| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Nimmy Varghese | + 1858 word(s) | 1858 | 2020-10-07 05:16:31 | | | |
| 2 | Bruce Ren | Meta information modification | 1858 | 2020-10-20 04:04:00 | | |
Video Upload Options
Recently, nutritional interventions have received attention as promising approaches to promote human health during a lifespan. The Mediterranean and Okinawan diets have been associated with longevity and decreasing risk for age-related diseases in contrast to the Western diet. The effect might be due to several antioxidative bioactive compounds highly consumed in both diets, namely, resveratrol, hydroxytyrosol, oleuropein, curcumin, and spermidine. This review aims to address the underlying mechanisms of these compounds to enhance mental fitness throughout life with a focus on brain mitophagy. Mitophagy is the autophagic clearance of dysfunctional, redundant, and aged mitochondria. In aging and neurodegenerative disorders, mitophagy is crucial to preserve the autophagy mechanism of the whole cell, especially during oxidative stress. Growing evidence indicates that curcumin, astaxanthin, resveratrol, hydroxytyrosol, oleuropein, and spermidine might exert protective functions via antioxidative properties and as well the enhanced induction of mitophagy mediators. The compounds seem to upregulate mitophagy and thereby alleviate the clearance of dysfunctional and aged mitochondria as well as mitogenesis. Thus, the Mediterranean or Okinawan diet could represent a feasible nutritional approach to reduce the risk of developing age-related cognitive impairment and corresponding disorders via the stimulation of mitophagy and thereby ensure a balanced redox state of brain cells.
1. Introduction
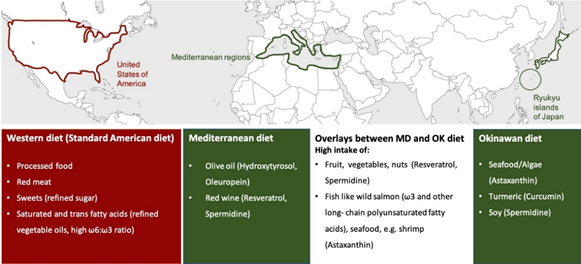
The interest in nutritional interventions as a promising approach to counteract pathological brain aging and age-related neurodegenerative disorders has been rising in the last decade [1]. In this light, several bioactive compounds present in different foods have been found to be beneficial for human health during a lifespan [2]. This review focuses on two different diets, the Mediterranean diet and the Okinawan diet, which are both linked to longevity and decreasing risk for chronic age-related brain disorders (Figure 1) [3][4].
Figure 1. Characteristic bioactive compounds of the Mediterranean and Asian/Okinawan diet. The figure shows the geographic origin of three different types of diets: the Western, the Mediterranean, and the Asian (Okinawan) diet. The Mediterranean diet is defined as a dietary pattern of people living in countries that border the Mediterranean Sea, whereas the Okinawan diet originates from the eating habits of the indigenous people of the Japanese Ryukyu Islands (main island Okinawa). The Western diet is prevalent in developed countries, especially in the USA. Typical of the Mediterranean diet is the consumption of red wine with its active ingredients resveratrol and spermidine, and the intake of olive oil with its active ingredients oleuropein and hydroxytyrosol. All four compounds are associated with beneficial effects on human health. Characteristic food items of the Okinawan diet that are also known for their health benefits are soy products such as tofu (spermidine), turmeric (curcumin), seafood, and algae (astaxanthin). Similarities of both diets include the intake of a broad spectrum of antioxidants, polyphenols (like resveratrol), and spermidine from fruit and vegetable and the moderate-to-high consumption of seafood and fish (astaxanthin, healthy fatty acids). In contrast to both diets, the Western diet could have detrimental health consequences due to the high consumption of unhealthy fatty acids (saturated fatty acids), red meat, sweets, and highly processed food items. Abbreviations: MD = Mediterranean diet, OK = Okinawan diet, ω = omega, USA = United States of America.
