Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Masanobu Kawai | -- | 2006 | 2022-08-29 06:42:28 | | | |
| 2 | Jessie Wu | + 10 word(s) | 2016 | 2022-08-29 07:54:08 | | | | |
| 3 | Jessie Wu | -1 word(s) | 2015 | 2022-08-29 07:57:58 | | | | |
| 4 | Jessie Wu | + 679 word(s) | 2694 | 2022-08-29 09:38:41 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Kawai, M.; Hasegawa, Y. Skeletal Characteristics with Turner Syndrome. Encyclopedia. Available online: https://encyclopedia.pub/entry/26584 (accessed on 30 March 2026).
Kawai M, Hasegawa Y. Skeletal Characteristics with Turner Syndrome. Encyclopedia. Available at: https://encyclopedia.pub/entry/26584. Accessed March 30, 2026.
Kawai, Masanobu, Yukihiro Hasegawa. "Skeletal Characteristics with Turner Syndrome" Encyclopedia, https://encyclopedia.pub/entry/26584 (accessed March 30, 2026).
Kawai, M., & Hasegawa, Y. (2022, August 29). Skeletal Characteristics with Turner Syndrome. In Encyclopedia. https://encyclopedia.pub/entry/26584
Kawai, Masanobu and Yukihiro Hasegawa. "Skeletal Characteristics with Turner Syndrome." Encyclopedia. Web. 29 August, 2022.
Copy Citation
Turner syndrome (TS) is a chromosomal disorder characterized by a short stature and gonadal dysgenesis, the latter of which requires estrogen replacement therapy (ERT) to induce and maintain secondary sexual characteristics. Insufficient ERT is associated with compromised skeletal health, including bone fragility, in adults with TS. Estrogen insufficiency during adolescence is critical because the acquisition of a defective bone mass during this period results in impaired bone strength later in the life.
bone
estrogen
children
1. Introduction
The complications of turner syndrome (TS) during adulthood also vary, with compromised bone health being one of the clinically evident complications [1][2][3][4][5]. Gravholt et al. previously reported the clinical characteristics of 594 subjects with TS in Denmark based on data from the Danish Cytogenetic Central Register and Danish National Registry of Patients, and reported an increased incidence of osteoporosis (RR 10.12 [95% CI 2.18–30.93]) and fractures (RR 2.16 [95% CI 1.50–3.00]) [5]. Multiple lines of evidence, including meta-analyses, indicate that estrogen insufficiency is a predominant risk factor for low bone mineral density (BMD) [6][7][8]. In addition, the early introduction of ERT to accrue better BMD during adulthood has been suggested [9][10][11][12][13][14]. As the acquisition of an adequate bone mass during adolescence and young adulthood is crucial for reducing the subsequent risk of osteoporosis, the appropriate introduction of ERT during adolescence is of clinical importance. Additionally, TS-associated comorbidities, including vitamin D deficiency, celiac disease, inflammatory bowel disease, and hyperthyroidism, are also risk factors for low BMD [15][16]. The genetic influence of haploinsufficiency of the short-stature homeobox (SHOX) gene on the skeletal characteristics in TS has also been reported [17]. Furthermore, the transcriptome analysis revealed a down-regulation of bone morphogenic protein 2 and insulin-like growth factor 2 expressions in human fibroblasts with 45, X karyotype compared with those with 46, XX karyotype [18], suggesting that the lack of one X chromosome may have a profound effect on bone metabolism beyond the effect of haploinsufficiency of the SHOX gene.
