Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Cezar Comanescu | -- | 2827 | 2022-09-01 23:11:10 | | | |
| 2 | Rita Xu | -153 word(s) | 2674 | 2022-09-02 03:31:14 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Comanescu, C. Justification and Design Strategies for Magnetic Nanoplatforms. Encyclopedia. Available online: https://encyclopedia.pub/entry/26805 (accessed on 29 March 2026).
Comanescu C. Justification and Design Strategies for Magnetic Nanoplatforms. Encyclopedia. Available at: https://encyclopedia.pub/entry/26805. Accessed March 29, 2026.
Comanescu, Cezar. "Justification and Design Strategies for Magnetic Nanoplatforms" Encyclopedia, https://encyclopedia.pub/entry/26805 (accessed March 29, 2026).
Comanescu, C. (2022, September 01). Justification and Design Strategies for Magnetic Nanoplatforms. In Encyclopedia. https://encyclopedia.pub/entry/26805
Comanescu, Cezar. "Justification and Design Strategies for Magnetic Nanoplatforms." Encyclopedia. Web. 01 September, 2022.
Copy Citation
Magnetic nanoparticles (MNPs) have evolved tremendously during recent years, in part due to the rapid expansion of nanotechnology and to their active magnetic core with a high surface-to-volume ratio, while their surface functionalization opened the door to a plethora of drug, gene and bioactive molecule immobilization. Taming the high reactivity of the magnetic core was achieved by various functionalization techniques, producing MNPs tailored for the diagnosis and treatment of cardiovascular or neurological disease, tumors and cancer.
magnetic nanoparticle
MRI
nanomedicine
drug delivery
1. Introduction
Ever since nanotechnology grew into a reckoned field of its own, the implications in medicine and pharmacology became obvious, and are today exploited commercially on many drug formulations. A busy lifestyle, erratic work schedule and oxidative stress, together with genetic and other risk factors have contributed to a surge in cancer incidence throughout the globe, and current estimates predict about 1.26 million deaths for 2022 in the EU due to cancer alone. Conventional approaches come with unavoidable side effects due to systemic exposure and response, and are less effective than directed therapy—a field where MNPs and SPIONs, in particular, have emerged as suitable candidates with more efficient delivery of the anticancer drugs and limited negative effects on neighboring tissues and organs.
Magnetic nanoparticles (MNPs) are at the very core of magnetic delivery systems and they aim to tackle site-specific tumors while ideally affording a controlled-release profile suitable for disease treatment. Their multifunctional dimensionality makes it possible for MNPs to be used in nanomedicine as elective candidates for drug targeting therapy when using an externally applied magnetic field.
With tunable physico-chemical properties and a very high surface-to-volume ratio typical for nanoparticles, MNPs can be engineered into drug-delivery systems with similar sizes to the organism’s own antibodies or proteins for improved biocompatibility, while incorporating therapeutic agents that would otherwise be difficult to deliver to the cancer cells. When superparamagnetic nanoparticles (SPIONs) are coated with biologically compatible polymers of fatty acids, systems with improved colloidal stability and reduced tendency of aggregation are obtained. MNPs were also used as contrast agents in magnetic resonance imaging (MRI). When functionalized with epithelial growth factor receptor antibodies or aptamers, an efficient diagnosis tool is created for many types of cancer or even detection of brain inflammation. MNPs are being used as alternative contrast agents in MR imaging owing to their superparamagnetic properties and high relaxivity, doubled by high biocompatibility upon surface functionalization and low toxicity, unlike the Gd complexes used traditionally as contract agent in MRI, which could potentially release Gd into the bloodstream.
2. Justification and Design Strategies for Magnetic Nanoplatforms
There are multiple reasons pleading for the use of MNPs as carriers for drug and gene delivery, making nanoparticle platforms superior to the traditional administration of the drug alone. Moreover, theranostics are able to integrate today’s MNPs in procedures capable of both diagnosis and disease treatment, which is unachievable via traditional drug administration. Using NPs for drug delivery allows modification of key aspects related to drug solubility, diffusivity, penetration and retention, pharmacokinetics, biodistribution, cytotoxicity, and half-life (controlled- and/or on-demand release).
Considering how time-consuming (~12 years on average) and extremely expensive (up to USD 2 billion) the drug development process can be, it may be surprising to note that, even when an efficient active component is identified, basic hiccups still plague the process, such as a lower grade in the biopharmaceutical system, which assesses the solubility and permeability of a drug. In other words, a highly efficient drug whose development took years of research may never reach the patient in need due to low solubility/permeability in the biological system. This severe shortcoming can be modulated by drug immobilization on magnetic nanocarriers, that are able to transport insoluble drugs to the target site by smart surface modification (for instance, with antibodies that would bind to molecules overexpressed at the tumoral site). Drug formulations employing stable nanoparticle dispersion of the drug can increase absorption, even when using much lower dosages—this advantage alone can make a drug with low bioavailability and poor penetration/absorption when used alone become suitably efficient for drug-delivery systems, with real prospects of reaching the market. Various groups reported that drug molecules conjugated to MNPs can exhibit efficiencies many times higher than using the drug by itself (Figure 1).
