
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Henry H.L. Wu | -- | 3195 | 2022-08-31 03:54:54 | | | |
| 2 | Vivi Li | -1 word(s) | 3194 | 2022-08-31 04:21:32 | | |
Video Upload Options
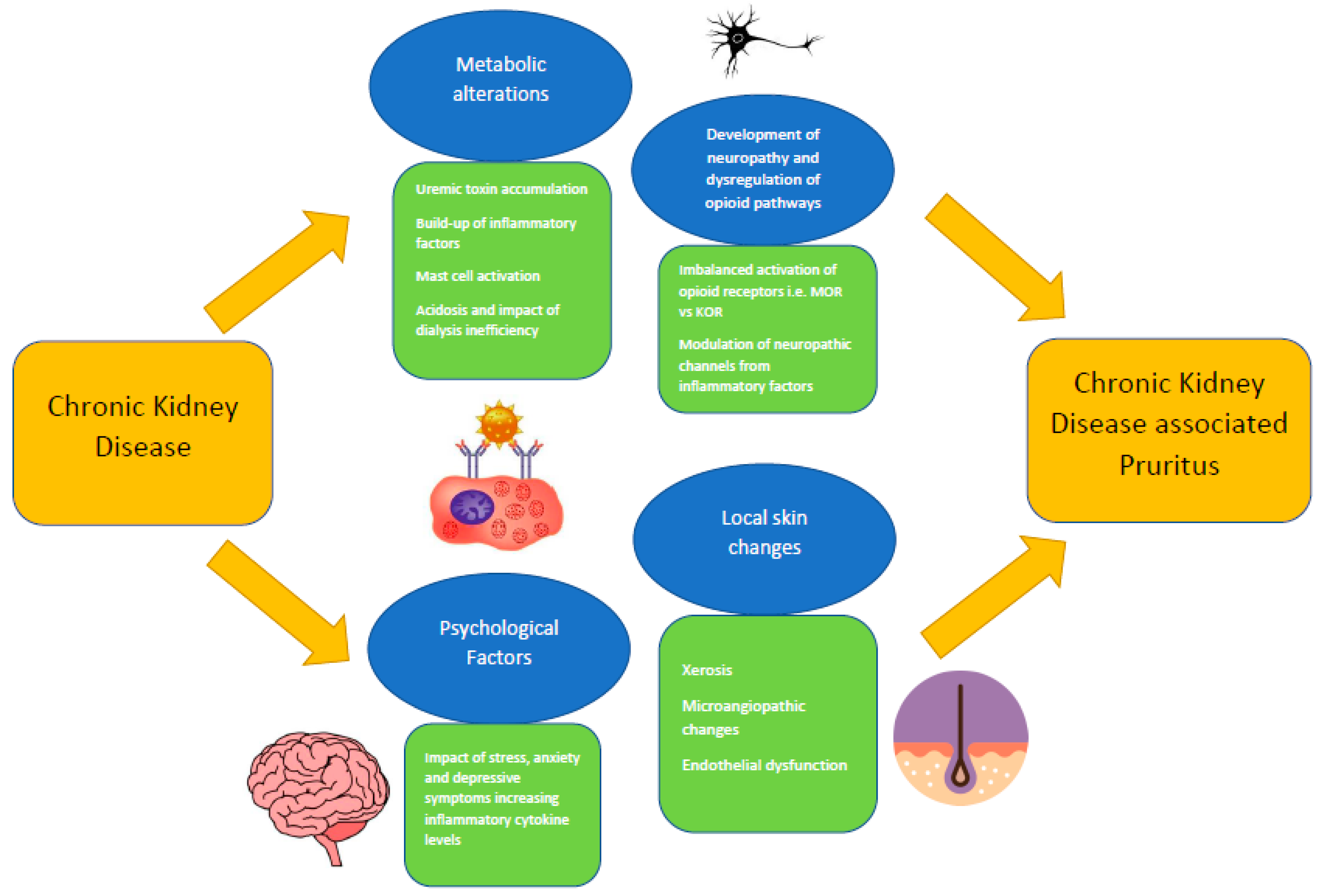
Chronic kidney disease-associated pruritus (CKDaP) is an often under-diagnosed and under-recognized condition, despite its considerable prevalence within the chronic kidney disease (CKD) population. Universally accepted guidelines are also lacking. The true prevalence of CKDaP worldwide therefore remains unknown, although its negative impact on mortality and health-related quality of life outcomes is very clear. The pathophysiological mechanisms leading to the onset of CKDaP are only partly understood. CKDaP is currently believed to be caused by a multifactorial process, from local skin changes, metabolic alterations, the development of neuropathy and dysregulation of opioid pathways, and psychological factors. Much work has been carried out towards a more systematic and structured approach to clinical diagnosis. Various tools are now available to assess the severity of CKDaP. Many of these tools require greater validation before they can be incorporated into the guidelines and into routine clinical practice. Further efforts are also needed in order to increase the awareness of clinicians and patients so that they can identify the CKDaP signs and symptoms in a timely manner.