2. Bone Geometry and Strength in Turner Syndrome during Pre-Pubertal and Pubertal Periods
Although BMD is an important surrogate marker for bone strength, other parameters of bone geometry also have significant impacts on bone strength; therefore, analyses of the bone geometry provide additional insights into the skeletal phenotype in children with TS. Cortical bones generally show age-dependent increases in the total cross-sectional area (CSA) and cortical thickness, and this is associated with increases in the strength−strain index (SSI) during puberty [19][20]. Pitukcheewanont et al. evaluated cortical bone geometry in 22 prepubertal TS with a mean (SD) age of 11.9 (3.3) years and found that the cortical bone CSA was similar to that of the controls after adjustments for weight, height, skeletal age, and pubertal stage [21]. Soucek et al. investigated 22 prepubertal girls with TS with a mean (SD) age of 10.3 (2.2) years and found a lower CSA Z-score in the TS group than in the controls, and this difference was not observed after adjustments for height. Therefore, the inclusion of prepubertal girls with delayed puberty may have affected the findings obtained. In a subpopulation of eight children younger than 10 years old, the cortical CSA was above the lower limit of the reference values in all of the subjects [22]. These findings indicate that cortical bone geometry, particularly cortical bone size, was unlikely affected during prepubertal ages in TS (Table 1) (Figure 1).
Table 1. Bone mineral density and geometry in children and adolescents with Turner syndrome.
| Author | Year | Age (Years) # | Bone Mineral Density | Bone Geometry | ||||
|---|---|---|---|---|---|---|---|---|
| Number | Methods | Trabecular Bone ## | Cortical Bone ## | |||||
| Prepubertal | ||||||||
| Ross et al. [19] | 1991 | 4–13 | 78 | DPA DXA | Lower LS aBMD by DPA, but the difference disappeared after adjustments for bone age or height age | Lower aBMD of the wrist after adjustments for chronological age, bone age, or BMI by SPA | na | |
| Nanao et al. [22] | 2002 | 4–6.9 | 5 | QCT | Similar LS vBMD to age-matched controls | na | na | |
| 7–9.9 | 8 | |||||||
| 10–12.9 | 15 | Lower LS vBMD than age-matched controls | ||||||
| Högler et al. [20] | 2004 | 10.46 ± 3.25 | 51 | DXA | LS vBMD Z score higher than −1.5 in 44 out of 51 | FN vBMD Z-score higher than −1.5 in 38 out of 51 | na | |
| <10 * | 21 | LS vBMD Z score higher than −1.5 in all subjects | FN vBMD Z-score higher than −1.5 in 16 out of 21 | |||||