The Mediterranean diet, a traditional dietary pattern of the people living in the Mediterranean region (as shown in Figure 1), is known for its high consumption of olive oil, fruits, and vegetables, which are especially high in antioxidants, as well as moderate to high intake of fish, whole grain cereals, and red wine [5], whereas, the consumption of sweets, meat, and dairy products is rather low [6]. There are specific plant bioactives (e.g., resveratrol from red wine, hydroxytyrosol and oleuropein from olive oil) that are especially characteristic of the Mediterranean diet and its beneficial effects on human health (Figure 1) [6][7]. Initially, the Mediterranean diet became popular because of its historical lower rate of cardiovascular-related diseases in comparison to the Western diet, which is prevalent in developed countries, especially in the US [6]. Different from the Mediterranean diet and Asian diet, Western-type diets are dominated by convenience and highly processed foods such as processed and red meat, desserts, sweets, fried food, and a high intake of dairy products, containing a higher number of saturated fatty acids and refined grains, so that consumption of a Western diet could have detrimental consequences on health, due to the reduced intake of fresh fruit and vegetables in contrast to the excess consumption of saturated fatty acids, sugar, and sodium (Figure 1) [8]. Nowadays, several epidemiological studies, as reviewed by Casas and coworkers, have shown the potential of the Mediterranean diet as protection against a wide range of disease conditions, such as cancer, diabetes, atherosclerosis, cognitive disorders, and Alzheimer disease (AD) [9][10]. Since 2010, the Mediterranean diet is considered as a heritage by humanity by UNESCO by virtue of its beneficial properties [11]. As part of an Asian diet, the Okinawan diet has been found to be particularly interesting since the residents of the Japanese islands of Ryukyu (main island Okinawa) also represent one of the healthiest and longest-living people in the world (as seen in Figure 1) [12]. Their diet is characterized by a low caloric and fatty acid intake, high consumption of vegetables and soy products, and a moderate-to-high intake of fish and sea vegetables. Le Couteur and coworkers named bitter melon, Okinawan tofu, turmeric, and seaweed as characteristics of the traditional Okinawan diet [12]. Similarities between the Mediterranean diet, as a European-based lifestyle, and the Okinawan diet, as part of an Asian-based lifestyle, are the high intake of antioxidants in the form of fruit and vegetables, the moderate-to-high consumption of fish, and the focus on healthy fats that are rich in omega 3 and lower in saturated fatty acids (Figure 1). In contrast to Asian diets, the Mediterranean diet is mostly abundant in isoflavones and curcumin. Although further research is required, Pallauf and colleagues hypothesized that combining foods from both diets could improve the overall health of aging populations. The removal of dysfunctional mitochondria called mitophagy is crucial for cell survival and health, especially for neurons, as impairments might generally happen with aging [13]. Recent studies have revealed abnormalities in mitochondrial function, including compromised clearing of dysfunctional mitochondria during brain aging and in the pathology of neurodegenerative disorders [14][15]. Therefore, as a next step, this review will target the underlying mechanisms of the most prominent bioactive compounds of both diets (namely, resveratrol from red wine, hydroxytyrosol and oleuropein from olive oil, astaxanthin from algae/seafood, curcumin from turmeric, as well as spermidine from soy products and vegetables) to promote health throughout life, with a specific focus on mitophagy and mitochondrial function in the brain.
2. Bioactive Compounds Promoting Health in the Mediterranean or Asian/Okinawan Diet
The Mediterranean diet and the Asian/Okinawan diet have both been linked to longevity and decreasing risk for chronic age-related diseases. Therefore, we selected the most prominent bioactive compounds of both diets to have a closer look at the biological properties that make them beneficial for human brain health during a lifespan. Due to the fact that turmeric and seaweed/seafood are traditional ingredients of the Okinawan diet, curcumin and astaxanthin were chosen as bioactive compounds; resveratrol (red wine) and hydroxytyrosol and oleuropein (olive oil) are well known in the Mediterranean diet (Figure 1). Although resveratrol is mostly associated with the Mediterranean diet, the highest natural concentration exists in Japanese knotweed, a spring vegetable from East Asia [16]. Hence, there might be an overlap of health-promoting bioactive compounds in the two types of diets, as astaxanthin can also be found in seafood (e.g., shrimp), which is as well consumed in Mediterranean areas. Spermidine is also present in both diets since it occurs in soy products and red wine but also in fresh fruit and vegetables, which are essential for the Mediterranean and Asian/Okinawan diets.
