
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Masaru Tanaka | -- | 2315 | 2022-10-24 12:21:15 | | | |
| 2 | Masaru Tanaka | + 7 word(s) | 2322 | 2022-10-24 12:33:37 | | | | |
| 3 | Masaru Tanaka | Meta information modification | 2322 | 2022-10-24 12:55:59 | | | | |
| 4 | Beatrix Zheng | Meta information modification | 2322 | 2022-10-25 03:06:01 | | |
Video Upload Options
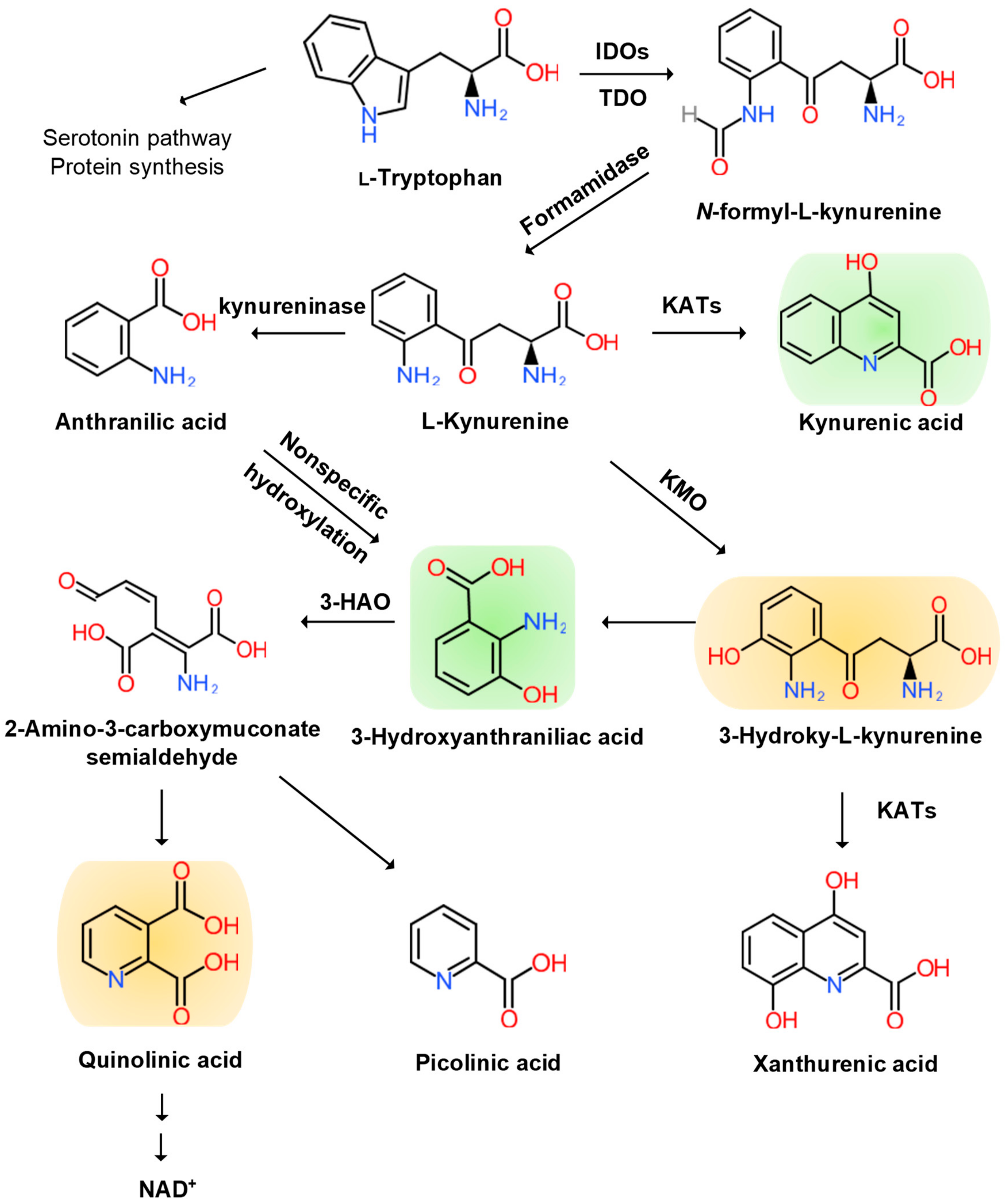
Worldwide, 50 million people suffer from dementia, a group of symptoms affecting cognitive and social functions, progressing severely enough to interfere with daily life. Alzheimer’s disease (AD) accounts for most of the dementia cases. Pathological and clinical findings have led to proposing several hypotheses of AD pathogenesis, finding a presence of positive feedback loops and additionally observing the disturbance of a branch of tryptophan metabolism, the kynurenine (KYN) pathway. Either causative or resultant of dementia, elevated levels of neurotoxic KYN metabolites are observed, potentially upregulating multiple feedback loops of AD pathogenesis. Memantine is an N-methyl-D-aspartate glutamatergic receptor (NMDAR) antagonist, which belongs to one of only two classes of medications approved for clinical use, but other NMDAR modulators have been explored so far in vain. An endogenous KYN pathway metabolite, kynurenic acid (KYNA), likewise inhibits the excitotoxic NMDAR. Besides its anti-excitotoxicity, KYNA is a multitarget compound that triggers anti-inflammatory and antioxidant activities. Modifying the KYNA level is a potential multitarget strategy to normalize the disturbed KYN pathway and thus to alleviate juxtaposing AD pathogeneses.