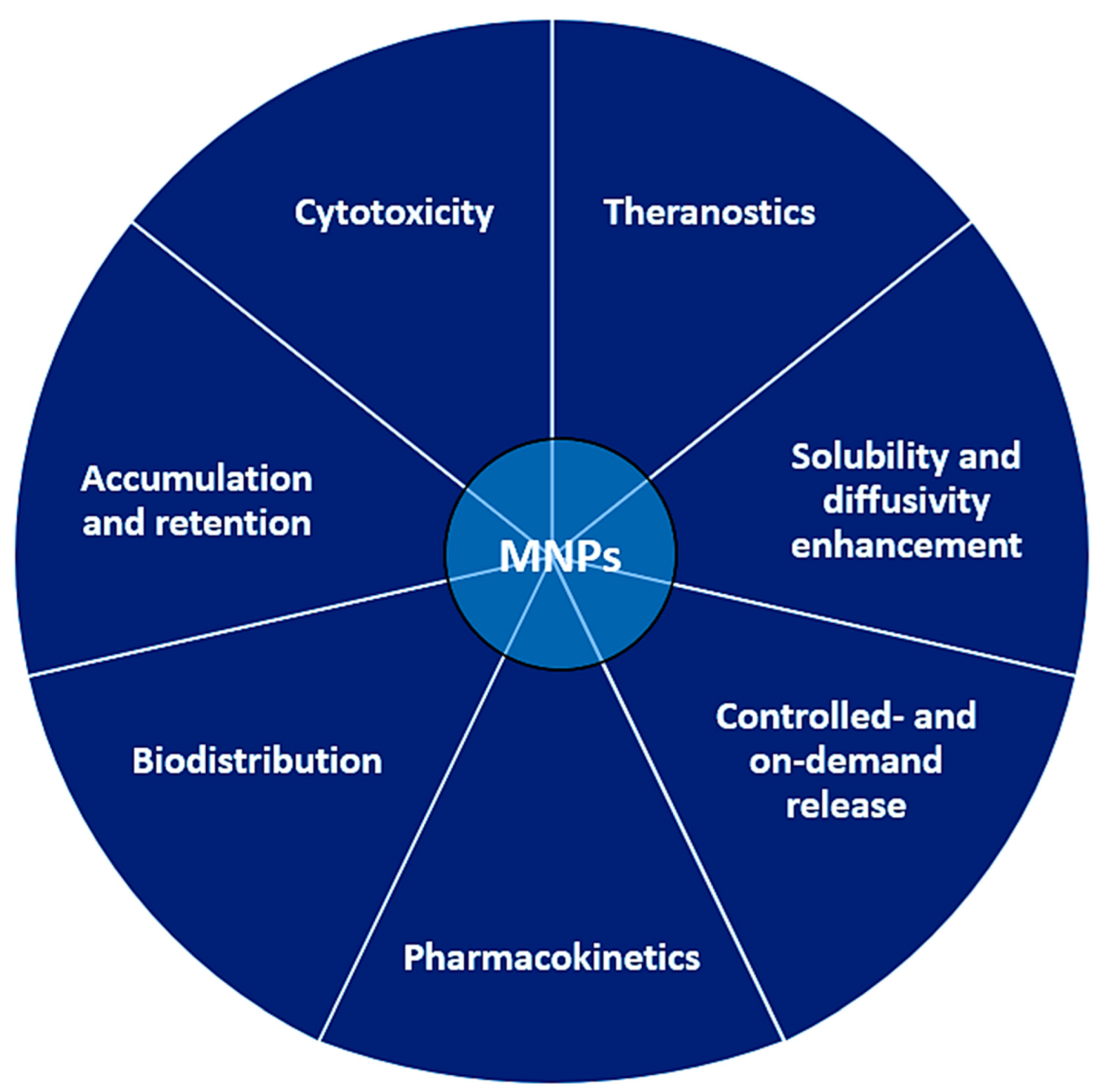
Figure 1. Advantages of using NP-based carrier platforms compared to classical drug administration protocols; improvements are recorded across all presented segments.
Conventional drug administration is typically carried out via oral, parental, pulmonary or transdermal routes, and usually requires several doses for maximum efficiency. However, a controlled release system based on drug-loaded NPs can bypass these shortcomings, by reducing drug quantities and related toxicity, and also the doses required for treatment. In a typical, conventional drug administration, the drug concentration in biological fluids can vary greatly, from subtherapeutic (no effect) to toxic levels; by contrast, the release profile of a drug in a controlled release system is rather constant, and within the limits required for maximal therapeutic effect. Such systems can be regarded as highly beneficial for patients using anti-inflammatory drugs (especially the elders), or diabetic patients; the former will not experience pain due to constant drug concentration in the blood, while the latter will have better glycemic control because insulin would be released on-demand contingent with blood sugar levels. The release can be prolonged to multiple weeks from a single administration, which would be unheard of in the case of traditional administration.
Another key parameter is the enhanced permeability and retention achievable due to specific hypervascularization at the tumor level (with the formation of epithelial pores), which translates into increased permeability to therapeutic drugs when conjugated to polymeric-coated NPs. For efficient drug accumulation, however, the nanoparticle-based drug formulation must be stable in biological fluids (it should not agglomerate/ precipitate), and should be tailored regarding size and concentration for optimal penetration through cellular membrane and cellular uptake.
2.1. Synthetic Strategies and Feedback-Driven Design
Synthetic methods currently utilized for SPION particle synthesis involve the hydrothermal route (with typically mesoporous NPs as the outcome [1][2][3][4][5][6][7][8][9]), as opposed to the classical co-precipitation route, which has a number of disadvantages including reproducibility issues regarding morphological parameters with direct influence over the magnetic properties [10][11][12] or the well-established sol–gel [13] and (auto)combustion [14] methods. Other synthetic routes focus on natural extracts as bio-inspired routes to MNPs based on iron oxides, with encouraging therapeutic potential [15] and other green synthesis strategies [16]. Other nature-inspired compounds such as magnetic zeolites are also under investigation today [17].
2.2. Physical Characterization
Physical characterization methods were employed for particle size determination, including analysis of magnetization curves [18], cation distribution in ferrites by X-ray absorption [19][20] and Mossbauer spectroscopy [21][22].
Magnetic measurements aim at providing a feedback-driven synthesis route for improving effective magnetic moment [23][24][25], especially when referring to ferrofluids since these target actual biologic systems [26][27][28]. Effective quantification of the heating ability of MNPs can be achieved via SAR determination experiments [29] and novel tools such as small-angle scattering can improve the design of functionalized MNPs [30].