1. Introduction
2. Epidemiology of CKDaP
3. Pathophysiology of CKDaP
4. Prevention and Treatment of CKDaP
References
- Levin, A.; Stevens, P.E.; Bilous, R.W.; Coresh, J.; De Francisco, A.L.; De Jong, P.E.; Griffith, K.E.; Hemmelgarn, B.R.; Iseki, K.; Lamb, E.J.; et al. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. Suppl. 2013, 3, 1–50.
- Romagnani, P.; Remuzzi, G.; Glassock, R.; Levin, A.; Jager, K.J.; Tonelli, M.; Massy, Z.; Wanner, C.; Anders, H.J. Chronic kidney disease. Nat. Rev. Dis. Primers 2017, 3, 17088.
- Anders, H.J.; Huber, T.B.; Isermann, B.; Schiffer, M. CKD in diabetes: Diabetic kidney disease versus nondiabetic kidney disease. Nat. Rev. Nephrol. 2018, 14, 361–377.
- Foreman, K.J.; Marquez, N.; Dolgert, A.; Fukutaki, K.; Fullman, N.; McGaughey, M.; Pletcher, M.A.; Smith, A.E.; Tang, K.; Yuan, C.W.; et al. Forecasting life expectancy, years of life lost, and all-cause and cause-specific mortality for 250 causes of death: Reference and alternative scenarios for 2016–40 for 195 countries and territories. Lancet 2018, 392, 2052–2090.
- Kalantar-Zadeh, K.; Jafar, T.H.; Nitsch, D.; Neuen, B.L.; Perkovic, V. Chronic kidney disease. Lancet 2021, 398, 786–802.
- Stefaniak, A.A.; Agelopoulos, K.; Bednarska-Chabowska, D.; Mazur, G.; Ständer, S.; Szepietowski, J.C. Small-fibre Neuropathy in Patients with Type 2 Diabetes Mellitus and its Relationship with Diabetic Itch: Preliminary Results. Acta Derm. Vener. 2022, 102, adv00719.
- U.S. Renal Data System. USRDS 2021 Annual Data Report: End Stage Renal Disease; National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases: Bethesda, MD, USA, 2021; Volume 2021.
- Australian and New Zealand Dialysis and Transplant Registry. Dialysis Centre Report 2015–2020; ANZDATA Registry: Adelaide, SA, Australia, 2021.
- Yang, M.; Fox, C.H.; Vassalotti, J.; Choi, M. Complications of progression of CKD. Adv. Chronic. Kidney Dis. 2011, 18, 400–405.
- Kim, D.; Pollock, C. Epidemiology and burden of chronic kidney disease-associated pruritus. Clin. Kidney J. 2021, 14, i1–i7.
- Hu, X.; Sang, Y.; Yang, M.; Chen, X.; Tang, W. Prevalence of chronic kidney disease-associated pruritus among adult dialysis patients: A meta-analysis of cross-sectional studies. Medicine 2018, 97, e10633.
- Cevikbas, F.; Lerner, E.A. Physiology and pathophysiology of itch. Physiol. Rev. 2020, 100, 945–982.
- Kremer, A.E.; Feramisco, J.; Reeh, P.W.; Beuers, U.; Elferink, R.P. Receptors, cells and circuits involved in pruritus of systemic disorders. Biochim. Biophys. Acta. 2014, 1842, 869–892.
- Martin, C.E.; Clotet-Freixas, S.; Farragher, J.F.; Hundemer, G.L. Have we just scratched the surface? A narrative review of uremic pruritus in 2020. Can. J. Kidney Health Dis. 2020, 7, 2054358120954024.