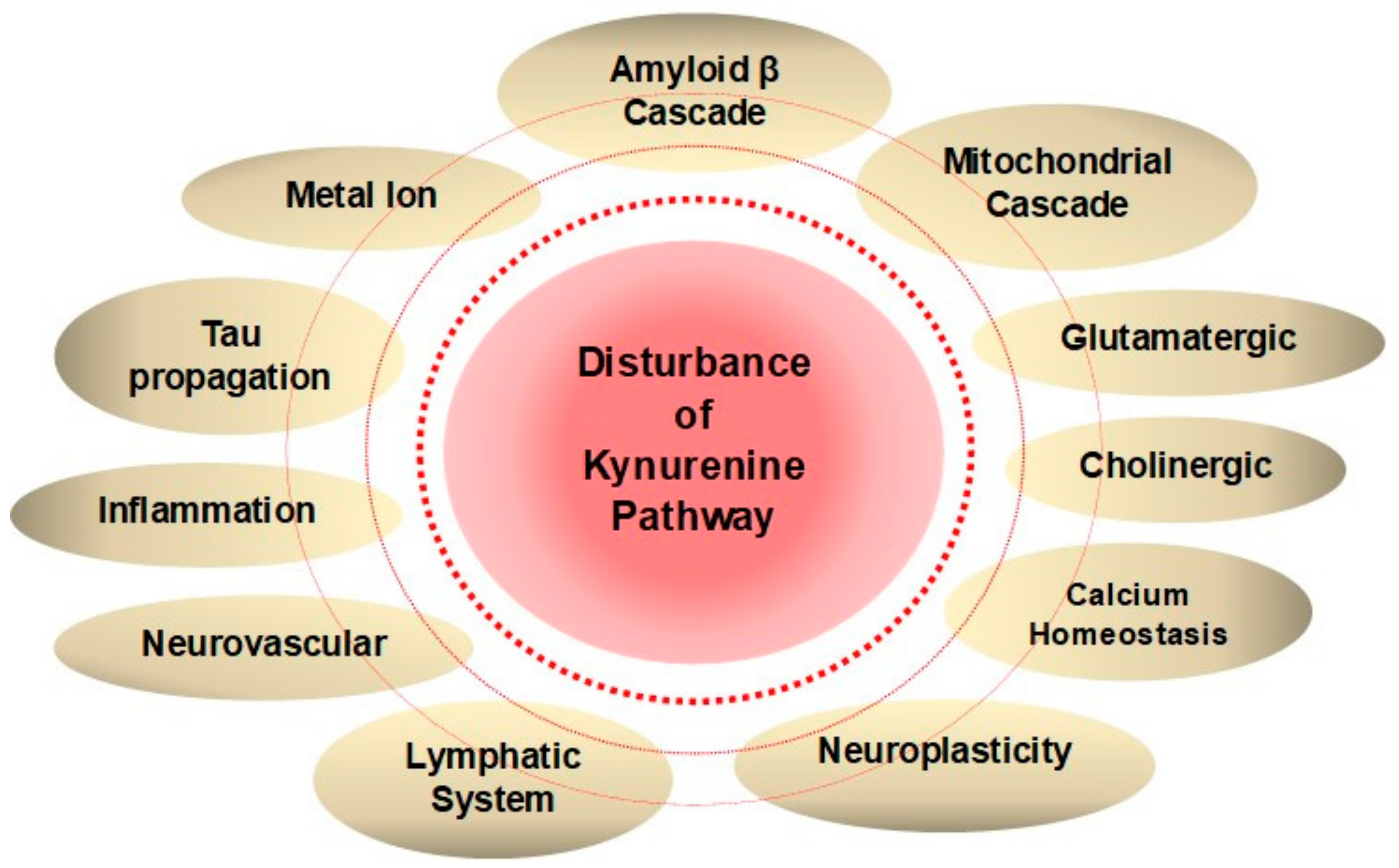
1. Multiple Positive Feedback Loops via Kynurenine Metabolites
2. Kynurenines in Major Neurocognitive Disorders
| Diseases | Study Types | Reference Numbers or Sample Numbers (Disease/Control) |
Samples | Risk of Bias |
|---|---|---|---|---|
| Neurodegenerative diseases | ||||
| Alzheimer’s disease [15][16][17][18][19] | ||||
| Guillemin et al., 2005 [16] | case-control study | 6/4 | brain tissue | High risk |
| Bonda et al., 2010 [17] | case-control study | 12/7 | brain tissue | |
| Gulaj et al., 2010 [18] | case-control study | 34/18 | serum | |
| Schwarcz et al., 2013 [19] | case-control study | 20/19 | serum | |
| Parkinson’s disease [20][21][22][23][24] | ||||
| Hartai et al., 2005 [22] | case-control study | 19/17 | plasma, RBC | High risk |
| Lewitt et al., 2013 [23] | case-control study | 48/57 | CSF | |
| Chang et al., 2018 [24] | case-control study | 118/37 | plasma | |
| Huntington’s disease [25][26][27][28][29] | ||||
| Reynolds and Pearson, 1989 [29] | case-control study | 12/11 | postmortem brain tissue | High risk |
| Beal et al., 1992 [27] | case-control study | 14–30/25–40 | postmortem brain tissue | |
| Jauch et al., 1995 [28] | case-control study | 17/17 | postmortem brain tissue | |
| Stoy et al., 2005 [26] | case-control study | 15/11 | plasma | |
| Vascular Cognitive Dementia | Unclear | |||
| Darlington et al., 2007 [30] | case-control study | 50/35 | serum | |
| Yan et al., 2015 [31] | case-control study | 28/20,11 | serum, CSF | |
| Psychiatric disorders | ||||