2.3. MNPs with Improved Magnetic Properties: Substitution/Doping Effect
Doping strategies have modified the therapeutic potential by altering the paramagnetic behavior of maghemite (γ-Fe2O3) [31], metal ferrites [32], as well as Gd3+ substitution of magnetite Fe3O4 with effect on superparamagnetic NPs [33].
Various strategies for anisotropy enhancement were explored, including layering of MNPs on amorphous substrates with perpendicular anisotropy [34].
Ferrites of spinel structure have been synthesized; BaFe2O4 [35], Mn–Zn ferrite [36], CuFe2O4 [37][38], Cu–Ni ferrite [39], Zn, Cu and Co ferrites [40][41], or Ni–Zn–Co ferrites with Gd3+ substitution [42].
2.4. Flow Characteristics and Simulated Models for MNPs in Biologic Fluids and High-Performance Ferrofluids
The flow parameters (magnetorheology) were reported recently [43][44][45][46][47][48], tackling also convection [49], location [50] and sedimentation processes [51][52] or multi-core MNPs [26], proper distribution in form of appropriate matrices such as gels [53], overall tracking efficiency [54], as well as guidance for cell behavior [55], and actual flow through artificial blood [56].
The effect of viscosity of the medium as a means to counteract the sedimentation tendency of MNPs was investigated [57], while the dispersibility of NPs was shown to be an efficient way to obtain stable ferrofluids and magnetic therapeutic fluids [58][59].
Simulation models of MNPs in the actual blood flow were reported very recently [60][61][62], on the magnetic susceptibility vs. frequency for MNPs [63][64], suitability of poly(vinyl) alcohol PVA coating of MNPs for drug delivery [65] or on the possible replacement of dopamine, the go-to drug used to treat Parkinson’s disease, by magnetite Fe3O4 NPs [66]. Alzheimer’s is also among the diseases targeted by functionalized SPIONs when conjugated with NIR dyes [67].
2.5. Morphology—Role of Size and Shape
Size control of MNPs [68] was been recently achieved in a green synthesis, bioinspired strategy by employing lysine in the synthesis of SPION magnetite materials [69]. Studies have shown that, for particle sizes higher than 100 nm, the MNPs are subject to macrophage phagocytosis by the spleen and liver, hence the requirements to design particles below 100 nm.
Size and shape effects on hyperthermia performance were also studied [70][71][72]. Results point to relatively lower toxicity when spherical nanoparticles are used (such as those obtained by the polyol method), rather than irregular/polyhedral-shaped NPs. I. Craciunescu et al. have investigated hydrophobic (oleic acid coating)/hydrophilic (azelaic acid-coated) magnetite (Fe3O4) and ferrites (MFe2O4, M = Mn, Zn) produced by the polyol method, of different shapes: spherical, cubic, hexagonal, octahedral and sizes (10–100 nm) with interesting findings linking shape and size of MNPs to the hyperthermia procedure. MFH (magnetic fluid hyperthermia) is a technique currently investigated which allows MNP-mediated conversion of alternating magnetic field energy into heat; moreover, this heat release event can be doubled by drug release at the tumor site which enhances the therapeutic chances of success in tumor treatment and should respect a maximal exposure criterion f × H ≤ 5 × 109 Hz × A/m for applicability in biological systems [70]. The SAR (specific absorption rate) is a key parameter quantifying the energy conversion process and is dependent on AC magnetic field amplitude, frequency and MNPs relaxation mechanisms. The magnetization at saturation increases to 90 emu/g in the case of cubic shapes and MNPs of 100 nm average size (Fe3O4 and MnFe2O4) and lower values (50–70 emu/g) for zinc ferrite nanoparticles [70]. Large-sized MNPs are expected to transfer heat in hyperthermia applications by means of hysteretic losses due to magnetic wall displacements, hence of prime interest parameters are magnetization at saturation MS and magnetic susceptibility in low AC magnetic fields—information that can be deduced by analysis of hysteresis loops [70]. Additional information regarding NPs size and size distribution, interparticle interactions and magnetic domain structure can be deduced by analysis of FC–ZFC curves (field-cooled–zero-field-cooled) or Mössbauer spectroscopy (for anisotropy energy determination, KV: K is the magnetic anisotropy constant, and V is the nanoparticle magnetic volume). It is worth noting that among the three types of samples analyzed (Fe3O4, MnFe2O4 and ZnFe2O4), the highest heating efficiency was that of the soft magnetic ZnFe2O4@azelaic acid (AZA) with a SAR of 175 W/g, more than double that of Fe3O4. AZA (SAR = 85 W/g), although it has the lowest saturation field among them. Increasing the heat efficiency is possible when using MNPs in a magnetically frozen regime at room temperature, which will not allow for the formation of moving magnetic domain walls [70].
Manganese ferrite MnFe2O4 was investigated extensively due to tunable magnetic properties, high biocompatibility and chemical stability [70][71]. Besides the essential role of hysteresis losses mentioned above (where it was the dominant heat transfer mechanism), two other mechanisms describe the heat transferred by NPs to the surroundings (SPL, specific power losses): Neel and Brownian relaxation [71]. SPL is also strongly influenced by particle size because this parameter alters the shape anisotropy. Chitosan-coated MnFe2O4 were obtained by co-precipitation of FeCl3 and MnCl2.4H2O with NH4OH, producing stable, functionalized MNPs with potential applications as positive/negative MRI contrast agents in the rat model [71]. A theoretical investigation of various sized Fe3O4 NPs (25, 50, 100 and 200 nm) showed that NPs with lower sizes produced a higher heat gradient in the tumor mesh (61, 49, 42 and 41, respectively), while those in the 50–100 nm size ranges were found to be the most promising candidates for hyperthermia and cellular uptake [72]. Considering the heat produced by hysteresis per volume unit P = μ0 f ∫H dM, one can expect better results when the alternating magnetic field frequency f is increased, within the biologically safe limit [72][73]. Theoretical simulations for correlating size with potential hyperthermia applications were also reported [73].
