
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Karla Ramírez Sánchez | + 3462 word(s) | 3462 | 2020-11-18 10:38:41 | | | |
| 2 | Vicky Zhou | Meta information modification | 3462 | 2020-11-24 03:13:33 | | |
Video Upload Options
Recently, tissue engineering and regenerative medicine studies have evaluated smart biomaterials as implantable scaffolds and their interaction with cells for biomedical applications. Porous materials have been used in tissue engineering as synthetic extracellular matrices, promoting the attachment and migration of host cells to induce the in vitro regeneration of different tissues. Biomimetic 3D scaffold systems allow control over biophysical and biochemical cues, modulating the extracellular environment through mechanical, electrical, and biochemical stimulation of cells, driving their molecular reprogramming. In this review, first we outline the main advantages of using polysaccharides as raw materials for porous scaffolds, as well as the most common processing pathways to obtain the adequate textural properties, allowing the integration and attachment of cells. The second approach focuses on the tunable characteristics of the synthetic matrix, emphasizing the effect of their mechanical properties and the modification with conducting polymers in the cell response. The use and influence of polysaccharide-based porous materials as drug delivery systems for biochemical stimulation of cells is also described. Overall, engineered biomaterials are proposed as an effective strategy to improve in vitro tissue regeneration and future research directions of modified polysaccharide-based materials in the biomedical field are suggested.
1. Introduction
The number of publications related to the tissue engineering field has increased dramatically in recent years, referring to the potential regenerative methods and strategies for almost every tissue and organ of the human body. Progress has been reached by the integration of interdisciplinary research from cell biology, biomaterial sciences, and medical fields [1]. Specifically, tissue engineering involves the design and synthesis of three-dimensional (3D) matrices from biomaterials to provide a structural framework and to facilitate the attachment and migration of host cells, inducing a successful in vitro and in vivo regeneration of tissues [2][3][4].
Biomimetic 3D scaffolds may allow the control and application of a multi-stimulus to cells, including mechanical, electrical, and biochemical stimulations, in order to trigger specific responses, such as cell differentiation and tissue repair [5][6][7][8].
Tissue regeneration is naturally mediated by molecular processes, which direct gene expression to control renewal, restoration, and cell proliferation [9]. Nevertheless, normal regeneration is affected by aging, diseases, or accidents [10][11]. Thus, the increasing incidence of skin, muscle, and bone disorders, suffered by many people around the world, has prompted a critical need to develop engineered strategies to improve the replacement and regeneration of biological materials [11][12][13].
While many repair techniques have been proposed over recent decades, most of the surgical interventions have been directed toward the treatment of clinical symptoms but none have successfully repaired damaged tissues [14]. Consequently, in recent years, tissue engineering and regenerative medicine studies are focused on using the regenerative abilities of cells, in combination with engineered biomaterials, to create implantable scaffolds for tissue regeneration and reparation [1][10].
Porous materials from polysaccharides have been used as extracellular matrices (ECM) in tissue engineering in order to generate diverse types of cell lineages, promoting regeneration [15][16], for instance, in stem cells [17], osteoblasts [18], skeletal muscle cells [19], and endothelial cells [20]. In the biomedical field, aerogels from different sources have found applications as implantable devices, dressings for wound healing, synthetic bone grafts, carriers for different drugs, biosensing, and biomedical imaging [6][21].
Our review summarizes the current status of smart 3D scaffold systems based on polysaccharides regarding their production, properties, and potential applications in the biomedical field. Although those topics have been extensively reviewed in the past, our approach will focus on the potential development of biomimetic 3D scaffold systems including the physical, mechanical, electrical, and biochemical properties of modified polysaccharide-based aerogels and cryogels. Moreover, novel research directions of these smart materials, including strategies for the impregnation of drugs and their subsequent release from porous materials, and modification with conductive polymers were covered to be applied in the biomedical field.
2. Processing Strategies for Polysaccharide-Based Aerogels
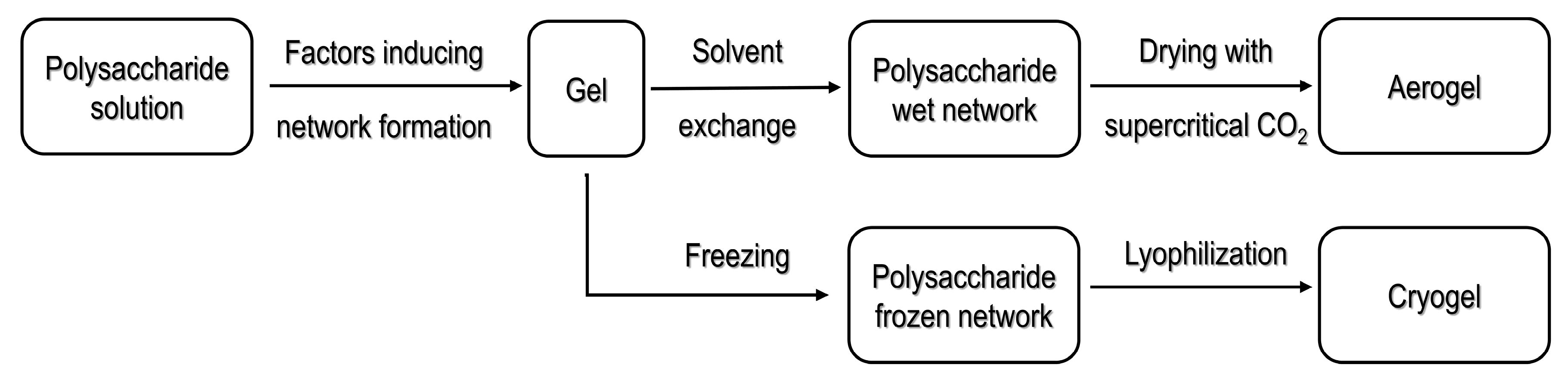
Diverse strategies have been used to obtain polysaccharide-based aerogels. The sol-gel method is commonly reported as an initial step in the processing pathways for organic or inorganic materials [22]. In the sol-gel process, a hydrogel formation is induced by crosslinking of the base material. Once the hydrogel is formed, it is necessary to select the drying method to be used; materials obtained from supercritical drying are commonly known as aerogels, whereas materials dried by freeze drying (lyophilization) are known as cryogels [21]. Figure 1 illustrates the scheme for supercritical drying and freeze drying, the most widely used methods in processing porous materials [21].
3. Polysaccharide-Based Porous Materials for Tissue Engineering
In recent years, tissue engineering and regenerative medicine studies have been based on the combination of specific types of cells and 3D porous scaffolds to induce a successful in vitro regeneration of diverse tissues [2][3][4].
The main efforts on engineered ECM in the biomedical field have been focused on the use and stimulation of pluripotent stem cells, which are special cells that have the ability to perpetuate themselves through a mechanism of self-renewal and to generate diverse types of cells through differentiation processes [15][16][17]. Nevertheless, osteoblasts [18], skeletal muscle cells [18], and endothelial cells [20] have been also studied.
3.1. Polysaccharide-Based Porous Materials as Extracellular Matrices
An extracellular matrix is an organized network composed by a mixture of cellular and non-cellular components. It plays an important role in tissue and organ morphogenesis, cell function, and structure maintenance. The biochemical and mechanical stimulus that cells receive from the matrix influences their growth, migration, differentiation, survival, and homeostasis [24].
Aerogels, as porous 3D matrices, possess a nanostructure that is able to mimic the extracellular matrix of the natural tissue, providing a favorable environment for the regeneration of tissues and organs [6][25]. Coupled with high porosity, low densities, and high inner surface areas, porous materials can provide appropriate morphology engineering, opening the possibility for their application as synthetic scaffolds for tissue engineering [26].
