| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alexander Govaris | + 4160 word(s) | 4160 | 2020-11-19 14:04:41 | | | |
| 2 | Peter Tang | -1290 word(s) | 2870 | 2020-12-03 02:29:13 | | |
Video Upload Options
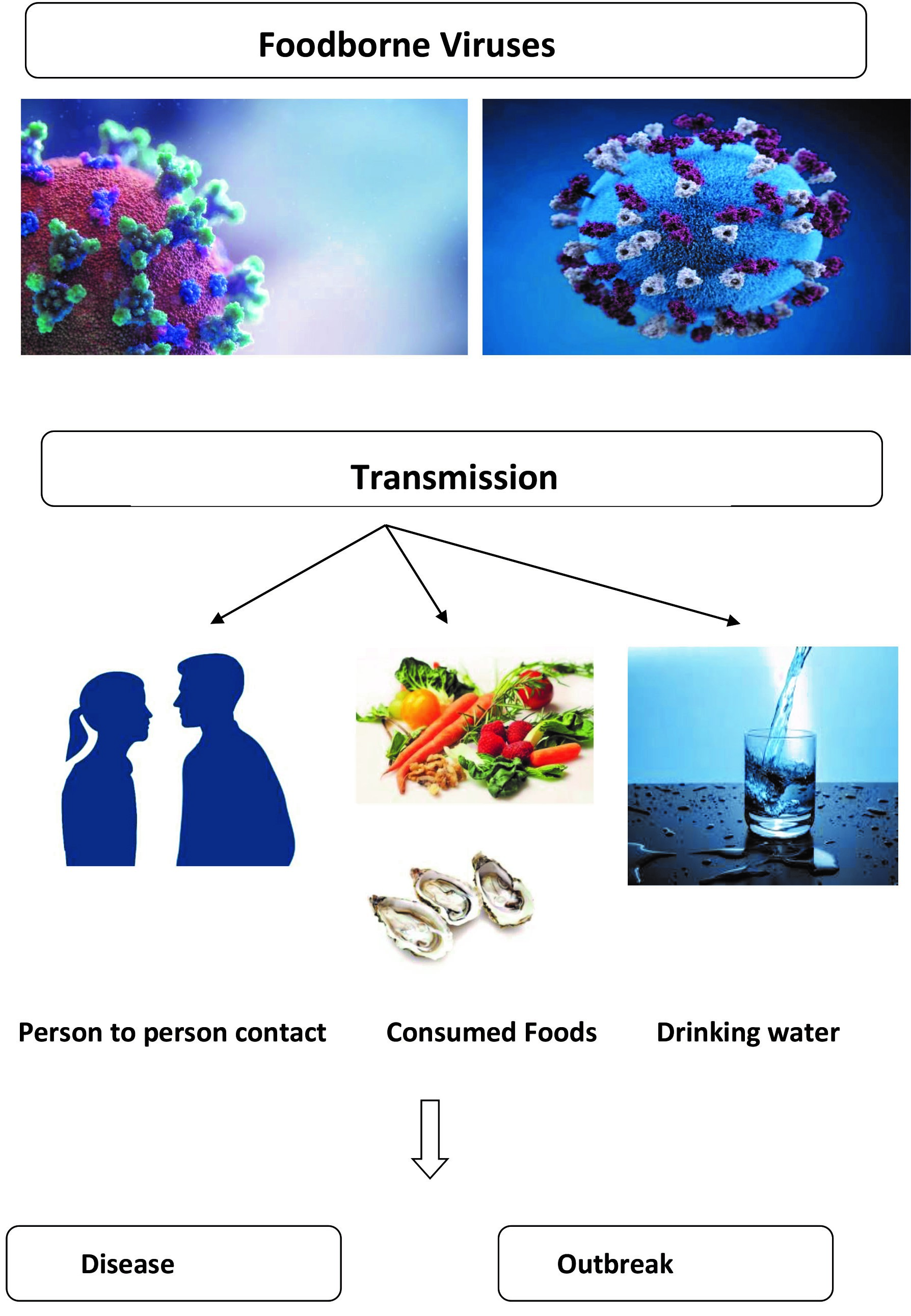
The human norovirus (HuNoV), human rotavirus (HRV), hepatitis A virus (HAV), hepatitis E virus (HEV), human astrovirus (HAstV), Aichi virus (AiV), sapovirus (SaV), human adenovirus (HAdV) and enterovirus (EV) are important foodborne viruses. The majority of the foodborne viruses have a low infection dose, are stable and can survive and persist in foods for a long time without loss of infectivity. Foodborne viruses are usually transmitted to human by consumption of contaminated food, person-to-person contact or drinking contaminated water.
