
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Shuiqiao Yuan | + 1745 word(s) | 1745 | 2020-11-17 03:26:33 | | | |
| 2 | Shuiqiao Yuan | + 10 word(s) | 1755 | 2020-11-17 09:08:15 | | | | |
| 3 | Dean Liu | -459 word(s) | 1296 | 2020-11-17 09:16:03 | | |
Video Upload Options
For a long time, the substantial significance of seminal plasma factors’ functions has been underestimated, which is restricted to spermatozoa transport and protection. Notably, significant advancements have been made in dissecting seminal plasma components, revealing new insights into multiple aspects of sperm function, as well as fertilization and pregnancy outcomes in recent years. In this review, we summarize the state-of-art discoveries regarding SP compositions and their implications in male fertility, particularly describing the novel understanding of seminal plasma components and related modifications using “omics” approaches and mainly focusing on proteome and RNA-seq data in the latest decade. Meanwhile, we highlighted the proposed mechanism of the regulation of SP molecules on immunomodulation in the female reproductive tract. Moreover, we also discussed the proteins investigated as non-invasive diagnosis biomarkers for male infertility in the clinic.
1. Introduction
To date, the majority of male infertility research has mainly focused on abnormality related to the testes, where the male gametes (spermatozoa) are produced through spermatogenesis and spermiogenesis[1]. For human male reproduction, apart from the testes, the accessory glands, including the prostate, seminal vesicles, and bulbourethral glands, are also essential, secreting a biological fluid termed as seminal plasma (SP) around sperm from ejaculation to the fertilization process[2]. Seminal plasma makes up more than 95% of human semen, whereas testicular secretions containing spermatozoa compose about 5%. Clinically, male fertility is often evaluated by routine semen analysis, which serves as a baseline marker with data regarding sperm quantity and quality, including sperm count, concentration, viability, motility, and morphology, while decreased fertility frequently associates with aberrant semen parameters[3].
However, limitations exist in investigating male infertility exclusively using routine semen analysis parameters for a certain patient since unexplained male infertility (UMI) cases have an average prevalence of approximately 15% among all male infertility cases[4]. Unexplained male infertility refers to the diagnosis for an individual whose semen fulfills WHO criteria but fails to conceive offspring. Thus, it is necessary and urgent to develop novel evaluation tests assessing male factor infertility besides examining the structure and function of spermatozoa and, furthermore, the possible involvement of seminal plasma factors should be considered[5]. Seminal plasma is the fluid part of semen, which carries spermatozoa passing throughout the male and female reproductive tract during ejaculation, eventually reaching the oocyte for successful fertilization. It is composed of a complicated set of heterogeneous molecular structures, including proteins, lipids, sugars (fructose), cell-free nucleic acid (DNA, microRNA, and LncRNA), and small-molecule metabolites as well as inorganic chemicals (ions). In general, seminal plasma factors provide energy for spermatozoa metabolism and motility and modulate spermatozoa function by regulating a cascade of molecular events, such as sperm maturation in the epididymis and capacitation during transport. Plasma molecules can give an idea about sperm concentration, motility, morphology, and cause of infertility. Notably, emerging evidence has indicated that seminal plasma is not merely the beneficial medium of spermatozoa but also contains essential spermatozoa function modulators (Figure 1). Recently, advancing technologies in the “omics” fields, such as genomics, transcriptomics, proteomics, and metabolomics, have allowed uncovering of novel aspects and improved understanding of seminal plasma involved in male sub- and infertility.