- Schricker, S.; Kimmel, M. Unravelling the pathophysiology of chronic kidney disease-associated pruritus. Clin. Kidney J. 2021, 14, i23–i31.
- Weisshaar, E.; Szepietowski, J.C.; Dalgard, F.J.; Garcovich, S.; Gieler, U.; Giménez-Arnau, A.M.; Lambert, J.; Leslie, T.; Mettang, T.; Misery, L.; et al. European S2k guideline on chronic pruritus. Acta Derm. Venereol. 2019, 99, 469–506.
- Manenti, L.; Leuci, E. Do you feel itchy? A guide towards diagnosis and measurement of chronic kidney disease-associated pruritus in dialysis patients. Clin. Kidney J. 2021, 14, i8–i15.
- Evangelidis, N.; Sautenet, B.; Madero, M.; Tong, A.; Ashuntantang, G.; Sanabria, L.C.; de Boer, I.H.; Fung, S.; Gallego, D.; Levey, A.S.; et al. Standardised Outcomes in Nephrology–Chronic Kidney Disease (SONG-CKD): A protocol for establishing a core outcome set for adults with chronic kidney disease who do not require kidney replacement therapy. Trials 2021, 22, 612.
- Fishbane, S.; Jamal, A.; Munera, C.; Wen, W.; Menzaghi, F. A phase 3 trial of difelikefalin in hemodialysis patients with pruritus. N. Engl. J. Med. 2020, 382, 222–232.
- Pisoni, R.L.; Wikström, B.; Elder, S.J.; Akizawa, T.; Asano, Y.; Keen, M.L.; Saran, R.; Mendelssohn, D.C.; Young, E.W.; Port, F.K. Pruritus in haemodialysis patients: International results from the Dialysis Outcomes and Practice Patterns Study (DOPPS). Nephrol. Dial. Transplant. 2006, 21, 3495–3505.
- Rayner, H.C.; Larkina, M.; Wang, M.; Graham-Brown, M.; van der Veer, S.N.; Ecder, T.; Hasegawa, T.; Kleophas, W.; Bieber, B.A.; Tentori, F.; et al. International comparisons of prevalence, awareness, and treatment of pruritus in people on hemodialysis. Clin. J. Am. Soc. Nephrol. 2017, 12, 2000–2007.
- Sukul, N.; Karaboyas, A.; Csomor, P.A.; Schaufler, T.; Wen, W.; Menzaghi, F.; Rayner, H.C.; Hasegawa, T.; Al Salmi, I.; Al-Ghamdi, S.M.; et al. Self-reported pruritus and clinical, dialysis-related, and patient-reported outcomes in hemodialysis patients. Kidney Med. 2021, 3, 42–53.
- Sukul, N.; Speyer, E.; Tu, C.; Bieber, B.A.; Li, Y.; Lopes, A.A.; Asahi, K.; Mariani, L.; Laville, M.; Rayner, H.C.; et al. Pruritus and patient reported outcomes in non-dialysis CKD. Clin. J. Am. Soc. Nephrol. 2019, 14, 673–681.
- Weiss, M.; Mettang, T.; Tschulena, U.; Passlick-Deetjen, J.; Weisshaar, E. Prevalence of chronic itch and associated factors in haemodialysis patients: A representative cross-sectional study. Acta Derm. Venereol. 2015, 95, 816–821.
- Aresi, G.; Rayner, H.C.; Hassan, L.; Burton, J.O.; Mitra, S.; Sanders, C.; van der Veer, S.N. Reasons for underreporting of uremic pruritus in people with chronic kidney disease: A qualitative study. J. Pain Symptom Manag. 2019, 58, 578–586.
- Wu, H.Y.; Huang, J.W.; Tsai, W.C.; Peng, Y.S.; Chen, H.Y.; Yang, J.Y.; Hsu, S.P.; Pai, M.F.; Ko, M.J.; Hung, K.Y.; et al. Prognostic importance and determinants of uremic pruritus in patients receiving peritoneal dialysis: A prospective cohort study. PLoS ONE 2018, 13, e0203474.
- Min, J.W.; Kim, S.H.; Kim, Y.O.; Jin, D.C.; Song, H.C.; Choi, E.J.; Kim, Y.L.; Kim, Y.S.; Kang, S.W.; Kim, N.H.; et al. Comparison of uremic pruritus between patients undergoing hemodialysis and peritoneal dialysis. Kidney Res. Clin. Pract. 2016, 35, 107–113.