1. Introduction
Among foodborne outbreaks reported worldwide, foodborne viruses' outbreaks have increased in recent years [1]. In contrast to many bacteria or fungi, foodborne viruses cannot grow in foods, they can survive during processing and storage of foods. Consumers can be infected by consumption of virus-contaminated foods.
Enteric viruses such as human noroviruses (HuNoV) and hepatitis A virus (HAV) have been linked with several recorded outbreaks worldwide.Occasional outbreaks worldwide, have been also caused by human astrovirus (HAstV), human rotavirus (HRV), sapovirus (SaV), enterovirus (EV), human adenovirus (HAdV) and Aichi virus (AiV). Human enteric viruses have been reported for 45% and 13.1% of the foodborne outbreaks in USA and EU, respectively [2]. Hepatitis E virus (HEV) has been also considered as an emerging foodborne virus. In 2014, viruses were ranked first (20.4%) among the foodborne outbreaks in the European Union (EU) for the first time [3]. Viruses are transmitted to human by consumption of contaminated food, person-to-person contact or environmental reasons, e.g., water [4].
Viruses can contaminate foods at various steps of their production from pre- to post-harvest. Humans can be infected by the consumption of foods contaminated with viruses. However, the transmissions of viruses via contaminated foods are complicated and usually not clear [5]. A viral transmission to humans from the consumption of contaminated foods depends on various parameters such as virus stability, food processing methods, the infection dose, and the susceptibility of the host [6]. Additionally, food components may protect the virus during the food processing methods and human ingestion. The dose of infection of foodborne viruses is generally low, and almost a small amount of virus can infect humans. Foodborne viruses can survive for a long time in foods without loss of infectivity [1]. In addition, many control strategies that rely on the intrinsic and extrinsic properties of foods. e.g., pH, water activity (aw) for control of bacterial population in food, is ineffective against foodborne viruses [1].
2. Foodborne Viruses
Foodborne viruses and their characteristics are listed in Table 1. These viruses can either be RNA or DNA and single- or double-stranded. They also have different pathology.
2.1. Human Norovirus (HuNoV)
HuNoV, previously known as Norwalk virus, is a non-segmented positive-sense RNA, non-enveloped virus, belonging in the family Caliciviridae. Noroviruses are divided into seven genogroups (GI to GVII) with more than 30 genotypes found worldwide, while GI, GII and GIV genogroups are usually infecting humans [7]. HuNoV is a human enteric pathogen and is recognized as the major etiological agent of acute gastroenteritis outbreaks worldwide; the majority of non-bacterial gastroenteritis outbreaks (90%) in the United States are associated to HuNoV [8]. In the European Union, ΗuNoV has been frequently identified in foodborne and waterborne outbreaks and has been the fourth etiological agent found in foodborne outbreaks [9].
HuNoV is highly contagious, requiring only a low infectious dose (< 102 copies/mL) and can be spread by water, aerosol vomitus droplets, person-to-person contact, and food [10]. The foodborne disease has an incubation period of 1 to 2 days and the symptoms include vomiting, gastralgia, fever, or diarrhea [11]. HuNoV infection may affect any age group, but children of less than 5 years old have the highest rates of HuNoV-associated medical visits [12], while older adults are at greatest risk for HuNoV-associated deaths [13]. Ahmed et al. (2014) reported that the pooled prevalence of norovirus in 187,336 patients with HV infections was 18% between the years 2008-2014 [14].
Raw shellfish, fruits and vegetables pose a significant risk for human HuNoV infection [15]. The prevalence of HuNoV in market shellfish ranged between 3.9% to 54% between the years 2000 to 2018 [16].
Table 1. Foodborne viruses.
