
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Tongtong Zhu | + 2700 word(s) | 2700 | 2020-09-04 08:27:52 | | | |
| 2 | Catherine Yang | Meta information modification | 2700 | 2020-12-03 05:30:25 | | |
Video Upload Options
Flaviviruses are arthropod-borne RNA viruses that have been used extensively to study host antiviral responses. Often selected just to represent standard single-stranded positive-sense RNA viruses in early studies, the Flavivirus genus over time has taught us how truly unique it is in its remarkable ability to target not just the RNA sensory pathways but also the cytosolic DNA sensing system for its successful replication inside the host cell. This review summarizes the main developments on the unexpected antagonistic strategies utilized by different flaviviruses, with RNA genomes, against the host cyclic GAMP synthase (cGAS)/stimulator of interferon genes (STING) cytosolic DNA sensing pathway in mammalian systems. On the basis of the recent advancements on this topic, we hypothesize that the mechanisms of viral sensing and innate immunity are much more fluid than what we had anticipated, and both viral and host factors will continue to be found as important factors contributing to the host innate immune system in the future.
1. Introduction
Flaviviruses are a group of important emerging and re-emerging pathogens that are mostly arthropod-borne. Several members of the genus, such as dengue virus (DENV), Zika virus (ZIKV), West Nile virus (WNV), yellow fever virus (YFV), and Japanese encephalitis virus (JEV), are major international public health concerns [1][2][3][4][5][6]. Flaviviruses’ viral particles consist of three structural proteins, a lipid envelope, and a positive-sense genomic RNA about 11 kb in length with structurally relevant non-coding regions on both the 3′ and 5′ ends. Their genomes encode a single polyprotein that is processed during and after translation by both host and viral proteases into ten individual proteins (Figure 1) [7].
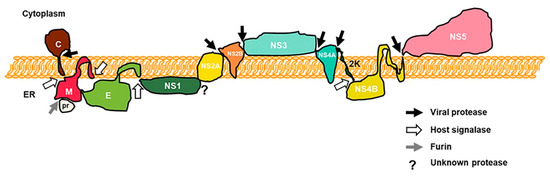
Figure 1. Schematic of a flavivirus polyprotein in the endoplasmic reticulum (ER). Capsid (C), prM, and envelop (E) proteins facilitate viral entry and maturation, while the other seven nonstructural (NS1, NS2A, NS2B, NS3, NS4A, NS4B, and NS5) proteins are responsible for viral replication, viral particle assembly, and maturation. Viral and host proteases cleavage sites are indicated by arrows. NS4A C-terminal transmembrane domain 2K is also shown (adapted from J.R Rodriguez-Madoz, unpublished).
Out of the ten, three are structural proteins (capsid, prM, and envelope) that facilitate viral entry and maturation, and seven are nonstructural proteins (NS1, NS2A, NS2B, NS3, NS4A, NS4B, and NS5) that are responsible for viral replication, viral particle assembly, and maturation [8]. Because of their limited number of proteins, flaviviruses must rely on clever strategies to antagonize host innate and adaptive immune systems to replicate successfully [3][9]. Stimulator of interferon genes (STING), an important adaptor of the human innate immune DNA sensory pathway, has been reported to be targeted for degradation by DENV, an RNA virus, to inhibit innate immune responses [10][11]. These observations led the way for additional studies on flaviviruses’ direct regulation on this host cytosolic DNA sensing system, which will be the focus of this review.
2. Innate Immune System
2.1. Overview
The clearing of an infection caused by pathogens (such as viruses, bacteria, fungi, or parasites) in humans is usually the result of a close collaboration between two major types of immune responses, namely the innate and adaptive immunity. During such an infection, a local innate immune response serves to quickly detect and contain the infection and then, through the help of a heterogeneous group of immune cells (such as Langerhans cells and other dendritic cells, macrophages, B cells, T cells, and others), activate an adaptive immune response that would further control and eventually eliminate the infection [12]. Given that innate immunity plays an essential role in the host’s defense against pathogen invasions, it is perhaps not surprising that many viruses have successfully evolved strategies to regulate or even antagonize the innate immune system during their early stages of infection [13][14]. Robust innate immune responses must rely on an extensive and sensitive network of surveillance proteins, or pattern recognition receptors (PRRs), to detect intruders exhibiting pathogen-associated molecular patterns (PAMPs) and mount proinflammatory and antimicrobial responses. In order to counter them, viruses have evolved ways to avoid detection and make sure they can “fly under the radar” and establish infection in the host [15][16][17].
The list for the currently known innate immune PRRs is extensive and has been steadily expanding in the past three decades since the discovery of the first Toll-like receptors (TLRs) by Hoffmann in 1996 after Janeway’s earlier prediction [18]. Now, the field has advanced tremendously beyond the TLR family, adding new sensors like the nucleotide-binding oligomerization domain-like receptor (NLR) family and its subfamilies (such as NLRP), RIG-I-like receptor (RLR) family, 2′-5′ oligoadenylate synthase (OAS) family, DEAD box polypeptide 41 (DDX41), stimulator of interferon genes (STING), cyclic GAMP synthase (cGAS), IFN-γ-inducible protein 16 (IFI16), and others to the long roster of molecular sentinels watching the host cellular space [19][20][21][22][23][24][25][26]. These receptors and their respective signaling pathways have been actively studied and described based on the pathogen components that they detect (foreign lipids, proteins, or nucleic acid structures or sequences) along with their proposed cellular localizations [27][28].