2.6. Intra- and Interparticle Interactions: Colloidal Stability and Size Differentiation
Interparticle interaction has long been seen as a possible cause of further aggregation [74]. R. Das et al. have shown the effect of shape (NRs nanorings of 55 nm length vs. NTs nanotubes of 470 nm length) on the MFH performance corresponding to Fe3O4 nanoparticles, while also raising the question of inter- and intraparticle interaction [74]. The morphology of NPs was controlled by the amount of NaH2PO4.2H2O used in the first precipitation step (higher concentration leads to NRs, lower concentration favors NTs). The iron oxide nanotubes NTs showed higher effective anisotropy and MS, but lower SAR value (80 W/g at 400 Oe and 300 Hz) than nanorings NRs featuring weaker intraparticle interactions (110 W/g). It becomes apparent that MFH is positively influenced by using MNPs of lower volume and weaker intraparticle interactions [74].
Colloidal stability is a key parameter to preventing undesired NP accumulation before ever reaching the target organ [75][76][77][78][79][80]. For instance, optical tracking of iron oxide ferrofluids at 10 T and a 100 T/m gradient has shown that aqueous ferrofluids are best investigated in high fields, which offer a reliable estimation of their behavior under lower, practical fields [75]: 0.25 vol% iron oxide as stabilized dispersion of citrate-coated maghemite nanoparticles (γ-Fe2O3), and commercial Fe3O4 ferrofluids [75]. Depending on the magnetic field strength (0.3–0.5 T and ~20 T/m for a neodymium magnet, 10 T and 100 T/m for a Bitter magnet), citrate-coated maghemite remains separately dispersed. However, when MNPs of higher polydispersity are used, the largest NPs separate rapidly from the solution while smaller NPs remain dispersed because of their low dipolar coupling energies [75].
V. Pilati et al. synthesized aqueous ferrofluids using the electric double-layer (EDL) strategy to maintain their solution stability [76]. These systems were based on biomagnetic core-shell ZnMn mixed ferrite@ maghemite shell out of which two specific compositions were further investigated, namely ZnδMn1 − δFe2O4@γ-Fe2O3 (δ = 0.2 and 0.5). The surface was further covered by a maghemite layer by exposing the ZnδMn1 − δFe2O4 core (co-precipitation) to HNO3 washing followed by hydrothermal treatment with Fe(NO3)3 0.5 M. The electrostatically stabilized ferrofluid was achieved by peptization of as-synthesized ferrite NPs in a dialysis bag, using HNO3 with pH fine tuning (final pH = 2.0) and solution ionic strength adjustment by means of NaNO3 formation [76]. Interestingly, dynamic light scattering (DLS) and small-angle X-ray scattering (SAXS) revealed that changing the NPs concentration from dilute to >25 mg/mL is accompanied by a change in global interaction forces from attractive (diluted) to repulsive (concentrated) [76]. L. L. e Castro et al. extended the applicability of EDL repulsive interactions by using Monte Carlo simulations to surfacted MNPs, where the charge is located typically at the extremities of the surfactant molecule (at the organic functionality, such as amino, carboxyl, etc.). They ran the simulations on spherically shaped magnetic NPs using a model proposed by Schnitzer and Morozov—an improvement over the traditional DLVO model traditionally used for modeling colloid stability [77].
J.C. Riedl et al. used maghemite (γ-Fe2O3) NPs dispersed in ionic liquids (ILs) based on ethylmethylimidazolium bistriflimide (EMIM TFSI) in a pursuit to obtain colloids stable from room temperature up to 200 °C; the dispersion of maghemite at concentrations up to 12 vol% was shown to be stable for several days at 200 °C [78]. M. Boskovic et al. synthesized Fe3 − xGdxO4 (x = 0, 0.1, 0.2) NPs of diameter ~8 nm by the coprecipitation method and by coating with citric acid (CA) with improved colloidal stability; the sample Fe2.80Gd0.20O4@CA embedded in human serum albumin afforded magnetic microspheres (MMS) as suitable carriers for drug-delivery applications [79]. Polymeric coatings of iron oxide nanoparticles such as silica-coated Fe3O4 NPs (diblock copolymers obtained by living cationic polymerization, PEO-b-PMAA) are oftentimes used because they lower Gibbs free energy of magnetic nanoparticles in solution, hence maintaining colloidal stability and preventing agglomeration [80].
References
- Fadli, A.; Amri, A.; Sari, E.O.; Sukoco, S.; Saprudin, D. Superparamagnetic nanoparticles with mesoporous structure prepared through hydrothermal technique. Mater. Sci. Forum 2020, 1000, 203–209.
- Ghaemi, A.; Mohave, F.; Farhadi, A.; Takassi, M.A.; Tavakkoli, H. Hydrothermal synthesis of mesoporous cobalt ferrite by ionic liquid-assisted process; catalytic performance, morphology, and magnetic studies. J. Aust. Ceram. Soc. 2021, 57, 1321–1330.
- Tombuloglu, H.; Khan, F.A.; Almessiere, M.A.; Aldakheel, S.; Baykal, A. Synthesis of niobium substituted cobalt-nickel nano-ferrite (Co0.5Ni0.5NbxFe2−xO4 (x ≤ 0.1) by hydrothermal approach show strong anti-colon cancer activities. J. Biomol. Struct. Dyn. 2021, 39, 2257–2265.