- Ramakrishnan, K.; Bond, T.C.; Claxton, A.; Sood, V.C.; Kootsikas, M.; Agnese, W.; Sibbel, S. Clinical characteristics and outcomes of end-stage renal disease patients with self-reported pruritus symptoms. Int. J. Nephrol. Renovasc. Dis. 2014, 7, 1–12.
- Afsar, B.; Afsar, R.E. HbA1c is related with uremic pruritus in diabetic and nondiabetic hemodialysis patients. Renal. Fail. 2012, 34, 1264–1269.
- Weisshaar, E.; Weiss, M.; Passlick-Deetjen, J.; Tschulena, U.; Maleki, K.; Mettang, T. Laboratory and dialysis characteristics in hemodialysis patients suffering from chronic itch-results from a representative cross-sectional study. BMC Nephrol. 2015, 16, 184.
- Stefaniak, A.A.; Krajewski, P.K.; Bednarska-Chabowska, D.; Bolanowski, M.; Mazur, G.; Szepietowski, J.C. Itch in Adult Population with Type 2 Diabetes Mellitus: Clinical Profile, Pathogenesis and Disease-Related Burden in a Cross-Sectional Study. Biology 2021, 10, 1332.
- Mathur, V.S.; Lindberg, J.; Germain, M.; Block, G.; Tumlin, J.; Smith, M.; Grewal, M.; McGuire, D.; ITCH National Registry Investigators. A longitudinal study of uremic pruritus in hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2010, 5, 1410–1419.
- Zhao, J.H.; Zhu, Q.S.; Li, Y.W.; Wang, L.L. Determinants of the intensity of uremic pruritus in patients receiving maintenance hemodialysis: A cross-sectional study. PLoS ONE 2021, 16, e0245370.
- Ozen, N.; Cinar, F.I.; Askin, D.; Mut, D. Uremic pruritus and associated factors in hemodialysis patients: A multi-center study. Kidney Res. Clin. Pract. 2018, 37, 138–147.
- Chen, H.Y.; Chiu, Y.L.; Hsu, S.P.; Pai, M.F.; Lai, C.F.; Yang, J.Y.; Peng, Y.S.; Tsai, T.J.; Wu, K.D. Elevated C-reactive protein level in hemodialysis patients with moderate/severe uremic pruritus: A potential mediator of high overall mortality. Q. J. Med. 2010, 103, 837–846.
- Grochulska, K.; Ofenloch, R.; Mettang, T.; Weisshaar, E. Mortality of Haemodialysis Patients with and without Chronic Itch: A Follow-up Study of the German Epidemiological Hemodialysis Itch Study (GEHIS). Acta Derm. Venereol. 2019, 99, 423–428.
- Plewig, N.; Ofenloch, R.; Mettang, T.; Weisshaar, E. The course of chronic itch in hemodialysis patients: Results of a 4-year follow-up study of GEHIS (German Epidemiological Hemodialysis Itch Study). J. Eur. Acad. Dermatol. Venereol. 2019, 33, 1429–1435.
- Rehman, I.U.; Lai, P.S.; Kun, L.S.; Lee, L.H.; Chan, K.G.; Khan, T.M. Chronic kidney disease-associated pruritus and quality of life in Malaysian patients undergoing hemodialysis. Ther. Apher. Dial. 2020, 24, 17–25.
- Ikonomou, M.; Skapinakis, P.; Balafa, O.; Eleftheroudi, M.; Damigos, D.; Siamopoulos, K.C. The impact of socioeconomic factors on quality of life of patients with chronic kidney disease in Greece. J. Renal. Care 2015, 41, 239–246.
- Seng, J.J.; Tan, J.Y.; Yeam, C.T.; Htay, H.; Foo, W.Y. Factors affecting medication adherence among pre-dialysis chronic kidney disease patients: A systematic review and meta-analysis of literature. Int. Urol. Nephrol. 2020, 52, 903–916.
- Weldegiorgis, M.; Smith, M.; Herrington, W.G.; Bankhead, C.; Woodward, M. Socioeconomic disadvantage and the risk of advanced chronic kidney disease: Results from a cohort study with 1.4 million participants. Nephrol. Dial. Transplant. 2020, 35, 1562–1570.