| Soucek et al. [21] | 2011 | 10.3 ± 2.2 | 22 | pQCT | Similar vBMD of the radius to age-matched controls | Lower vBMD of the radius than age-matched controls | Higher radial CSA than age-matched controls after adjustments for height. Lower cortical thickness than age- matched controls | |
| <10 * | 8 | Normal vBMD of the radius in all subjects | Lower vBMD of the radius than the lower limit of the age-matched reference in 3 out of 10 | CSA above the lower limit in all subjects. Cortical thickness: the lower half of the normal range in 6, lower than the lower limit in 1 | ||||
| Pitukcheewanont et al. [26] | 2011 | 11.9 ± 3.3 | 22 | DXA pQCT | Similar LS vBMD to age-matched controls after adjustments for weight, height, skeletal age, and pubertal stage | Similar femoral vBMD with age-matched controls after adjustments for weight, height, skeletal age, and pubertal stage | Similar cortical bone CSA to age-matched controls after adjustments for weight, height, skeletal age, and pubertal stage | |
| Prepubertal/Pubertal | ||||||||
| Shaw et al. [27] | 1997 | 4–17 | 18 | DXA | Lower LS aBMD than the age-matched controls, but the difference disappeared after adjustments for body weight and pubertal status | na | na | |
| Holroyd et al. [28] | 2010 | 12.7 ± 3.8 7–19 | 22 | DXA pQCT | Similar LS BMAD and radial vBMD Z-scores as the age- and Tanner-stage-matched controls | Lower FN BMAD and radial vBMD Z-scores than the age- and Tanner-stage-matched controls | A similar radial cortical thickness Z-score as the age- and Tanner-stage-matched controls | |
| Soucek et al. [24] | 2018 | 10.0 ± 2.2 13.5 ± 1.5 16.1 ± 0.4 | 15 14 3 | pQCT | Normal height-specific z-score of the radius vBMD during the prepubertal period. Age-dependent decrease in the radial vBMD Z-score thereafter |
Normal vBMD during prepubertal ages, followed by a decrease during peripubertal ages. Thereafter, an increase was observed during pubertal initiation/induction | Comparable height-specific CSA during prepubertal period. The height-specific CSA Z-score showed an age- dependent increase | |
| Pubertal | ||||||||
| Högler et al. [20] | 2004 | 13.89 ± 2.23 | 16 | DXA | Age-dependent decrease during the pre- and peripubertal, and pubertal periods | The FN vBMD Z-score is constant during the pre- and peripubertal, and pubertal periods | na | |
| 18.98 ± 2.40 | 16 | |||||||
| Soucek et al. [21] | 2011 | 14.3 ± 1.7 17.4 ± 1.2 | 25 20 | pQCT | Lower radial vBMD Z-score than the reference value | Lower radial vBMD Z-score than the reference value | Similar height-adjusted CSA as the reference value | |