2.2. Viral Nucleic Acid Sensing
Because in most types of cells, active replication of viruses results in an accumulation of intracellular nucleic acids, both host cytosolic DNA and RNA sensors and their respective pathways have been areas of active investigation in the innate immunity field after their discoveries [29][30]. These nucleic acid sensors are capable of detecting their respective ligands, initiating signaling cascades through interaction with adaptor proteins, triggering expression of type I interferons (IFN-I), and mounting an innate immune response by the release of cellular proinflammatory cytokines and chemokines, all of which are essential in defending against viral infection [14][17][31][32][33][34]. Additionally, in response to IFN-I production, there is an induction of IFN stimulated genes (ISGs) in the same cell and bystander cells that results in an inhibition of viral replication in those cells, which is termed the antiviral state [35]. More recently, the sensors’ ability (or inability) to differentiate self versus non-self elements in the cellular space has caught the attention of a wide number of groups that are actively investigating their roles in mutagenesis and autoimmune diseases [36][37][38][39].
2.3. Innate Immune DNA Sensors
Host DNA sensors known to play important innate immune functions include TLR9, IFI16, absent in melanoma 2 (AIM2), DDX41, cGAS, and others. Detailed reviews of the current list of DNA sensors have been published extensively by different groups [14][34][40][41]. In this review, we will highlight some key players that have been intensely studied in the field of viral innate immunity.
It is worth pointing out that TLR9 mainly serves as the endosomal DNA sensor, while others are known to play parts in detection of DNA in the cytosolic space. After activation by unmethylated CpG-DNA in the endolysosomal system, TLR9 responds by either engaging interferon regulatory factors (IRFs) to induce IFN-I production or recruiting nuclear factor NF-κB, which leads to the production of proinflammatory cytokines [42][43]. IFI16, a member of the IFN-inducible Pyrin and HIN domain (PYHIN) gene family, was first characterized as a nuclear protein as it harbors a nuclear localization signal (NLS) in the N-terminus [44]. Since then, reports have surfaced finding IFI16 in both nuclear and cytoplasm compartments across different cell types [45]. Although more studies are needed to understand the exact mechanisms governing its subcellular localization, IFI16 is known to induce the activation of inflammasomes as well as the IFN-I response via a STING-dependent manner after detecting dsDNA, or other nucleotide structures [46][47][48]. More on the STING activation pathway will be discussed in the next section. AIM2, also a member of the PYHIN family, was characterized as the activator of the inflammasome and pyroptosome responses after cytoplasmic DNA detection [49]. The assembly of inflammasomes leads to the activation of downstream inflammatory caspases, which in turn can mediate important proinflammatory cytokine interleukin-l beta (IL-lβ) and IL-18 production and pyroptosis, a proinflammatory type of cell death [50][51]. DDX41 was first identified as an intracellular DNA sensor in dendritic cells during a siRNA screen among 59 members in the DEAD box family [52]. It can directly bind DNA and STING and activate IFN-I production through a STING-dependent pathway [24]. Z-DNA binding protein 1 (ZBP1, also known as DAI) is a cytosolic DNA sensor that has been implicated in RNA virus sensing (more on ZBP1 and Flavivirus will be discussed later in this review) [53][54][55]. The current list of proposed cytosolic DNA sensors will eventually include proteins that are previously known to have been involved in DNA damage response pathways, transcription, inflammasome assemblies, and other cellular functions [56][57][58][59][60]. Although there are a few publications on some of these DNA sensors and their proposed roles in controlling Flavivirus infection in different biological systems, there is yet to be any evidence of antagonism of these pathways by flaviviruses, other than the cGAS/STING pathway [61][62][63][64].
Among all the known innate immune sensors that recognize DNA, the cGAS/STING pathway has served as the principal human cytosolic DNA sensing system (RIG-I/MAVS pathway being the main cytosolic RNA sensing counterpart) ever since their respective discoveries in 2013 and 2008 [14][23][65][66][67]. It is also the host DNA sensing pathway that has been shown to be most implicated during flaviviruses replication and studied in that context. This innate immune sensory system surveys the host cytosolic space for double-stranded DNA and RNA/DNA hybrids regardless of the specific sequence, whose presence usually indicates a pathogen infection or DNA damage inside the nucleus [37][68][69][70]. Activated cGAS produces secondary messenger 2′3′-cGAMP through dimerization that can diffuse across neighboring cells and bind and activate downstream adaptor STING [71][72].
STING (also known as MITA, MPYS, ERIS, or TMEM173) is a transmembrane innate immune signaling adaptor that relays alert signals from activated cGAS to IFN-I and NF-κB pathways in order to mount an inflammatory response against the detected danger signal [31][32]. At resting state, STING associates with stromal interaction molecule 1 (STIM1) until it interacts with cGAMP (produced by cGAS or endogenously by bacteria), which disrupts this interaction and induces STING to translocate from the ER to the ER-Golgi intermediate compartment (ERGIC) to recruit and activate TANK-binding kinase 1 (TBK1), followed by the recruitment and activation of IRF3. The dimerized IRF3 can then translocate into the nucleus and promote IFN-I expression, which leads to a global upregulation of a set of ISGs critical for mounting an effective innate immune defense against sources of danger [72][73][74]. Aside from cGAS, STING seems to also play a part in downstream signaling of other DNA sensors (such as DDX41 and IFI16) [20][24][56][75].
In addition to its canonical role played in the cytosolic DNA sensory pathway, STING also was reported to interact with mitochondrial antiviral signaling protein (MAVS, also known as VISA, IPS-1, or Cardif) both in the presence and absence of Sendai virus (SeV) infection and with RIG-I during SeV infection [65]. In another report, endogenous STING was shown to associate with RIG-I in normal human umbilical vein endothelial cells (HUVECs) either directly or indirectly as a complex [67]. Other groups have since reported more evidence on this interaction [76][77]. However, the exact role STING plays in cytosolic RNA sensing and anti-RNA virus defense is still unclear and needs further exploration [78].