|
Viruses |
Particle/ Genome |
Genus/ Family |
Disease |
Infection Dose |
Disease burden |
Associated Foods |
|
Human norovirus (HuNoV) |
Non-enveloped/ ssRNA |
Norovirus/ Caliciviridae |
Acute gastroenteritis |
100 copies/mL |
The leading cause of foodborne illness worldwide. 685 million cases, ca. 200,000 deaths/year. Children < 5 years old: 200 million cases, 70,000 deaths/ year (mostly in developing countries) |
Shellfish oysters, fish, buffet meals, vegetables |
|
Human rotavirus (HRV) |
Non-enveloped/ segmented dsRNA |
Rotavirus/ Reoviridae |
Viral gastroenteritis in children, adult diarrhea |
10–100 infectious viral particles |
The most common cause of severe diarrhoeal disease in children <5 years old: >2 million cases, >200,000 deaths/ year (90% in Africa or Asia) |
Clams and oysters, fruits, vegetables |
|
Hepatitis A (HAV) |
Non-enveloped/ ssRNA |
Hepatovirus/ Picornaviridae |
Hepatitis A |
10–100 viral particles |
ca. 5% of recorded foodborne outbreaks. The leading cause of viral hepatitis. 1.4 million cases, >7,000 deaths / year
|
Sandwiches, fruits, vegetables, milk, shellfish |
|
Human astrovirus (HAtVs) |
Non-enveloped/ ssRNA |
Mamastrovirus/ Astroviridae |
Gastroenteritis |
Unknown; relatively low |
5% infection 2-9% of all acute nonbacterial gastroenteritis in children
|
Bivalve mollusks, fruits, vegetables |
|
Aichi virus (AiV) |
Non-enveloped/ ssRNA |
Kobuvirus/ Picornaviridae |
Gastroenteritis |
Unknown |
0.5–1.8% of gastroenteritis sporadic cases |
Raw shellfish |
|
Hepatitis E (HEV) |
Non-enveloped/ ssRNA |
Orthohepevirus/ Hepeviridae |
Hepatitis E |
Unknown |
20 million infections, ca. 3.3 million symptomatic cases, > 40,000 deaths/year (ca. .3% of the mortality due to viral hepatitis) |
Raw/undercooked boar, deer and pork meat, livers and liver sausages |
|
Sapovirus (SaV) |
Non-enveloped/ ssRNA |
Sapovirus/ Caliciviridae |
Gastroenteritis |
Unknown; likely a low infectious dose similar to that of HuNoV |
2-12% of cases of acute gastroenteritis |
Shellfish (oysters and clams) |
|
Human adenovirus (HAdV) |
Non-enveloped/ dsDNA |
Mastadenovirus/Adenoviridae |
Gastroenteritis, fever, respiratory disease, conjunctivitis, hemorrhagic cystitis, meningoencephalitis |
Unknown |
5–7% of respiratory tract infections in children. The exact prevalence and incidence are unknown
|
Seafoods (shellfish) |
|
Enterovirus (EV) |
Non-enveloped/ ssRNA |
Enterovirus/ Picornaviridae |
Heart disorders hand-foot-and-mouth disease (HFMD), natal sepsis, meningitis/ encephalitis |
Low; 1–10 infectious viral particles |
Millions infections/year, > 90% asymptomatic > 80% in children < 15 years old |
Shellfish (mainly oysters) |
ssRNA: single-stranded RNA, ssDNA: single-stranded DNA.
2.2. Human Rotavirus (HRV)
Rotaviruses are double-stranded non-enveloped RNA viruses, belonging in the family Reoviridae [17]. They are divided into seven groups (A–G); humans are infected by groups A–C, while animals are infected by the rest of the groups. Human rotavirus (HRV) causes severe childhood gastroenteritis and diarrhea in infants and children of less than 5 years old [18]. Group B rotavirus, also known as adult diarrhea rotavirus, has caused major epidemics of severe diarrhea, in thousands of individuals of all ages in China. Group C rotavirus has been associated with rare and sporadic cases of diarrhea in children in many countries and the first outbreak was reported in Japanin 2005 [18].
The most common mode of transmission for HRV is through fecal-oral route, either from person-to-person or contact with contaminated environmental surfaces or through contaminated water and food with faeces [19]. Human and animal rotavirus shed into sewage and the environment can contaminate surface waters, seafood, soft fruits and vegetables. Also infected food handlers may contaminate foods. Outbreaks of waterborne and foodborne HRV infection have been reported in several countries [8]. Transmission through respiratory droplets has also been suggested. In many cases, the root of contaminated food for the HRV transmission to infected persons was unclear [20].
Very low doses of the viruses can be infectious as such as doses of 10-100 viral particles in food or water can cause infection in humans [8]. The incubation period for HRV infection is about 1-3 days [21]. Τhe disease caused by HRV is self-limiting in general, lasting for about 4-7 days and the typical symptoms include watery diarrhea, abdominal pain, vomiting, and dehydration that can lead to a high mortality rate [18].