BMD: bone mineral density; BMI: body mass index; BMAD: bone mineral apparent density; aBMD: areal BMD; vBMD: volumetric BMD; CSA: total cross-sectional area; SPA: single photon absorptiometry; DPA: dual photon absorptiometry; DXA: dual energy X-ray absorptiometry; QCT: quantitative computed tomography; pQCT: peripheral QCT; LS: lumbar spine; FN: femoral neck; na: not available. #: Age is shown as the mean ± standard deviation, a range, or both. ##: Data on aBMD of the lumbar spine by DXA are regarded as trabecular bone BMD, whereas those on the femoral neck are regarded as cortical bone BMD. *: subpopulation of those younger than 10 years old collected based on the figures reported in the study.
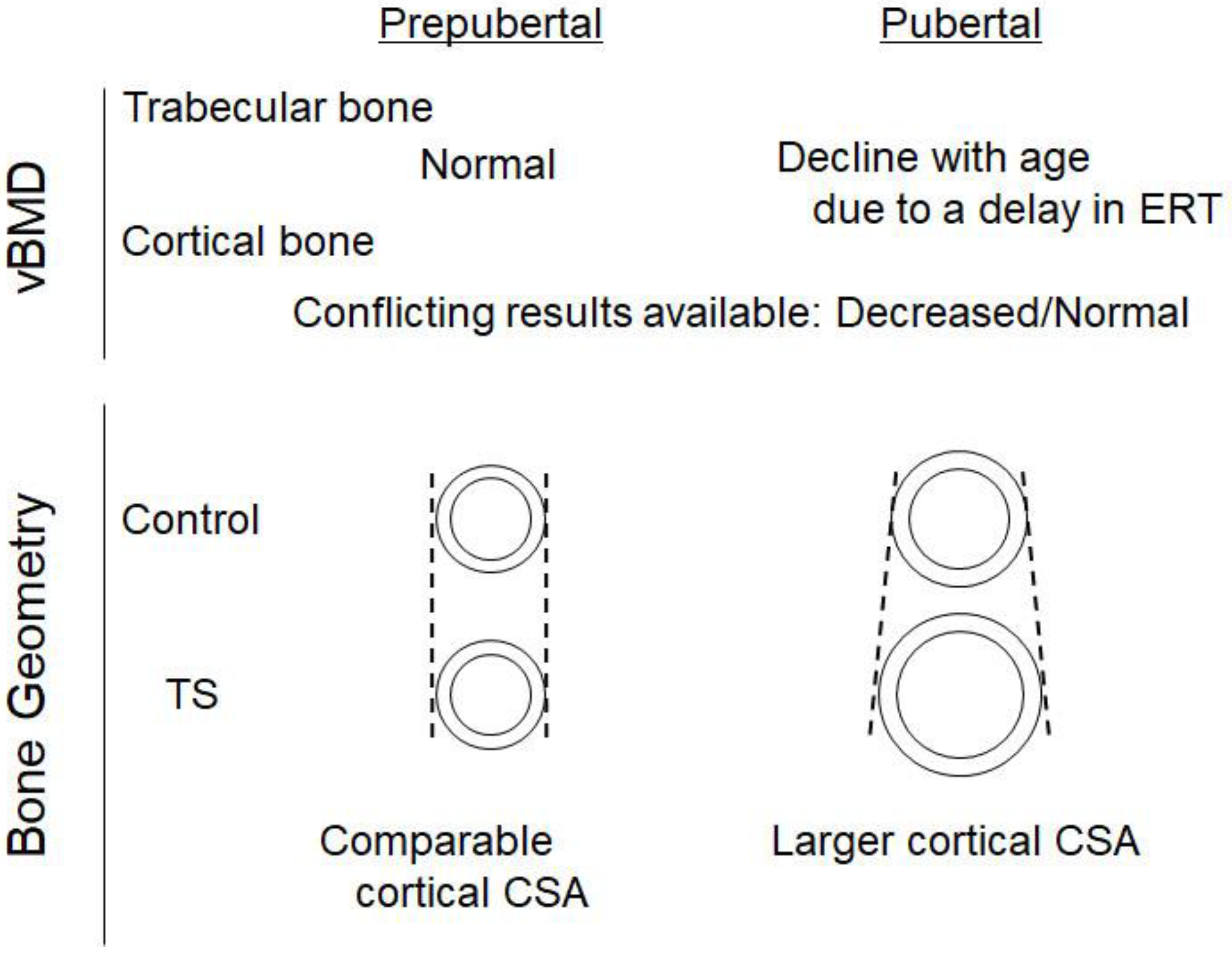
Figure 1. Summary of the skeletal characteristics of TS. Trabecular volumetric bone mineral density (vBMD) is normal in Turner syndrome (TS) during the prepubertal period, whereas it declines thereafter due to a delay in the commencement of estrogen replacement therapy (ERT). Cortical vBMD is low or normal in TS, depending on the confounding variables included in the study design. The inclusion of bone size results in the normal cortical vBMD in TS. The cortical cross-sectional area (CSA) is normal in TS during the prepubertal period, but a larger CSA is observed in TS probably due to a delay in ERT commencement, which may confer bone strength. Growth hormone treatment may also be responsible for a larger CSA in TS.
To obtain a more detailed understanding of the alterations in bone geometry in TS during the pubertal period, Soucek et al. performed a longitudinal analysis in TS in which ERT was initiated based on the current protocol, and found that the height-specific CSA Z-score was not altered in prepubertal girls, whereas age-dependent elevations were observed thereafter that were associated with increases in the SSI Z-score, suggesting that those with TS acquired larger bones during the pubertal period [23]. This characteristic of bone geometry may be explained by the unique effects of estrogen on the acquisition of cortical bone. Estrogen has been shown to inhibit periosteal apposition, whereas endocortical apposition is stimulated [20][24][25][26], which results in the acquisition of smaller bones with a narrower marrow cavity in girls than in boys; therefore, larger cortical bones in TS may be a reflection of the delayed administration of estrogen. In accordance with this notion, in a clinical setting of delayed puberty, the delayed increase in estrogen levels resulted in normal or larger cortical bones with a thinner cortex [20]. As larger cortical bones generally confer more strength to bones [27], the SSI Z-score is considered to be higher in the TS patients than in the controls during adolescence [23].