As we learn more about the mechanisms behind cGAS/STING sensing and activation, it has become increasingly clear that this pathway plays an essential role in host innate immunity, especially in regulating antiviral responses [79]. This is perhaps why many viruses, mostly DNA viruses, are found to have evolved strategies to target this pathway for disruption. An excellent review of different ways DNA viruses sabotage this cellular information relay highway was published by Ahn and Barber [80]. For more on the history of the cGAS/STING signaling pathway’s discovery and what we currently know about its various broader cellular functions, readers are suggested to consult this detailed review [81]. For the remainder of this review, the focus will be on how one specific genus of RNA virus, namely flaviviruses, has helped widen our understanding of the scope of cGAS/STING innate immune sensing in the context of antiviral defense. We will focus on several members of this genus and their different strategies in regulating cGAS or STING to illustrate the current knowledge on this topic (summarized in Table 1).
Table 1. Mechanisms of known cyclic GAMP synthase (cGAS)/stimulator of interferon genes (STING) antagonism by flaviviruses. NS, nonstructural; DENV, dengue virus; ZIKV, Zika virus; WNV, West Nile virus; YFV, yellow fever virus; JEV, Japanese encephalitis virus; IFN, interferon; TBK1, TANK-binding kinase 1.
| Genus | Species | Protein | Mechanism of cGAS/STING Antagonism | References |
|---|---|---|---|---|
| Flavivirus | DENV | NS2B | Targets cGAS for degradation through an autophagy–lysosome-dependent mechanism. | [82] |
| NS2B3 | Proteolytically degrades human STING, but not mouse STING. | [10][11][83][84][85][86] | ||
| ZIKV | NS1 | Indirectly degrades cGAS via the enhanced stabilization of caspase-1. | [87] | |
| NS2B3 | Proteolytically degrades human STING, but not mouse STING. | [85] | ||
| WNV | NS2B3 | Proteolytically degrades human STING, but not mouse STING. | [85] | |
| YFV | NS4B | Unknown | [19] | |
| JEV | NS2B3 | Proteolytically degrades human STING, but not mouse STING. | [85] | |
| DTMUV * | NS2A | Competes with duck TBK1 to bind with duck STING, thus disrupting its dimerization and inhibiting downstream IFN production. | [88] | |
| NS2B3 | Proteolytically degrades duck STING through an NS2B dependent manner with the binding of NS2B with duck STING being required for cleaving | [89] |
3. Perspectives
It remains elusive why a whole genus of single-stranded positive-sensed RNA viruses has evolved a seemingly conserved mechanism targeting an important, mostly cytosolic DNA sensing system. This is also the case for some other RNA viruses, including coronaviruses (SARS-CoV), that are capable of targeting STING [77][90][91][92].
If a DNA virus evolves mechanisms to co-opt, for example, the RIG-I/MAVS pathway, it could be reasoned that DNA viruses have RNA forms as an intermediary step during active infection and that could serve as ligands for the host cytosolic RNA sensing mechanism and alert the cell to mount an antiviral response [93]. Nevertheless, none of the flaviviruses mentioned are known to have a DNA intermediary phase during infection. If the cGAS/STING pathway is triggered during flaviviruses replication, what could serve as the danger signal?
We offered the hypothesis that host mtDNA leakage can occur during DENV replication, and the resultant misplaced mtDNA in the cytosol can serve as ligands that alert the cytosolic innate immune DNA sensors [82][94]. This hypothesis was based on the previous observation that DENV NS4B can disrupt the integrity of mitochondria and dampen innate immune signaling [95]. After using transduced cGAS cells and cGAS pulldown assays, our group provided evidence that mtDNA can be readily detected by cGAS during DENV infection [82]. Together with the recent report that IL-lβ is responsible for inducing the release of mtDNA during DENV infection and subsequent innate immune response activation, mtDNA’s role in acting as the host ligand for cytosolic DNA sensors is further confirmed [96][97]. Nevertheless, is mtDNA the only source of agonist inducing cGAS/STING response during Flavivirus infection?
To our knowledge, a comprehensive screening followed by functional validation of putative cGAS ligands of either viral or host origin under viral infection conditions has not yet been done, even when new evidence of host nucleic acid elements modulating and regulating immune responses is increasingly surfacing in recent years [98][99][100][101][102][103][104][105]. One particular field that we propose would broaden our understanding of innate immunity is at the interdisciplinary space between host innate immune sensing and endogenous retroelements (EREs). Although these elements consist of a large part of our genome, we know remarkably little about their potential regulations and functions, especially during viral infections. Growing evidence suggests that viral infections can impact ERE expression levels, and some non-retroviral RNA viruses can even be reverse-transcribed by the host EREs under certain conditions [100][103][105][106][107].
In particular, EREs derived from non-retroviral RNA viruses in insect systems have been actively studied in recent years and are found to provide partial protection against parent viruses. The Saleh group has reported this phenomenon in the form of finding reverse-transcribed viral DNA (from RNA of insect flaviviruses) capable of inducing virus-specific siRNA responses amplification in a range of insect cells [108][109][110][111]. Even when more studies are warranted before a definitive antiviral or proviral role can be ascribed to EREs in mammalian systems during infection, we hypothesize that future studies may show more host nucleic acids can act as agonists for the cGAS/STING pathway during RNA virus infection. This would not only serve to explain further the seemingly paradoxical conserved strategy in silencing the cytosolic DNA sensing pathway by flaviviruses, but also open up a new direction for cGAS/STING research in general.
References
- Tomori, O. Yellow fever: The recurring plague. Crit. Rev. Clin. Lab. Sci. 2004, 41, 391–427.
- Mackenzie, J.S.; Gubler, D.J.; Petersen, L.R. Emerging flaviviruses: The spread and resurgence of Japanese encephalitis, West Nile and dengue viruses. Nat. Med. 2004, 10, S98–S109.
- Fernandez-Garcia, M.-D.; Mazzon, M.; Jacobs, M.; Amara, A. Pathogenesis of Flavivirus Infections: Using and Abusing the Host Cell. Cell Host Microbe 2009, 5, 318–328.