2.3. Hepatitis A Virus (HAV)
HAV is a 27 nm, non-enveloped, positive-stranded RNA virus, a member of the Picornaviridae family [8]. Human strains are grouped into three genotypes (I–III) and seven subgenotypes (IA–IIIB) based on their genomic characterization. The infectious dose of HAV is low, with 10–100 viral particles.
During the incubation time (range 15–50 days, approximately 28 days), the virus is discarded from the body. HAV can infect humans through the fecal–oral route, direct person-to-person contact or ingestion of contaminated water or food, such as shellfish, fruits or uncooked vegetables [6]. HAV survived for 42 and 20 days in spinach and green onions, respectively [22][23]. Various prevalence ranges for HAV in market shellfish have been reported such as 1% and 26% in Spain (2015) [24] and Tunisia (2006) [25], respectively. In shellfish, HAV survived for almost one month in refrigerated oysters [8]. HAV infection from frozen contaminated shellfish and fruits was also reported [26].
The symptoms of HAV infection include fever, headache, fatigue, nausea, and abdominal discomfort followed by signs of hepatitis 2 to 3 weeks [27]. A lifelong immunity occurs after infection and since HAV is a single serotype, the vaccine of HAV can prevent the disease. AHAV vaccine is commercially available since 1995 [28].
Waterborne and foodborne HAV is estimated to infect 2–7% of the human gastroenteritis patients, and foodborne infection can often result in a larger and prolonged outbreak [8]. In the EU, HAV ranked second, after Salmonella, for the number of hospitalizations with 6.8% of all outbreak-related hospitalizations reported in 2018 [9]. It is important to know that HAV is the most heat-resistant viral pathogen [29].
2.4. Hepatitis E Virus (HEV)
HEV is a single-stranded, non-enveloped, icosahedral RNA virus in the family Hepeviridae, a diverse family of viruses infecting animals and human [30]. Strains of HEV infecting humans belong to the Orthohepevirus genus which is divided into four species (A–D), with human disease being caused by strains within species A, which comprises eight genotypes [31]. Two of these are obligate human pathogens (HEV1, HEV2), and two are endemic in several animal species, such as pigs and wild boar, causing zoonotic infections in humans (HEV3, HEV4). The remaining genotypes appear to be restricted to wild boar (HEV5, HEV6) and camels (HEV7, HEV8), although a case of human HEV7 infection has been reported [32].
HEV spreads via the fecal-oral route from contaminated food and water [33]. The infectious dose of HEV has not been accurately determined; the incubation period is approximately 15 to 60 days. HEV infection usually leads to acute hepatitis. In the first phase (1-10 days), the disease manifests as influenza-like syndrome with abdominal pain, tenderness, nausea, vomiting, and fever, while in the second phase (15–40 days) is characterized with concurrent jaundice, anorexia, hepatomegaly, myalgia, and dark urine [18]. Typically, disease is self-limiting; but mortality rates can become high, particularly among pregnant women and in patients with preexisting liver disease, or may even evolve to a chronic state, especially in immunosuppressed individuals. HEV has been shown to produce a range of extra-hepatic manifestations [34].
HEV is now recognized as the most common cause of acute viral hepatitis worldwide with increasing numbers of autochthonous events occurring globally, including Europe [8][32]. HEVcan be transmitted by contaminated food, as increasingly reported in small outbreaks and sporadic cases. Infection is related to the consumption of undercooked or raw meat from deer and boar [27]. HEV is also present in porcine muscle and has also been detected in commercial livers, sausages, and pork derived foods [33][35]. In addition, since large amounts of viruses excreted in feces, animal manure land application and runoffs can contaminate irrigation and drinking water with concomitant contamination of vegetables or shellfish [36]. Therefore, the animal strains of HEV pose not only a zoonotic risk but also a food safety risk.
2.5. Human Astrovirus (HAstV)
HAstV are small non-enveloped single-stranded positive RNA viruses belonging to the Astroviridae family, genus Mamastrovirus. Classic HAstV is grouped in 8 serotypes and is responsible for 2% to 9% of all acute nonbacterial gastroenteritis infections in the pediatric population worldwide. However, infections in immunocompromised individuals and elderly people are also reported [37]. Type 1 astroviruses are the most prevalent of 8 serotypes across epidemiologic contexts [37]. The infection is transmitted essentially through the fecal-oral route, either directly or by ingestion of food. HAstV is stable in drinking water, fresh surface water, and seawater, indicating another means of transmission. HAstV infection can be caused by a relatively low dose and following an incubation period of 3–4 days, typically, induces a mild, watery diarrhea that lasts 2 to 3 days, associated with vomiting, fever, anorexia, and abdominal pain. Infections are usually self-limiting but can also spread systemically and cause severe infections in immunocompromised patients [34]. In recent years, large foodborne HAstV outbreaks have been observed worldwide. Consumption of contaminated bivalve mollusks were usually associated in several outbreaks, due to their contamination with HAstVs in polluted water [18].