3. Fracture Risks in Children and Adolescents with Turner Syndrome
The larger CSA in TS indicates that bone strength is not impaired in children with TS; however, conflicting findings are available on this issue. Ross et al. examined the fracture incidence in 78 children with TS aged between 4 and 13 years old in 1991, and found that the annual incidence of fractures was similar in the TS patients and controls (19.9/1000 in TS vs. 27.8/1000 in controls), whereas the annual incidence of wrist fractures was higher in TS (9.1/1000 in TS vs. 3.5/1000 in controls) [28]. A recent longitudinal analysis of the fracture incidence in children with TS aged between 6 and 16 years demonstrated that the fracture rate of the limbs and spine was not higher in TS individuals [23]. The discrepancy between these two studies may be due to differences in the percentage of individuals receiving ERT. In the former study, 13 out of 78 subjects received ERT briefly for a mean duration of 6 months, whereas ERT was initiated in a defined protocol in the latter study, indicating that the former may have included more individuals for whom ERT was insufficiently performed; however, this difference may not have been a major factor that created a distinct result between two studies, because the age of the subjects in the former was younger than that in the later study, which may justify the increased percentage of subjects without ERT in the former study. Therefore, further accumulation of studies is clearly necessary to unravel whether the fracture risk is increased in children and adolescence with TS.
4. Effects of Growth Hormone Treatment on Bone Mass and Geometry in Children with Turner Syndrome
Growth hormone (GH) has been widely utilized to increase adult height in TS with successful outcomes [29][30][31][32][33]. In addition to its growth-promoting effects, GH has been shown to exert anabolic effects on the skeleton largely through its action to induce the expression of insulin-like growth factor 1, which was based on findings obtained from individuals with GH deficiency [34][35][36]. Additionally, a pharmacological dose of GH in short-statured children born with SGA increased the bone mass [37]. Although the effects of GH on BMD have been extensively examined in TS, conflicting findings have been obtained. Sas et al. investigated the effects of a 7-year treatment with GH on phalangeal vBMD and reported increases after adjustments for bone age [38]. In contrast, Ari et al. compared BMD between TS children treated with or without GH, and showed that after adjustments for bone size and bone age, the aBMD of LS and the radius did not significantly differ between the two groups [39]. Aycan et al. examined the effects of a 1-year GH treatment in prepubertal girls with TS with a mean age (SD) of 9.8 (2.5) years, and showed that GH treatment did not affect the vBMD of LS [40]. Similar findings were obtained on the effects of GH on BMD during childhood and adolescence [41][42][43]. These findings indicate that GH treatment in TS girls did not have a beneficial impact on BMD. However, as discussed earlier, bone strength is not affected by BMD only, bone geometry also plays important roles. Accordingly, Nour et al. recently investigated bone geometry using high-resolution pQCT and found that GH increased the bone size and the mechanical index of the polar moment of inertia, while DXA-based BMD was unaffected [44], which implies the beneficial effects of GH on bone strength, potentially resulting in reductions in the risk of fracture. Similar findings were reported on the effects of GH on decreases in the risk of fracture in age-related osteoporosis [45]. In addition to its effects on bone geometry, GH indirectly affected bone strength by improving muscle strength. Despite these benefits of GH on bone geometry and strength, limited information is currently available and, thus, further studies are needed to elucidate the effects of GH on fracture risk in adults with TS.
5. Effects of Estrogen Replacement Therapy during Adolescence on Bone Mineral Density in Young Adults with Turner Syndrome
aBMD has been extensively examined in adults with TS [3][4][11][16][46]. Itonaga et al. analyzed LS aBMD in TS patients aged between 15 and 49 years old and found that it was lower than the age-matched reference values [11]. Han et al. investigated aBMD in 177 subjects with TS aged between 19 and 60 years, and found that 55 and 9% of subjects showed T-scores for LS aBMD that were lower than −1.0 and −2.5, respectively [4]. Similarly, Freriks et al. recently evaluated BMD in adult women with TS (N = 150) with a mean age (SD) of 31.0 (10.4) years old, and showed that 52 and 12% of subjects had osteopenia with T-scores between −1.0 and −2.5 and lower than −2.5, respectively [3].
