
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | DAVID MORALES-MORALES | -- | 3625 | 2022-09-21 05:51:19 | | | |
| 2 | Lindsay Dong | -12 word(s) | 3613 | 2022-09-21 07:47:20 | | |
Video Upload Options
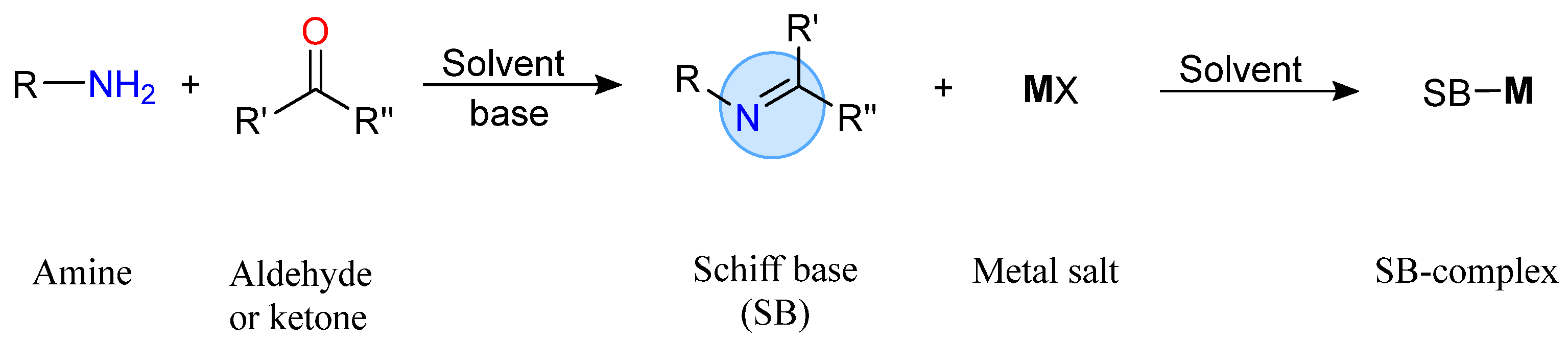
The azomethine group is the common structural feature of SBs, where the substituents can be alkyl, cycloalkyl, aryl, or heterocyclic groups. The carbon atom of the C=Nimine bond is prone to nucleophilic addition, while the nitrogen atom possesses a highly reactive free electron pair that can form stable complexes with metal ions. SBs are among the most widely used organic compounds, showing a wide range of applications as intermediates in organic synthesis, chemosensors, and polymeric stabilizers, in the food, dye, and pigment industry, as well as as catalysts and, in recent years, for their recognized biological properties. The use of tridentate SBs ligands in different organometallic and coordination complexes containing main-group metals and transition metals has been an option to study the biological activity of new possible metallopharmaceuticals that contribute to increase activity and to counteract the effect of microbial resistance.