A scaffold acts as a template for new tissue formation [27] and its 3D structure guides the proliferation and colonization of cells, promoting tissue growth [28]. The configuration of the scaffold topology is critical in controlling cellular function, it should match the endogenous topology of the cell membrane in order to enhance signaling and function [29].
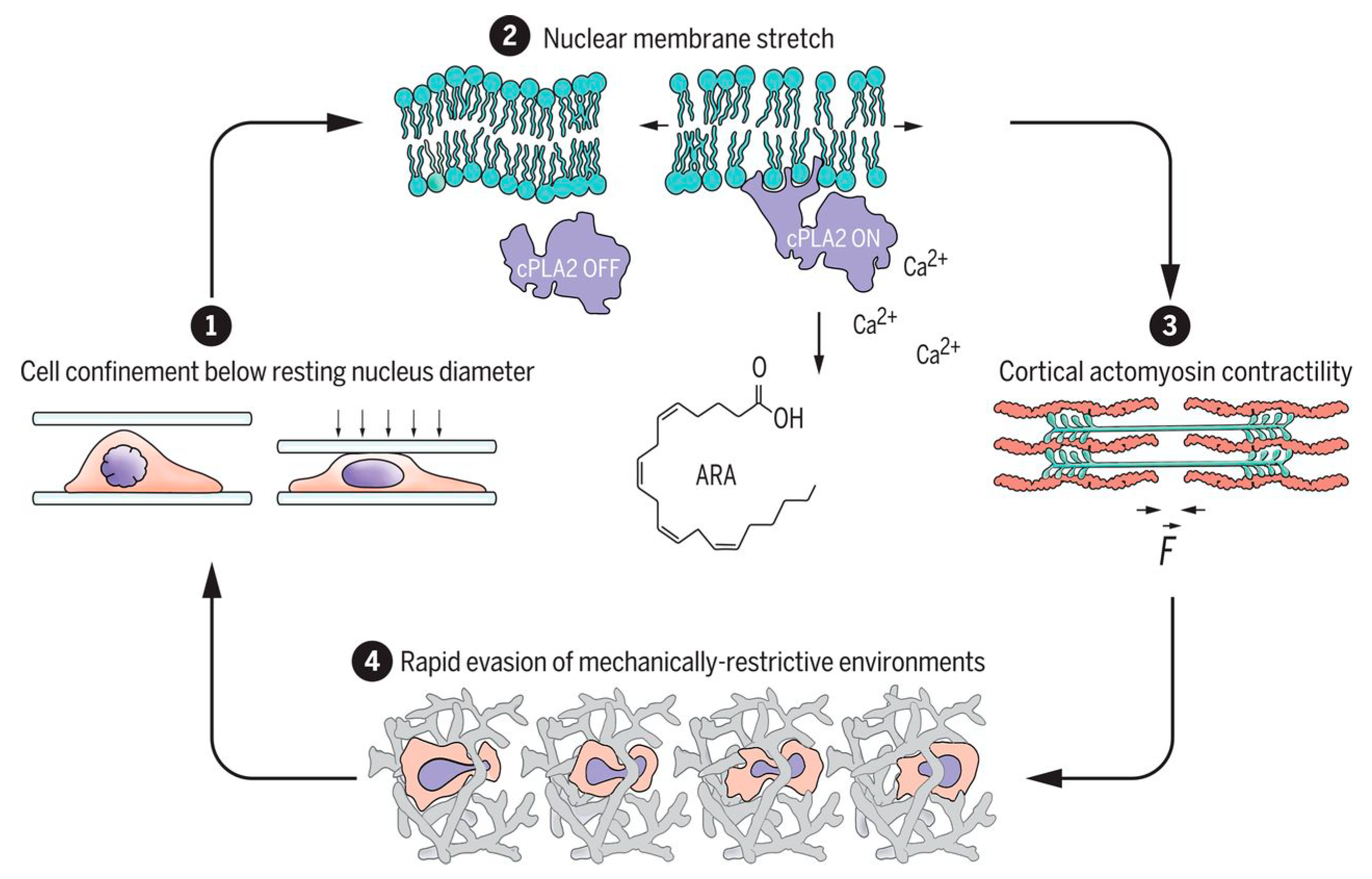
The spatial arrangement, porosity, biocompatibility, and proper scale of the ECM are some of the most important features that must be adjusted for use in nervous tissue, skin, bone, and muscle [30]. Nevertheless, several other factors, such as mechanical properties and chemical modification of scaffolds, significantly influence cellular behavior [5][31]. For example, recent studies have shown that the cell nucleus works as a fast mechanical respondent in cell contractility events because of the three-dimensional extracellular matrix restriction environment, inducing deformation and the movement of cells through the activation of cytosolic phospholipase A2 and arachidonic acid, which regulate myosin activity [32][33] (Figure 2).
3.2. Influence of the Mechanical Properties of the Scaffold in Cells and Tissues Behavior
The main goal of tissue engineering and regenerative medicine is to create strategies for replacing defective tissue. The use of polymeric scaffolds as extracellular matrices tries to mimic the in vivo host conditions to restore or improve the regeneration of damaged tissues. An extracellular matrix requires not only pore size control to induce cell adhesion and the ingress of nutrients and oxygen but also the incorporation of signal molecules, such as growth and differentiation factors, as well as a proper matrix architecture and mechanical properties to keep the implanted cells alive [34][35][36][37][38][39].
The mechanical characteristics of a scaffold for in vitro or in vivo cell studies may ultimately impact how the hosted tissue responds to the scaffold [31]. In this regard, the architecture, chemistry, topography, and physical properties of the employed scaffold as an ECM influence the structure and function of the surrounding tissue. Cells are constantly subjected to physical forces from their microenvironment. Mechanical properties of the porous materials are indispensable to determine the viability of a tissue and play a crucial role in cellular phenotype and homeostasis [40]. The mechanoresponsive cells include chondrocytes [32], cardiomyocytes [39], osteoblasts [41], muscle cells [42], endothelial cells [43], stem cells [7], and other tissue connective cells.
Cells may sense physical cues, such as osmotic pressure, shear force, and compression loading, as well as architecture, rigidity, and other several properties of the ECM, through a process known as mechanotransduction [40]. Thus, mechanotransduction corresponds to the cell capacity to transform a mechanical stimulus into biochemical signals. There are surface proteins in cell membranes which detect a force differential and then amplify and propagate this mechanical signal to elicit a change in cell behavior [44][40].
Compression and shear stress, caused by the synthetic ECM in a cell culture, transfer mechanical stimulation to the cells and enhance their biochemical signaling. The upregulation of gene expression and the changes in cellular metabolism during mechanical stimulation are regulated by mechanically sensitive surface receptors on cell membranes. There are several proteins related with the mechanotransduction to biochemical events, integrins are the best proteins studied so far [44].
Scaffold stiffness has been shown to have a significant impact on numerous cells and their fate [31]. The stiffness of an ECM in 2D cell cultures may influence the differentiation pattern of a same cell type; it has been reported that a soft matrix (0.1–1 kPa) promotes neurogenic differentiation, matrices with a medium stiffness (8–17 kPa) promote myogenic differentiation, and matrices with high stiffness (25–40 kPa) promote the osteogenic differentiation of mesenchymal stem cells [9]. Several authors have reported that the stiffness of a synthetic ECM induces mechanical stimulation of cells and the subsequent expression of cellular differentiation markers [38], tissue organization [42], causes the synthesis of extracellular matrix components [38], changes cell morphology, and improves their adhesion to synthetic scaffolds [38][43]. Additionally, the positive inotropic and chronotropic responses to both ion concentration (i.e., calcium, Ca2+) and temperature after mechanical stimulation of cardiomyocytes are also reported [39].