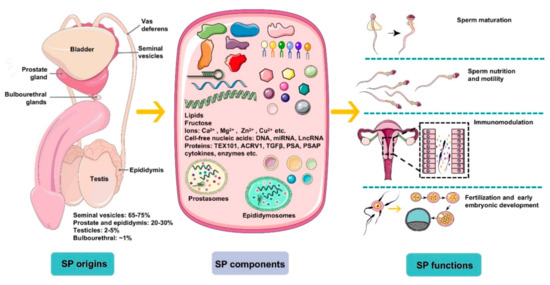
Figure 1. An overview of the origins, components, and main functions of seminal plasma (SP). Seminal plasma makes up more than 95% of the human ejaculate volume, whereas testicular secretions containing spermatozoa compose about 2–5% of semen. Seminal plasma is mainly derived from seminal vesicles, the prostate and epididymis, which produce ~65–75% and ~20–30% of the volume of semen, respectively, with a small proportion generated by bulbourethral glands (~1%). SP is composed of a complicated set of heterogeneous molecular structures, including proteins (enzymes, cytokines, TEX101, ACRV1, TGFΒβ, prostate-specific antigen (PSA), prostatic-specific acid phosphatase (PSAP), etc.), lipids, sugars (fructose), cell-free nucleic acid (DNA, microRNA, and LncRNA), ions (Ca2+, Mg2+, Zn2+, Cu2+, etc.), and small-molecule metabolites. In general, SP is a beneficial medium for spermatozoa maturation, nutrition, and motility and modulates spermatozoa function by regulating a cascade of molecular events, such as sperm maturation in the epididymis and capacitation during transport. More importantly, plasma molecules, such as cytokines, directly recognize receptors on epithelial cells lining the cervix and uterus to induce synthesis of pro-inflammatory cytokines and chemokines that recruit and activate inflammatory leukocytes. Besides, SP modulates the release of cytokines and growth factors that regulate embryo development in the oviduct and uterus before implantation, which is critical for early embryo development and implantation.
2. Potential Application of Seminal Plasma Factors as Bio-Markers
It is promising that seminal plasma proteins might be sources of biomarkers for the noninvasive diagnosis of male fertility disorders due to their relatively high levels and ease of collection. Aberrant protein concentration changes in seminal plasma can indicate pathological process stages and discriminate different types of male infertility. The verified and validated protein biomarkers are L-PGDS, ACRV1, ECM1, and TEX10. L-PGDS is a 26KDa enzyme and glycoprotein, mainly secreted by Sertoli cells, with unclear male fertility function. ELISA determined the seminal concentration of this enzyme ranging from 0.3 to 42 μg/mL, which was significantly lower in the oligozoospermic group (2.47 ± 0.51 μg/mL) than in the normozoospermic group (9.75 ± 1.49 μg/mL)[6]. A more detailed association study revealed that the concentration of seminal L-PGDS is significantly positively correlated with sperm concentration, sperm motility, and percentage of normal morphology and decreased progressive motility from normal to oligospermic patients [7]. Male infertility can be classified into oligozoospermia, asthenospermia, and azoospermia, based on the number and motility of sperm, among which azoospermia is the most severe of male infertility, with near absence of sperm in semen. SP-10 is an acrosomal matrix protein encoded by the acrosomal vesicle protein 1 (ACRV1) gene, which is specifically expressed in spermatids after meiosis and sperm [8]. ELISA using monoclonal antibody revealed a direct relationship between sperm count and seminal concentration of SP-10, which permits it to be a biomarker for oligozoospermic diagnosis. SpermCheck Fertility, a reliable and straightforward immunodiagnostic test based on the above idea, was developed, which quickly provides information for males on whether their sperm number is normal or not to evaluate their fertility [9], and the clinical and consumer trial of SpermCheck Fertility has been applied to detect extreme oligospermia or azoospermia[10].
To date, testicular biopsy is the only valuable method to detect whether spermatozoa are in the testis or not and to distinguish obstructive azoospermia (OA) and nonobstructive azoospermia (NOA). However, surgical exploration of random testicular tissue may not accurately reflect NOA histopathology because of the spatial distribution of spermatogenesis [11]. Searching for biomarkers may allow for avoiding the painful biopsy and deviation of the diagnosis results. Fortunately, some proteins can potentially differentiate normal, OA, and NOA patients. Seminal L-PGDS level could be applied as a biomarker for azoospermia, and high L-PGDS (more than 100 g/L) in men with azoospermia could be potentially diagnosed as nonobstructive azoospermia without biopsy, which contributes to infertility in almost 30% of these men [12]. The concentrations of epididymis-specific expressed ECM1 in OA patients are significantly lower than in control and NOA cases and, more importantly, could distinguish OA cases from normal ones with 100% specificity and sensitivity, and OAs from NOAs with 73% specificity, at 100% sensitivity at a cutoff of 2.3 mg/mL[11].