1. Schiff Bases
1.1. Biological Importance of Schiff Bases
1.2. Schiff Bases as Tridentate Ligands
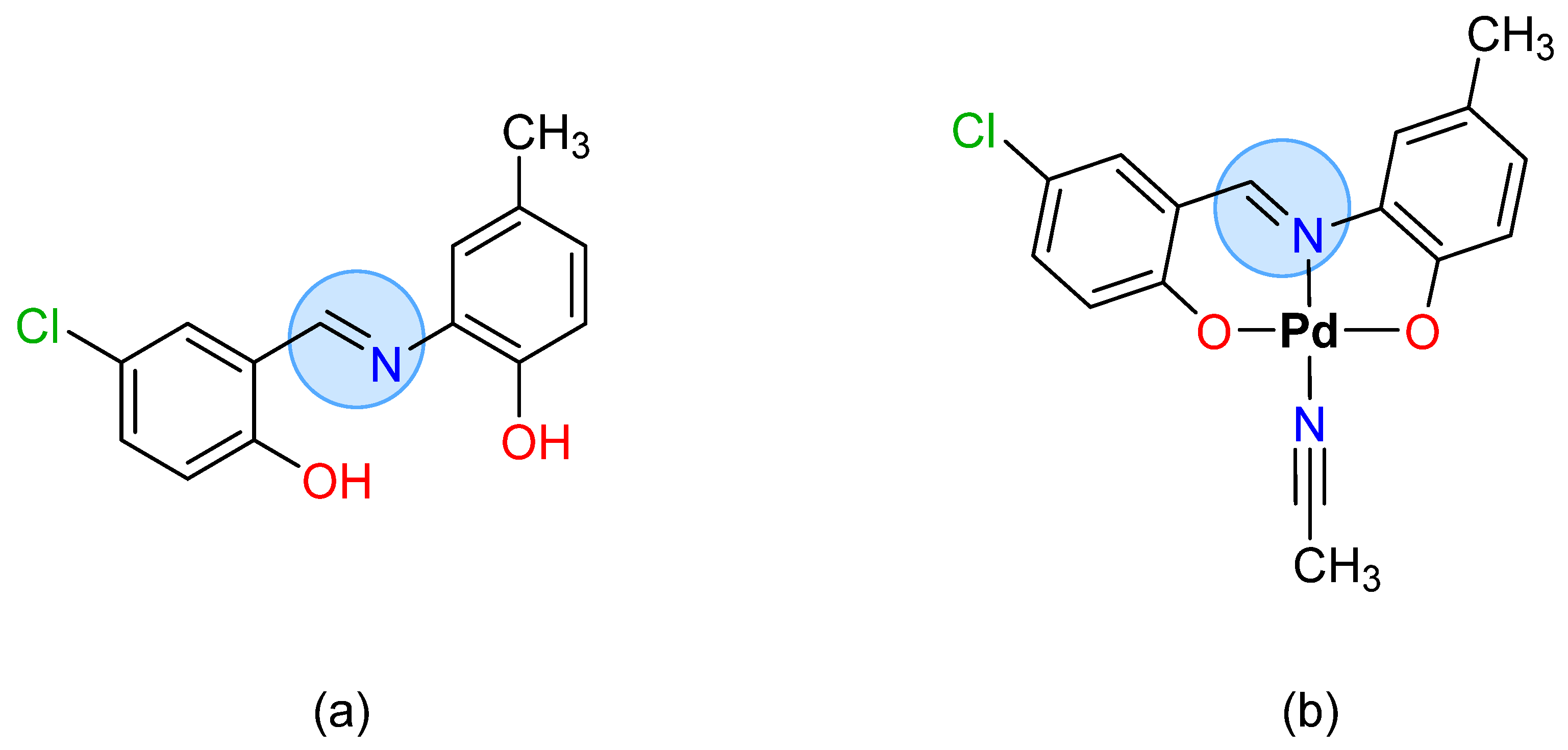
1.3. Synthesis of Metal Complexes Using Schiff Bases
To prepare the metal complexes derived from SBs, triethylamine is generally used to guarantee a basic medium and thus promote the deprotonation of ligands, while the metal is protected by adding weak acids that prevent the precipitation.
2. Antimicrobial Metallopharmaceuticals with Tridentate Schiff Bases
2.1. Main Group Elements
Medicinal inorganic chemistry is a growing field that has proven to be very effective in the treatment and diagnostic of many diseases [13]. Nowadays, transition metal complexes are the most known and most used compounds in the design of metallodrugs. However, main group elements have received extensive attention since the discovery of salvarsan, also known as arsphenamine or Ehrlich 606, a mixture of 3-amino-4-hydroxyphenyl-As(I) and As(V) compounds synthetized by Paul Ehrlich as an effective cure for syphilis. Salvarsan is acknowledged as the first pharmaceutic cure for a disease and opened the door to the research of new molecules that target specific cells to treat many infections [14]. Arsenic was widely used to treat parasite infections, skin diseases, and anaemia in the early 20th century. Livingstone reported the effective action of arsenic in trypanosomiasis, and subsequent reports showed the beneficial action of arsenic against the parasites in the blood stream, but also acknowledged that since the known forms of arsenic were toxic, its use could be lethal to the patient. Organic arsenicals were recognized as less toxic that their inorganic counterpart and were used to treat syphilis and sleeping sickness [15]. Atoxyl, melarsoprol, and melarsonyl are examples of chemotherapeutic drugs for the treatment of infectious diseases [15][16][17]. Furthermore, antimony-based chemotherapy drugs are the main treatment for Leishmaniosis [18]. The pentavalent antimonial compounds that had traditionally been used as treatment for Leishmaniosis (meglumine antimoniate and sodium stibogluconate) had important side effects, and over time the parasite became resistant, so the available drug was not safe enough nor effective [18][19].