Complexity of the mechanotransduction induced by integrins is multifaceted as the proteins can form 24 possible functional distinct dimers and each dimer forms diverse complexes with multiple intracellular adaptor proteins to dictate the interplay between biochemical and cytoskeletal elements to determine their contribution to cellular mechanoresponses [38][44]. Nevertheless, it is well known that efficient force transfer and associated cytoskeleton changes are correlated with focal adhesion formation, as defined by the recruitment of talin, vinculin, and α-actinin to the stimulated integrin; these focal adhesion proteins form the molecular bridge that physically interlinks integrins with actin microfilaments [9][37][45].
Cytoskeletal changes caused by mechanical stimulation of cells are influenced by several biochemical pathways. It has been reported that maturation of focal adhesions causes activation of focal adhesion kinase (FAK); the scaffold protein, associated with adhesion plaque, triggered the Rho-associated protein kinase cascade (ROCK), which enhanced cellular tension through engagement of actomyosin contractility [40][46]. ROCK protein involves several downstream signals, including extracellular signal-regulated kinases (ERKs) and the hippo pathway, which is related with yes-associated protein 1 (YAP1); both biochemical pathways translocate some activated proteins to the nucleus and associated with transcriptional factors to regulate cell proliferation, tissue growth, and differentiation, as well as cell migration [33][47][48]. The chronical cellular tension reinforces these downstream signaling pathways to potentiate the production of ECM and ECM remodeling proteins that stiffen the local microenvironment and reinforce mechanosignaling [40].
Experiments related with the mechanical stimulation of cells have been carried out since 1938, when Glücksmann studied endosteal cells from embryonic chick tibiae [49]. Cells were grown on substrates of explanted intercostal muscle, to which pairs of neighboring ribs were left attached [49]. After several days, cells were compressed when the ribs were drawn near toward one another as the muscle tissue degenerated.
The mechanical characteristics of scaffolds can be adjusted using adequate dynamic biomaterials in order to create matrices with an appropriate stiffness to direct specific cellular responses. Mechanical properties of synthetic scaffolds are also used to design stimulation protocols to induce the controlled release of responsive drugs potentially used for tissue regeneration.
3.3. Polysaccharide-Based Porous Materials as Scaffolds for Electrical Stimulation of Cells
Another research field of interest is focused on the preparation of electrical systems to induce specific cellular responses. Diverse tissues (e.g., nerve, muscle, and glandular) make use of endogenous electric fields (EF) to transmit electrical signals. The endogenously-generated EF exists in both the cytoplasm and extracellular space [50]. Ionic currents and EFs in living cells play critical roles in important biological processes as they generate electromotive force, maintain a required electric potential, and allow some cellular functions [51][52]. These bioelectric signals are generated by gap junctional connections and ion channels or pumps moving ions across the membrane [51][53][54].
Currently, exogenous electrical stimulation of cells is a widely used method to improve their biological functions. Many authors have reported the use of nerve [55], bone [56], muscle [57], and neural stem cells [58], because their extensively recognized piezoelectric characteristics make them attractive for research on the role of exogenous electrical stimulation.
Coupling of an electromagnetic field with a live cell can occur via field interaction with charged molecules and proteins in the cell membrane [52]. It is reported that the application of the EF in a culture medium affects the migration [59], orientation [60], proliferation, and differentiation of cells [61][62]. Nevertheless, in most cases, it is used specifically to revive damaged or disabled tissues in the neuromuscular system as well as to accelerate the healing of injured musculoskeletal tissues, such as bone, ligament, and articular cartilage [50].
In this regard, biomaterials may receive considerable attention for their influence on cellular behaviors, ability to mimic biological functions, and, more recently, as electronic conductive systems with a potential use as tissue engineering scaffolds [5][63].
Some electroactive materials, such as conductive polymers (CPs) (e.g., poly(3,4-ethylenedioxythiophene) (PEDOT)) are currently being studied in combination with aerogels or cryogels as a promising field in regenerative medicine (Figure 3). Nevertheless, in the past, research studies have extensively used this kind of polymer to create organic conductive interfaces, neuroprosthetic devices, neural probes, and controlled drug-delivery systems [5][64][65].

Figure 3. Porous material microtomography (micro-CT) image (a) and aerogel images before (b) and after conductive polymer (i.e., poly(3,4-ethylenedioxythiophene) (PEDOT)) modification (c). Reproduced from [26][66] under Elsevier Copyright Clearance Center (CCC) licenses.
Conductive polymers can be structured with porous systems using different techniques [67][68][69]. Starch and starch/κ-carrageenan aerogels have been used as templates for the obtention of nanoporous conductive materials [26][66]. In the biomedical field, conductive nanoporous materials have been applied not only as physical support but also as a medium to provide electrical stimulation of a cell culture. Electrical stimulation in neural cells has shown great potential for function restoring and wound healing [70].
On the other hand, the incorporation of anionic drugs and κ-carrageenan on the structure of starch porous materials is particularly interesting since both compounds may act as dopant agents for the conductive matrix, as it was shown recently [29][66][71]. Dexamethasone, a well-known glucocorticoid anionic drug, has recently been the object of research from an electrochemical point of view, regarding its doping properties on conductive matrices [72] and for its ability to be released by electrochemical stimulation from a PEDOT/κ-carrageenan film [29]. The above opens the possibility to create scaffolds from conductive porous materials and the incorporation of specific drugs in their structure to be applied as stimulation systems in tissue engineering.
3.4. Polysaccharide-Based Porous Materials as Drug-Delivery Systems
One of the main approaches and most relevant applications of biopolymer-based aerogels is their use as drug-delivery systems [6][73]. The application of these materials as controlled drug-release matrices has gained interest in the last years due to aerogel properties, such as its high surface area, high porosity, and biocompatibility [73]. Aerogels can act as a carrier for bioactive compounds, showing high loading capacity, enhanced stability upon storage, and accelerated drug release, if required [74]. Along with the high loading capacity, biopolymer-based aerogels also show an improved dissolution rate of poorly water-soluble drugs [6].
The biocompatibility of natural polymers along with the outstanding performance of aerogels as carriers for active compounds, such as drugs, have promoted the systems as scaffolds in body implants to accelerate tissue formation by providing a suitable porous structure that promotes cell colonization [75][76]. Diverse authors have also studied the incorporation of drugs and growth factors to promote the attachment, proliferation, and differentiation of cells, in order to provide both substitutes for damaged tissues and therapeutic schemes that reduce post-implantation inflammation and infections [12][76][77][78].
Controllable drug-release systems may be categorized as mechanical methods, which are mainly in vivo implantable pump delivery systems built from biocompatible nanomaterials [79][80], and as polymeric drug delivery systems. The last one makes use of biopolymers, in which the delivery of drugs is mainly dominated via diffusion and recently by electrochemical methods [80]. Hence, the incorporation of drugs within these kinds of porous scaffolds has been studied previously for osteogenic differentiation, bone repair activity, and the stimulation of neural tissues [65][76].
3.4.1. Diffusive Phenomena on the Controlled Release of Drugs on Polysaccharide-Based Aerogels
Different methods for drug impregnation or loading can be found in literature regarding porous materials from polysaccharides. Supercritical technology employing scCO2 has been defined as the most innovative technique for producing polymer/drug composite systems for pharmaceutical applications [81]. By means of supercritical fluid technology, the impregnation of aerogel particles with drugs such as ketoprofen was achieved [75]. This process consists of placing aerogel particles and ketoprofen in a closed autoclave under agitation; the ketoprofen was dissolved in scCO2 and adsorbed in the aerogel matrix [75]. The same procedure was reproduced for obtaining poly(ɛ-Caprolactone) (PCL) scaffolds loaded with ketoprofen [82] and for alginate-based aerogel microparticles for mucosal drug-delivery [73]. In addition, maize starch aerogels and calcium alginate aerogels were impregnated with different non-steroidal anti-inflammatory drugs, such as nimesulide, ketoprofen, and diclefenac sodium [81].
