healthy men and women" (in en). https://www.isrctn.com/ISRCTN17072692.
- "Evaluation of the Safety and Immunogenicity of a SARS-CoV-2 rS (COVID-19) Nanoparticle Vaccine With/Without Matrix-M Adjuvant". https://clinicaltrials.gov/ct2/show/NCT04368988.
- "An Open Study of the Safety, Tolerability and Immunogenicity of "Gam-COVID-Vac Lyo@ Vaccine". https://clinicaltrials.gov/ct2/show/NCT04437875.
- "Safety and Immunogenicity Study of GX-19, a COVID-19 Preventive DNA Vaccine in Healthy Adults". https://clinicaltrials.gov/ct2/show/NCT04445389.
- "S. Korea's Genexine begins human trial of coronavirus vaccine". Reuters. 2020-06-19. https://www.reuters.com/article/health-coronavirus-genexine-vaccine-idUSL4N2DW1T3.
- "SCB-2019 as COVID-19 Vaccine". https://clinicaltrials.gov/ct2/show/NCT04405908.
- "Clover Biopharmaceuticals starts Phase I Covid-19 vaccine trial". Clinical Trials Arena. 2020-06-20. https://www.clinicaltrialsarena.com/news/clover-vaccine-covid-19-trial/.
- "Monovalent Recombinant COVID19 Vaccine". https://clinicaltrials.gov/ct2/show/NCT04453852.
- "A Study to Evaluate the Safety, Reactogenicity and Immunogenicity of Vaccine CVnCoV in Healthy Adults". https://clinicaltrials.gov/ct2/show/NCT04449276.
- "Chinese Clinical Trial Register (ChiCTR) - The world health organization international clinical trials registered organization registered platform". http://www.chictr.org.cn/showprojen.aspx?proj=55524.
- "Phase I Clinical Study of Recombinant Novel Coronavirus Vaccine". https://clinicaltrials.gov/ct2/show/NCT04445194.
- "Safety, Tolerability and Immunogenicity of a Coronavirus-Like Particle COVID-19 Vaccine in Adults Aged 18-55 Years". https://clinicaltrials.gov/ct2/show/NCT04450004.
- St. Philip, Elizabeth; Favaro, Avis; MacLeod, Meredith (2020-07-14). "The hunt for a vaccine: Canadian company begins human testing of COVID-19 candidate". CTV News. https://www.ctvnews.ca/health/coronavirus/the-hunt-for-a-vaccine-canadian-company-begins-human-testing-of-covid-19-candidate-1.5022960.
- Vishwadha Chander (2020-07-14). "Canada's Medicago begins human trials of plant-based COVID-19 vaccine". National Post (Reuters). https://nationalpost.com/news/canada/canadas-medicago-begins-human-trials-of-plant-based-covid-19-vaccine.
- "COVID-19 vaccine and therapeutics tracker". BioRender. 2020-06-21. https://biorender.com/covid-vaccine-tracker.
- Zimmer, Carl (2020-05-20). "Prototype Vaccine Protects Monkeys From Coronavirus" (in en-US). The New York Times. ISSN 0362-4331. https://www.nytimes.com/2020/05/20/health/coronavirus-vaccine-harvard.html.
- "Public statement for collaboration on COVID-19 vaccine development". World Health Organization. 2020-04-13. https://www.who.int/news-room/detail/13-04-2020-public-statement-for-collaboration-on-covid-19-vaccine-development.
- "Estimating the cost of vaccine development against epidemic infectious diseases: a cost minimisation study". Lancet Global Health 6 (12): e1386–96. December 2018. doi:10.1016/S2214-109X(18)30346-2. PMID 30342925. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7164811
- "Early Drug Discovery and Development Guidelines: For Academic Researchers, Collaborators, and Start-up Companies". Assay Guidance Manual. Eli Lilly & Company and the National Center for Advancing Translational Sciences. 1 July 2016. https://www.ncbi.nlm.nih.gov/books/NBK92015/.