Research about the synergic effect of SBs with metals has been addressed mainly for transition metals, and the main group has received less attention, mostly due to the toxicological effects that these kinds of compounds have historically shown. However, in recent decades, the number of reports of medicinal research of SBs with the main group have been increasing. Tin is a main group metal that shares some characteristics with the transition metals. It can form complexes with several organic molecules bound through donor heteroatoms as oxygen, sulphur, or nitrogen, in a wide range of geometries.
SBs with Sn(IV) in vitro antimicrobial activities have been studied against Gram-positive bacteria, Gram-negative bacteria, and fungal strains. In many cases, these complexes showed better activity against Gram-positive strains, such as S. aureus and B. subtilis, than Gram-negative strains, such as E. coli and P. aeruginosa [20][21][22][23]. This result can be explained by the chelation theory that sustains that chelation increases the lipophilic character of the complexes, allowing the permeation of the compound through the cell wall of the bacteria. Through chelation, the metal polarity is reduced by sharing its positive charge with the heteroatom in the ligand, originating an electron delocalization over the metallo-ring. As such, the biological activity of organotin(IV) complexes depend on the donor ligand, the geometry, and the coordination number over the tin atom [21][24].
Along with the chemistry of organotin(IV) complexes, the biological activities of hypercoordinated silicon compounds with SB ligands have also been studied [25][26]. Silicon compounds with tridentate SBs are scarce, with some early reports by Puri et al. looking to examine the physical and chemical properties of these compounds. In this report, a series of complexes, which exhibit a hexacoordinate Si(IV) atom and the ligand N,N′-diethylenetriamine-bis(salicylideneimine), are reported [27].
2.2. Titanium Group
2.3. Vanadium Group
2.4. Chromium Group
2.5. Manganese Group
2.6. Iron Group
2.7. Cobalt Group
2.8. Nickel, Copper, and Zinc Groups
3. Conclusions
References
- Jia, Y.; Li, J. Molecular Assembly of Schiff Base Interactions: Construction and Application. Chem. Rev. 2015, 115, 1597–1621.
- Ritter, E.; Przybylski, P.; Brzezinski, B.; Bartl, F. Schiff Bases in Biological Systems. Curr. Org. Chem. 2009, 13, 241–249.
- Uddin, M.N.; Ahmed, S.S.; Alam, S.M.R. REVIEW: Biomedical applications of Schiff base metal complexes. J. Coord. Chem. 2020, 73, 3109–3149.
- Andiappan, K.; Sanmugam, A.; Deivanayagam, E.; Karuppasamy, K.; Kim, H.-S.; Vikraman, D. In vitro cytotoxicity activity of novel Schiff base ligand–lanthanide complexes. Sci. Rep. 2018, 8, 3054.
- Banerjee, A.; Chattopadhyay, S. Synthesis and characterization of mixed valence cobalt(III)/cobalt(II) complexes with N,O-donor Schiff base ligands. Polyhedron 2019, 159, 1–11.
- Alterhoni, E.; Tavman, A.; Hacioglu, M.; Şahin, O.; Seher Birteksöz Tan, A. Synthesis, structural characterization and antimicrobial activity of Schiff bases and benzimidazole derivatives and their complexes with CoCl2, PdCl2, CuCl2 and ZnCl2. J. Mol. Struct. 2021, 1229, 129498.