| Major depressive disorder | Low risk | |||
| Ogawa et al., 2014 [32] | meta-analysis | 10 | plasma | |
| Réus et al., 2015 [33] | systematic review | 29 | plasma, blood, serum, CSF, urine, brain tissue |
|
| Ogyu et al., 2018 [34] | meta-analysis | 22 | plasma | |
| Bipolar disorder [35][36][37][38][39][40][41][42] | ||||
| Birner et al., 2017 [40] | case-control study | 143/101 | blood | Unclear |
| Wang et al., 2018 [41] | meta-analysis | 16 | CSF | |
| Arnone et al., 2018 [42] | meta-analysis | 5 | serum | |
| Generalized anxiety disorder | ||||
| Orlikov et al., 1994 [43] | case-control study | 16/15 | plasma | Unclear |
| Altmaier et al., 2013 [44] | case-control study | 386/116 | serum | |
| Autism spectrum disorder | ||||
| Lim et al., 2016 [45] | case-control study | 15/12 | blood | Unclear |
| Bryn et al., 2017 [46] | case-control study | 30/30 | serum | |
2.1. Kynurenines in Neuodegenerative Diseases
| Diseases | Neurotoxic Kynurenines |
Neuromodulatory Kynurenines |
|---|---|---|
| Neurodegenerative diseases | ||
| Alzheimer’s disease | ↑ | ↓ |
| Parkinson’s disease | ↑ | ↓ |
| Huntington’s disease | ↑ | ↓ |
| Vascular cognitive dementia | ↑ | ↑ |
| Psychiatric disorders | ||
| Major depressive disorder | ↑ | ↓ |
| Bipolar disorder | ? | ? |
| Generalized anxiety disorder | ↓ | ? |
| Autism spectrum disorder | ↑ | ? |
2.2. Kynurenines in Psychiatric Disorders
References
- Bohár, Z.; Toldi, J.; Fülöp, F.; Vécsei, L. Changing the Face of Kynurenines and Neurotoxicity: Therapeutic Considerations. Int. J. Mol. Sci. 2015, 16, 9772–9793.