These findings indicate that aBMD is decreased in adults with TS, which is partly attributable to insufficient ERT, as evidenced in multiple observational studies and meta-analyses [7][8]. However, limited information is currently available on the effects of ERT during adolescence, particularly the age at which to initiate ERT, on BMD during adulthood. Cameron-Pimblett et al. analyzed the effects of the starting age of ERT on LS aBMD in adults with TS ranging between 18.1 and 70.3 years, and found a negative association between the starting age of ERT and LS aBMD [9]. Nguyen et al. also investigated the relationship between ERT and the spinal trabecular bone score (TBS) [13], an indirect measurement of the spinal trabecular architecture calculated based on DXA images that have been negatively associated with major fracture risks [47], and showed that TBS in TS patients aged between 20 and 49 years old was adversely influenced by a delay in the initiation of ERT [13]. Similar findings were obtained in other studies [12][13][14][48].
Therefore, these findings clearly indicate the importance of the early introduction of ERT in TS, where appropriate; however, the inclusion of aged individuals in the study design may create a bias when interpreting the effects of ERT on BMD because multiple variables, including medication for osteoporosis and age-dependent decreases in bone mass, may have affected BMD in aged populations. To avoid these limitations, the influence of the timing of ERT on peak bone mass needs to be examined. This strategy is also supported by the peak bone mass attained during young adulthood being an important factor affecting the risk of osteoporotic fractures later in life [49][50][51]. In the general population, LS aBMD has been shown to peak in women in their twenties and thirties [52]; however, age-dependent alterations in aBMD have not been examined in detail in TS. Based on these findings, researchers initially investigated age-dependent alterations in LS aBMD in adults with TS and found that aBMD peaked at between 30 and 34 years of age in TS, and thereafter decreased, similar to the reference population [11].
These results imply the importance of investigating aBMD during ages between 30 and 34 years of age in TS patients; however, because of the paucity of data during this period, researchers alternatively investigated the relationship between the starting age of ERT and aBMD in young adults aged between 18 and 30 years old, during which the peak bone mass of LS appeared to be acquired in TS [10]. Researchers also included individuals who had never been treated with anti-osteoporotic drugs, including the active form of vitamin D and bisphosphonates. Using these populations, researchers initially found that individuals with spontaneous menarche had higher LS aBMD than those without, suggesting that estrogen sufficiency during the pubertal period is important to accrue better LS aBMD during young adulthood. In addition, researchers revealed that the starting age of the adult dose of estrogen was negatively associated with LS aBMD. Although the starting age of ERT was not associated with low LS aBMD when the duration between the ages at which ERT and the adult dose of estrogen were initiated was incorporated into the study design as a confounding variable, the starting age of ERT was negatively correlated with LS aBMD, indicating that the early initiation and appropriate dose escalation of estrogen are important for the improved accrual of LS aBMD in young adulthood for patients with TS.
References
- Gravholt, C.H.; Viuff, M.H.; Brun, S.; Stochholm, K.; Andersen, N.H. Turner syndrome: Mechanisms and management. Nat. Rev. Endocrinol. 2019, 15, 601–614.
- Gravholt, C.H.; Vestergaard, P.; Hermann, A.P.; Mosekilde, L.; Brixen, K.; Christiansen, J.S. Increased fracture rates in Turner’s syndrome: A nationwide questionnaire survey. Clin. Endocrinol. 2003, 59, 89–96.
- Freriks, K.; Timmermans, J.; Beerendonk, C.C.; Verhaak, C.M.; Netea-Maier, R.T.; Otten, B.J.; Braat, D.D.; Smeets, D.F.; Kunst, D.H.; Hermus, A.R.; et al. Standardized multidisciplinary evaluation yields significant previously undiagnosed morbidity in adult women with Turner syndrome. J. Clin. Endocrinol. Metab. 2011, 96, E1517–E1526.
- Han, T.S.; Cadge, B.; Conway, G.S. Hearing impairment and low bone mineral density increase the risk of bone fractures in women with Turner’s syndrome. Clin. Endocrinol. 2006, 65, 643–647.
- Gravholt, C.H.; Juul, S.; Naeraa, R.W.; Hansen, J. Morbidity in Turner syndrome. J. Clin. Epidemiol. 1998, 51, 147–158.