- Tilak, R.; Ray, S.; Tilak, V.W.; Mukherji, S. Dengue, chikungunya … and the missing entity—Zika fever: A new emerging threat. Med. J. Armed. Forces India 2016, 72, 157–163.
- Habarugira, G.; Suen, W.W.; Hobson-Peters, J.; Hall, R.A.; Bielefeldt-Ohmann, H. West Nile Virus: An Update on Pathobiology, Epidemiology, Diagnostics, Control and “One Health” Implications. Pathogens 2020, 9, 589.
- Simon, L.V.; Sandhu, D.S.; Goyal, A.; Kruse, B. Encephalitis, Japanese. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020.
- Pierson, T.C.; Diamond, M.S. Flaviviruses. In Fields Virology, 6th ed.; Knipe, D.M., Howley, P.M., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; Volume 1, pp. 747–794.
- Shah, P.S.; Link, N.; Jang, G.M.; Sharp, P.P.; Zhu, T.; Swaney, D.L.; Johnson, J.R.; Von Dollen, J.; Ramage, H.R.; Satkamp, L.; et al. Comparative Flavivirus-Host Protein Interaction Mapping Reveals Mechanisms of Dengue and Zika Virus Pathogenesis. Cell 2018, 175, 1931–1945.
- Miorin, L.; Maestre, A.M.; Fernandez-Sesma, A.; García-Sastre, A. Antagonism of type I interferon by flaviviruses. Biochem. Biophys. Res. Commun. 2017, 492, 587–596.
- Yu, C.-Y.; Chang, T.-H.; Liang, J.-J.; Chiang, R.-L.; Lee, Y.-L.; Liao, C.-L.; Lin, Y.-L. Dengue Virus Targets the Adaptor Protein MITA to Subvert Host Innate Immunity. PLoS Pathog. 2012, 8, e1002780.
- Aguirre, S.; Maestre, A.M.; Pagni, S.; Patel, J.R.; Savage, T.; Gutman, D.; Maringer, K.; Bernal-Rubio, D.; Shabman, R.S.; Simon, V.; et al. DENV Inhibits Type I IFN Production in Infected Cells by Cleaving Human STING. PLoS Pathog. 2012, 8, e1002934.
- Murphy, K.; Weaver, C. Janeway’s Immunobiology, 9th ed.; Garland Science: New York, NY, USA; London, UK, 2017.
- Lei, J.; Hilgenfeld, R. RNA-virus proteases counteracting host innate immunity. FEBS Lett. 2017, 591, 3190–3210.
- Ma, Z.; Ni, G.; Damania, B. Innate Sensing of DNA Virus Genomes. Annu. Rev. Virol. 2018, 5, 341–362.
- Chan, Y.K.; Gack, M.U. Viral evasion of intracellular DNA and RNA sensing. Nat. Rev. Microbiol. 2016, 14, 360–373.
- Christensen, M.H.; Paludan, S.R. Viral evasion of DNA-stimulated innate immune responses. Cell Mol. Immunol. 2017, 14, 4–13.
- Chen, N.; Xia, P.; Li, S.; Zhang, T.; Wang, T.T.; Zhu, J. RNA sensors of the innate immune system and their detection of pathogens. IUBMB Life 2017, 69, 297–304.
- Lemaitre, B.; Nicolas, E.; Michaut, L.; Reichhart, J.-M.; Hoffmann, J.A. The Dorsoventral Regulatory Gene Cassette spätzle/Toll/cactus Controls the Potent Antifungal Response in Drosophila Adults. Cell 1996, 86, 973–983.
- Ishikawa, H.; Ma, Z.; Barber, G.N. STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature 2009, 461, 788–792.
- Unterholzner, L.; Keating, S.E.; Baran, M.; Horan, K.A.; Jensen, S.B.; Sharma, S.; Sirois, C.M.; Jin, T.; Latz, E.; Xiao, T.S.; et al. IFI16 is an innate immune sensor for intracellular DNA. Nat. Immunol. 2010, 11, 997–1004.
- Kumar, H.; Kawai, T.; Akira, S. Pathogen Recognition by the Innate Immune System. Int. Rev. Immunol. 2011, 30, 16–34.
- Loo, Y.-M.; Gale, M., Jr. Immune signaling by RIG-I-like receptors. Immunity 2011, 34, 680–692.
- Sun, L.; Wu, J.; Du, F.; Chen, X.; Chen, Z.J. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science 2013, 339, 786–791.
- Jiang, Y.; Zhu, Y.; Liu, Z.-J.; Ouyang, S. The emerging roles of the DDX41 protein in immunity and diseases. Protein Cell 2017, 8, 83–89.
- Dolasia, K.; Bisht, M.K.; Pradhan, G.; Udgata, A.; Mukhopadhyay, S. TLRs/NLRs: Shaping the landscape of host immunity. Int. Rev. Immunol. 2018, 37, 3–19.
- Schwartz, S.L.; Conn, G.L. RNA regulation of the antiviral protein 2’-5’-oligoadenylate synthetase. Wiley Interdiscip. Rev. RNA 2019, 10, e1534.
- Kumar, H.; Kawai, T.; Akira, S. Toll-like receptors and innate immunity. Biochem. Biophys. Res. Commun. 2009, 388, 621–625.
- Takeuchi, O.; Akira, S. Pattern Recognition Receptors and Inflammation. Cell 2010, 140, 805–820.
- Vasseur, E.; Patin, E.; Laval, G.; Pajon, S.; Fornarino, S.; Crouau-Roy, B.; Quintana-Murci, L. The selective footprints of viral pressures at the human RIG-I-like receptor family. Hum. Mol. Genet. 2011, 20, 4462–4474.
- Dempsey, A.; Bowie, A.G. Innate immune recognition of DNA: A recent history. Virology 2015, 479–480, 146–152.