- Sas, K.; Szabó, E.; Vécsei, L. Mitochondria, Oxidative Stress and the Kynurenine System, with a Focus on Ageing and Neuroprotection. Molecules 2018, 23, 191.
- Badawy, A.A. Kynurenine pathway of tryptophan metabolism: regulatory and functional aspects. Int. J. Tryptophan Res. 2017, 10, 1178646917691938.
- Merlo, L.M.; Pigott, E.; Duhadaway, J.B.; Grabler, S.; Metz, R.; Prendergast, G.C.; Mandik-Nayak, L. IDO2 is a critical mediator of autoantibody production and inflammatory pathogenesis in a mouse model of autoimmune arthritis. J. Immunol. 2014, 92, 2082–2090.
- Boros, F.A.; Bohár, Z.; Vécsei, L. Genetic alterations affecting the genes encoding the enzymes of the kynurenine pathway and their association with human diseases. Mutat. Res. 2018, 776, 32–45.
- Boros, F.A.; Vécsei, L. Immunomodulatory Effects of Genetic Alterations Affecting the Kynurenine Pathway. Front. Immunol. 2019, 10, 2570.
- Parrott, J.M.; O’Connor, J.C. Kynurenine 3-Monooxygenase: An Influential Mediator of Neuropathology. Front. Psychiatry 2015, 6, 16.
- Herédi, J.; Berkó, A.M.; Jankovics, F.; Iwamori, T.; Iwamori, N.; Ono, E.; Horváth, S.; Kis, Z.; Toldi, J.; Vécsei, L.; et al. Astrocytic and neuronal localization of kynurenine aminotransferase-2 in the adult mouse brain. Brain Struct. Funct. 2017, 222, 1663–1672.
- Robotka, H.; Toldi, J.; Vecsei, L. L-Kynurenine: Metabolism and mechanism of neuroprotection. Future Neurol. 2008, 3, 169–188.
- Stone, T.W. Does kynurenic acid act on nicotinic receptors? An assessment of the evidence. J. Neurochem. 2019.
- Vécsei, L.; Szalárdy, L.; Fülöp, F.; Toldi, J. Kynurenines in the CNS: Recent advances and new questions. Nat. Rev. Drug Discov. 2013, 12, 64–82.