2.1. Curcumin (Turmeric)
Turmeric grows widely in different parts of India and Southeast Asia, as well as in tropical regions, and is well known as a spice in human diets [17]. Curcumin, a bright yellow-colored polyphenol that is derived from the rhizome of turmeric (Curcuma longa), is the main active compound of turmeric [18][19]. Turmeric or turmeric compounds like curcumin are used in traditional Indian medicine (Ayurvedic medicine) and other traditional medical systems for the treatment of a wide variety of diseases and conditions, such as pulmonary or gastrointestinal diseases [17]. Moreover, it is believed to possess a wide range of biological properties, including anti-inflammatory, antioxidant, as well as neuroprotective effects [17][18][19].
2.2. Astaxanthin (Algae, Seafood)
Astaxanthin is a xanthophyll carotenoid that is found in marine microorganisms, microalgae, fungi, yeast, and crustaceans like shrimp and lobster, as well as in reddish-colored fish like salmon [20][21]. Astaxanthin is considered to be one of the most potent antioxidants in nature and, as such, it is not only able to decrease oxidative stress in cell- and animal models but is also believed to have a positive effect on aging [21][22]. Besides its antioxidant effects, astaxanthin seems to exert anti-inflammatory, antitumor, and antiaging effects [23].
2.3. Resveratrol (Red Wine, Japanese Knotweed, Grapes)
Resveratrol is a natural stilbene polyphenolic compound that is prominent in red wine as well as in other food sources, including fruits like grapes, bananas, and blueberries, but also in spinach, peanuts, and cocoa [24][25][26]. Japanese knotweed possesses the highest naturally occurring levels of resveratrol [16]. Numerous biological properties of resveratrol have been reported, such as antioxidant, anti-inflammatory, antitumor, antiaging, and neuro- and cardioprotective effects [27][28].
2.4. Hydroxytyrosol, Oleuropein (Olive Oil)
Hydroxytyrosol and oleuropein are the major phenolic compounds found in olive oil and a major source of unsaturated fatty acids in the Mediterranean diet [29][30]. The relative amount of oleuropein and hydroxytyrosol in extra virgin olive can vary as it is influenced by different factors such as the variety and maturity of the olive fruits, the climate, and processing [31]. Hydroxytyrosol is a product of oleuropein hydrolysis that occurs during the maturation and storage of olive oil, as well by microbiota action in the organism after the consumption of olive oil [31]. Hydroxytyrosol can also be found in wine due to the ability of yeast to produce it during alcoholic fermentation . Several in vitro and in vivo studies have shown the various health benefits of oleuropein and its derivates, including antioxidant, anti-inflammatory, antiviral, antitumor, cardioprotective, antidiabetic, and neuroprotective effects [31].
2.5. Spermidine (Fruit, Vegetable, Soybean)
Spermidine is a polyamine which exists in all living cells, including microorganism, plants, and animals [32]. There are three different sources for spermidine in humans: endogenous biosynthesis, exogenous food intake, and microbial activity in the intestines [33][34]. Additional supplementation of polyamine precursors, such as ornithine and related amino acids, is supposed to enhance polyamine synthesis [35]. Spermidine-rich sources are, on the one hand, unprocessed plant-derived foods, e.g., fresh green pepper, cauliflower, broccoli, nuts, wheat germ, and mushrooms and, on the other hand, products deriving from fermentation processes such as soybean, cheese, and red wine [36][37]. According to Madeo and colleagues, the highest amount of spermidine is found in the Mediterranean diet [38]. Spermidine exerts antioxidant, anti-inflammatory, and cardioprotective actions and is furthermore believed to preserve high-order brain functions [39]. Both Jing and coworkers and Madeo and coworkers have mentioned the potential of exogenous spermidine supply in prolonging the life span of several model organisms, including yeast, nematodes, and flies [40][41]. Jing and group postulate that spermidine might have an antiaging effect due to its autophagy-enhancing properties [40].
References
- Gensous, N.; Garagnani, P.; Santoro, A.; Giuliani, C.; Ostan, R.; Fabbri, C.; Milazzo, M.; Gentilini, D.; Di Blasio, A.M.; Pietruszka, B.; et al. One-year Mediterranean diet promotes epigenetic rejuvenation with country- and sex-specific effects: A pilot study from the NU-AGE project. GeroScience 2020, 42, 687–701, doi:10.1007/s11357-019-00149-0.