2.6. Aichi Virus (AiV)
AiV is a spherical (ca. 30 nm in diameter) non-enveloped, single-stranded positive sense RNA genome virus classified in the genus Kobuvirus, family Picornaviridae. It was initially found in the Aichi region of Japan, in 1989, from patients suffering from gastro-enteritis infection associated with the consumption of contaminated raw oysters [38].
It has been proposed as causative agentof human gastroenteritis potentially transmitted by fecal–oral routes through contaminated food or water; however, the epidemiology and etiological role of AiV is, to a large extent, unknown [38].
Clinical signs and symptoms of AiV infection, following an incubation period 0.5–2.5 days, include diarrhea, abdominal pain, nausea, vomiting, and fever, reflecting gastroenteritis [39]. Serologic studies indicate that up to 90% of the human population has been exposed to AiV by the age of 40 years indicating widespread exposure. However, a low incidence of AiV infection in patients with either sporadic or epidemic gastroenteritis has been reported [40].
Recent epidemiological studies revealed that the virus could be detected in a higher frequency and abundance than other human enteric viruses. AiV excreted with human feces contaminate waters and it is frequently found in surface waters, in wastewater, in sewage or river water [41]. Humans could be exposed to these viruses in surface waters used for the production of drinking water (after insufficient hygienic treatment) or for recreational purposes and after consumption of raw shellfish cultivated in contaminated surface waters [42]. The vast majority of the recorded AiV outbreaks between the years 1987 to 2007 were associated with the consumptions of contaminated raw oysters [43].
2.7. Sapovirus (SaV)
SaV is a small (30–38 nm in diameter), positive single-stranded RNA genome virus belonging to the genus Sapovirus, and a member of the Caliciviridae family. To date, five genogroups of SaV are recognized, GI to GV. The saVsGI, GII, GIV and GV genogroups infect humans, while the GIII genogroup infects pigs [44].
Originally SaV was found to cause gastroenteritis in children, but it was also observed in gastroenteritis cases in elderly people. SaV is usually transmitted through the fecal–oral route. However, SaV can be also transmitted via contaminated drinking water and food or person-to-person contact [45]. The incubation period is 0.5–2 days and SaV infection manifests with typical viral gastroenteritissymptoms, characterized primarily by diarrhea and vomiting often accompanied with fever, nausea, and abdominal cramps [46]. Duration of disease ranges from 2–6 days [47]. The infectious dose is estimated at 1,015 to 2,800 genomic copies [45].
SaV outbreaks have been increased recently, especially in Japan. SaV have been found in sewage (treated and untreated), river water and shellfish (oysters and clams) [44]. Sporadic cases of SaV infections caused by consumption of contaminated food have been reported [48].
2.8. Human Adenovirus (HAdV)
HAdVs are non-enveloped, double-stranded DNA viruses that can infect humans. They belong to the genus Mastadenovirus, family Adenoviridae. HAdV causes several different clinical syndromes such as gastroenteritis, respiratory disease, hemorrhagic cystitis, hepatitis, meningoencephalitis, and exanthema [49]. It rarely causes serious illness or death, mostly in infants and immunocompromised hosts, or patients who have respiratory or cardiac diseases [49]. HAdV is currently divided into 9 subgroups (A to I); 90 genotypes have been also recognized [50]. The serotypes causing gastroenteritis are 40–41, which belong to species F and are the most common etiological agents (5–20%) of acute gastroenteritis in young children [50] and of several outbreaks in communities such as schools, nurseries and military camps [51].
Usually, after an incubation period of 8 to 10 days, periodic diarrhea occurs, with a low-grade fever, vomiting, abdominal pains, or dehydration and complications of the respiratory system [49]. HAdV infection is likely to be caused via several routes. However, the most common HAdV infection to humans is via the fecal–oral route. Food, particularly contaminated seafoods (shellfish), and water are also vectors of transmission and were also associated with several foodborne outbreaks [51].