- Zádori, D.; Nyiri, G.; Szonyi, A.; Szatmári, I.; Fülöp, F.; Toldi, J.; Freund, T.F.; Vécsei, L.; Klivényi, P. Neuroprotective effects of a novel kynurenic acid analogue in a transgenic mouse model of Huntington’s disease. J. Neural Transm. 2011, 118, 865–875.
- Majláth, Z.; Toldi, J.; Vécsei, L. The potential role of kynurenines in Alzheimer’s disease: pathomechanism and therapeutic possibilities by influencing the glutamate receptors. J. Neural Transm. 2014, 121, 881–889.
- Raison, C.L.; Dantzer, R.; Kelley, K.W.; Lawson, M.A.; Woolwine, B.J.; Vogt, G.; Spivey, J.R.; Saito, K.; Miller, A.H. CSF concentrations of brain tryptophan and kynurenines during immune stimulation with IFN-alpha: relationship to CNS immune responses and depression. Mol. Psychiatry 2010, 15, 393–403.
- Sorgdrager, F.J.H.; Vermeiren, Y.; Van Faassen, M.; Van Der Ley, C.; Nollen, E.A.A.; Kema, I.P.; De Deyn, P.P. Age- and disease-specific changes of the kynurenine pathway in Parkinson’s and Alzheimer’s disease. J. Neurochem. 2019, 151, 656–668.
- Guillemin, G.J.; Brew, B.J.; Noonan, C.E.; Takikawa, O.; Cullen, K.M. Indoleamine 2,3 hippocampus dioxygenase and quinolinic acid immunoreactivity in Alzheimer’s disease. Neuropathol. Appl. Neurobiol. 2005, 31, 395–404.
- Bonda, D.; Mailankot, M.; Stone, J.G.; Garrett, M.R.; Staniszewska, M.; Castellani, R.J.; Siedlak, S.L.; Zhu, X.; Lee, H.; Perry, G.; et al. Indoleamine 2,3-dioxygenase and 3-hydroxykynurenine modifications are found in the neuropathology of Alzheimer’s disease. Redox Rep. 2010, 15, 161–168.
- Gulaj, E.; Pawlak, K.; Bien, B.; Pawlak, D. Kynurenine and its metabolites in Alzheimer’s disease patients. Adv. Med. Sci. 2010, 55, 204–211.
- Schwarcz, M.J.; Guillemin, G.J.; Teipel, S.J.; Buerger, K.; Hampel, H. Increased 3-hydroxykynurenine serum concentrations differentiate Alzheimer’s disease patients from controls. Eur. Arch. Psychiatry Clin. Neurosci. 2013, 263, 345.
- Jacobs, K.; Lim, C.K.; Blennow, K.; Zetterberg, H.; Chatterjee, P.; Martins, R.N.; Brew, B.; Guillemin, G.; Lovejoy, D. Correlation between plasma and CSF concentrations of kynurenine pathway metabolites in Alzheimer’s disease and relationship to amyloid-β and tau. Neurobiol. Aging 2019, 80, 11–20.
- Hafstad Solvang, S.E.; Nordrehaug, J.E.; Aarsland, D.; Lange, J.; Ueland, P.M.; McCann, A.; Midttun, Ø.; Tell, G.; Giil, L.M. Kynurenines, Neuropsychiatric Symptoms, and Cognitive Prognosis in Patients with Mild Dementia. Int. J. Tryptophan Res. 2019, 12, 1178646919877883.
- Hartai, Z.; Klivenyi, P.; Janaky, T.; Penke, B.; Dux, L.; Vecsei, L. Kynurenine metabolism in plasma and in red blood cells in Parkinson’s disease. J. Neurol. Sci. 2005, 239, 31–35.
- Lewitt, P.A.; Li, J.; Lu, M.; Beach, T.G.; Adler, C.H.; Guo, L. Arizona Parkinson’s Disease Consortium. 3-hydroxykynurenine and other Parkinson’s disease biomarkers discovered by metabolomic analysis. Mov. Disord. 2013, 28, 1653–1660.