- Stander, S.; Weisshaar, E.; Mettang, T.; Szepietowski, J.C.; Carstens, E.; Ikoma, A.; Bergasa, N.V.; Gieler, U.; Misery, L.; Wallengren, J.; et al. Clinical classification of itch: A position paper of the International Forum for the Study of Itch. Acta Derm. Venereol. 2007, 87, 291–294.
- Attia, E.A.; Hassan, S.I.; Youssef, N.M. Cutaneous disorders in uremic patients on hemodialysis: An Egyptian case-controlled study. Int. J. Dermatol. 2010, 49, 1024–1030.
- Kurban, M.S.; Boueiz, A.; Kibbi, A.G. Cutaneous manifestations of chronic kidney disease. Clin. Dermatol. 2008, 26, 255–264.
- Szepietowski, J.C.; Reich, A.; Schwartz, R.A. Uraemic xerosis. Nephrol. Dial. Transplant. 2004, 19, 2709–2712.
- Wojtowicz-Prus, E.; Kilis-Pstrusinska, K.; Reich, A.; Zachwieja, K.; Miklaszewska, M.; Szczepanska, M.; Szepietowski, J.C. Disturbed skin barrier in children with chronic kidney disease. Pediatr. Nephrol. 2015, 30, 333–338.
- Yosipovitch, G.; Reis, J.; Tur, E.; Sprecher, E.; Yarnitsky, D.; Boner, G. Sweat secretion, stratum corneum hydration, small nerve function and pruritus in patients with advanced chronic renal failure. Br. J. Dermatol. 1995, 133, 561–564.
- Chorążyczewska, W.; Reich, A.; Szepietowski, J.C. Lipid content and barrier function analysis in uraemic pruritus. Acta Derm. Venereol. 2016, 96, 402–403.
- Prommer, H.U.; Maurer, J.; von Websky, K.; Freise, C.; Sommer, K.; Nasser, H.; Samapati, R.; Reglin, B.; Guimarães, P.; Pries, A.R.; et al. Chronic kidney disease induces a systemic microangiopathy, tissue hypoxia and dysfunctional angiogenesis. Sci. Rep. 2018, 8, 5317.
- Gilchrest, B.A.; Rowe, J.W.; Mihm, M.C., Jr. Clinical and histological skin changes in chronic renal failure: Evidence for a dialysis-resistant, transplant-responsive microangiopathy. Lancet 1980, 2, 1271–1275.
- Bose, C.; Shah, S.V.; Karaduta, O.K.; Kaushal, G.P. Carbamylated low-density lipoprotein (cLDL)-mediated induction of autophagy and its role in endothelial cell injury. PLoS ONE 2016, 11, e0165576.
- Querfeld, U.; Mak, R.H.; Pries, A.R. Microvascular disease in chronic kidney disease: The base of the iceberg in cardiovascular comorbidity. Clin. Sci. 2020, 134, 1333–1356.
- Makhlough, A.; Emadi, N.; Sedighi, O.; Khademloo, M.; Bicmohamadi, A.R. Relationship between serum intact parathyroid hormone and pruritus in hemodialysis patients. Iran. J. Kidney Dis. 2013, 7, 42–46.
- Duque, M.I.; Thevarajah, S.; Chan, Y.H.; Tuttle, A.B.; Freedman, B.I.; Yosipovitch, G. Uremic pruritus is associated with higher Kt/V and serum calcium concentration. Clin. Nephrol. 2006, 66, 184–191.
- Dashti-Khavidaki, S.; Khalili, H.; Vahedi, S.M.; Lessan-Pezeshki, M. Serum zinc concentrations in patients on maintenance hemodialysis and its relationship with anemia, parathyroid hormone concentrations and pruritus severity. Saudi J. Kidney Dis. Transpl. 2010, 21, 641–645.
- Narita, I.; Alchi, B.; Omori, K.; Sato, F.; Ajiro, J.; Saga, D.; Kondo, D.; Skatsume, M.; Maruyama, S.; Kazama, J.J.; et al. Etiology and prognostic significance of severe uremic pruritus in chronic hemodialysis patients. Kidney Int. 2006, 69, 1626–1632.
- Momose, A.; Kudo, S.; Sato, M.; Saito, H.; Nagai, K.; Katabira, Y.; Funyu, T. Calcium ions are abnormally distributed in the skin of haemodialysis patients with uraemic pruritus. Nephrol. Dial. Transplant. 2004, 19, 2061–2066.