- Gupta, D.; Jamwal, D.; Rana, D.; Katoch, A. Microwave Synthesized Nanocomposites for Enhancing Oral Bioavailability of Drugs; Elsevier Inc.: Amsterdam, The Netherlands, 2018; ISBN 9780128137581.
- Mishra, A.P.; Jain, R.K. Conventional and microwave synthesis, spectral, thermal and antimicrobial studies of some transition metal complexes containing 2-amino-5-methylthiazole moiety. J. Saudi Chem. Soc. 2014, 18, 814–824.
- Fahmi, N.; Shrivastava, S.; Meena, R.; Joshi, S.C.; Singh, R.V. Microwave assisted synthesis, spectroscopic characterization and biological aspects of some new chromium(iii) complexes derived from N⁁O donor Schiff bases. New J. Chem. 2013, 37, 1445–1453.
- More, R.B.; Mahale, R.G.; Patil, S.R. Ultrasound assisted Schiff base metal complexes, organic reactions and nano-particles synthesis study—A Review. J. Res. Pharm. Sci. 2020, 6, 35–40.
- Merzougui, M.; Ouari, K.; Weiss, J. Ultrasound assisted synthesis, characterization and electrochemical study of a tetradentate oxovanadium diazomethine complex. J. Mol. Struct. 2016, 1120, 239–244.
- Abdel-Rahman, L.H.; Abu-Dief, A.M.; El-Khatib, R.M.; Abdel-Fatah, S.M. Some new nano-sized Fe(II), Cd(II) and Zn(II) Schiff base complexes as precursor for metal oxides: Sonochemical synthesis, characterization, DNA interaction, in vitro antimicrobial and anticancer activities. Bioorg. Chem. 2016, 69, 140–152.
- Mjos, K.D.; Orvig, C. Metallodrugs in Medicinal Inorganic Chemistry. Chem. Rev. 2014, 114, 4540–4563.
- Lloyd, N.C.; Morgan, H.W.; Nicholson, B.K.; Ronimus, R.S. The composition of Ehrlich’s Salvarsan: Resolution of a century-old debate. Angew. Chem. Int. Ed. 2005, 44, 941–944.
- Jolliffe, D.M. A history of the use of arsenicals in man. J. R. Soc. Med. 1993, 86, 287–289.
- Wéry, M. Drug used in the treatment of sleeping sickness (human African trypanosomiasis: HAT). Int. J. Antimicrob. Agents 1994, 4, 227–238.
- Breinl, A.; Todd, J.L. Atoxyl in the treatment of trypanosomiasis. BMJ 1907, 1, 132–134.
- Davies-Bolorunduro, O.F.; Osuolale, O.; Saibu, S.; Adeleye, I.A.; Aminah, N.S. Bioprospecting marine actinomycetes for antileishmanial drugs: Current perspectives and future prospects. Heliyon 2021, 7, e07710.
- Mohammadi, M.; Zaki, L.; Saryazdi, A.K.P.; Tavakoli, P.; Tavajjohi, A.; Poursalehi, R.; Delavari, H.; Ghaffarifar, F. Efficacy of green synthesized silver nanoparticles via ginger rhizome extract against Leishmania major in vitro. PLoS ONE 2021, 16, e0255571.
- Bhatra, P.; Sharma, J.; Sharma, R.A.; Singh, Y. Synthesis, characterization and antimicrobial activity of diorganotin(IV) derivatives of some bioactive bifunctional tridentate Schiff base ligands. Main Gr. Met. Chem. 2016, 39, 1–8.
- Devi, J.; Yadav, J.; Singh, N. Synthesis, characterisation, in vitro antimicrobial, antioxidant and anti-inflammatory activities of diorganotin(IV) complexes derived from salicylaldehyde Schiff bases. Res. Chem. Intermed. 2019, 45, 3943–3968.