2.9. Enterovirus (EV)
Enteroviruses (EVs) are non-enveloped, single-stranded RNA viruses, members of the Picornaviridae family. Four groups of EVs were recognized, namely, Coxsackie A, Coxsackie B, polioviruses and echoviruses, based on the clinical symptoms. Due to sequence diversity, EVs have been also classified into 15 species: rhinovirus A to C and enterovirus A to L. Three rhinovirus species (A to C) and four enterovirus species (A to D) infect millions of individuals worldwide every year [52]. They are known to cause a diverse range of acute infections in humans, such as herpangina, myocarditis and pericarditis, hand-foot-and-mouth disease (HFMD), and neonatal sepsis [53]. They are the most common etiology of meningitis/encephalitis [54].
EVs are transmitted predominantly via the fecal–oral route, but some species can be spread through respiratory secretions. The infection dose is low, 1–10 infectious viral particles. The incubation period is usually 2–5 days, with replication initially occurring in the upper respiratory tract (via inhalation or contact with oral or nasal mucous membranes), or the gastrointestinal tract (via ingestion) [53]. The vast majority of people infected with EV—over 90%—will either have no symptoms or have non-specific symptoms, such as sudden fever. However, EV infection could also manifest with a wide range of symptoms such as mild respiratory symptoms, flu-like illness with fever and muscle aches, fever with a rash, and gastrointestinal symptoms. Most EV infections are shelf-limited within 7–10 days.Foodborne EV outbreaks are linked to the consumption of raw shellfish, mainly oysters from harvest area contaminated by human sewage [2].
References
- Sánchez, G.; Bosch, A. Survival of Enteric Viruses in the Environment and Food. Viruses Foods 2016, 26, 367–392.
- Yeargin, T.; Gibson, K.E. Key characteristics of foods with an elevated risk for viral enteropathogen contamination. J. Appl. Microbiol. 2018, 26, 996–1010.
- EFSA and ECDC (European Food Safety Authority and European Centre for Disease Prevention and Control). The European Union Summary Report on Trends and Sources of Zoonoses, Zoonotic Agents and Food-borne Outbreaks in 2013. EFSA J. 2015, 13, 3991.
- Koopmans, M.; Duizer, E. Foodborne viruses: An emerging problem. Int. J. Food Microbiol. 2004, 90, 23–41.
- Marsh, Z.; Shah, M.P.; Wikswo, M.E.; Barclay, L.; Kisselburgh, H.; Kambhampati, A.; Cannon, J.L.; Parashar, U.D.; Vinjé, J.; Hall, A.J. Epidemiology of foodborne norovirus outbreaks–United States, 2009–2015. Food Saf. 2018, 6, 58–66.
- Bosch, A.; Gkogka, E.; Le Guyader, F.S.; Loisy-Hamon, F.; Lee, A.; van Lieshout, L.; Marthi, B.; Myrmel, M.; Sansom, A.; Schultz, A.C.; et al. Foodborne viruses: Detection, risk assessment, and control options in food processing. Int. J. Food Microbiol. 2018, 285, 110–128.
- Vinje´ J. Advances in laboratory methods for detection and typing of norovirus. J Clin Microbiol. 2015, 53, 373-381.
- Neethirajan, S.; Ahmed, S.R.; Chand, R.; Buozis, J.; Nagy, É. Recent Advances in Biosensor Development for Foodborne Virus Detection. Nanotheranostics 2017, 1, 272–295.
- EFSA (European Food Safety Authority). The European Union One Health 2018 Zoonoses Report. EFSA J. 2019, 17, e05926.
- Lee, HM.; Kwon, J.; Choi, JS., Lee, KH., Yang, S.; Ko, SM.; Chung, J.K.; Cho, SY.; Kim D. Rapid detection of norovirus from fresh lettuce using immunomagnetic separation and a quantum dots assay. J Food Prot. 2013, 76, 707-11.
- Koopmans, M.; von Bonsdorff, C.H.; Vinji, J.; de Medici, D.; Monoroe, S. Foodborne viruses. FEMS Microbiol. Rev. 2002, 26, 187–205.
- Shah, M.P.; Hall, A.J. Norovirus Illnesses in Children and Adolescents. Infect. Dis. Clin. North. Am. 2018, 32, 103–118.
- Cardemil, C.V.; Parashar, U.D.; Hall, A.J. Norovirus Infection in Older Adults: Epidemiology, Risk Factors, and Opportunities for Prevention and Control. Infect. Dis. Clin. North. Am. 2017, 31, 839–870.
- Ahmed, S.M.; Hall, A.J.; Robinson, A.E.; Verhoef, L.; Premkumar, P.; Parashar, U.; Koopmans, M.; Lopman, B.A. Global prevalence of norovirus in cases of gastroenteritis: A systematic review and meta-analysis Lancet. Infect. Dis. 2014, 14(8), pp. 725-730.