- Chou, F.F.; Ho, J.C.; Huang, S.C.; Sheen-Chen, S.M. A study on pruritus after parathyroidectomy for secondary hyperparathyroidism. J. Am. Coll. Surg. 2000, 190, 65–70.
- Ganz, T.; Nemeth, E. Iron balance and the role of hepcidin in chronic kidney disease. Semin. Nephrol. 2016, 36, 87–93.
- Kimmel, M.; Alscher, D.M.; Dunst, R.; Braun, N.; Machleidt, C.; Kiefer, T.; Stülten, C.; van der Kuip, H.; Pauli-Magnus, C.; Raub, U.; et al. The role of micro-inflammation in the pathogenesis of uraemic pruritus in haemodialysis patients. Nephrol. Dial. Transplant. 2006, 21, 749–755.
- Fallahzadeh, M.K.; Roozbeh, J.; Geramizadeh, B.; Namazi, M.R. Interleukin-2 serum levels are elevated in patients with uremic pruritus: A novel finding with practical implications. Nephrol. Dial. Transplant. 2011, 26, 3338–3344.
- Ko, M.J.; Peng, Y.S.; Chen, H.Y.; Hsu, S.P.; Pai, M.F.; Yang, J.Y.; Wen, S.Y.; Jee, S.H.; Wu, H.Y.; Chiu, H.C. Interleukin-31 is associated with uremic pruritus in patients receiving hemodialysis. J. Am. Acad. Dermatol. 2014, 71, 1151–1159.
- Szepietowski, J.; Thepen, T.; Van Vloten, W.A.; Szepietowski, T.; Bihari, I.C. Pruritus and mast cell proliferation in the skin of haemodialysis patients. Inflamm. Res. 1995, 44, S84–S85.
- Jiang, Y.M.; Huang, C.; Peng, Z.; Han, S.L.; Li, W.G.; Zhu, M.X.; Xu, T.L. Acidosis counteracts itch tachyphylaxis to consecutive pruritogen exposure dependent on acid-sensing ion channel 3. Mol. Pain. 2017, 13, 1744806917721114.
- Peng, Z.; Li, W.G.; Huang, C.; Jiang, Y.M.; Wang, X.; Zhu, M.X.; Cheng, X.; Xu, T.L. ASIC3 mediates itch sensation in response to coincident stimulation by acid and nonproton ligand. Cell Rep. 2015, 13, 387–398.
- Malekmakan, L.; Malekmakan, A.; Sayadi, M.; Pakfetrat, M.; Sepaskhah, M.; Roozbeh, J. Association of high-sensitive C-reactive protein and dialysis adequacy with uremic pruritus. Saudi J. Kidney Dis. Transpl. 2015, 26, 890–895.
- Ko, M.J.; Wu, H.Y.; Chen, H.Y.; Chiu, Y.L.; Hsu, S.P.; Pai, M.F.; Lai, C.F.; Lu, H.M.; Huang, S.C.; Yang, S.Y.; et al. Uremic pruritus, dialysis adequacy, and metabolic profiles in hemodialysis patients: A prospective 5-year cohort study. PLoS ONE 2013, 8, e71404.
- Reszke, R.; Szepietowski, J.C. End-stage renal disease chronic itch and its management. Dermatol. Clin. 2018, 36, 277–292.
- Zakrzewska-Pniewska, B.; Jedras, M. Is pruritus in chronic uremic patients related to peripheral somatic and autonomic neuropathy? Study by R-R interval variation test (RRIV) and by sympathetic skin response (SSR). Neurophysiol. Clin. 2001, 31, 181–193.
- Mettang, T.; Kremer, A.E. Uremic pruritus. Kidney Int. 2015, 87, 685–691.
- Paul, L.B.; Mei, B.Q. Itch: Mechanisms and Treatment; Akiyama, T., Carstens, E., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2014.
- Schulze, E.; Witt, M.; Fink, T.; Hofer, A.; Funk, R.H. Immunohistochemical detection of human skin nerve fibers. Acta Histochem. 1997, 99, 301–309.
- Ständer, S.; Gunzer, M.; Metze, D.; Luger, T.; Steinhoff, M. Localization of μ-opioid receptor 1A on sensory nerve fibers in human skin. Regul. Pept. 2002, 110, 75–83.