- "Innovation in the pharmaceutical industry: New estimates of R&D costs". Journal of Health Economics 47: 20–33. May 2016. doi:10.1016/j.jhealeco.2016.01.012. PMID 26928437. https://dukespace.lib.duke.edu/dspace/bitstream/handle/10161/12742/DiMasi-Grabowski-Hansen-RnD-JHE-2016.pdf;sequence=1. Retrieved 21 April 2020.
- Kleinnijenhuis, J; van Crevel, R; Netea, MG (January 2015). "Trained immunity: consequences for the heterologous effects of BCG vaccination.". Transactions of the Royal Society of Tropical Medicine and Hygiene 109 (1): 29–35. doi:10.1093/trstmh/tru168. PMID 25573107. https://dx.doi.org/10.1093%2Ftrstmh%2Ftru168
- de Vrieze, Jop (23 March 2020). "Can a century-old TB vaccine steel the immune system against the new coronavirus?". Science. doi:10.1126/science.abb8297. https://dx.doi.org/10.1126%2Fscience.abb8297
- "BCG-induced trained immunity: can it offer protection against COVID-19?". Nat. Rev. Immunol. 20 (6): 335–337. June 2020. doi:10.1038/s41577-020-0337-y. PMID 32393823. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7212510
- "BCG vaccine protection from severe coronavirus disease 2019 (COVID-19)". Proc. Natl. Acad. Sci. U.S.A.. July 2020. doi:10.1073/pnas.2008410117. PMID 32647056. https://dx.doi.org/10.1073%2Fpnas.2008410117
- "BCG vaccine and COVID-19: implications for infection prophylaxis and cancer immunotherapy". J Immunother Cancer 8 (2). July 2020. doi:10.1136/jitc-2020-001119. PMID 32636240. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7342862
- "Bacille Calmette-Guérin (BCG) vaccination and COVID-19". 12 April 2020. https://www.who.int/news-room/commentaries/detail/bacille-calmette-gu%C3%A9rin-(bcg)-vaccination-and-covid-19.
- "EudraCT 2020-000919-69". https://www.clinicaltrialsregister.eu/ctr-search/trial/2020-000919-69/NL.
- "Murdoch Children's Research Institute to trial preventative vaccine for COVID-19 healthcare workers". https://www.mcri.edu.au/news/murdoch-children%E2%80%99s-research-institute-trial-preventative-vaccine-covid-19-healthcare-workers.
- "BCG Vaccination to Protect Healthcare Workers Against COVID-19". US National Library of Medicine, National Institutes of Health. https://clinicaltrials.gov/ct2/show/NCT04327206.
- "BCG Vaccine for Health Care Workers as Defense Against SARS-COV2". US National Library of Medicine, National Institutes of Health. https://clinicaltrials.gov/ct2/show/NCT04348370.
- "Application of BCG Vaccine for Immune-prophylaxis Among Egyptian Healthcare Workers During the Pandemic of COVID-19". US National Library of Medicine, National Institutes of Health. https://clinicaltrials.gov/ct2/show/NCT04350931.
- "EudraCT Number 2020-001591-15". https://www.clinicaltrialsregister.eu/ctr-search/trial/2020-001591-15/NL.
- "Performance Evaluation of BCG vs COVID-19". US National Library of Medicine, National Institutes of Health. 24 April 2020. https://clinicaltrials.gov/ct2/show/NCT04362124.
- "COVID-19: BCG As Therapeutic Vaccine, Transmission Limitation, and Immunoglobulin Enhancement". US National Library of Medicine, National Institutes of Health. https://clinicaltrials.gov/ct2/show/NCT04369794.
- "EudraCT Number 2020-001678-31". EU. https://www.clinicaltrialsregister.eu/ctr-search/trial/2020-001678-31/FR.
- "Using BCG Vaccine to Protect Health Care Workers in the COVID-19 Pandemic". US National Library of Medicine, National Institutes of Health. https://clinicaltrials.gov/ct2/show/NCT04373291.
- "BCG Vaccination for Healthcare Workers in COVID-19 Pandemic – Full Text View – ClinicalTrials.gov". US National Library of Medicine, National Institutes of Health. https://clinicaltrials.gov/ct2/show/NCT04379336.
- "Bacillus Calmette-guérin Vaccination to Prevent COVID-19". U.S. National Library of Medicine, National Institutes of Health. https://clinicaltrials.gov/ct2/show/NCT04414267.