- Robilotti, E.; Deresinski, S.; Pinsky, B.A. Norovirus. Clin. Microbiol. Rev. 2015, 28, 134-164.
- Razafimahefa, R.M.; Ludwig-Begall, L.F.; Thiry, E. Cockles and mussels, alive, alive, oh* The role of bivalve molluscs as transmission vehicles for human norovirus infections. Transbound. Emerg. Dis. 2020, 67(S2), 9-25.
- Esona, M.D.; Gautam, R. Rotavirus. Clin. Lab. Med. 2015, 35, 363–391.
- Todd, E.C.D.; Greig, J.D. Viruses of foodborne origin: A review. Virus Adapt. Treat. 2015, 7, 25–45.
- White, P.A.; Netzler, N.E.; Hansman, G.S. Foodborne viral pathogens. Florida, USA: CRC Press. 2017.
- Koopmans, M.; Brown, D. (1999). "Seasonality and diversity of Group A rotaviruses in Europe". Acta Paediatr. 1999, 88(Suppl 426), 14–19.
- Bernstein, D.I. (2009). Rotavirus overview. Pediatr. Infect. Dis. J. 2009, 28, S50-3.
- Shieh, Y.C.; Stewart, D.S.; Laird, D.T. Survival of hepatitis A virus in spinach during low temperature storage. J. Food Prot. 2009, 72, 2390–2393.
- Sun, Y.; Laird, D.T; Shieh, Y.C. Temperature dependent survival of hepatitis A virus during storage of contaminated onions. Appl. Environ. Microbiol. 2012, 78, 4976–4983.
- Polo, D.; Varela, M.F.; Romalde, J.L. Detection and quantification of hepatitis A virus and norovirus in Spanish authorized shellfish harvesting areas. Int. J. Food Microbiol. 2015, 193, 43–50.
- Elamri, D.E.; Aouni, M.; Parnaudeau, S.; Le Guyader, F.S. Detection of human enteric viruses in shellfish collected in Tunisia. Lett. Appl. Microbiol. 2006, 43, 399–404.
- Randazzo, W.; Sanchez, G. Hepatitis A infections from food. J. Appl. Microbiol. 2020, 129, 1120-1132.
- Sanchez-Vega, R.; Elez-Martínez, P.; Martín-Belloso, O. (2014). Influence of high intensity pulsed electric field processing parameters on antioxidant compounds of broccoli juice. Innov. Food Sci. Emerg. Technol. 2014, 29, 70–77.
- Sanchez, G. Processing strategies to inactivate hepatitis A virus in food products: a critical review. Compr. Rev. Food Sci. Food Saf. 2015, 14, 771–784.
- Peng, J.; Tang, J.; Barrett, D.M.; Sablani, S.; Anderson, N.; Powers, J.R. Thermal pasteurization of ready-to-eat foods and vegetables: Critical factors for process design and effects on quality. Crit. Rev. Food Sci. Nutr. 2017, 57, 2970–2995.
- Chen, Q.; Yuan, L.; Wan, J.; Yanlin, C.; Chaoyang, D. Colorimetric detection of hepatitis E virus based on reverse transcription loop mediated isothermal amplification (RT-LAMP) assay. J. Virol. Methods 2014, 197, 29–33.
- Purdy, M.A.; Harrison, T.J.; Jameel, S.; Meng, X.J.; Okamoto, H.; Van der Poel W.H.M.; Smith, D.S. ICTV virus taxonomy profile: hepeviridae. J. Gen. Virol. 2017, 98, 2645–2646.
- Webb, G.W.; Dalton, H.R. Hepatitis E: an underestimated emerging threat. Ther. Adv. Infect. Dis. 2019, 6, 2049936119837162.
- Di Bartolo, I.; Angeloni, G.; Ponterio, E.; Ostanello F., Ruggeri F.M. Detection of hepatitis E virus in pork liver sausages. Int. J. Food Microbiol. 2015, 193, 29-33.
- Bosch, A.; Pinto, R.M; Guix S. Foodborne viruses. Curr. Opin. Food Sci. 2016, 8, 110–119.
- Di Bartolo, I.; Diez-Valcarce, M.; Vasickova, P.; Kralik, P.; Hernandez, M. Angeloni, G.; Ostanello, F.; Bouwknegt, M.; Rodriguez-Lazaro, D.; Pavlik, I.; et al. Hepatitis E virus in pork production chain in Czech Republic, Italy, and Spain, 2010. Emerg. Infect. Dis. 2012; 18, 1282-1289.