- Labuz, D.; Berger, S.; Mousa, S.A.; Zöllner, C.; Rittner, H.L.; Shaqura, M.A.; Segovia-Silvestre, T.; Przewlocka, B.; Stein, C.; Machelska, H. Peripheral antinociceptive effects of exogenous and immune cell-derived endomorphins in prolonged inflammatory pain. J. Neurosci. 2006, 26, 4350–4358.
- Verma–Gandhu, M.; Bercik, P.; Motomura, Y.; Verdu, E.F.; Khan, W.I.; Blennerhassett, P.A.; Wang, L.; El–Sharkawy, R.T.; Collins, S.M. CD4+ T-cell modulation of visceral nociception in mice. Gastroenterology 2006, 130, 1721–1728.
- Mousa, S.A.; Machelska, H.; Schäfer, M.; Stein, C. Immunohistochemical localization of endomorphin-1 and endomorphin-2 in immune cells and spinal cord in a model of inflammatory pain. J. Neuroimmunol. 2002, 126, 5–15.
- Smith, E.M. Opioid peptides in immune cells. Adv. Exp. Med. Biol. 2003, 521, 51–68.
- Wieczorek, A.; Krajewski, P.; Kozioł-Gałczyńska, M.; Szepietowski, J.C. Opioid receptors expression in the skin of haemodialysis patients suffering from uraemic pruritus. J. Eur. Acad. Derm. Venereol. 2020, 34, 2368–2372.
- Świerczyńska, K.; Krajewski, P.K.; Nowicka-Suszko, D.; Białynicki-Birula, R.; Krajewska, M.; Szepietowski, J.C. The Serum Level of IL-31 in Patients with Chronic Kidney Disease-Associated Pruritus: What Can We Expect? Toxins 2022, 14, 197.
- Philippe, D.; Dubuquoy, L.; Groux, H.; Brun, V.; Van Chuoï-Mariot, M.T.; Gaveriaux-Ruff, C.; Colombel, J.F.; Kieffer, B.L.; Desreumaux, P. Anti-inflammatory properties of the μ opioid receptor support its use in the treatment of colon inflammation. J. Clin. Investig. 2003, 111, 1329–1338.
- Tegeder, I.; Geisslinger, G. Opioids as modulators of cell death and survival—unraveling mechanisms and revealing new indications. Pharmacol. Rev. 2004, 56, 351–369.
- Schricker, S.; Heider, T.; Schanz, M.; Dippon, J.; Alscher, M.D.; Weiss, H.; Mettang, T.; Kimmel, M. Strong associations between inflammation, Pruritus and mental health in dialysis patients. Acta Derm. Venereol. 2019, 99, 524–529.
- Taraz, M.; Taraz, S.; Dashti-Khavidaki, S. Association between depression and inflammatory/anti-inflammatory cytokines in chronic kidney disease and end-stage renal disease patients: A review of literature. Hemodial. Int. 2015, 19, 11–22.
- Yamamoto, Y.; Hayashino, Y.; Yamazaki, S.; Akiba, T.; Akizawa, T.; Asano, Y.; Saito, A.; Kurokawa, K.; Miyachi, Y.; Fukuhara, S. Depressive symptoms predict the future risk of severe pruritus in haemodialysis patients: Japan Dialysis Outcomes and Practice Patterns Study. Br. J. Dermatol. 2009, 161, 384–389.
- Morton, C.A.; Lafferty, M.; Hau, C.; Henderson, I.; Jones, M.; Lowe, J.G. Pruritus and skin hydration during dialysis. Nephrol. Dial. Transplant. 1996, 11, 2031–2036.
- Lipman, Z.M.; Paramasivam, V.; Yosipovitch, G.; Germain, M.J. Clinical management of chronic kidney disease-associated pruritus: Current treatment options and future approaches. Clin. Kidney J. 2021, 14, i16–i22.
- Okada, K.; Matsumoto, K. Effect of skin care with an emollient containing a high water content on mild uremic pruritus. Therapher. Dial. 2004, 8, 419–422.
- Balaskas, E.; Szepietowski, J.C.; Bessis, D.; Ioannides, D.; Ponticelli, C.; Ghienne, C.; Taberly, A.; Dupuy, P. Randomized, double-blind study with glycerol and paraffin in uremic xerosis. Clin. J. Am. Soc. Nephrol. 2011, 6, 748–752.