- Panda, J.; Das, S.; Kumar, S.; Tudu, B.; Sarkar, R. Investigation of antibacterial, antioxidant, and anticancer properties of hydrothermally synthesized cobalt ferrite nanoparticles. Appl. Phys. A Mater. Sci. Process. 2022, 128, 562.
- Albornoz, C.A.; Paulin, M.A.; Cristóbal, A.A.; Vega, D.R.; Leyva, A.G.; Ramos, C.P. Microwave-assisted hydrothermal nanoarchitectonics of polyethyleneimine-coated iron oxide nanoparticles. Appl. Phys. A Mater. Sci. Process. 2022, 128, 68.
- Fayazzadeh, S.; Khodaei, M.; Arani, M.; Mahdavi, S.R.; Nizamov, T.; Majouga, A. Magnetic Properties and Magnetic Hyperthermia of Cobalt Ferrite Nanoparticles Synthesized by Hydrothermal Method. J. Supercond. Nov. Magn. 2020, 33, 2227–2233.
- Park, Y.; Yoon, H.J.; Lee, S.E.; Lee, L.P. Multifunctional Cellular Targeting, Molecular Delivery, and Imaging by Integrated Mesoporous-Silica with Optical Nanocrescent Antenna: MONA. ACS Nano 2022, 16, 2013–2023.
- Wang, F.; Qi, X.; Geng, J.; Liu, X.; Li, D.; Zhang, H.; Zhang, P.; He, X.; Li, B.; Li, Z.; et al. Template-free construction of hollow mesoporous Fe3O4 nanospheres as controlled drug delivery with enhanced drug loading capacity. J. Mol. Liq. 2022, 347, 118000.
- Gao, Y.; Shi, X.; Shen, M. Intelligent Design of Ultrasmall Iron Oxide Nanoparticle-Based Theranostics. ACS Appl. Mater. Interfaces 2021, 13, 45119–45129.
- Saputra, O.A.; Wibowo, F.R.; Lestari, W.W.; Handayani, M. Highly monodisperse and colloidal stable of L-serine capped magnetite nanoparticles synthesized via sonochemistry assisted co-precipitation method. Adv. Nat. Sci. Nanosci. Nanotechnol. 2020, 11, 025012.
- Kafi-Ahmadi, L.; Khademinia, S.; Nansa, M.N.; Alemi, A.A.L.I.; Mahdavi, M.; Marjani, A.P. Co-precipitation synthesis, characterization of CoFe2O4 nanomaterial and evaluation of its toxicity behavior on human leukemia cancer K562 cell line. J. Chil. Chem. Soc. 2020, 65, 4845–4848.
- Tadic, M.; Lazovic, J.; Panjan, M.; Kralj, S. Hierarchical iron oxide nanocomposite: Bundle-like morphology, magnetic properties and potential biomedical application. Ceram. Int. 2022, 48, 16015–16022.
- Porrawatkul, P.; Nuengmatcha, P.; Tangwatanakul, W.; Chanthai, S. Effect of Zn, Ni, and Mn doping ions on magnetic properties of MFe2O4 (M = Mn, Zn, and Ni) nanoparticles synthesized via sol–gel autocombustion using PVA/sago starch blend as a chelating agent. J. Korean Ceram. Soc. 2020, 57, 676–683.
- Qayoom, M.; Bhat, R.; Shah, K.A.; Pandit, A.H.; Firdous, A.; Dar, G.N. Modified Solution Combustion Synthesis of Nickel-Doped Magnetite Nanoparticles and the Influence of Annealing on Their Optical, Electrical, and Magnetic Properties. J. Electron. Mater. 2020, 49, 1215–1229.
- Haydar, M.S.; Das, D.; Ghosh, S.; Mandal, P. Implementation of mature tea leaves extract in bioinspired synthesis of iron oxide nanoparticles: Preparation, process optimization, characterization, and assessment of therapeutic potential. Chem. Pap. 2022, 76, 491–514.
- Mangamma, J.L.; Devi, D.R.; Sagar, P.S.R.V.; Babu, M.R.; Basavaiah, K. Review on Plant Mediated Green Synthesis of Magnetite Nanoparticles for Pollution Abatement, Biomedical and Electronic Applications. Asian J. Chem. 2022, 34, 1047–1054.
- Loiola, A.R.; Bessa, R.A.; Oliveira, C.P.; Freitas, A.D.L.; Soares, S.A.; Bohn, F.; Pergher, S.B.C. Magnetic zeolite composites: Classification, synthesis routes, and technological applications. J. Magn. Magn. Mater. 2022, 560, 169651.
- Bertran, A.; Sandoval, S.; Oro-Sole, J.; Sanchez, A.; Tobias, G. Particle size determination from magnetization curves in reduced graphene oxide decorated with monodispersed superparamagnetic iron oxide nanoparticles. J. Colloid Interface Sci. 2020, 566, 107–119.
- Harada, M.; Kuwa, M.; Sato, R.; Teranishi, T.; Takahashi, M.; Maenosono, S. Cation Distribution in Monodispersed MFe2O4 (M = Mn, Fe, Co, Ni, and Zn) Nanoparticles Investigated by X-ray Absorption Fine Structure Spectroscopy: Implications for Magnetic Data Storage, Catalysts, Sensors, and Ferrofluids. ACS Appl. Nano Mater. 2020, 3, 8389–8402.
- Wahba, A.M.; Mohamed, M.B. Correlating cation distribution with the structural and magnetic properties of Co0.5Zn0.5AlxFe2–xO4 nanoferrites. Appl. Phys. A Mater. Sci. Process. 2020, 126, 488.
- Palade, P.; Comanescu, C.; Kuncser, A.; Berger, D.; Matei, C.; Iacob, N.; Kuncser, V. Mesoporous Cobalt Ferrite Nanosystems Obtained by Surfactant-Assisted Hydrothermal Method: Tuning Morpho-structural and Magnetic Properties via pH-Variation. Nanomaterials 2020, 10, 476.