- Davinelli, S.; De Stefani, D.; De Vivo, I.; Scapagnini, G. Polyphenols as Caloric Restriction Mimetics Regulating Mitochondrial Biogenesis and Mitophagy. Trends Endocrinol. Metab. 2020, 31, 536–550, doi:10.1016/j.tem.2020.02.011.
- Willcox, D.C.; Scapagnini, G.; Willcox, B.J. Healthy aging diets other than the Mediterranean: A focus on the Okinawan diet. Mech. Ageing Dev. 2014, 136–137, 148–162, doi:10.1016/j.mad.2014.01.002.
- Hidalgo-Mora, J.J.; García-Vigara, A.; Sanchez-Sanchez, M.L.; García-Pérez, M.-Á.; Tarín, J.J.; Cano, A. The Mediterranean diet: A historical perspective on food for health. Maturitas 2020, 132, 65–69, doi:10.1016/j.maturitas.2019.12.002.
- Martínez-González, M.A.; Hershey, M.S.; Zazpe, I.; Trichopoulou, A. Transferability of the Mediterranean Diet to Non-Mediterranean Countries. What Is and What Is Not the Mediterranean Diet. Nutrients 2017, 9, 1226, doi:10.3390/nu9111226.
- Huhn, S.; Masouleh, S.K.; Stumvoll, M.; Villringer, A.; Witte, A.V. Components of a Mediterranean diet and their impact on cognitive functions in aging. Front. Aging Neurosci. 2015, 7, 132, doi:10.3389/fnagi.2015.00132.
- Pallauf, K.; Giller, K.; Huebbe, P.; Rimbach, G. Nutrition and healthy ageing: Calorie restriction or polyphenol-rich “MediterrAsian” diet? Oxid. Med. Cell. Longev. 2013, 2013, 1–14, doi:10.1155/2013/707421.
- Guilleminault, L.; Williams, E.A.; Scott, H.; Berthon, B.S.; Jensen, M.E.; Wood, L.G. Diet and Asthma: Is It Time to Adapt Our Message? Nutrients 2017, 9, 1227, doi:10.3390/nu9111227.
- Casas, R.; Sacanella, E.; Estruch, R. The immune protective effect of the Mediterranean diet against chronic low-grade inflammatory diseases. Endocr. Metab. Immune Disord. Drug Targets 2014, 14, 245–254.
- Tosti, V.; Bertozzi, B.; Fontana, L. Health Benefits of the Mediterranean Diet: Metabolic and Molecular Mechanisms. J. Gerontol. A Biol. Sci. Med. Sci. 2018, 73, 318–326.
- de Pablos, R.M.; Espinosa-Oliva, A.M.; Hornedo-Ortega, R.; Cano, M.; Arguelles, S. Hydroxytyrosol protects from aging process via AMPK and autophagy; a review of its effects on cancer, metabolic syndrome, osteoporosis, immune-mediated and neurodegenerative diseases. Pharmacol. Res. 2019, 143, 58–72, doi:10.1016/j.phrs.2019.03.005.
- Le Couteur, D.; Solon-Biet, S.M.; Wahl, D.; Cogger, V.C.; Willcox, B.J.; Willcox, D.C.; Raubenheimer, D.; Simpson, S.J. New Horizons: Dietary protein, ageing and the Okinawan ratio. Age Ageing 2016, 45, 443–447, doi:10.1093/ageing/afw069.
- Reddy, P.H.; Oliver, D.M. Amyloid Beta and Phosphorylated Tau-Induced Defective Autophagy and Mitophagy in Alzheimer’s Disease. Cells 2019, 8, 488, doi:10.3390/cells8050488.
- Kerr, J.S.; Adriaanse, B.A.; Greig, N.H.; Mattson, M.P.; Cader, M.Z.; Bohr, V.A.; Fang, E.F. Mitophagy and Alzheimer’s Disease: Cellular and Molecular Mechanisms. Trends Neurosci. 2017, 40, 151–166, doi:10.1016/j.tins.2017.01.002.
- Bakula, D.; Scheibye-Knudsen, M. MitophAging: Mitophagy in Aging and Disease. Front. Cell Dev. Boil. 2020, 8, 239, doi:10.3389/fcell.2020.00239.