3.4.2. Controlled Drug Release by Electrical Stimulation Employing Conductive Porous Materials
In order to prevent the negative effects resulting from exposure to high dosages of drugs, local electronically-controlled release of pharmaceutical compounds from implantable devices appears as a promising option [83]. Drugs anchored inside the conductive materials have been reported using supercritical technology and electropolymerization [65][84].
Electrochemical methods involve the use of conductive polymers, which are electrochemically oxidized during the polymerization processes, generating charge carriers, and, thus, allowing ionic drugs impregnation based on electrostatic interactions [85]. There are two main electrochemical methods to induce the immobilization of drugs. One-step immobilization or in situ immobilization: an ionic drug (preferably anionic) acts as a doping agent and its anchoring proceeds simultaneously with the process of matrix formation [84][86][87]. Anti-cancer drugs, anti-inflammatory compounds, and hormones have been fixed on conductive materials using one-step immobilization, mainly for the development of neural devices [88][89][90].
The second method corresponds to the two-step or ex situ immobilization. The incorporation of the drug is carried out after the synthesis of the matrix, through ion exchange processes taking place at their surface. First, the polymer film is synthesized from a solution consisting of the monomer and a small ionic molecule as doping agent, without the drug. The obtained film is later reduced and oxidized by an electrical stimulus [86][87]. Reduction induces the removal of the dopant from the film; meanwhile, the drug, which acts as the second doping agent, is incorporated during the process of matrix oxidation [87].
Drug delivery is caused by electrochemical stimulation of the conductive matrix, which induces the oxidation and/or reduction of the film. By applying a negative potential, the polymeric matrix is reduced and the cationic charge of the polymer backbone is neutralized, causing the release of the anionic drug by electrostatic mechanisms [86]. In a similar procedure, applying negative and positive cyclic potentials induces the reduction and oxidation of the polymeric film, respectively; meanwhile, the matrix experiments expansion and contraction, which force the release of the drug. Although cyclic stimulation allows a greater amount of drug release in comparison with other methods, some authors have reported that the application of the stimulus may cause delamination, cracks, and breakdowns of the matrix, mainly in one-step immobilization systems [65][91][92].
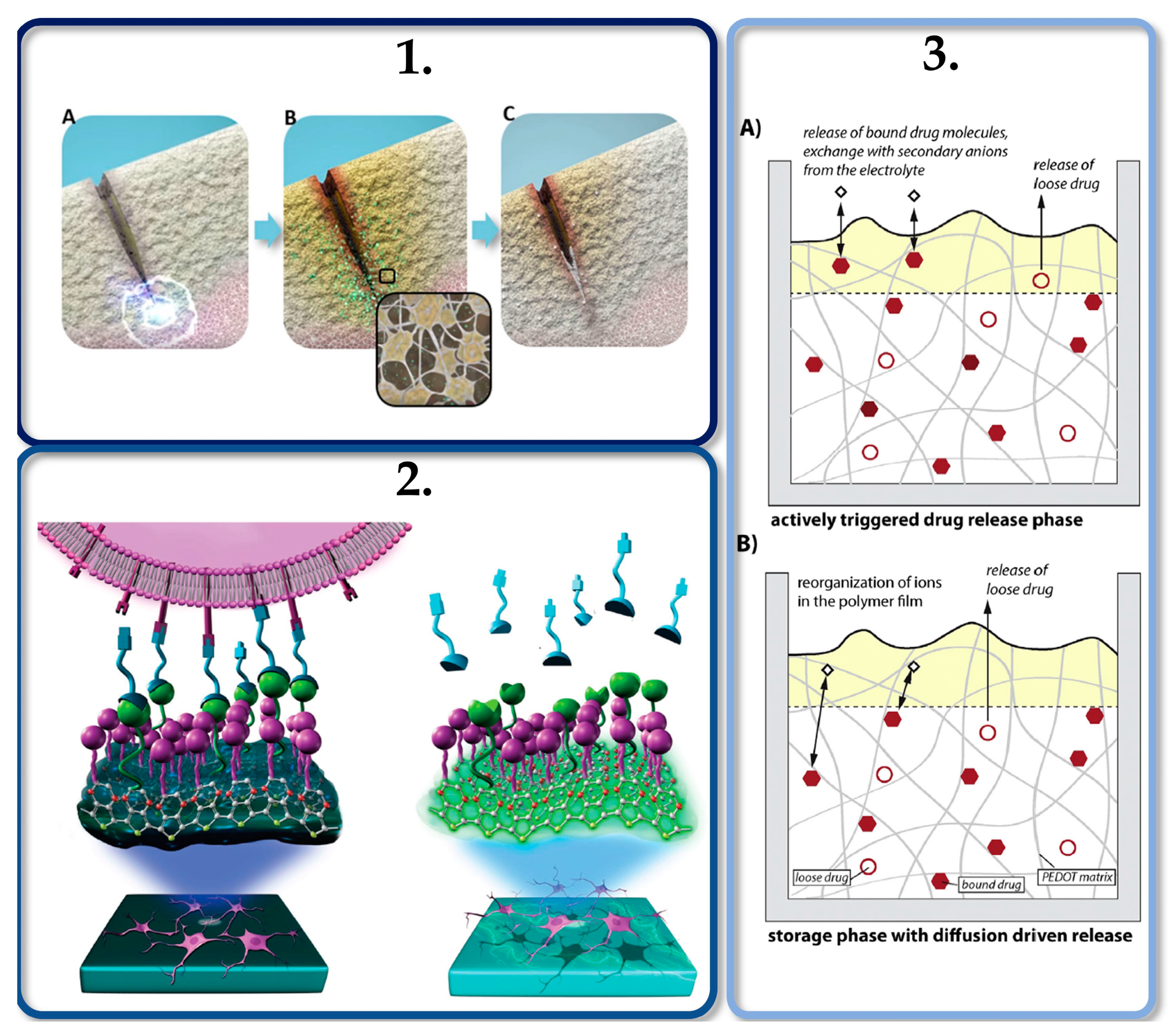
The controlled release of drugs using electrical stimulation from conductive polymer films [29][65][89] opens the door for a different approach regarding the application of polysaccharide aerogels on drug delivery. Since these materials can be coated with an electrically conductive material while incorporating active compounds, those composites may be used in the controlled release of bioactive molecules by electrical stimulation [93][94]. These biochemical release systems are the main focus of several research groups and further investigations should follow this path in order to promote smart scaffolds that merge mechanical, electrical, and biochemical stimulation processes, mimicking the in vivo ECM conditions, in order to promote specific cell behavior, as shown in Figure 4.
References
- Brahatheeswaran Dhandayuthapani; Yasuhiko Yoshida; Toru Maekawa; D. Sakthi Kumar; Polymeric Scaffolds in Tissue Engineering Application: A Review. International Journal of Polymer Science 2011, 2011, 1-19, 10.1155/2011/290602.
- Scott Stratton; Namdev B. Shelke; Kazunori Hoshino; Swetha Rudraiah; Sangamesh G. Kumbar; Bioactive polymeric scaffolds for tissue engineering. Bioactive Materials 2016, 1, 93-108, 10.1016/j.bioactmat.2016.11.001.
- Marco Santoro; Sarita R. Shah; Jennifer L. Walker; Antonios G. Mikos; Poly(lactic acid) nanofibrous scaffolds for tissue engineering. Advanced Drug Delivery Reviews 2016, 107, 206-212, 10.1016/j.addr.2016.04.019.
- Hyerim Jo; Myeongbu Sim; Semin Kim; Sumi Yang; Youngjae Yoo; Jin-Ho Park; T. H. Yoon; Min‐Gon Kim; Jae Young Lee; Electrically conductive graphene/polyacrylamide hydrogels produced by mild chemical reduction for enhanced myoblast growth and differentiation. Acta Biomaterialia 2016, 48, 100-109, 10.1016/j.actbio.2016.10.035.