TEX101 is a membrane glycoprotein and is specifically expressed in germ cells without any expression in other tissues and cells, which can be cleaved from the spermatozoa surface and released into seminal plasma during sperm maturation into epididymis[13]. The notably decreased concentration in azoospermia makes it valuable as a biomarker to discriminate azoospermia from normal controls. Furthermore, the specific expression pattern allows it to differentiate various histopathological NOA subtypes, including hypospermatogenesis (HS), maturation arrest (MA), and Sertoli cell-only syndrome (SCO). In fact, the average concentration of seminal TEX101 from normal men is approximately 2 mg/mL, whereas there are low levels in SP (<120 ng/mL) in NOA cases with HS and MA, and it is not detectable in SCO without germ cells[11].
References
- Krausz, C.; Riera-Escamilla, A. Genetics of male infertility. Nat. Rev. Urol. 2018, 15, 369–384.
- Gilany, K.; Minai-Tehrani, A.; Savadi-Shiraz, E.; Rezadoost, H.; Lakpour, N. Exploring the human seminal plasma proteome: An unexplored gold mine of biomarker for male infertility and male reproduction disorder. J. Reprod. Infertil. 2015, 16, 61–71.
- World Health Organization. WHO Laboratory Manual for The Examination And Processing of Human Semen, 5th ed.; World Health Organization: Geneva, Swizerland, 2010.
- Hamada, A.; Esteves, S.C.; Nizza, M.; Agarwal, A. Unexplained male infertility: Diagnosis and management. Int. Braz. J. Urol. Off. J. Braz. Soc. Urol. 2012, 38, 576–594.
- Szczykutowicz, J.; Kaluza, A.; Kazmierowska-Niemczuk, M.; Ferens-Sieczkowska, M. The Potential Role of Seminal Plasma in the Fertilization Outcomes. Biomed. Res. Int. 2019, 2019, 5397804.
- Tokugawa, Y.; Kunishige, I.; Kubota, Y.; Shimoya, K.; Nobunaga, T.; Kimura, T.; Saji, F.; Murata, Y.; Eguchi, N.; Oda, H.; et al. Lipocalin-type prostaglandin D synthase in human male reproductive organs and seminal plasma. Biol. Reprod. 1998, 58, 600–607.
- Diamandis, E.P.; Arnett, W.P.; Foussias, G.; Pappas, H.; Ghandi, S.; Melegos, D.N.; Mullen, B.; Yu, H.; Srigley, J.; Jarvi, K. Seminal plasma biochemical markers and their association with semen analysis findings. Urology 1999, 53, 596–603.
- Kurth, B.E.; Wright, R.M.; Flickinger, C.J.; Herr, J.C. Stage-specific detection of mRNA for the sperm antigen SP-10 in human testes. Anat. Rec. 1993, 236, 619–625.
- Coppola, M.A.; Klotz, K.L.; Kim, K.A.; Cho, H.Y.; Kang, J.; Shetty, J.; Howards, S.S.; Flickinger, C.J.; Herr, J.C. SpermCheck Fertility, an immunodiagnostic home test that detects normozoospermia and severe oligozoospermia. Hum. Reprod. 2010, 25, 853–861.
- Klotz, K.L.; Coppola, M.A.; Labrecque, M.; Brugh, V.M., 3rd; Ramsey, K.; Kim, K.A.; Conaway, M.R.; Howards, S.S.; Flickinger, C.J.; Herr, J.C. Clinical and consumer trial performance of a sensitive immunodiagnostic home test that qualitatively detects low concentrations of sperm following vasectomy. J. Urol. 2008, 180, 2569–2576.
- Drabovich, A.P.; Dimitromanolakis, A.; Saraon, P.; Soosaipillai, A.; Batruch, I.; Mullen, B.; Jarvi, K.; Diamandis, E.P. Differential diagnosis of azoospermia with proteomic biomarkers ECM1 and TEX101 quantified in seminal plasma. Sci. Transl. Med. 2013, 5, 212ra160.
- Heshmat, S.M.; Mullen, J.B.; Jarvi, K.A.; Soosaipillai, A.; Diamandis, E.P.; Hamilton, R.J.; Lo, K.C. Seminal plasma lipocalin-type prostaglandin D synthase: A potential new marker for the diagnosis of obstructive azoospermia. J. Urol. 2008, 179, 1077–1080.
- Fujihara, Y.; Tokuhiro, K.; Muro, Y.; Kondoh, G.; Araki, Y.; Ikawa, M.; Okabe, M. Expression of TEX101, regulated by ACE, is essential for the production of fertile mouse spermatozoa. Proc. Natl. Acad. Sci. USA 2013, 110, 8111–8116.