- Kotecha, R.; Takami, A.; Espinoza, J.L. Dietary phytochemicals and cancer chemoprevention: A review of the clinical evidence. Oncotarget 2016, 7, 52517–52529, doi:10.18632/oncotarget.9593.
- de Oliveira, M.R.; Jardim, F.R.; Setzer, W.N.; Nabavi, S.M.; Nabavi, S.F. Curcumin, mitochondrial biogenesis, and mitophagy: Exploring recent data and indicating future needs. Biotechnol. Adv. 2016, 34, 813–826, doi:10.1016/j.biotechadv.2016.04.004.
- Liao, V.H.; Yu, C.W.; Chu, Y.J.; Li, W.H.; Hsieh, Y.C.; Wang, T.T. Curcumin-mediated lifespan extension in Caenorhabditis elegans. Mech. Ageing Dev. 2011, 132, 480–487, doi:10.1016/j.mad.2011.07.008.
- Forouzanfar, F.; Read, M.I.; Barreto, G.E.; Sahebkar, A. Neuroprotective effects of curcumin through autophagy modulation. IUBMB Life 2020, 72, 652–664, doi:10.1002/iub.2209.
- Galasso, C.; Orefice, I.; Pellone, P.; Cirino, P.; Miele, R.; Ianora, A.; Brunet, C.; Sansone, C. On the Neuroprotective Role of Astaxanthin: New Perspectives? Mar. Drugs 2018, 16, doi:10.3390/md16080247.
- Yan, T.; Zhao, Y.; Zhang, X.; Lin, X. Astaxanthin Inhibits Acetaldehyde-Induced Cytotoxicity in SH-SY5Y Cells by Modulating Akt/CREB and p38MAPK/ERK Signaling Pathways. Mar. Drugs 2016, 14, doi:10.3390/md14030056.
- Yang, J.P.; Shin, J.H.; Seo, S.H.; Kim, S.G.; Lee, S.H.; Shin, E.H. Effects of Antioxidants in Reducing Accumulation of Fat in Hepatocyte. Int. J. Mol. Sci. 2018, 19, doi:10.3390/ijms19092563.
- Lee, H.; Lim, J.W.; Kim, H. Effect of Astaxanthin on Activation of Autophagy and Inhibition of Apoptosis in Helicobacter pylori-Infected Gastric Epithelial Cell Line AGS. Nutrients 2020, 12, doi:10.3390/nu12061750.
- Zhang, Q.; Zhang, C.; Ge, J.; Lv, M.W.; Talukder, M.; Guo, K.; Li, Y.H.; Li, J.L. Ameliorative effects of resveratrol against cadmium-induced nephrotoxicity via modulating nuclear xenobiotic receptor response and PINK1/Parkin-mediated Mitophagy. Food Funct. 2020, 11, 1856–1868, doi:10.1039/c9fo02287b.
- Gomes, B.A.Q.; Silva, J.P.B.; Romeiro, C.F.R.; Dos Santos, S.M.; Rodrigues, C.A.; Goncalves, P.R.; Sakai, J.T.; Mendes, P.F.S.; Varela, E.L.P.; Monteiro, M.C. Neuroprotective Mechanisms of Resveratrol in Alzheimer's Disease: Role of SIRT1. Oxid. Med. Cell. Longev. 2018, 2018, 8152373, doi:10.1155/2018/8152373.
- Cao, S.; Shen, Z.; Wang, C.; Zhang, Q.; Hong, Q.; He, Y.; Hu, C. Resveratrol improves intestinal barrier function, alleviates mitochondrial dysfunction and induces mitophagy in diquat challenged piglets(1). Food Funct. 2019, 10, 344–354, doi:10.1039/c8fo02091d.
- Shakeri, F.; Bianconi, V.; Pirro, M.; Sahebkar, A. Effects of Plant and Animal Natural Products on Mitophagy. Oxid. Med. Cell. Longev. 2020, 2020, 6969402, doi:10.1155/2020/6969402.
- Naoi, M.; Wu, Y.; Shamoto-Nagai, M.; Maruyama, W. Mitochondria in Neuroprotection by Phytochemicals: Bioactive Polyphenols Modulate Mitochondrial Apoptosis System, Function and Structure. Int. J. Mol. Sci. 2019, 20, doi:10.3390/ijms20102451.