- Salvador D. Aznar-Cervantes; Ana Pagan Bernabeu; Jose G. Martinez; Antonia Bernabeu-Esclapez; Toribio F. Otero; Luis Meseguer-Olmo; Juan I. Paredes; Jose L. Cenis; Electrospun silk fibroin scaffolds coated with reduced graphene promote neurite outgrowth of PC-12 cells under electrical stimulation. Materials Science and Engineering: C 2017, 79, 315-325, 10.1016/j.msec.2017.05.055.
- Carlos A. García-González; Tatiana Budtova; Luisa Durães; Can Erkey; Pasquale Del Gaudio; Pavel Gurikov; Matthias M. Koebel; Falk Liebner; Monica Neagu; Irina Smirnova; et al. An Opinion Paper on Aerogels for Biomedical and Environmental Applications.. Molecules 2019, 24, 1815, 10.3390/molecules24091815.
- K.T. Shalumon; Han-Tsung Liao; Chang-Yi Kuo; Chak-Bor Wong; Chien-Ju Li; Mini P.A.; Jyh-Ping Chen; Rational design of gelatin/nanohydroxyapatite cryogel scaffolds for bone regeneration by introducing chemical and physical cues to enhance osteogenesis of bone marrow mesenchymal stem cells. Materials Science and Engineering: C 2019, 104, 109855, 10.1016/j.msec.2019.109855.
- Dzmitry Afanasenkau; Daria Kalinina; Vsevolod Lyakhovetskii; Christoph Tondera; Oleg Gorsky; Seyyed Moosavi; Natalia Pavlova; Natalia Merkulyeva; Allan V. Kalueff; Ivan R. Minev; et al.Pavel Musienko Rapid prototyping of soft bioelectronic implants for use as neuromuscular interfaces. Nature Biomedical Engineering 2020, 4, 1-13, 10.1038/s41551-020-00615-7.
- Akhilesh K. Gaharwar; Irtisha Singh; Ali Khademhosseini; Engineered biomaterials for in situ tissue regeneration. Nature Reviews Materials 2020, 5, 1-20, 10.1038/s41578-020-0209-x.
- Maliheh Jahromi; Shahnaz Razavi; Abbas Bakhtiari; The advances in nerve tissue engineering: From fabrication of nerve conduit to in vivo nerve regeneration assays. Journal of Tissue Engineering and Regenerative Medicine 2019, 13, 2077-2100, 10.1002/term.2945.
- Hang Lin; JiHee Sohn; He Shen; Mark T. Langhans; Rocky S. Tuan; Bone marrow mesenchymal stem cells: Aging and tissue engineering applications to enhance bone healing. Biomaterials 2019, 203, 96-110, 10.1016/j.biomaterials.2018.06.026.
- Irina Alexandra Paun; M. Zamfirescu; Catalin Romeo Luculescu; Adriana Maria Acasandrei; Cosmin Catalin Mustaciosu; Mona Mihailescu; Maria Dinescu; Electrically responsive microreservoires for controllable delivery of dexamethasone in bone tissue engineering. Applied Surface Science 2016, 392, 321-331, 10.1016/j.apsusc.2016.09.027.
- Víctor Santos-Rosales; Inés Ardao; Carmen Alvarez-Lorenzo; Nilza Ribeiro; J. M. Oliveira; C.A. García-González; Sterile and Dual-Porous Aerogels Scaffolds Obtained through a Multistep Supercritical CO₂-Based Approach.. Molecules 2019, 24, 871, 10.3390/molecules24050871.
- J.-K Francis Suh; Howard W.T Matthew; Application of chitosan-based polysaccharide biomaterials in cartilage tissue engineering: a review. Biomaterials 2000, 21, 2589-2598, 10.1016/s0142-9612(00)00126-5.
- Zahia Hamidouche; Eric Haÿ; Pascal Vaudin; Pierre Charbord; Roland Schüle; Pierre J. Marie; Olivia Fromigué; FHL2 mediates dexamethasone‐induced mesenchymal cell differentiation into osteoblasts by activating Wnt/β‐catenin signaling‐dependent Runx2 expression. The FASEB Journal 2008, 22, 3813-3822, 10.1096/fj.08-106302.
- Irving L. Weissman; Sean J. Morrison; Michael F. Clarke; Irving L. Weissman; Stem cells, cancer, and cancer stem cells. Nature 2001, 414, 105-111, 10.1038/35102167.
- J. Sammons; N. Ahmed; M. El-Sheemy; H T Hassan; The Role of BMP-6, IL-6, and BMP-4 in Mesenchymal Stem Cell–Dependent Bone Development: Effects on Osteoblastic Differentiation Induced by Parathyroid Hormone and Vitamin D3. Stem Cells and Development 2004, 13, 273-280, 10.1089/154732804323099208.
- Iwona M. Wojak-Ćwik; Łucja Rumian; Małgorzata Krok-Borkowicz; Ricarda Hess; Ricardo Bernhardt; Piotr Dobrzyński; Stephanie Möller; Matthias Schnabelrauch; Vera Hintze; Dieter Scharnweber; et al.Elżbieta Pamuła Synergistic effect of bimodal pore distribution and artificial extracellular matrices in polymeric scaffolds on osteogenic differentiation of human mesenchymal stem cells. Materials Science and Engineering: C 2019, 97, 12-22, 10.1016/j.msec.2018.12.012.
- Umber Cheema; S.-Y. Yang; Vivek Mudera; G Goldspink; R.A. Brown; 3-D in vitro model of early skeletal muscle development. Cell Motility and the Cytoskeleton 2003, 54, 226-236, 10.1002/cm.10095.
- Hongkun He; Marianna Sofman; Alex J-S Wang; Caroline C. Ahrens; Wade Wang; Linda G. Griffith; Paula T. Hammond; Engineering Helical Modular Polypeptide-Based Hydrogels as Synthetic Extracellular Matrices for Cell Culture. Biomacromolecules 2019, 21, 566-580, 10.1021/acs.biomac.9b01297.
- Mehrez E. El-Naggar; Sarah I. Othman; Ahmed A. Allam; Osama M. Morsy; Synthesis, drying process and medical application of polysaccharide-based aerogels. International Journal of Biological Macromolecules 2020, 145, 1115-1128, 10.1016/j.ijbiomac.2019.10.037.
- C.A. García-González; M. Alnaief; I. Smirnova; Polysaccharide-based aerogels—Promising biodegradable carriers for drug delivery systems. Carbohydrate Polymers 2011, 86, 1425-1438, 10.1016/j.carbpol.2011.06.066.
- Kathirvel Ganesan; Tatiana Budtova; Lorenz Ratke; Pavel Gurikov; Victor Baudron; Imke Preibisch; Philipp Niemeyer; Irina Smirnova; Barbara Milow; Review on the Production of Polysaccharide Aerogel Particles. Materials 2018, 11, 2144, 10.3390/ma11112144.
- Achilleas D. Theocharis; Spyros S. Skandalis; Chrysostomi Gialeli; Nikos K. Karamanos; Extracellular matrix structure. Advanced Drug Delivery Reviews 2016, 97, 4-27, 10.1016/j.addr.2015.11.001.
- Fergal J. O'brien; Biomaterials & scaffolds for tissue engineering. Materials Today 2011, 14, 88-95, 10.1016/s1369-7021(11)70058-x.
- Ricardo Starbird-Perez; Carlos A. García-González; Irina Smirnova; Wolfgang H. Krautschneider; Wolfgang Bauhofer; Synthesis of an organic conductive porous material using starch aerogels as template for chronic invasive electrodes. Materials Science and Engineering: C 2014, 37, 177-183, 10.1016/j.msec.2013.12.032.