- Prasad, K.S.; Kumar, L.S.; Chandan, S.; Jayalakshmi, B.; Revanasiddappa, H.D. Diorganotin(IV) complexes of biologically potent 4(3H)-quinazolinone derived Schiff bases: Synthesis, spectroscopic characterization, DNA interaction studies and antimicrobial activity. Spectrochim. Acta-Part A Mol. Biomol. Spectrosc. 2011, 81, 276–282.
- Sedaghat, T.; Aminian, M.; Bruno, G.; Amiri Rudbari, H. Binuclear organotin(IV) complexes with adipic dihydrazones: Synthesis, spectral characterization, crystal structures and antibacterial activity. J. Organomet. Chem. 2013, 737, 26–31.
- Manju; Mishra, N.; Kumar, D. Coordination chemistry of Schiff base tin complexes. Russ. J. Coord. Chem. Khimiya 2014, 40, 343–357.
- Bhanuka, S.; Khaturia, S.; Chahar, M.; Singh, H.L. Design, Spectroscopic Characterization and Theoretical Studies of Organotin(IV) and Organosilicon(IV) Complexes with Schiff Base Ligands Derived from Amino Acids. Asian J. Chem. 2020, 32, 2821–2828.
- Singh, K.; Puri, P.; Kumar, Y.; Sharma, C. Hypercoordinated Organosilicon(IV) and Organotin(IV) Complexes: Syntheses, Spectral Studies, and Antimicrobial Activity In Vitro. ISRN Inorg. Chem. 2013, 2013, 356802.
- Puri, J.K.; Singh, R.; Chahal, V.K.; Sharma, R.P. Novel hexacoordinate organosilicon(IV) complexes of diethylenetriamine schiff base with SiO2N3 skeleton. Arkivoc 2009, 2009, 247–256.
- Mendes, I.C.; Moreira, J.P.; Ardisson, J.D.; dos Santos, R.G.; da Silva, P.R.O.; Garcia, I.; Castiñeiras, A.; Beraldo, H. Organotin(IV) complexes of 2-pyridineformamide-derived thiosemicarbazones: Antimicrobial and cytotoxic effects. Eur. J. Med. Chem. 2008, 43, 1454–1461.
- El-Saied, F.; Ayad, M.; Aly, S. Tin(IV), titanium(IV) and hafnium(IV) complexes of some aromatic Schiff bases derived from 4-aminoantipyrine. Transit. Met. Chem. 1993, 18, 279–282.
- Al-Hakimi, A.N.; Alminderej, F.; Aroua, L.; Alhag, S.K.; Alfaifi, M.Y.; Samir, O.M.; Mahyoub, J.A.; Elbehairi, S.E.I.; Alnafisah, A.S. Design, synthesis, characterization of zirconium (IV), cadmium (II) and iron (III) complexes derived from Schiff base 2-aminomethylbenzimidazole, 2-hydroxynaphtadehyde and evaluation of their biological activity. Arab. J. Chem. 2020, 13, 7378–7389.
- Prakash, A.; Ahmad, S.I.M. Physico-chemical and antimicrobial studies on Ni(II), Cu (II) and Ti(III) Schiff base complexes derived from 2- furfuraldehyde. Orient. J. Chem. 2009, 25, 391–395.
- Yaul, A.R.; Dhande, V.V.; Yaul, S.R.; Aswar, A.S. Transition metal complexes containing tridentate hydrazone Schiff bases: Synthesis, characterization and biological activity. J. Indian Chem. Soc. 2011, 88, 775–780.
- Shashidhara, G.M.; Goudar, T.R. Oxovanadium(IV) and Niobium(V) Complexes with Some New Schiff Bases. Synth. React. Inorg. Met. Chem. 2000, 30, 1581–1599.
- Panchal, P.K.; Parekh, H.M.; Pansuriya, P.B.; Patel, M.N. Bactericidal activity of different oxovanadium(IV) complexes with Schiff bases and application of chelation theory. J. Enzyme Inhib. Med. Chem. 2006, 21, 203–209.