- Burdette, D.L.; Vance, R.E. STING and the innate immune response to nucleic acids in the cytosol. Nat. Immunol. 2013, 14, 19–26.
- Maringer, K.; Fernandez-Sesma, A. Message in a bottle: Lessons learned from antagonism of STING signalling during RNA virus infection. Cytokine Growth Factor Rev. 2014, 25, 669–679.
- Patel, J.R.; García-Sastre, A. Activation and regulation of pathogen sensor RIG-I. Cytokine Growth Factor Rev. 2014, 25, 513–523.
- Abe, T.; Marutani, Y.; Shoji, I. Cytosolic DNA-sensing immune response and viral infection. Microbiol. Immunol. 2019, 63, 51–64.
- Jin, S. The Cross-Regulation Between Autophagy and Type I Interferon Signaling in Host Defense. Adv. Exp. Med. Biol. 2019, 1209, 125–144.
- Smith, S.; Jefferies, C. Role of DNA/RNA sensors and contribution to autoimmunity. Cytokine Growth Factor Rev. 2014, 25, 745–757.
- Gao, D.; Li, T.; Li, X.-D.; Chen, X.; Li, Q.-Z.; Wight-Carter, M.; Chen, Z.J. Activation of cyclic GMP-AMP synthase by self-DNA causes autoimmune diseases. Proc. Natl. Acad. Sci. USA 2015, 112, E5699–E5705.
- Yang, H.; Wang, H.; Ren, J.; Chen, Q.; Chen, Z.J. cGAS is essential for cellular senescence. Proc. Natl. Acad. Sci. USA 2017, 114, E4612–E4620.
- Liu, H.; Zhang, H.; Wu, X.; Ma, D.; Wu, J.; Wang, L.; Jiang, Y.; Fei, Y.; Zhu, C.; Tan, R.; et al. Nuclear cGAS suppresses DNA repair and promotes tumorigenesis. Nature 2018, 563, 131–136.
- Takaoka, A.; Shinohara, S. DNA sensors in innate immune system. Uirusu 2008, 58, 37–46.
- Unterholzner, L. The interferon response to intracellular DNA: Why so many receptors? Immunobiology 2013, 218, 1312–1321.
- Sasai, M.; Linehan, M.M.; Iwasaki, A. Bifurcation of Toll-Like Receptor 9 Signaling by Adaptor Protein 3. Science 2010, 329, 1530.
- Marongiu, L.; Gornati, L.; Artuso, I.; Zanoni, I.; Granucci, F. Below the surface: The inner lives of TLR4 and TLR9. J. Leukoc. Biol. 2019, 106, 147–160.
- Briggs, L.J.; Johnstone, R.W.; Elliot, R.M.; Xiao, C.Y.; Dawson, M.; Trapani, J.A.; Jans, D.A. Novel properties of the protein kinase CK2-site-regulated nuclear- localization sequence of the interferon-induced nuclear factor IFI 16. Biochem. J. 2001, 353, 69–77.
- Veeranki, S.; Choubey, D. Interferon-inducible p200-family protein IFI16, an innate immune sensor for cytosolic and nuclear double-stranded DNA: Regulation of subcellular localization. Mol. Immunol. 2012, 49, 567–571.
- Jakobsen, M.R.; Bak, R.O.; Andersen, A.; Berg, R.K.; Jensen, S.B.; Jin, T.; Laustsen, A.; Hansen, K.; Østergaard, L.; Fitzgerald, K.A.; et al. IFI16 senses DNA forms of the lentiviral replication cycle and controls HIV-1 replication. Proc. Natl. Acad. Sci. USA 2013, 110, E4571–E4580.
- Jakobsen, M.R.; Paludan, S.R. IFI16: At the interphase between innate DNA sensing and genome regulation. Cytokine Growth Factor Rev. 2014, 25, 649–655.
- Dell’Oste, V.; Gatti, D.; Giorgio, A.G.; Gariglio, M.; Landolfo, S.; De Andrea, M. The interferon-inducible DNA-sensor protein IFI16: A key player in the antiviral response. New Microbiol. 2015, 38, 5–20.
- Fernandes-Alnemri, T.; Yu, J.W.; Datta, P.; Wu, J.; Alnemri, E.S. AIM2 activates the inflammasome and cell death in response to cytoplasmic DNA. Nature 2009, 458, 509–513.
- Man, S.M.; Karki, R.; Kanneganti, T.D. AIM2 inflammasome in infection, cancer, and autoimmunity: Role in DNA sensing, inflammation, and innate immunity. Eur. J. Immunol. 2016, 46, 269–280.
- Lugrin, J.; Martinon, F. The AIM2 inflammasome: Sensor of pathogens and cellular perturbations. Immunol. Rev. 2018, 281, 99–114.
- Zhang, Z.; Yuan, B.; Bao, M.; Lu, N.; Kim, T.; Liu, Y.J. The helicase DDX41 senses intracellular DNA mediated by the adaptor STING in dendritic cells. Nat. Immunol. 2011, 12, 959–965.
- Takaoka, A.; Wang, Z.; Choi, M.K.; Yanai, H.; Negishi, H.; Ban, T.; Lu, Y.; Miyagishi, M.; Kodama, T.; Honda, K.; et al. DAI (DLM-1/ZBP1) is a cytosolic DNA sensor and an activator of innate immune response. Nature 2007, 448, 501–505.
- Thapa, R.J.; Ingram, J.P.; Ragan, K.B.; Nogusa, S.; Boyd, D.F.; Benitez, A.A.; Sridharan, H.; Kosoff, R.; Shubina, M.; Landsteiner, V.J.; et al. DAI Senses Influenza A Virus Genomic RNA and Activates RIPK3-Dependent Cell Death. Cell Host Microbe 2016, 20, 674–681.