- Gao, S.; Li, D.; Zha, E.; Zhou, T.; Wang, S.; Yue, X. Surveillance of hepatitis E virus contamination in shellfish in China. Int. J. Environ. Res. Public. Health 2015, 12, 2026-2036.
- Burbelo, P.D.; Ching, K.H.; Esper, F.; Iadarola, M.J.; Delwart, E.; Lipkin, W.I.; Kapoor, A. Serological studies confirm the novel astrovirus HMOAstV-C as a highly prevalent human infectious agent. PLoS ONE 2011, 6, e22576.
- Kitajima, M.; Gerba, C.P. Aichi virus 1: Environmental occurrence and behavior. Pathogens 2015, 4, 256–268.
- Yamashita, T.; Ito, M.; Tsuzuki, H. Identification of Aichi virus infection by measurement of immunoglobulin responses in an enzyme-linked immunosorbent assay. J. Clin. Microbiol. 2001, 39, 4178–4180.
- Reuter, G.; Boros, A.; Pankovics, P. Kobuviruses—a comprehensive review. Rev. Med. Virol. 2011, 21, 32–41.
- Knight, A.; Li, D.; Uyttendaele, M.; Jaykus, L-A. A critical review of methods for detecting human noroviruses and predicting their infectivity. Crit. Rev. Microbiol. 2013, 39, 295-309.
- Lodder, W.J.; Rutjes, S.A.; Takumi, K.; de Roda Husman, A.M. Aichi virus in sewage and surface water, the Netherlands. Emerg. Infect. Dis. 2013, 19, 1222–1230.
- Rivadulla, E.; Romalde, J.L. A Comprehensive Review on Human Aichi Virus. Virol. Sin.2020, In press.
- D’Souza, D.H. 5-Update on foodborne viruses: Types, concentration and sampling methods. Adv. Microbial. Food Saf. 2015, 2, 102–116.
- Oka, T.; Wang, Q.; Katayama, K.; Saif, L.J. Comprehensive review of human sapoviruses. Clin. Microbiol. Rev. 2015, 28, 32–53.
- Lee, L.E.; Cebelinski, E.A.; Fuller, C.; Keene, W.E.; Smith, K.; Vinjé, J.; Besser, J.M. Sapovirus outbreaks in long-term care facilities, Oregon and Minnesota, USA, 2002-2009. Emerg. Infect. Dis. 2012, 18, 873–6.
- Usuku, S.; Kumazaki, M.; Kitamura, K.; Tochikubo, O.; Noguchi, Y. An outbreak of food-borne gastroenteritis due to sapovirus among junior high school students. Jpn. J. Infect. Dis. 2008, 61, 438-41.
- Miranda, R.C.; Schaffner, D.W. Virus risk in the food supply chain. Curr. Opin. Food Sci.2019, 30, 43-48.
- Sanaei Dashti, A.; Ghahremani, P.; Hashempoor, T.; Karimi, A. Molecular Epidemiology of Enteric Adenovirus Gastroenteritis in under-Five-Year-Old Children in Iran. Gastroenter. Res. Prac. 2016, 2045697.
- Banerjee, A.; De, P.; Manna, B.; Chawla-Sarkar, M. Molecular characterization of enteric adenovirus genotypes 40 and 41 identified in children with acute gastroenteritis in Kolkata, India during 2013–2014. J. Med. Virol. 2017, 89, 606–614.
- Kumthip, K.; Khamrin, P.; Ushijima, H.; Maneekarn, N. Enteric and non-enteric adenoviruses associated with acute gastroenteritis in pediatric patients in Thailand, 2011 to 2017. PLoS ONE 2019, 14, e0220263.
- Chen, B.-S.; Lee, H.-C.; Lee, K.-M.; Gong, Y.-N.; Shih, S.-R. Enterovirus and Encephalitis. Front. Microbiol. 2020, 11, 261.
- Tang, J.W.; Holmes, C.W. Acute and chronic disease caused by enteroviruses. Virulence 2017, 8, 1062–1065.
- Balada-Llasat, J.M.; Rosenthal, N.; Hasbun, R.; Zimmer, L.; Bozzette, S.; Duff, S.; Allison, J.; Bozzette, S. Cost of managing meningitis and encephalitis among infants and children in the United States. Diagn. Microbiol. Infect. Dis. 2019, 93, 349–354.