- Chang, K.H.; Cheng, M.L.; Tang, H.Y.; Huang, C.Y.; Wu, Y.R.; Chen, C.M. Alternations of Metabolic Profile and Kynurenine Metabolism in the Plasma of Parkinson’s Disease. Mol. Neurobiol. 2018, 55, 6319–6328.
- Obál, I.; Majláth, Z.; Toldi, J.; Vécsei, L. Mental disturbances in Parkinson’s disease and related disorders: The role of excitotoxins. J. Parkinsons Dis. 2014, 4, 139–150.
- Stoy, N.; Mackay, G.M.; Forrest, C.M.; Christofides, J.; Egerton, M.; Stone, T.W.; Darlington, L.G. Tryptophan metabolism and oxidative stress in patients with Huntington’s disease. J. Neurochem. 2005, 93, 611–623.
- Beal, M.F.; Matson, W.R.; Storey, E.; Milbury, P.; Ryan, E.A.; Ogawa, T.; Bird, E.D. Kynurenic acid concentrations are reduced in Huntington’s disease cerebral cortex. J. Neurol. Sci. 1992, 108, 80–87.
- Jauch, D.; Urbańska, E.M.; Guidetti, P.; Bird, E.D.; Vonsattel, J.P.; Whetsell, W.O., Jr.; Schwarcz, R. Dysfunction of brain kynurenic acid metabolism in Huntington’s disease: focus on kynurenine aminotransferases. J. Neurol. Sci. 1995, 130, 39–47.
- Reynolds, G.P.; Pearson, S.J. Increased brain 3-hydroxykynurenine in Huntington’s disease. Lancet 1989, 2, 979–980.
- Darlington, L.G.; Mackay, G.M.; Forrest, C.M.; Stoy, N.; George, C.; Stone, T.W. Altered kynurenine metabolism correlates with infarct volume in stroke. Eur. J. Neurosci. 2007, 26, 2211–2221.
- Yan, E.B.; Frugier, T.; Lim, C.K.; Heng, B.; Sundaram, G.; Tan, M.; Rosenfeld, J.V.; Walker, D.W.; Guillemin, G.J.; Morganti-Kossmann, M.C. Activation of the kynurenine pathway and increased production of the excitotoxin quinolinic acid following traumatic brain injury in humans. J. Neuroinflamm. 2015, 30, 110.
- Ogawa, S.; Fujii, T.; Koga, N.; Hori, H.; Teraishi, T.; Hattori, K.; Noda, T.; Higuchi, T.; Motohashi, N.; Kunugi, H. Plasma L-tryptophan concentration in major depressive disorder: new data and meta-analysis. J. Clin. Psychiatry 2014, 75, e906–e915.
- Réus, G.; Jansen, K.; Titus, S.; Carvalho, A.F.; Gabbay, V.; Quevedo, J. Kynurenine pathway dysfunction in the pathophysiology and treatment of depression: evidences from animal and human studies. J. Psychiatry Res. 2015, 68, 316–328.
- Ogyu, K.; Kubo, K.; Noda, Y.; Iwata, Y.; Tsugawa, S.; Omura, Y.; Wada, M.; Tarumi, R.; Plitman, E.; Moriguchi, S.; et al. Kynurenine pathway in depression: A systematic review and meta-analysis. Neurosci. Biobehav. Rev. 2018, 90, 16–25.
- Serafini, G.; Hayley, S.; Pompili, M.; Dwivedi, Y.; Brahmachari, G.; Girardi, P.; Amore, M. Hippocampal neurogenesis, neurotrophic factors and depression: Possible therapeutic targets? CNS Neurol. Disord. Drug Targets 2014, 13, 1708–1721.
- Serafini, G.; Adavastro, G.; Canepa, G.; Capobianco, L.; Conigliaro, C.; Pittaluga, F.; Murri, M.B.; Valchera, A.; De Berardis, D.; Pompili, M.; et al. Abnormalities in Kynurenine Pathway Metabolism in Treatment-Resistant Depression and Suicidality: A Systematic Review. CNS Neurol. Disord. Drug Targets 2017, 16, 440–453.