- Ebosie, N.P.; Ogwuegbu, M.O.C.; Onyedika, G.O.; Onwumere, F.C. Biological and analytical applications of Schiff base metal complexes derived from salicylidene-4-aminoantipyrine and its derivatives: A review. J. Iran. Chem. Soc. 2021, 18, 3145–3175.
- Zhang, J.; Jiang, P.; Shen, Y.; Zhang, W.; Li, X. Molybdenum(VI) complex with a tridentate Schiff base ligand immobilized on SBA-15 as effective catalysts in epoxidation of alkenes. Microporous Mesoporous Mater. 2015, 206, 161–169.
- Cindrić, M.; Strukan, N.; Vrdoljak, V.; Kajfež, T.; Kamenar, B. A Series of Molybdenum(VI) Complexes with Tridentate Schiff Base Ligands. Z. Anorg. Allg. Chem. 2002, 628, 2113–2117.
- McAuliffe, C.A.; Sayle, B.J. Coordination chemistry of molybdenum and tungsten—VIII. Oxomolybdenum(V) complexes of 8-hydroxyquinoline and relevance of EPR spectra to binding sites in flavoenzymes. Bioinorg. Chem. 1978, 8, 331–339.
- Agarwal, D.D.; Rastogi, R.; Gupta, V.K.; Singh, S. Olefin eooxidation catalyzed by dioxo- tungsten(VI) schiff base complexes 4. Indian J. Chem. 1999, 38, 369–372.
- Amini, M.; Bagherzadeh, M.; Eftekhari-Sis, B.; Ellern, A.; Keith Woo, L. Catalytic efficacy of an oxido-peroxido tungsten(VI) complex: Synthesis, X-ray structure and oxidation of sulfides and olefins. J. Coord. Chem. 2013, 66, 1897–1905.
- Suresh, M.S.; Prakash, V. Preparation and characterization of Cr(III), Mn(II), Co(III), Ni(II), Cu(II), Zn(II) and Cd(II) chelates of schiffs base derived from vanillin and 4-amino antipyrine. Int. J. Phys. Sci. 2010, 5, 2203–2211.
- Abu-Dief, A.M.; El-khatib, R.M.; Aljohani, F.S.; Alzahrani, S.O.; Mahran, A.; Khalifa, M.E.; El-Metwaly, N.M. Synthesis and intensive characterization for novel Zn(II), Pd(II), Cr(III) and VO(II)-Schiff base complexes; DNA-interaction, DFT, drug-likeness and molecular docking studies. J. Mol. Struct. 2021, 1242, 130693.
- Zhang, L.Z.; Ding, T.; Chen, C.L.; Li, M.X.; Zhang, D.; Niu, J.Y. Biological activities of pyridine-2-carbaldehyde Schiff bases derived from S-methyl- and S-benzyldithiocarbazate and their zinc(II) and manganese(II) complexes. Crystal Structure of the Manganese(II) complex of pyridine-2-carbaldehyde S-benzyldithiocarbaz. Russ. J. Coord. Chem. 2011, 37, 356–361.
- Jamuna, K.; Naik, B.R.; Sreenu, B.; Seshaiah, K. Synthesis, characterization and antibacterial activity of Cu(II) and Fe(III) complexes of a new tridentate Schiff base ligand. J. Chem. Pharm. Res. 2012, 4, 4275–4282.
- El-Sherif, A.A.; Shoukry, M.M.; Abd-Elgawad, M.M.A. Synthesis, characterization, biological activity and equilibrium studies of metal(II) ion complexes with tridentate hydrazone ligand derived from hydralazine. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 98, 307–321.
- Shebl, M.; Saif, M.; Nabeel, A.I.; Shokry, R. New non-toxic transition metal nanocomplexes and Zn complex-silica xerogel nanohybrid: Synthesis, spectral studies, antibacterial, and antitumor activities. J. Mol. Struct. 2016, 1118, 335–343.