- "Measles Vaccine in HCW". US National Library of Medicine, National Institutes of Health. https://clinicaltrials.gov/ct2/show/NCT04357028.
- Diamond, Michael S; Pierson, Theodore C (2020-05-13). "The challenges of vaccine development against a new virus during a pandemic". Cell Host and Microbe. doi:10.1016/j.chom.2020.04.021. PMID 32407708. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7219397
- Thorp, H. Holden (27 March 2020). "Underpromise, overdeliver". Science 367 (6485): 1405. doi:10.1126/science.abb8492. PMID 32205459. https://dx.doi.org/10.1126%2Fscience.abb8492
- "Ten health issues WHO will tackle this year". World Health Organization. 2019. https://www.who.int/emergencies/ten-threats-to-global-health-in-2019.
- Dubé, Eve; Laberge, Caroline; Guay, Maryse; Bramadat, Paul; Roy, Réal; Bettinger, Julie (2013-08-01). "Vaccine hesitancy: an overview". Human Vaccines and Immunotherapeutics 9 (8): 1763–1773. doi:10.4161/hv.24657. ISSN 2164-554X. PMID 23584253. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=3906279
- Iwasaki, Akiko; Yang, Yexin (2020-04-21). "The potential danger of suboptimal antibody responses in COVID-19". Nature Reviews Immunology. doi:10.1038/s41577-020-0321-6. ISSN 1474-1733. PMID 32317716. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=7187142
- Wiedermann, Ursula; Garner-Spitzer, Erika; Wagner, Angelika (2016). "Primary vaccine failure to routine vaccines: Why and what to do?". Human Vaccines and Immunotherapeutics 12 (1): 239–243. doi:10.1080/21645515.2015.1093263. ISSN 2164-554X. PMID 26836329. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=4962729
- Cohen, Jon (2020-06-19). "Pandemic vaccines are about to face the real test". Science 368 (6497): 1295-6. doi:10.1126/science.368.6497.1295. https://science.sciencemag.org/content/368/6497/1295. Retrieved 20 June 2020.
- Hu, Yuwei; Leng, Shumei (2020-06-16). "Brazil joins China in quest for vaccine". Global Times-China. https://www.globaltimes.cn/content/1191829.shtml.
- Jacqueline Howard; Veronica Stracqualursi (2020-06-18). "Fauci warns of 'anti-science bias' being a problem in US". CNN. https://www.cnn.com/2020/06/18/politics/anthony-fauci-coronavirus-anti-science-bias/index.html.
- "How flu vaccine effectiveness and efficacy are measured". Centers for Disease Control and Prevention, National Center for Immunization and Respiratory Diseases, US Department of Health and Human Services. 2016-01-29. https://www.cdc.gov/flu/vaccines-work/effectivenessqa.htm?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fflu%2Fprofessionals%2Fvaccination%2Feffectivenessqa.htm.
- "Principles of epidemiology, Section 8: Concepts of disease occurrence". Centers for Disease Control and Prevention, Center for Surveillance, Epidemiology, and Laboratory Services, US Department of Health and Human Services. 2012-05-18. https://www.cdc.gov/csels/dsepd/ss1978/lesson1/section8.html.
- Alimuddin Zumla; David S Hui; Stanley Perlman (2015-09-11). "Middle East respiratory syndrome". Lancet 386: 995–1007. doi:10.1016/S0140-6736(15)60454-8. PMID 26049252. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=4721578
- "Is the coronavirus mutating? Yes. But here’s why you don’t need to panic". Science News. 26 May 2020. https://www.sciencenews.org/article/coronavirus-covid19-mutations-strains-variants.
- Gates, Bill (2020-04-23). "The first modern pandemic: The scientific advances we need to stop COVID-19". The Gates Notes. https://www.gatesnotes.com/Health/Pandemic-Innovation.
- Stevis-Gridneff, Matina; Jakes, Lara (2020-05-04). "World Leaders Join to Pledge $8 Billion for Vaccine as U.S. Goes It Alone" (in en-US). The New York Times. ISSN 0362-4331. https://www.nytimes.com/2020/05/04/world/europe/eu-coronavirus-vaccine.html.