- Chin, K.Y.; Pang, K.L. Therapeutic Effects of Olive and Its Derivatives on Osteoarthritis: From Bench to Bedside. Nutrients 2017, 9, doi:10.3390/nu9101060.
- Cetrullo, S.; D’Adamo, S.; Guidotti, S.; Borzi, R.M.; Flamigni, F. Hydroxytyrosol prevents chondrocyte death under oxidative stress by inducing autophagy through sirtuin 1-dependent and -independent mechanisms. Biochim. Biophys. Acta 2016, 1860, 1181–1191, doi:10.1016/j.bbagen.2016.03.002.
- Cordero, J.G.; Garcia-Escudero, R.; Avila, J.; Gargini, R.; Garcia-Escudero, V. Benefit of Oleuropein Aglycone for Alzheimer's Disease by Promoting Autophagy. Oxid. Med. Cell. Longev. 2018, 2018, 5010741, doi:10.1155/2018/5010741.
- Munoz-Esparza, N.C.; Latorre-Moratalla, M.L.; Comas-Baste, O.; Toro-Funes, N.; Veciana-Nogues, M.T.; Vidal-Carou, M.C. Polyamines in Food. Front. Nutr. 2019, 6, 108, doi:10.3389/fnut.2019.00108.
- Saiki, S.; Sasazawa, Y.; Fujimaki, M.; Kamagata, K.; Kaga, N.; Taka, H.; Li, Y.; Souma, S.; Hatano, T.; Imamichi, Y.; et al. A metabolic profile of polyamines in parkinson disease: A promising biomarker. Ann. Neurol. 2019, 86, 251–263, doi:10.1002/ana.25516.
- Schwarz, C.; Stekovic, S.; Wirth, M.; Benson, G.; Royer, P.; Sigrist, S.J.; Pieber, T.; Dammbrueck, C.; Magnes, C.; Eisenberg, T.; et al. Safety and tolerability of spermidine supplementation in mice and older adults with subjective cognitive decline. Aging (Albany N. Y.) 2018, 10, 19–33, doi:10.18632/aging.101354.
- Ramani, D.; De Bandt, J.P.; Cynober, L. Aliphatic polyamines in physiology and diseases. Clin. Nutr. 2014, 33, 14–22, doi:10.1016/j.clnu.2013.09.019.
- Madeo, F.; Bauer, M.A.; Carmona-Gutierrez, D.; Kroemer, G. Spermidine: A physiological autophagy inducer acting as an anti-aging vitamin in humans? Autophagy 2019, 15, 165–168, doi:10.1080/15548627.2018.1530929.
- Kiechl, S.; Pechlaner, R.; Willeit, P.; Notdurfter, M.; Paulweber, B.; Willeit, K.; Werner, P.; Ruckenstuhl, C.; Iglseder, B.; Weger, S.; et al. Higher spermidine intake is linked to lower mortality: A prospective population-based study. Am. J. Clin. Nutr. 2018, 108, 371–380, doi:10.1093/ajcn/nqy102.
- Madeo, F.; Eisenberg, T.; Pietrocola, F.; Kroemer, G. Spermidine in health and disease. Science 2018, 359, doi:10.1126/science.aan2788.
- Wirth, M.; Schwarz, C.; Benson, G.; Horn, N.; Buchert, R.; Lange, C.; Kobe, T.; Hetzer, S.; Maglione, M.; Michael, E.; et al. Effects of spermidine supplementation on cognition and biomarkers in older adults with subjective cognitive decline (SmartAge)-study protocol for a randomized controlled trial. Alzheimers Res. Ther. 2019, 11, 36, doi:10.1186/s13195-019-0484-1.
- Jing, Y.H.; Yan, J.L.; Wang, Q.J.; Chen, H.C.; Ma, X.Z.; Yin, J.; Gao, L.P. Spermidine ameliorates the neuronal aging by improving the mitochondrial function in vitro. Exp. Gerontol. 2018, 108, 77–86, doi:10.1016/j.exger.2018.04.005.
- Madeo, F.; Eisenberg, T.; Buttner, S.; Ruckenstuhl, C.; Kroemer, G. Spermidine: A novel autophagy inducer and longevity elixir. Autophagy 2010, 6, 160–162, doi:10.4161/auto.6.1.10600.