- Gleeson, J. P.; O'Brien, F. J. . Advances in Composite Materials for Medicine and Nanotechnology; Attaf, Brahim, Eds.; Intech Open Access: Croatia, 2011; pp. 39-59.
- Carlos A. García-González; Angel Concheiro; Carmen Alvarez-Lorenzo; Processing of Materials for Regenerative Medicine Using Supercritical Fluid Technology. Bioconjugate Chemistry 2015, 26, 1159-1171, 10.1021/bc5005922.
- Karla Ramírez-Sánchez; Aura Ledezma-Espinoza; Andrés Sánchez-Kopper; Esteban Avendano; Monica Prado; Ricardo Starbird-Perez; Polysaccharide κ-Carrageenan as Doping Agent in Conductive Coatings for Electrochemical Controlled Release of Dexamethasone at Therapeutic Doses. Molecules 2020, 25, 2139, 10.3390/molecules25092139.
- Haoxiang Chen; Juchang Zhong; Jian Wang; Ruiying Huang; Xiaoyin Qiao; Honghui Wang; Zhikai Tan; Enhanced growth and differentiation of myoblast cells grown on E-jet 3D printed platforms. International Journal of Nanomedicine 2019, 14, 937-950, 10.2147/ijn.s193624.
- Basak E. Uygun; Sarah E. Stojsih; Howard W. T. Matthew; Effects of Immobilized Glycosaminoglycans on the Proliferation and Differentiation of Mesenchymal Stem Cells. Tissue Engineering Part A 2009, 15, 3499-3512, 10.1089/ten.tea.2008.0405.
- Valeria Venturini; Fabio Pezzano; Frederic Català Castro; Hanna-Maria Häkkinen; Senda Jiménez-Delgado; Mariona Colomer-Rosell; Monica Marro; Queralt Tolosa-Ramon; Sonia Paz-López; Miguel A. Valverde; et al.Julian WeghuberPablo Loza-AlvarezMichael KriegStefan WieserVerena Ruprecht The nucleus measures shape changes for cellular proprioception to control dynamic cell behavior. Science 2020, 370, eaba2644, 10.1126/science.aba2644.
- A. J. Lomakin; C. J. Cattin; D. Cuvelier; Z. Alraies; M. Molina; G. P. F. Nader; N. Srivastava; P. J. Sáez; J. M. Garcia-Arcos; I. Y. Zhitnyak; et al.A. BhargavaM. K. DriscollE. S. WelfR. FiolkaR. J. PetrieN. S. De SilvaJ. M. González-GranadoN. ManelA. M. Lennon-DuménilD. J. MüllerM. Piel The nucleus acts as a ruler tailoring cell responses to spatial constraints. Science 2020, 370, eaba2894, 10.1126/science.aba2894.
- Yusuke Nagai; Hidenori Yokoi; Keiko Kaihara; Keiji Naruse; The mechanical stimulation of cells in 3D culture within a self-assembling peptide hydrogel. Biomaterials 2012, 33, 1044-1051, 10.1016/j.biomaterials.2011.10.049.
- Niamh Fahy; Mauro Alini; Martin Stoddart; Mechanical stimulation of mesenchymal stem cells: Implications for cartilage tissue engineering. Journal of Orthopaedic Research 2017, 36, 52-63, 10.1002/jor.23670.
- Angelo S. Mao; David J. Mooney; Regenerative medicine: Current therapies and future directions. Proceedings of the National Academy of Sciences 2015, 112, 14452-14459, 10.1073/pnas.1508520112.
- John A. Pedersen; Melody A. Swartz; Mechanobiology in the Third Dimension. Annals of Biomedical Engineering 2005, 33, 1469-1490, 10.1007/s10439-005-8159-4.
- Vanessa Terraciano; Nathaniel Hwang; Lorenzo Moroni; Hyung Bin Park; Zijun Zhang; Joseph Mizrahi; Dror Seliktar; Jennifer Elisseeff; Differential Response of Adult and Embryonic Mesenchymal Progenitor Cells to Mechanical Compression in Hydrogels. Stem Cells 2007, 25, 2730-2738, 10.1634/stemcells.2007-0228.
- W. Zhang; C.W. Kong; M.H. Tong; W.H. Chooi; Nan Huang; R.A. Li; Barbara P. Chan; Maturation of human embryonic stem cell-derived cardiomyocytes (hESC-CMs) in 3D collagen matrix: Effects of niche cell supplementation and mechanical stimulation. Acta Biomaterialia 2017, 49, 204-217, 10.1016/j.actbio.2016.11.058.
- J. Matthew Barnes; Laralynne Przybyla; Valerie M. Weaver; Tissue mechanics regulate brain development, homeostasis and disease. Journal of Cell Science 2016, 130, 71-82, 10.1242/jcs.191742.
- Chao Liu; Xin Cui; Thomas M. Ackermann; Vittoria Flamini; Weiqiang Chen; Alesha B. Castillo; Osteoblast-derived paracrine factors regulate angiogenesis in response to mechanical stimulation. Integrative Biology 2015, 8, 785-794, 10.1039/c6ib00070c.
- Nina Bono; D. Pezzoli; L. Lévesque; C. Loy; Gabriele Candiani; G. B. Fiore; Diego Mantovani; Unraveling the role of mechanical stimulation on smooth muscle cells: A comparative study between 2D and 3D models. Biotechnology and Bioengineering 2016, 113, 2254-2263, 10.1002/bit.25979.
- Elham Hasanzadeh; Ghassem Amoabediny; Nooshin Haghighipour; Nasim Gholami; Javad Mohammadnejad; Shahrokh Shojaei; Nasim Salehi-Nik; The stability evaluation of mesenchymal stem cells differentiation toward endothelial cells by chemical and mechanical stimulation. In Vitro Cellular & Developmental Biology - Plant 2017, 53, 818-826, 10.1007/s11626-017-0165-y.
- Aban Shuaib; Daniyal Motan; Pinaki Bhattacharya; Alex McNabb; Timothy M. Skerry; Damien Lacroix; Heterogeneity in The Mechanical Properties of Integrins Determines Mechanotransduction Dynamics in Bone Osteoblasts. Scientific Reports 2019, 9, 1-14, 10.1038/s41598-019-47958-z.
- N Wang; J P Butler; D E Ingber; Mechanotransduction across the cell surface and through the cytoskeleton. Science 1993, 260, 1124-1127, 10.1126/science.7684161.
- H. Maldonado; C. Calderon; F. Burgos-Bravo; O. Kobler; W. Zuschratter; O. Ramirez; S. Härtel; P. Schneider; A.F.G. Quest; Rodrigo Herrera-Molina; et al.Lisette Leyton Astrocyte-to-neuron communication through integrin-engaged Thy-1/CBP/Csk/Src complex triggers neurite retraction via the RhoA/ROCK pathway. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research 2017, 1864, 243-254, 10.1016/j.bbamcr.2016.11.006.
- Hyun Jung Lee; Miguel F. Diaz; Katherine M. Price; Joyce A. Ozuna; Songlin Zhang; Eva M. Sevick-Muraca; John P. Hagan; Pamela L. Wenzel; Fluid shear stress activates YAP1 to promote cancer cell motility. Nature Communications 2017, 8, 14122, 10.1038/ncomms14122.
- Xueran Chen; Shangrong Zhang; Zhen Wang; Fengsong Wang; Xinwang Cao; Quan Wu; Chenggang Zhao; Huihui Ma; Fang Ye; Hongzhi Wang; et al.Zhiyou Fang Supervillin promotes epithelial-mesenchymal transition and metastasis of hepatocellular carcinoma in hypoxia via activation of the RhoA/ROCK-ERK/p38 pathway. Journal of Experimental & Clinical Cancer Research 2018, 37, 128, 10.1186/s13046-018-0787-2.