- Kuriakose, T.; Man, S.M.; Malireddi, R.K.; Karki, R.; Kesavardhana, S.; Place, D.E.; Neale, G.; Vogel, P.; Kanneganti, T.D. ZBP1/DAI is an innate sensor of influenza virus triggering the NLRP3 inflammasome and programmed cell death pathways. Sci. Immunol. 2016, 1, aag2045.
- Ferguson, B.J.; Mansur, D.S.; Peters, N.E.; Ren, H.; Smith, G.L. DNA-PK is a DNA sensor for IRF-3-dependent innate immunity. Elife 2012, 1, e00047.
- Kondo, T.; Kobayashi, J.; Saitoh, T.; Maruyama, K.; Ishii, K.J.; Barber, G.N.; Komatsu, K.; Akira, S.; Kawai, T. DNA damage sensor MRE11 recognizes cytosolic double-stranded DNA and induces type I interferon by regulating STING trafficking. Proc. Natl. Acad. Sci. USA 2013, 110, 2969–2974.
- Wang, J.; Kang, L.; Song, D.; Liu, L.; Yang, S.; Ma, L.; Guo, Z.; Ding, H.; Wang, H.; Yang, B. Ku70 Senses HTLV-1 DNA and Modulates HTLV-1 Replication. J. Immunol. 2017, 199, 2475–2482.
- Chiu, Y.H.; Macmillan, J.B.; Chen, Z.J. RNA polymerase III detects cytosolic DNA and induces type I interferons through the RIG-I pathway. Cell 2009, 138, 576–591.
- Muruve, D.A.; Pétrilli, V.; Zaiss, A.K.; White, L.R.; Clark, S.A.; Ross, P.J.; Parks, R.J.; Tschopp, J. The inflammasome recognizes cytosolic microbial and host DNA and triggers an innate immune response. Nature 2008, 452, 103–107.
- Ekchariyawat, P.; Hamel, R.; Bernard, E.; Wichit, S.; Surasombatpattana, P.; Talignani, L.; Thomas, F.; Choumet, V.; Yssel, H.; Desprès, P.; et al. Inflammasome signaling pathways exert antiviral effect against Chikungunya virus in human dermal fibroblasts. Infect. Genet. Evol. 2015, 32, 401–408.
- Lai, J.H.; Wang, M.Y.; Huang, C.Y.; Wu, C.H.; Hung, L.F.; Yang, C.Y.; Ke, P.Y.; Luo, S.F.; Liu, S.J.; Ho, L.J. Infection with the dengue RNA virus activates TLR9 signaling in human dendritic cells. EMBO Rep. 2018, 19, e46182.
- Daniels, B.P.; Kofman, S.B.; Smith, J.R.; Norris, G.T.; Snyder, A.G.; Kolb, J.P.; Gao, X.; Locasale, J.W.; Martinez, J.; Gale, M., Jr.; et al. The Nucleotide Sensor ZBP1 and Kinase RIPK3 Induce the Enzyme IRG1 to Promote an Antiviral Metabolic State in Neurons. Immunity 2019, 50, 64–76.
- Rothan, H.A.; Arora, K.; Natekar, J.P.; Strate, P.G.; Brinton, M.A.; Kumar, M. Z-DNA-Binding Protein 1 Is Critical for Controlling Virus Replication and Survival in West Nile Virus Encephalitis. Front. Microbiol. 2019, 10, 2089.
- Zhong, B.; Yang, Y.; Li, S.; Wang, Y.-Y.; Li, Y.; Diao, F.; Lei, C.; He, X.; Zhang, L.; Tien, P.; et al. The Adaptor Protein MITA Links Virus-Sensing Receptors to IRF3 Transcription Factor Activation. Immunity 2008, 29, 538–550.
- Jin, L.; Waterman, P.M.; Jonscher, K.R.; Short, C.M.; Reisdorph, N.A.; Cambier, J.C. MPYS, a Novel Membrane Tetraspanner, Is Associated with Major Histocompatibility Complex Class II and Mediates Transduction of Apoptotic Signals. Mol. Cell. Biol. 2008, 28, 5014–5026.
- Ishikawa, H.; Barber, G.N. STING is an endoplasmic reticulum adaptor that facilitates innate immune signalling. Nature 2008, 455, 674–678.
- Shu, C.; Li, X.; Li, P. The mechanism of double-stranded DNA sensing through the cGAS-STING pathway. Cytokine Growth Factor Rev. 2014, 25, 641–648.
- Mankan, A.K.; Schmidt, T.; Chauhan, D.; Goldeck, M.; Honing, K.; Gaidt, M.; Kubarenko, A.V.; Andreeva, L.; Hopfner, K.P.; Hornung, V. Cytosolic RNA:DNA hybrids activate the cGAS-STING axis. EMBO J. 2014, 33, 2937–2946.
- Cheng, Z.; Dai, T.; He, X.; Zhang, Z.; Xie, F.; Wang, S.; Zhang, L.; Zhou, F. The interactions between cGAS-STING pathway and pathogens. Signal Transduct. Target. Ther. 2020, 5, 91.
- Ablasser, A.; Goldeck, M.; Cavlar, T.; Deimling, T.; Witte, G.; Röhl, I.; Hopfner, K.P.; Ludwig, J.; Hornung, V. cGAS produces a 2′-5′-linked cyclic dinucleotide second messenger that activates STING. Nature 2013, 498, 380–384.
- Wan, D.; Jiang, W.; Hao, J. Research Advances in How the cGAS-STING Pathway Controls the Cellular Inflammatory Response. Front. Immunol. 2020, 11, 615.
- Liu, S.; Cai, X.; Wu, J.; Cong, Q.; Chen, X.; Li, T.; Du, F.; Ren, J.; Wu, Y.T.; Grishin, N.V.; et al. Phosphorylation of innate immune adaptor proteins MAVS, STING, and TRIF induces IRF3 activation. Science 2015, 347, aaa2630.