- Forouhari, A.; Moghtaderi, M.; Raeisi, S.; Shahidi, S.; Parin Hedayati, Z.; Zaboliyan, J.; Ani, S.; Moeinzadeh, F.; Mortazavi, M. Pruritus-reducing effects of omega-3 fatty acids in hemodialysis patients: A cross-over randomized clinical trial. Hemodial. Int. 2022, in press.
- Panahi, Y.; Dashti-Khavidaki, S.; Farnood, F.; Noshad, H.; Lotfi, M.; Gharekhani, A. Therapeutic effects of omega-3 fatty acids on chronic kidney disease-associated pruritus: A literature review. Adv. Pharm. Bull. 2016, 6, 509–514.
- Pele, M.; Waluyo, A. Use of olive oil and warm water in bathing intervention in preventing risk of skin integrity damage in total care patients with chronic disease: A case study. J. Pendidik. Keperawatan Indo. 2019, 5, 1–6.
- Elmariah, S.B.; Lerner, E.A. Topical therapies for pruritus. Semin. Cutan. Med. Surg. 2011, 30, 118–126.
- Chen, Z.J.; Cao, G.; Tang, W.X.; Lv, X.Y.; Huang, S.M.; Qin, W.; Ping, F.; Ye, T. A randomized controlled trial of high-permeability haemodialysis against conventional haemodialysis in the treatment of uraemic pruritus. Clin. Exp. Dermatol. 2009, 34, 679–683.
- Gotch, F.A.; Sargent, J.A.; Keen, M.L. Whither goest kt/v? Kidney Int. 2000, 58, S3–S18.
- AlSahow, A.; Muenz, D.; Al-Ghonaim, M.A.; Al Salmi, I.; Hassan, M.; Al Aradi, A.H.; Hamad, A.; Al-Ghamdi, S.M.; Shaheen, F.A.; Alyousef, A.; et al. Kt/V: Achievement, predictors and relationship to mortality in hemodialysis patients in the Gulf Cooperation Council countries: Results from DOPPS (2012–18). Clin. Kidney J. 2021, 14, 820–830.
- Eloot, S.; Van Biesen, W.; Glorieux, G.; Neirynck, N.; Dhondt, A.; Vanholder, R. Does the adequacy parameter kt/v urea reflect uremic toxin concentrations in hemodialysis patients? PLoS ONE 2013, 8, e76838.
- Krajewski, P.K.; Krajewska, M.; Szepietowski, J.C. Pruritus in renal transplant recipients: Current state of knowledge. Adv. Clin. Exp. Med. 2020, 29, 769–772.
- Panuccio, V.; Tripepi, R.; Bellantoni, M.; Saporito, L.; Quattrone, S.; Lacava, V.; Parlongo, G.; Tripepi, G.; Mallamaci, F.; Zoccali, C. Pruritus and quality of life in renal transplant patients. Clin. Transplant. 2017, 31, e12893.
- Schricker, S.; Weisshaar, E.; Kupfer, J.; Mettang, T. Prevalence of pruritus in a single cohort of long-term kidney transplant recipients. Acta Derm. Venereol. 2020, 100, adv00066.
- Ketteler, M.; Block, G.A.; Evenepoel, P.; Fukagawa, M.; Herzog, C.A.; McCann, L.; Moe, S.M.; Shroff, R.; Tonelli, M.A.; Toussaint, N.D.; et al. Executive summary of the 2017 KDIGO Chronic Kidney Disease–Mineral and Bone Disorder (CKD-MBD) Guideline Update: What’s changed and why it matters. Kidney Int. 2017, 92, 26–36.
- Apetrii, M.; Goldsmith, D.; Nistor, I.; Siriopol, D.; Voroneanu, L.; Scripcariu, D.; Vervloet, M.; Covic, A. Impact of surgical parathyroidectomy on chronic kidney disease-mineral and bone disorder (CKD-MBD)–a systematic review and meta-analysis. PLoS ONE 2017, 12, e0187025.
- Amro, A.; Waldum-Grevbo, B.; von der Lippe, N.; Brekke, F.B.; Miaskowski, C.; Os, I. Symptom clusters from dialysis to renal transplantation: A five-year longitudinal study. J. Pain Symptom Manag. 2016, 51, 512–519.