- A. Glücksmann; Studies on bone mechanics in vitro. I. Influence of pressure on orientation of structure. The Anatomical Record 1938, 72, 97-113, 10.1002/ar.1090720109.
- Greeshma Thrivikraman; Sunil Kumar Boda; Sunil Kumar Boda; Unraveling the mechanistic effects of electric field stimulation towards directing stem cell fate and function: A tissue engineering perspective. Biomaterials 2017, 150, 60-86, 10.1016/j.biomaterials.2017.10.003.
- Jaatinen, L. The Effect of an Applied Electric Current on Cell Proliferation, Viability, Morphology, Adhesion,and Stem Cell Differentiation. Ph.D. Thesis, Tampere University of Technology, Tampere, Finland, 2017.
- T. Taghian; D. A. Narmoneva; A. B. Kogan; Modulation of cell function by electric field: a high-resolution analysis. Journal of The Royal Society Interface 2015, 12, 20150153, 10.1098/rsif.2015.0153.
- Wang, L.; Liu, Y.; Jin, H.; Steinacker, J. Electrical stimulation induced Hsp70 response in C2C12 cells. Exerc. Immunol. Rev. 2010, 16, 86–97.
- Laleh Ghasemi-Mobarakeh; Molamma P Prabhakaran; Mohammad Morshed; Mohammad Hossein Nasr-Esfahani; Hossein Baharvand; Sahar Kiani; Salem S Al-Deyab; Seeram Ramakrishna; Application of conductive polymers, scaffolds and electrical stimulation for nerve tissue engineering. Journal of Tissue Engineering and Regenerative Medicine 2011, 5, e17-e35, 10.1002/term.383.
- Peter C. Sherrell; Brianna C. Thompson; Jonathan K. Wassei; Amy A. Gelmi; Michael J. Higgins; Richard B. Kaner; Gordon G. Wallace; Maintaining Cytocompatibility of Biopolymers Through a Graphene Layer for Electrical Stimulation of Nerve Cells. Advanced Functional Materials 2013, 24, 769-776, 10.1002/adfm.201301760.
- Ilyas S. Aleem; Idris Aleem; Nathan Evaniew; Jason W. Busse; Michael Yaszemski; Arnav Agarwal; Thomas Einhorn; Mohit Bhandari; Efficacy of Electrical Stimulators for Bone Healing: A Meta-Analysis of Randomized Sham-Controlled Trials. Scientific Reports 2016, 6, 31724, 10.1038/srep31724.
- Jenny W.H. Lou; Austin J. Bergquist; Abdulaziz Aldayel; Jennifer Czitron; David F. Collins; Interleaved neuromuscular electrical stimulation reduces muscle fatigue. Muscle & Nerve 2016, 55, 179-189, 10.1002/mus.25224.
- Wei Zhu; Tao Ye; Se-Jun Lee; Haitao Cui; Shida Miao; Xuan Zhou; Danmeng Shuai; Lijie Grace Zhang; Enhanced neural stem cell functions in conductive annealed carbon nanofibrous scaffolds with electrical stimulation. Nanomedicine: Nanotechnology, Biology and Medicine 2018, 14, 2485-2494, 10.1016/j.nano.2017.03.018.
- Xiaoning Yuan; Derya E. Arkonac; Pen-Hsiu Grace Chao; Gordana Vunjak-Novakovic; Electrical stimulation enhances cell migration and integrative repair in the meniscus. Scientific Reports 2014, 4, nd, 10.1038/srep03674.
- Tai, G.; Reid, B.; Cao, L.; Zhao, M. Electrotaxis and wound healing: Experimental methods to study electricfields as a directional signal for cell migration. Methods Mol. Biol. 2009, 571, 77–97.
- Piyush Bajaj; Bobby Reddy; Larry Millet; Chunan Wei; Pinar Zorlutuna; Gang Bao; Rashid Bashir; Patterning the differentiation of C2C12 skeletal myoblasts. Integrative Biology 2010, 3, 897-909, 10.1039/c1ib00058f.
- Weibo Guo; Xiaodi Zhang; Xin Yu; Shu Wang; Jichuan Qiu; Wei Tang; Linlin Li; Hong Liu; Zhong Lin Wang; Self-Powered Electrical Stimulation for Enhancing Neural Differentiation of Mesenchymal Stem Cells on Graphene–Poly(3,4-ethylenedioxythiophene) Hybrid Microfibers. ACS Nano 2016, 10, 5086-5095, 10.1021/acsnano.6b00200.
- Jing Wang; Lingling Tian; Nuan Chen; Seeram Ramakrishna; Xiumei Mo; The cellular response of nerve cells on poly-l-lysine coated PLGA-MWCNTs aligned nanofibers under electrical stimulation. Materials Science and Engineering: C 2018, 91, 715-726, 10.1016/j.msec.2018.06.025.
- Filipa Pires; Quirina Ferreira; Carlos A.V. Rodrigues; J. Morgado; Frederico Castelo Ferreira; Neural stem cell differentiation by electrical stimulation using a cross-linked PEDOT substrate: Expanding the use of biocompatible conjugated conductive polymers for neural tissue engineering. Biochimica et Biophysica Acta (BBA) - General Subjects 2015, 1850, 1158-1168, 10.1016/j.bbagen.2015.01.020.
- C. Boehler; C. Kleber; N. Martini; Y. Xie; I. Dryg; T. Stieglitz; U.G. Hofmann; M. Asplund; Actively controlled release of Dexamethasone from neural microelectrodes in a chronic in vivo study. Biomaterials 2017, 129, 176-187, 10.1016/j.biomaterials.2017.03.019.
- Roy Zamora-Sequeira; Inés Ardao; Ricardo Starbird-Perez; C.A. García-González; Conductive nanostructured materials based on poly-(3,4-ethylenedioxythiophene) (PEDOT) and starch/κ-carrageenan for biomedical applications. Carbohydrate Polymers 2018, 189, 304-312, 10.1016/j.carbpol.2018.02.040.
- Hao, H.B.; Yuan, L.; Fu, Z.B.; Wang, C.Y.; Yang, X.; Zhu, J.; Qu, J.; Chen, H.; Schiraldi, D. Biomass-basedmechanically-strong and electrically-conductive polymer aerogels and their application for supercapacitors.ACS Appl. Mater. Interfaces 2016, 8, 9917–9924.
- Insang You; Unyong Jeong; Electromechanical Decoupling by Porous Aerogel Conducting Polymer. Matter 2019, 1, 24-25, 10.1016/j.matt.2019.06.008.
- Xuetong Zhang; Dongwu Chang; Jiren Liu; Yunjun Luo; Conducting polymer aerogels from supercritical CO2 drying PEDOT-PSS hydrogels. Journal of Materials Chemistry 2009, 20, 5080, 10.1039/c0jm00050g.
- Christopher Bertucci; Ryan Koppes; Courtney Dumont; Abigail N. Koppes; Neural responses to electrical stimulation in 2D and 3D in vitro environments. Brain Research Bulletin 2019, 152, 265-284, 10.1016/j.brainresbull.2019.07.016.
- Priscila Hernandez-Suarez; Karla Ramirez; Fernando Alvarado; E. Avendano; Karla Ramírez Sánchez; Electrochemical characterization of poly(3,4-ethylenedioxythiophene)/κ-carrageenan as a biocompatible conductive coat for biologic applications. MRS Communications 2018, 9, 218-223, 10.1557/mrc.2018.189.
- Eimear M. Ryan; Carmel B. Breslin; Formation of polypyrrole with dexamethasone as a dopant: Its cation and anion exchange properties. Journal of Electroanalytical Chemistry 2018, 824, 188-194, 10.1016/j.jelechem.2018.07.052.