- Blanchfield, Mike (2020-04-30). "Global philanthropists, experts call for COVID-19 vaccine distribution plan". The Toronto Star. https://www.thestar.com/news/canada/2020/04/30/global-philanthropists-experts-call-for-covid-19-vaccine-distribution-plan.html.
- Eyal, Nir; Lipsitch, Marc; Smith, Peter G. (31 March 2020). "Human challenge studies to accelerate coronavirus vaccine licensure". The Journal of Infectious Diseases 221 (11): 1752–1756. doi:10.1093/infdis/jiaa152. PMID 32232474. PMC 7184325. https://academic.oup.com/jid/advance-article/doi/10.1093/infdis/jiaa152/5814216. Retrieved 19 April 2020.
- "Should scientists infect healthy people with the coronavirus to test vaccines?". Nature 580 (7801): 17. April 2020. doi:10.1038/d41586-020-00927-3. PMID 32218549. https://www.nature.com/articles/d41586-020-00927-3. Retrieved 19 April 2020.
- Eric Boodman (13 March 2020). "Coronavirus vaccine clinical trial starting without usual animal data". STAT. https://www.statnews.com/2020/03/11/researchers-rush-to-start-moderna-coronavirus-vaccine-trial-without-usual-animal-testing/.
- Cohen, Jon (31 March 2020). "Speed coronavirus vaccine testing by deliberately infecting volunteers? Not so fast, some scientists warn". Science. doi:10.1126/science.abc0006. https://www.sciencemag.org/news/2020/03/speed-coronavirus-vaccine-testing-deliberately-infecting-volunteers-not-so-fast-some. Retrieved 19 April 2020.
- Walker, P.; Whittaker, C.; Watson, O.; Baguelin, M.; Ainslie, K.; Bhatia, S.; Bhatt, S.; Boonyasiri, A. et al. (26 March 2020). The global impact of COVID-19 and strategies for mitigation and suppression. Imperial College COVID-19 Response Team. doi:10.25561/77735. https://spiral.imperial.ac.uk:8443/bitstream/10044/1/77735/10/2020-03-26-COVID19-Report-12.pdf. Retrieved 19 April 2020.
- "Key criteria for the ethical acceptability of COVID-19 human challenge studies". World Health Organization. 2020-05-06. https://apps.who.int/iris/bitstream/handle/10665/331976/WHO-2019-nCoV-Ethics_criteria-2020.1-eng.pdf.
- Weintraub, Rebecca; Yadav, Prashant; Berkley, Seth (2020-04-02). "A COVID-19 vaccine will need equitable, global distribution". Harvard Business Review. ISSN 0017-8012. https://hbr.org/2020/04/a-covid-19-vaccine-will-need-equitable-global-distribution.
- "COVID-19 pandemic reveals the risks of relying on private sector for life-saving vaccines, says expert". CBC Radio. 2020-05-08. https://www.cbc.ca/radio/thesundayedition/the-sunday-edition-for-may-10-2020-1.5554451/covid-19-pandemic-reveals-the-risks-of-relying-on-private-sector-for-life-saving-vaccines-says-expert-1.5554463.
- Ahmed, Darius D. (2020-06-04). "Oxford, AstraZeneca COVID-19 deal reinforces 'vaccine sovereignty'". STAT. https://www.statnews.com/2020/06/04/oxford-astrazeneca-covid-19-deal-reinforces-vaccine-sovereignty/.
- Blankenship, Kyle (2020-06-04). "AstraZeneca unveils massive $750M deal in effort to produce billions of COVID-19 shots". FiercePharma. https://www.fiercepharma.com/manufacturing/astrazeneca-unveils-massive-750m-deal-effort-to-produce-billions-covid-19-shots.
- Cohen, Jon (2020-06-04). "Top U.S. scientists left out of White House selection of COVID-19 vaccine short list". Science. doi:10.1126/science.abd1719. ISSN 0036-8075. https://www.sciencemag.org/news/2020/06/top-us-scientists-left-out-white-house-selection-covid-19-vaccine-shortlist. Retrieved 10 June 2020.