- Cintron, D.; Rodriguez-Gutierrez, R.; Serrano, V.; Latortue-Albino, P.; Erwin, P.J.; Murad, M.H. Effect of estrogen replacement therapy on bone and cardiovascular outcomes in women with turner syndrome: A systematic review and meta-analysis. Endocrine 2017, 55, 366–375.
- Landin-Wilhelmsen, K.; Bryman, I.; Windh, M.; Wilhelmsen, L. Osteoporosis and fractures in Turner syndrome-importance of growth promoting and oestrogen therapy. Clin. Endocrinol. 1999, 51, 497–502.
- Klein, K.O.; Rosenfield, R.L.; Santen, R.J.; Gawlik, A.M.; Backeljauw, P.F.; Gravholt, C.H.; Sas, T.C.J.; Mauras, N. Estrogen Replacement in Turner Syndrome: Literature Review and Practical Considerations. J. Clin. Endocrinol. Metab. 2018, 103, 1790–1803.
- Cameron-Pimblett, A.; Davies, M.C.; Burt, E.; Talaulikar, V.S.; La Rosa, C.; King, T.F.J.; Conway, G.S. Effects of Estrogen Therapies on Outcomes in Turner Syndrome: Assessment of Induction of Puberty and Adult Estrogen Use. J. Clin. Endocrinol. Metab. 2019, 104, 2820–2826.
- Nishigaki, S.; Itonaga, T.; Hasegawa, Y.; Kawai, M. Starting age of oestrogen-progestin therapy is negatively associated with bone mineral density in young adults with Turner syndrome independent of age and body mass index. Clin. Endocrinol. 2021, 95, 84–91.
- Itonaga, T.; Koga, E.; Nishigaki, S.; Kawai, M.; Sakakibara, H.; Hasegawa, Y. A retrospective multicenter study of bone mineral density in adolescents and adults with Turner syndrome in Japan. Endocr. J. 2020, 67, 1023–1028.
- Kodama, M.; Komura, H.; Kodama, T.; Nishio, Y.; Kimura, T. Estrogen therapy initiated at an early age increases bone mineral density in Turner syndrome patients. Endocr. J. 2012, 59, 153–159.
- Nguyen, H.H.; Wong, P.; Strauss, B.J.; Ebeling, P.R.; Milat, F.; Vincent, A. A Cross-Sectional and Longitudinal Analysis of Trabecular Bone Score in Adults With Turner Syndrome. J. Clin. Endocrinol. Metab. 2018, 103, 3792–3800.
- Nguyen, H.H.; Wong, P.; Strauss, B.J.; Jones, G.; Ebeling, P.R.; Milat, F.; Vincent, A. Delay in estrogen commencement is associated with lower bone mineral density in Turner syndrome. Climacteric 2017, 20, 436–441.
- Gravholt, C.H.; Andersen, N.H.; Conway, G.S.; Dekkers, O.M.; Geffner, M.E.; Klein, K.O.; Lin, A.E.; Mauras, N.; Quigley, C.A.; Rubin, K.; et al. Clinical practice guidelines for the care of girls and women with Turner syndrome: Proceedings from the 2016 Cincinnati International Turner Syndrome Meeting. Eur. J. Endocrinol. 2017, 177, G1–G70.
- Augoulea, A.; Zachou, G.; Lambrinoudaki, I. Turner syndrome and osteoporosis. Maturitas 2019, 130, 41–49.
- Soucek, O.; Zapletalova, J.; Zemkova, D.; Snajderova, M.; Novotna, D.; Hirschfeldova, K.; Plasilova, I.; Kolouskova, S.; Rocek, M.; Hlavka, Z.; et al. Prepubertal girls with Turner syndrome and children with isolated SHOX deficiency have similar bone geometry at the radius. J. Clin. Endocrinol. Metab. 2013, 98, E1241–E1247.
- Rajpathak, S.N.; Vellarikkal, S.K.; Patowary, A.; Scaria, V.; Sivasubbu, S.; Deobagkar, D.D. Human 45,X fibroblast transcriptome reveals distinct differentially expressed genes including long noncoding RNAs potentially associated with the pathophysiology of Turner syndrome. PLoS ONE 2014, 9, e100076.