- Orozco-Henao, J.M.; Muraca, D.; Sánchez, F.H.; Mendoza Zélis, P. Determination of the effective anisotropy of magnetite/maghemite nanoparticles from Mössbauer effect spectra. J. Phys. D Appl. Phys. 2022, 55, 335302.
- Kahmann, T.; Ludwig, F. Magnetic field dependence of the effective magnetic moment of multi-core nanoparticles. J. Appl. Phys. 2020, 127, 233901.
- Wang, S.; Xu, J.; Li, W.; Sun, S.; Gao, S.; Hou, Y. Magnetic Nanostructures: Rational Design and Fabrication Strategies toward Diverse Applications. Chem. Rev. 2022, 122, 5411–5475.
- Pyatakov, A.; Pyatakova, Z.; Tishin, A.M. Short history overview of magnetism and magnetic technologies for medical applications. In Materials and Technologies for Medical Applications; Woodhead Publishing: Cambridge, UK, 2022; pp. 3–21.
- Krasia-Christoforou, T.; Socoliuc, V.; Knudsen, K.D.; Tombácz, E.; Turcu, R.; Vekas, L. From single-core nanoparticles in ferrofluids to multi-core magnetic nanocomposites: Assembly strategies, structure and magnetic behavior. Nanomaterials 2020, 10, 2178.
- Scanone, A.C.; Gsponer, N.S.; Alvarez, M.G.; Heredia, D.A.; Durantini, A.M.; Durantini, E.N. Magnetic Nanoplatforms for in Situ Modification of Macromolecules: Synthesis, Characterization, and Photoinactivating Power of Cationic Nanoiman–Porphyrin Conjugates. ACS Appl. Bio Mater. 2020, 3, 5930–5940.
- Shi, Y.; Jyoti, D.; Gordon-Wylie, S.W.; Weaver, J.B. Quantification of magnetic nanoparticles by compensating for multiple environment changes simultaneously. Nanoscale 2020, 12, 195–200.
- Papadopoulos, C.; Efthimiadou, E.K.; Pissas, M.; Fuentes, D.; Boukos, N.; Psycharis, V.; Kordas, G.; Loukopoulos, V.C.; Kagadis, G.C. Magnetic fluid hyperthermia simulations in evaluation of SAR calculation methods. Phys. Med. 2020, 71, 39–52.
- Honecker, D.; Bersweiler, M.; Erokhin, S.; Berkov, D.; Chesnel, K.; Venero, D.A.; Qdemat, A.; Disch, S.; Jochum, J.K.; Michels, A.; et al. Using small-angle scattering to guide functional magnetic nanoparticle design. Nanoscale Adv. 2022, 4, 1026–1059.
- Priyadarshi, H.; Gaur, U. Paramagnetism-enhanced doped maghemite nanoparticles for targeted drug delivery. Mater. Today Proc. 2021, 43, 3030–3033.
- Pardo, A.; Pelaz, B.; Gallo, J.; Bañobre-López, M.; Parak, W.J.; Barbosa, S.; Del Pino, P.; Taboada, P. Synthesis, Characterization, and Evaluation of Superparamagnetic Doped Ferrites as Potential Therapeutic Nanotools. Chem. Mater. 2020, 32, 2220–2231.
- Dhillon, G.; Kumar, N.; Chitkara, M.; Sandhu, I.S. Effect of Gd3+ substitution on physicochemical properties of superparamagnetic Fe3O4 nanoparticles. J. Mater. Sci. Mater. Electron. 2021, 32, 22387–22397.
- Saini, A.; Borchers, J.A.; George, S.; Maranville, B.B.; Krycka, K.L.; Dura, J.A.; Theis-Bröhl, K.; Wolff, M. Layering of Magnetic Nanoparticles at Amorphous Magnetic Templates with Perpendicular Anisotropy. Soft Matter 2020, 16, 7676.
- Zhao, P.; Zhao, J.; Deng, Y.; Zeng, G.; Jiang, Y.; Liao, L.; Zhang, S.; Tao, Q.; Liu, Z.; Tang, X.; et al. Application of iron/barium ferrite/carbon-coated iron nanocrystal composites in transcatheter arterial chemoembolization of hepatocellular carcinoma. J. Colloid Interface Sci. 2021, 601, 30–41.
- Hölscher, J.; Petrecca, M.; Albino, M.; Garbus, P.G.; Saura-Múzquiz, M.; Sangregorio, C.; Christensen, M. Magnetic Property Enhancement of Spinel Mn-Zn Ferrite through Atomic Structure Control. Inorg. Chem. 2020, 59, 11184–11192.
- Riaz, H.; Hashmi, R.; Abid, S.; Shareef, N.; Faqir, A.; Amir, A.; Shahzad, M.S.; Shakeel, M.; Akhtar, S.; Ashiq, M.N.; et al. Intraperitoneal injections of copper ferrite nanoparticles disturb blood, plasma, and antioxidant parameters of Wistar rats in a sex-specific manner. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2020, 393, 2019–2028.
- Sarkar, B.J.; Bandyopadhyay, A. Studies of magnetic behavior of chemically synthesized interacting superparamagnetic copper ferrite nanoparticles. J. Mater. Sci. Mater. Electron. 2021, 32, 1491–1505.
- Manju, B.G.; Raji, P. Green synthesis, characterization, and antibacterial activity of lime-juice-mediated copper–nickel mixed ferrite nanoparticles. Appl. Phys. A Mater. Sci. Process. 2020, 126, 156.
- Althubayti, M.M.; Hjiri, M.; Alonizan, N.H.; Lemine, O.M.; Aida, M.S. Influence of divalent metals (Zn, Cu and Co) on the synthesis and magnetic properties of spinel ferrite nanopowders. J. Mater. Sci. Mater. Electron. 2020, 31, 8194–8205.