- Suzuki, H.; Ohgidani, M.; Kuwano, N.; Chrétie, F.; Lorin de la Grandmaison, G.; Onaya, M.; Tominaga, I.; Setoyama, D.; Kang, D.; Mimura, M.; et al. Suicide and Microglia: Recent Findings and Future Perspectives Based on Human Studies. Front. Cell Neurosci. 2019, 13, 31.
- Martinez-Arán, A.; Vieta, E.; Colom, F.; Torrent, C.; Sánchez-Moreno, J.; Reinares, M.; Benabarre, A.; Goikolea, J.M.; Brugué, E.; Daban, C.; et al. Cognitive impairment in euthymic bipolar patients: implications for clinical and functional outcome. Bipolar. Disord. 2004, 6, 224–232.
- Malhi, G.; Ivanovski, B.; Hadzi-Pavlovic, D.; Mitchell, P.B.; Vieta, E.; Sachdev, P. Neuropsychological deficits and functional impairment in bipolar depression, hypomania and euthymia. Bipolar Disord. 2007, 9, 114–125.
- Birner, A.; Platzer, M.; Bengesser, S.A.; Dalkner, N.; Fellendorf, F.T.; Queissner, R.; Pilz, R.; Rauch, P.; Maget, A.; Hamm, C. Increased breakdown of kynurenine towards its neurotoxic branch in bipolar disorder. PLoS ONE 2017, 12, e0172699.
- Wang, A.K.; Miller, B.J. Meta-analysis of Cerebrospinal Fluid Cytokine and Tryptophan Catabolite Alterations in Psychiatric Patients: Comparisons Between Schizophrenia, Bipolar Disorder, and Depression. Schizophr. Bull. 2018, 44, 75–83.
- Arnone, D.; Saraykar, S.; Salem, H.; Teixeira, A.L.; Dantzer, R.; Selvaraj, S. Role of Kynurenine pathway and its metabolites in mood disorders: A systematic review and meta-analysis of clinical studies. Neurosci. Biobehav. Rev. 2018, 92, 477–485.
- Orlikov, A.B.; Prakhye, I.B.; Ryzov, I.V. Kynurenine in blood plasma and DST in patients with endogenous anxiety and endogenous depression. Biol. Psychiatry 1994, 36, 97–102.
- Altmaier, E.; Emeny, R.; Krumsiek, J.; Lacruz, M.E.; Lukaschek, K.; Häfner, S.; Kastenmüller, G.; Römisch-Margl, W.; Prehn, C.; Mohney, R.P.; et al. Metabolomic profiles in individuals with negative affectivity and social inhibition: a population-based study of Type D personality. Psychoneuroendocrinology 2013, 38, 1299–1309.
- Lim, C.K.; Essa, M.M.; de Paula Martins, R.; Lovejoy, D.B.; Bilgin, A.A.; Waly, M.I.; Al-Farsi, Y.M.; Al-Sharbati, M.; Al-Shaffae, M.A.; Guillemin, G.J. Altered kynurenine pathway metabolism in autism: Implication for immune-induced glutamatergic activity. Autism Res. 2016, 9, 621–631.
- Bryn, V.; Verkerk, R.; Skjeldal, O.H.; Saugstad, O.D.; Ormstad, H. Kynurenine Pathway in Autism Spectrum Disorders in Children. Neuropsychobiology 2017, 76, 82–88.
- Plitman, E.; Iwata, Y.; Caravaggio, F.; Nakajima, S.; Chung, J.K.; Gerretsen, P.; Kim, J.; Takeuchi, H.; Chakravarty, M.M.; Remington, G.; et al. Kynurenic acid in schizophrenia: A systematic review and meta-analysis. Schizophr. Bull. 2017, 43, 764–777.