- Bollyky, Thomas J.; Gostin, Lawrence O.; Hamburg, Margaret A. (2020-05-07). "The equitable distribution of COVID-19 therapeutics and vaccines". JAMA. doi:10.1001/jama.2020.6641. https://jamanetwork.com/journals/jama/fullarticle/2765944. Retrieved 10 June 2020.
- Aakash, B; Faulconbridge, Guy; Holton, Kate (2020-05-22). "U.S. secures 300 million doses of potential AstraZeneca COVID-19 vaccine". The Guardian. Reuters. https://www.theguardian.pe.ca/news/world/astrazeneca-gets-first-supply-agreements-for-covid-19-vaccine-452225/.
- Paton, James; Griffin, Riley; Koons, Cynthia. "U.S. likely to get Sanofi vaccine first if it succeeds". Bloomberg. https://www.bloombergquint.com/business/u-s-to-get-sanofi-covid-vaccine-first-if-it-succeeds-ceo-says.
- "Serum Institute investing US$100 million on potential COVID-19 vaccine". The Economic Times. 2020-06-09. https://economictimes.indiatimes.com/industry/healthcare/biotech/pharmaceuticals/serum-institute-investing-usd-100-million-on-potential-covid-19-vaccine/articleshow/76288408.cms.
- Roy, Abhirup; Rocha, Euan (2020-05-22). "How one Indian company could be world's door to a COVID-19 vaccine" (in en). Reuters. https://www.reuters.com/article/us-health-coronavirus-india-serum-insigh-idUSKBN22Y2BI.
- Khamsi, Roxanne (2020-04-09). "If a coronavirus vaccine arrives, can the world make enough?" (in en). Nature 580: 578–580. doi:10.1038/d41586-020-01063-8. https://www.nature.com/articles/d41586-020-01063-8. Retrieved 10 June 2020.
- Gretler, Corinne (2020-05-18). "China pledges to make its coronavirus vaccine a 'public good'". The National Post (Bloomberg). https://nationalpost.com/news/world/china-pledges-to-make-its-coronavirus-vaccine-a-public-good.
- "CEPI welcomes UK Government's funding and highlights need for $2 billion to develop a vaccine against COVID-19". Coalition for Epidemic Preparedness Innovations, Oslo, Norway. 6 March 2020. https://cepi.net/news_cepi/2-billion-required-to-develop-a-vaccine-against-the-covid-19-virus/.
- Huneycutt, Brenda; Lurie, Nicole; Rotenberg, Sara; Wilder, Richard; Hatchett, Richard (2020-02-24). "Finding equipoise: CEPI revises its equitable access policy". Vaccine 38 (9): 2144–2148. doi:10.1016/j.vaccine.2019.12.055. PMID 32005536. PMC 7130943. https://linkinghub.elsevier.com/retrieve/pii/S0264410X19317190. Retrieved 10 June 2020.
- "Vaccine for COVID-19". The Center for Artistic Activism. 2020-03-22. https://c4aa.org/2020/03/join-us-free-the-vaccine-for-covid-19.
- "UAEM response to COVID-19". Universities Allied for Essential Medicines. 2020. https://uaem.org/.
- Azar, Alex (4 February 2020). "Notice of Declaration under the Public Readiness and Emergency Preparedness Act for medical countermeasures against COVID-19". https://www.phe.gov/Preparedness/legal/prepact/Pages/COVID19.aspx.
- Kertscher, Tom (23 January 2020). "No, there is no vaccine for the Wuhan coronavirus". Poynter Institute. https://www.politifact.com/factchecks/2020/jan/23/facebook-posts/there-outbreak-china-wuhan-coronavirus-there-not-v/.
- McDonald, Jessica (24 January 2020). "Social Media Posts Spread Bogus Coronavirus Conspiracy Theory". Annenberg Public Policy Center. https://www.factcheck.org/2020/01/social-media-posts-spread-bogus-coronavirus-conspiracy-theory/.
- "WARNING LETTER – North Coast Biologics – MARCS-CMS 607532". U.S. Food and Drug Administration (FDA). 21 May 2020. https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/north-coast-biologics-607532-05212020.