- Silvestri, N.; Gavilán, H.; Guardia, P.; Brescia, R.; Fernandes, S.; Samia, A.C.S.; Teran, F.J.; Pellegrino, T. Di- and tri-component spinel ferrite nanocubes: Synthesis and their comparative characterization for theranostic applications. Nanoscale 2021, 13, 13665–13680.
- Hossain, M.D.; Khan, M.N.I.; Nahar, A.; Ali, M.A.; Matin, M.A.; Hoque, S.M.; Hakim, M.A.; Jamil, A.T.M.K. Tailoring the properties of Ni-Zn-Co ferrites by Gd3+ substitution. J. Magn. Magn. Mater. 2020, 497, 165978.
- Vangijzegem, T.; Stanicki, D.; Panepinto, A.; Socoliuc, V.; Vekas, L.; Muller, R.N.; Laurent, S. Influence of Experimental Parameters of a Continuous Flow Process on the Properties of Very Small Iron Oxide Nanoparticles (VSION) Designed for T1-Weighted Magnetic Resonance Imaging (MRI). Nanomaterials 2020, 10, 757.
- Morillas, J.R.; de Vicente, J. Magnetorheology: A review. Soft Matter 2020, 16, 9614–9642.
- Zablotsky, D.; Kralj, S.; Maiorov, M.M. Features of magnetorheology of biocompatible chain-forming ferrofluids with multi-core magnetic nanoparticles: Experiment and simulation. Colloids Surf. A 2020, 603, 125079.
- Craciunescu, I.; Chiţanu, E.; Codescu, M.M.; Iacob, N.; Kuncser, A.; Kuncser, V.; Socoliuc, V.; Susan-Resiga, D.; Bălănean, F.; Ispas, G.; et al. High performance magnetorheological fluids: Very high magnetization FeCo-Fe3O4 nanoclusters in ferrofluid carrier. Soft Matter 2022, 18, 626–639.
- Lu, Q.; Choi, K.; Nam, J.-D.; Choi, H.J. Magnetic Polymer Composite Particles: Design and Magnetorheology. Polymers 2021, 13, 512.
- Willis, A.J.; Pernal, S.P.; Gaertner, Z.A.; Lakka, S.S.; Sabo, M.E.; Creighton, F.M.; Engelhard, H.H. Rotating magnetic nanoparticle clusters as microdevices for drug delivery. Int. J. Nanomed. 2020, 15, 4105–4123.
- Esfe, M.H.; Afrand, M.; Esfandeh, S. Investigation of the effects of various parameters on the natural convection of nanofluids in various cavities exposed to magnetic fields: A comprehensive review. J. Therm. Anal. Calorim. 2020, 140, 2055–2075.
- Ma, W.; Xu, M.; Zhong, Z.; Li, X.; Huan, Z. Closed-Loop Control for Trajectory Tracking of a Microparticle Based on Input-to-State Stability Through an Electromagnetic Manipulation System. IEEE Access 2020, 8, 46537–46545.
- van Silfhout, A.M.; Engelkamp, H.; Erné, B.H. Magnetic Sedimentation Velocities and Equilibria in Dilute Aqueous Ferrofluids. J. Phys. Chem. B 2020, 124, 7989–7998.
- Socoliuc, V.; Turcu, R. Large scale aggregation in magnetic colloids induced by high frequency magnetic fields. J. Magn. Magn. Mater. 2020, 500, 166348.
- Ganguly, S.; Margel, S. Review: Remotely controlled magneto-regulation of therapeutics from magnetoelastic gel matrices. Biotechnol. Adv. 2020, 44, 107611.
- Podoliak, N.; Richardson, G. Correctly computing targeting efficiency in magnetically targeted delivery from particle tracking models. J. Magn. Magn. Mater. 2022, 549, 168960.
- Russo, T.; Peluso, V.; Gloria, A.; Oliviero, O.; Rinaldi, L.; Improta, G.; de Santis, R.; D’Antò, V. Combination Design of Time-Dependent Magnetic Field and Magnetic Nanocomposites to Guide Cell Behavior. Nanomaterials 2020, 10, 577.
- Bhatt, A.; Sakai, K.; Madhyastha, R.; Murayama, M.; Madhyastha, H.; Rath, S.N. Biosynthesis and characterization of nano magnetic hydroxyapatite (nMHAp): An accelerated approach using simulated body fluid for biomedical applications. Ceram. Int. 2020, 46, 27866–27876.
- Vijayakanth, V.; Chintagumpala, K. A review on an effect of dispersant type and medium viscosity on magnetic hyperthermia of nanoparticles. Polym. Bull. 2022.
- Zuluaga-Parra, J.D.; Sánchez-Valdés, S.; Ramos-deValle, L.F.; Beltrán-Ramírez, F.I.; da-Silva, L.; Ramírez-Vargas, E.; Vázquez-Rodríguez, S.; Flores-Gallardo, S.; Méndez-Nonell, J.; Valera-Zaragoza, M.; et al. A novel method for the modification of magnetite nanoparticles for the enhancement of its dispersibility in hydrophobic media. J. Magn. Magn. Mater. 2020, 514, 167169.
- Park, Y.; Cho, H. Improvement in the dispersion stability of iron oxide nanoparticles in highly concentrated brine solution using encapsulation with polymer-polymer crosslinked shells. Adv. Powder Technol. 2020, 31, 4743–4750.
- Roa-Barrantes, L.M.; Patarroyo, D.J.R. Magnetic Field Effect on the Magnetic Nanoparticles Trajectories in Pulsating Blood Flow: A Computational Model. BioNanoScience 2022, 12, 571–581.