- Srikanth, S.; Woo, J.S.; Wu, B.; El-Sherbiny, Y.M.; Leung, J.; Chupradit, K.; Rice, L.; Seo, G.J.; Calmettes, G.; Ramakrishna, C.; et al. The Ca2+ sensor STIM1 regulates the type I interferon response by retaining the signaling adaptor STING at the endoplasmic reticulum. Nat. Immunol. 2019, 20, 152–162.
- Wang, L.; Wen, M.; Cao, X. Nuclear hnRNPA2B1 initiates and amplifies the innate immune response to DNA viruses. Science 2019, 365, eaav0758.
- Nazmi, A.; Mukhopadhyay, R.; Dutta, K.; Basu, A. STING Mediates Neuronal Innate Immune Response Following Japanese Encephalitis Virus Infection. Sci. Rep. 2012, 2, 347.
- Nitta, S.; Sakamoto, N.; Nakagawa, M.; Kakinuma, S.; Mishima, K.; Kusano-Kitazume, A.; Kiyohashi, K.; Murakawa, M.; Nishimura-Sakurai, Y.; Azuma, S.; et al. Hepatitis C virus NS4B protein targets STING and abrogates RIG-I-mediated type I interferon-dependent innate immunity. Hepatology 2013, 57, 46–58.
- Zevini, A.; Olagnier, D.; Hiscott, J. Crosstalk between Cytoplasmic RIG-I and STING Sensing Pathways. Trends Immunol. 2017, 38, 194–205.
- Dhanwani, R.; Takahashi, M.; Sharma, S. Cytosolic sensing of immuno-stimulatory DNA, the enemy within. Curr. Opin. Immunol. 2018, 50, 82–87.
- Ahn, J.; Barber, G.N. STING signaling and host defense against microbial infection. Exp. Mol. Med. 2019, 51, 1–10.
- Hopfner, K.-P.; Hornung, V. Molecular mechanisms and cellular functions of cGAS–STING signalling. Nat. Rev. Mol. Cell Biol. 2020, 21, 501–521.
- Aguirre, S.; Luthra, P.; Sanchez-Aparicio, M.T.; Maestre, A.M.; Patel, J.; Lamothe, F.; Fredericks, A.C.; Tripathi, S.; Zhu, T.; Pintado-Silva, J.; et al. Dengue virus NS2B protein targets cGAS for degradation and prevents mitochondrial DNA sensing during infection. Nat. Microbiol. 2017, 2, 17037.
- Dalrymple, N.A.; Cimica, V.; Mackow, E.R. Dengue Virus NS Proteins Inhibit RIG-I/MAVS Signaling by Blocking TBK1/IRF3 Phosphorylation: Dengue Virus Serotype 1 NS4A Is a Unique Interferon-Regulating Virulence Determinant. MBio 2015, 6, e00553-15.
- Stabell, A.C.; Meyerson, N.R.; Gullberg, R.C.; Gilchrist, A.R.; Webb, K.J.; Old, W.M.; Perera, R.; Sawyer, S.L. Dengue viruses cleave STING in humans but not in nonhuman primates, their presumed natural reservoir. Elife 2018, 7, e31919.
- Ding, Q.; Gaska, J.M.; Douam, F.; Wei, L.; Kim, D.; Balev, M.; Heller, B.; Ploss, A. Species-specific disruption of STING-dependent antiviral cellular defenses by the Zika virus NS2B3 protease. Proc. Natl. Acad. Sci. USA 2018, 115, E6310–E6318.
- Su, C.I.; Kao, Y.T.; Chang, C.C.; Chang, Y.; Ho, T.S.; Sun, H.S.; Lin, Y.L.; Lai, M.M.C.; Liu, Y.H.; Yu, C.Y. DNA-induced 2′3′-cGAMP enhances haplotype-specific human STING cleavage by dengue protease. Proc. Natl. Acad. Sci. USA 2020, 117, 15947–15954.
- Zheng, Y.; Liu, Q.; Wu, Y.; Ma, L.; Zhang, Z.; Liu, T.; Jin, S.; She, Y.; Li, Y.-P.; Cui, J. Zika virus elicits inflammation to evade antiviral response by cleaving cGAS via NS1-caspase-1 axis. EMBO J. 2018, 37, e99347.
- Zhang, W.; Jiang, B.; Zeng, M.; Duan, Y.; Wu, Z.; Wu, Y.; Wang, T.; Wang, M.; Jia, R.; Zhu, D.; et al. Binding of Duck Tembusu Virus Nonstructural Protein 2A to Duck STING Disrupts Induction of Its Signal Transduction Cascade To Inhibit Beta Interferon Induction. J. Virol. 2020, 94.
- Wu, Z.; Zhang, W.; Wu, Y.; Wang, T.; Wu, S.; Wang, M.; Jia, R.; Zhu, D.; Liu, M.; Zhao, X.; et al. Binding of the Duck Tembusu Virus Protease to STING Is Mediated by NS2B and Is Crucial for STING Cleavage and for Impaired Induction of IFN-β. J. Immunol. 2019, 203, 3374–3385.
- Ding, Q.; Cao, X.; Lu, J.; Huang, B.; Liu, Y.J.; Kato, N.; Shu, H.B.; Zhong, J. Hepatitis C virus NS4B blocks the interaction of STING and TBK1 to evade host innate immunity. J. Hepatol. 2013, 59, 52–58.
- Chen, X.; Yang, X.; Zheng, Y.; Yang, Y.; Xing, Y.; Chen, Z. SARS coronavirus papain-like protease inhibits the type I interferon signaling pathway through interaction with the STING-TRAF3-TBK1 complex. Protein Cell 2014, 5, 369–381.