- V.S.S. Gonçalves; Pavel Gurikov; J. Poejo; A.A. Matias; S. Heinrich; C.M.M. Duarte; I. Smirnova; Alginate-based hybrid aerogel microparticles for mucosal drug delivery. European Journal of Pharmaceutics and Biopharmaceutics 2016, 107, 160-170, 10.1016/j.ejpb.2016.07.003.
- Clara López-Iglesias; Joana Barros; Inés Ardao; Fernando J. Monteiro; Carmen Alvarez-Lorenzo; José L. Gómez-Amoza; Carlos A. García-González; Vancomycin-loaded chitosan aerogel particles for chronic wound applications. Carbohydrate Polymers 2018, 204, 223-231, 10.1016/j.carbpol.2018.10.012.
- C.A. García-González; Irina Smirnova; Use of supercritical fluid technology for the production of tailor-made aerogel particles for delivery systems. The Journal of Supercritical Fluids 2013, 79, 152-158, 10.1016/j.supflu.2013.03.001.
- Leticia Goimil; Víctor Santos-Rosales; Araceli Delgado; Carmen Évora; R. Reyes; A. Abel Lozano-Pérez; Salvador D. Aznar-Cervantes; Jose Luis Cenis; Francisco J. Otero Espinar; Angel Concheiro; et al.Carmen Alvarez-LorenzoC.A. García-González scCO2-foamed silk fibroin aerogel/poly(ε-caprolactone) scaffolds containing dexamethasone for bone regeneration. Journal of CO2 Utilization 2019, 31, 51-64, 10.1016/j.jcou.2019.02.016.
- Pedro F. Costa; Ana M. Puga; Luis Diaz-Gomez; Angel Concheiro; Dirk H. Busch; Carmen Alvarez-Lorenzo; Additive manufacturing of scaffolds with dexamethasone controlled release for enhanced bone regeneration. International Journal of Pharmaceutics 2015, 496, 541-550, 10.1016/j.ijpharm.2015.10.055.
- Qihai Liu; Lian Cen; Heng Zhou; Shuo Yin; Guangpeng Liu; Wei Liu; Yilin Cao; Lei Cui; The Role of the Extracellular Signal-Related Kinase Signaling Pathway in Osteogenic Differentiation of Human Adipose-Derived Stem Cells and in Adipogenic Transition Initiated by Dexamethasone. Tissue Engineering Part A 2009, 15, 3487-3497, 10.1089/ten.tea.2009.0175.
- M. Danckwerts; A. Fassihi; Implantable Controlled Release Drug Delivery Systems: A Review. Drug Development and Industrial Pharmacy 1990, 17, 1465-1502, 10.3109/03639049109026629.
- Chizhu Ding; Zibiao Li; A review of drug release mechanisms from nanocarrier systems. Materials Science and Engineering: C 2017, 76, 1440-1453, 10.1016/j.msec.2017.03.130.
- Paola Franco; Iolanda De Marco; Supercritical CO2 adsorption of non-steroidal anti-inflammatory drugs into biopolymer aerogels. Journal of CO2 Utilization 2020, 36, 40-53, 10.1016/j.jcou.2019.11.001.
- Leticia Goimil; Mara E.M. Braga; Ana M.A. Dias; José L. Gómez-Amoza; Angel Concheiro; Patricia Diaz-Rodriguez; Hermínio C. De Sousa; Carlos A. García-González; Supercritical processing of starch aerogels and aerogel-loaded poly(ε-caprolactone) scaffolds for sustained release of ketoprofen for bone regeneration. Journal of CO2 Utilization 2017, 18, 237-249, 10.1016/j.jcou.2017.01.028.
- K Krukiewicz; Tailorable drug capacity of dexamethasone-loaded conducting polymer matrix. IOP Conference Series: Materials Science and Engineering 2018, 369, 012002, 10.1088/1757-899x/369/1/012002.
- Susanne Löffler; Silke Seyock; Rolf Nybom; Gunilla B. Jacobson; Agneta Richter-Dahlfors; Electrochemically triggered release of acetylcholine from scCO2 impregnated conductive polymer films evokes intracellular Ca2+ signaling in neurotypic SH-SY5Y cells. Journal of Controlled Release 2016, 243, 283-290, 10.1016/j.jconrel.2016.10.020.
- Richard Balint; Nigel J. Cassidy; Sarah H. Cartmell; Conductive polymers: Towards a smart biomaterial for tissue engineering. Acta Biomaterialia 2014, 10, 2341-2353, 10.1016/j.actbio.2014.02.015.
- B. Alshammary; Frank C. Walsh; P. Herrasti; Carlos Ponce De Leon; Electrodeposited conductive polymers for controlled drug release: polypyrrole. Journal of Solid State Electrochemistry 2015, 20, 839-859, 10.1007/s10008-015-2982-9.
- Katarzyna Krukiewicz; Barbara Gniazdowska; Tomasz Jarosz; Artur P. Herman; Slawomir Boncel; Roman Turczyn; Effect of immobilization and release of ciprofloxacin and quercetin on electrochemical properties of poly(3,4-ethylenedioxypyrrole) matrix. Synthetic Metals 2019, 249, 52-62, 10.1016/j.synthmet.2019.02.001.
- Naader Alizadeh; Ehsan Shamaeli; Electrochemically controlled release of anticancer drug methotrexate using nanostructured polypyrrole modified with cetylpyridinium: Release kinetics investigation. Electrochimica Acta 2014, 130, 488-496, 10.1016/j.electacta.2014.03.055.
- C. Boehler; Felix Oberueber; Maria Asplund; Tuning drug delivery from conducting polymer films for accurately controlled release of charged molecules.. Journal of Controlled Release 2019, 304, 173-180, 10.1016/j.jconrel.2019.05.017.
- Darren Svirskis; Manisha Sharma; Yang Yu; Sanjay Garg; Electrically switchable polypyrrole film for the tunable release of progesterone. Therapeutic Delivery 2013, 4, 307-313, 10.4155/tde.12.166.
- Lucas Leprince; Audrey Dogimont; Delphine Magnin; Sophie Demoustier-Champagne; Dexamethasone electrically controlled release from polypyrrole-coated nanostructured electrodes. Journal of Materials Science: Materials in Electronics 2010, 21, 925-930, 10.1007/s10856-010-4008-6.
- Grace Stevenson; Simon E. Moulton; Peter C. Innis; Gordon G. Wallace; Polyterthiophene as an electrostimulated controlled drug release material of therapeutic levels of dexamethasone. Synthetic Metals 2010, 160, 1107-1114, 10.1016/j.synthmet.2010.02.035.
- Jin Qu; Yongping Liang; Mengting Shi; Baolin Guo; Y. Gao; Zhanhai Yin; Biocompatible conductive hydrogels based on dextran and aniline trimer as electro-responsive drug delivery system for localized drug release. International Journal of Biological Macromolecules 2019, 140, 255-264, 10.1016/j.ijbiomac.2019.08.120.
- Jin Qu; Xin Zhao; Peter X. Ma; Baolin Guo; Injectable antibacterial conductive hydrogels with dual response to an electric field and pH for localized “smart” drug release. Acta Biomaterialia 2018, 72, 55-69, 10.1016/j.actbio.2018.03.018.
- Raquel Núñez-Toldrà; Fabian Vasquez-Sancho; Nathalie Barroca; Gustau Catalan; Investigation of The Cellular Response to Bone Fractures: Evidence for Flexoelectricity. Scientific Reports 2020, 10, 1-10, 10.1038/s41598-019-57121-3.
- Hsing-An Lin; Bo Zhu; Yu-Wei Wu; Jun Sekine; Aiko Nakao; Shyh-Chyang Luo; Yoshiro Yamashita; Hsiao-Hua Yu; Dynamic Poly(3,4-ethylenedioxythiophene)s Integrate Low Impedance with Redox-Switchable Biofunction. Advanced Functional Materials 2018, 28, nd, 10.1002/adfm.201703890.