- Salem, S.F.; Tuchin, V.V. Numerical simulation of magnetic nanoparticles in the blood stream. In Proceedings of the Saratov Fall Meeting 2019: Optical and Nano-Technologies for Biology and Medicine, Saratov, Russia, 23–27 September 2019; Volume 11457, p. 114571N.
- Badfar, H.; Motlagh, S.Y.; Sharifi, A. Numerical Simulation of Magnetic Drug Targeting to the Stenosis Vessel Using Fe3O4 Magnetic Nanoparticles Under the Effect of Magnetic Field of Wire. Cardiovasc. Eng. Technol. 2020, 11, 162–175.
- Kreissl, P.; Holm, C.; Weeber, R. Frequency-dependent magnetic susceptibility of magnetic nanoparticles in a polymer solution: A simulation study. Soft Matter 2021, 17, 174–183.
- Chen, H.-H.; Hsu, M.-H.; Lee, K.-H.; Chen, W.-Y.; Yang, S.-Y. Real-time changes in the AC magnetic susceptibility of reagents during immunomagnetic reduction assays. AIP Adv. 2022, 12, 065220.
- Labbafzadeh, M.R.; Vakili, M.H. Application of magnetic electrospun polyvinyl alcohol/collagen nanofibres for drug delivery systems. Mol. Simul. 2022, 48, 1–7.
- Harris, R.A. Simulation study on the physicochemical properties of Fe3O4 nanoparticles as drug delivery vehicles for dopamine replacement therapy of Parkinson’s disease. Mater. Today Commun. 2022, 31, 103829.
- Cai, J.; Dao, P.; Chen, H.; Yan, L.; Li, Y.L.; Zhang, W.; Li, L.; Du, Z.; Dong, C.-Z.; Meunier, B. Ultrasmall superparamagnetic iron oxide nanoparticles-bound NIR dyes: Novel theranostic agents for Alzheimer’s disease. Dye Pigment. 2020, 173, 107968.
- Sadeghi-Goughari, M.; Jeon, S.; Kwon, H.-J. Magnetic nanoparticles-enhanced focused ultrasound heating: Size effect, mechanism, and performance analysis. Nanotechnology 2020, 31, 245101.
- Contreras-Montoya, R.; Jabalera, Y.; Blanco, V.; Cuerva, J.M.; Jimenez-Lopez, C.; Alvarez De Cienfuegos, L. Lysine as Size-Control Additive in a Bioinspired Synthesis of Pure Superparamagnetic Magnetite Nanoparticles. Cryst. Growth Des. 2020, 20, 533–542.
- Craciunescu, I.; Palade, P.; Iacob, N.; Ispas, G.M.; Stanciu, A.E.; Kuncser, V.; Turcu, R.P. High Performance Functionalized Magnetic Nanoparticles with Tailored Sizes and Shapes for Localized Hyperthermia Applications. J. Phys. Chem. C 2021, 125, 11132–11146.
- Islam, K.; Haque, M.; Kumar, A.; Hoq, A.; Hyder, F.; Hoque, S.M. Manganese ferrite nanoparticles (MnFe2O4): Size dependence for hyperthermia and negative/positive contrast enhancement in MRI. Nanomaterials 2020, 10, 2297.
- Malekie, S.; Rajabi, A. Study on Fe3O4 Magnetic Nanoparticles Size Effect on Temperature Distribution of Tumor in Hyperthermia: A Finite Element Method. Int. J. Nanosci. Nanotechnol. 2020, 16, 181–188.
- Bilmez, B.; Toker, M.Ö.; Toker, O.; İçelli, O. Monte Carlo study on size-dependent radiation enhancement effects of spinel ferrite nanoparticles. Radiat. Phys. Chem. 2022, 199, 110364.
- Das, R.; Masa, J.; Kalappattil, V.; Nemati, Z.; Rodrigo, I.; Garaio, E.; García, J.; Phan, M.-H.; Srikanth, H. Iron Oxide Nanorings and Nanotubes for Magnetic Hyperthermia: The Problem of Intraparticle Interactions. Nanomaterials 2021, 11, 1380.
- van Silfhout, A.M.; Engelkamp, H.; Erné, B.H. Colloidal Stability of Aqueous Ferrofluids at 10 T. J. Phys. Chem. Lett. 2020, 11, 5908–5912.
- Pilati, V.; Gomide, G.; Gomes, R.C.; Goya, G.F.; Depeyrot, J. Colloidal Stability and Concentration Effects on Nanoparticle Heat Delivery for Magnetic Fluid Hyperthermia. Langmuir 2021, 37, 1129–1140.
- Castro, L.L.; Amorim, C.C.C.; Miranda, J.P.V.; Cassiano, T.S.A.; Paula, F.L.O. The role of small separation interactions in ferrofluid structure. Colloids Surf. A 2022, 635, 128082.
- Riedl, J.C.; Sarkar, M.; Fiuza, T.; Cousin, F.; Depeyrot, J.; Dubois, E.; Mériguet, G.; Perzynski, R.; Peyre, V. Design of concentrated colloidal dispersions of iron oxide nanoparticles in ionic liquids: Structure and thermal stability from 25 to 200 °C. J. Colloid Interface Sci. 2022, 607, 584–594.
- Boskovic, M.; Fabián, M.; Vranjes-Djuric, S.; Antic, B. Magnetic nano- and micro-particles based on Gd-substituted magnetite with improved colloidal stability. Appl. Phys. A Mater. Sci. Process. 2021, 127, 372.
- Aguilar, N.M.; Perez-Aguilar, J.M.; González-Coronel, V.J.; Moro, J.G.S.; Sanchez-Gaytan, B.L. Polymers as Versatile Players in the Stabilization, Capping, and Design of Inorganic Nanostructures. ACS Omega 2021, 6, 35196–35203.
More
Information
Subjects:
Nanoscience & Nanotechnology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
657
Revisions:
2 times
(View History)
Update Date:
02 Sep 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