- Holm, C.K.; Rahbek, S.H.; Gad, H.H.; Bak, R.O.; Jakobsen, M.R.; Jiang, Z.; Hansen, A.L.; Jensen, S.K.; Sun, C.; Thomsen, M.K.; et al. Influenza A virus targets a cGAS-independent STING pathway that controls enveloped RNA viruses. Nat. Commun. 2016, 7, 10680.
- Zhao, Y.; Karijolich, J. Know Thyself: RIG-I-Like Receptor Sensing of DNA Virus Infection
- Aguirre, S.; Fernandez-Sesma, A. Collateral Damage during Dengue Virus Infection: Making Sense of DNA by cGAS. J. Virol. 2017, 91, e01081-16.
- Chatel-Chaix, L.; Cortese, M.; Romero-Brey, I.; Bender, S.; Neufeldt, C.J.; Fischl, W.; Scaturro, P.; Schieber, N.; Schwab, Y.; Fischer, B.; et al. Dengue Virus Perturbs Mitochondrial Morphodynamics to Dampen Innate Immune Responses. Cell Host Microbe 2016, 20, 342–356.
- Chang, D.M.; Shaio, M.F. Production of interleukin-1 (IL-1) and IL-1 inhibitor by human monocytes exposed to dengue virus. J. Infect. Dis. 1994, 170, 811–817.
- Aarreberg, L.D.; Esser-Nobis, K.; Driscoll, C.; Shuvarikov, A.; Roby, J.A.; Gale, M., Jr. Interleukin-1β Induces mtDNA Release to Activate Innate Immune Signaling via cGAS-STING. Mol. Cell. 2019, 74, 801–815.
- Nellåker, C.; Yao, Y.; Jones-Brando, L.; Mallet, F.; Yolken, R.H.; Karlsson, H. Transactivation of elements in the human endogenous retrovirus W family by viral infection. Retrovirology 2006, 3, 44.
- Volkman, H.E.; Stetson, D.B. The enemy within: Endogenous retroelements and autoimmune disease. Nat. Immunol. 2014, 15, 415.
- Honda, T.; Tomonaga, K. Endogenous non-retroviral RNA virus elements evidence a novel type of antiviral immunity. Mob. Genet. Elem. 2016, 6, e1165785.
- Manghera, M.; Ferguson-Parry, J.; Lin, R.; Douville, R.N. NF-κB and IRF1 Induce Endogenous Retrovirus K Expression via Interferon-Stimulated Response Elements in Its 5′ Long Terminal Repeat. J. Virol. 2016, 90, 9338.
- Mu, X.; Ahmad, S.; Hur, S. Chapter Two—Endogenous Retroelements and the Host Innate Immune Sensors. In Advances in Immunology; Alt, F.W., Ed.; Academic Press: Cambridge, MA, USA, 2016; Volume 132, pp. 47–69.
- Grandi, N.; Tramontano, E. Human Endogenous Retroviruses Are Ancient Acquired Elements Still Shaping Innate Immune Responses. Front. Immunol. 2018, 9, 2039.
- Chen, S.; Hu, X.; Cui, I.H.; Wu, S.; Dou, C.; Liu, Y.; Sun, Z.; Xue, S.; Geng, T.; Liu, Z.; et al. An endogenous retroviral element exerts an antiviral innate immune function via the derived lncRNA lnc-ALVE1-AS1. Antivir. Res. 2019, 170, 104571.
- Schmidt, N.; Domingues, P.; Golebiowski, F.; Patzina, C.; Tatham, M.H.; Hay, R.T.; Hale, B.G. An influenza virus-triggered SUMO switch orchestrates co-opted endogenous retroviruses to stimulate host antiviral immunity. Proc. Natl. Acad. Sci. USA 2019, 116, 17399–17408.
- Shimizu, A.; Nakatani, Y.; Nakamura, T.; Jinno-Oue, A.; Ishikawa, O.; Boeke, J.D.; Takeuchi, Y.; Hoshino, H. Characterisation of cytoplasmic DNA complementary to non-retroviral RNA viruses in human cells. Sci. Rep. 2014, 4, 5074.
- Dunker, W.; Zhao, Y.; Song, Y.; Karijolich, J. Recognizing the SINEs of Infection: Regulation of Retrotransposon Expression and Modulation of Host Cell Processes. Viruses 2017, 9, 386.
- Raquin, V.; Merkling, S.H.; Gausson, V.; Moltini-Conclois, I.; Frangeul, L.; Varet, H.; Dillies, M.A.; Saleh, M.C.; Lambrechts, L. Individual co-variation between viral RNA load and gene expression reveals novel host factors during early dengue virus infection of the Aedes aegypti midgut. PLoS Negl. Trop. Dis. 2017, 11, e0006152.
- Suzuki, Y.; Frangeul, L.; Dickson, L.B.; Blanc, H.; Verdier, Y.; Vinh, J.; Lambrechts, L.; Saleh, M.C. Uncovering the Repertoire of Endogenous Flaviviral Elements in Aedes Mosquito Genomes. J. Virol. 2017, 91, e00571-17.
- Poirier, E.Z.; Goic, B.; Tomé-Poderti, L.; Frangeul, L.; Boussier, J.; Gausson, V.; Blanc, H.; Vallet, T.; Loyd, H.; Levi, L.I.; et al. Dicer-2-Dependent Generation of Viral DNA from Defective Genomes of RNA Viruses Modulates Antiviral Immunity in Insects. Cell Host Microbe 2018, 23, 353–365.
- Suzuki, Y.; Baidaliuk, A.; Miesen, P.; Frangeul, L.; Crist, A.B.; Merkling, S.H.; Fontaine, A.; Lequime, S.; Moltini-Conclois, I.; Blanc, H.; et al. Non-retroviral Endogenous Viral Element Limits Cognate Virus Replication in Aedes aegypti Ovaries. Curr. Biol. 2020.