- Rauch, F.; Tutlewski, B.; Schonau, E. The bone behind a low areal bone mineral density: Peripheral quantitative computed tomographic analysis in a woman with osteogenesis imperfecta. J. Musculoskelet. Neuronal Interact. 2002, 2, 306–308.
- Seeman, E. Clinical review 137: Sexual dimorphism in skeletal size, density, and strength. J. Clin. Endocrinol. Metab. 2001, 86, 4576–4584.
- Pitukcheewanont, P.; Numbenjapon, N.; Safani, D.; Rossmiller, S.; Gilsanz, V.; Costin, G. Bone size and density measurements in prepubertal children with Turner syndrome prior to growth hormone therapy. Osteoporos. Int. 2011, 22, 1709–1715.
- Soucek, O.; Lebl, J.; Snajderova, M.; Kolouskova, S.; Rocek, M.; Hlavka, Z.; Cinek, O.; Rittweger, J.; Sumnik, Z. Bone geometry and volumetric bone mineral density in girls with Turner syndrome of different pubertal stages. Clin. Endocrinol. 2011, 74, 445–452.
- Soucek, O.; Schonau, E.; Lebl, J.; Willnecker, J.; Hlavka, Z.; Sumnik, Z. A 6-Year Follow-Up of Fracture Incidence and Volumetric Bone Mineral Density Development in Girls With Turner Syndrome. J. Clin. Endocrinol. Metab. 2018, 103, 1188–1197.
- Zhang, X.Z.; Kalu, D.N.; Erbas, B.; Hopper, J.L.; Seeman, E. The effects of gonadectomy on bone size, mass, and volumetric density in growing rats are gender-, site-, and growth hormone-specific. J. Bone Miner. Res. 1999, 14, 802–809.
- Yeh, J.K.; Chen, M.M.; Aloia, J.F. Ovariectomy-induced high turnover in cortical bone is dependent on pituitary hormone in rats. Bone 1996, 18, 443–450.
- Turner, R.T.; Hannon, K.S.; Demers, L.M.; Buchanan, J.; Bell, N.H. Differential effects of gonadal function on bone histomorphometry in male and female rats. J. Bone Miner. Res. 1989, 4, 557–563.
- Turner, C.H.; Burr, D.B. Basic biomechanical measurements of bone: A tutorial. Bone 1993, 14, 595–608.
- Ross, J.L.; Long, L.M.; Feuillan, P.; Cassorla, F.; Cutler, G.B., Jr. Normal bone density of the wrist and spine and increased wrist fractures in girls with Turner’s syndrome. J. Clin. Endocrinol. Metab. 1991, 73, 355–359.
- Dantas, N.C.B.; Braz, A.F.; Malaquias, A.; Lemos-Marini, S.; Arnhold, I.J.P.; Silveira, E.R.; Antonini, S.R.; Guerra-Junior, G.; Mendonca, B.; Jorge, A.; et al. Adult Height in 299 Patients with Turner Syndrome with or without Growth Hormone Therapy: Results and Literature Review. Horm. Res. Paediatr. 2021, 94, 63–70.
- Ahn, J.M.; Suh, J.H.; Kwon, A.R.; Chae, H.W.; Kim, H.S. Final Adult Height after Growth Hormone Treatment in Patients with Turner Syndrome. Horm. Res. Paediatr. 2019, 91, 373–379.
- Lanes, R.; Lindberg, A.; Carlsson, M.; Chrysis, D.; Aydin, F.; Camacho-Hubner, C.; King, D.; Davenport, M. Near Adult Height in Girls with Turner Syndrome Treated with Growth Hormone Following Either Induced or Spontaneous Puberty. J. Pediatr. 2019, 212, 172–179.e1.
- Cleemann Wang, A.; Hagen, C.P.; Nedaeifard, L.; Juul, A.; Jensen, R.B. Growth and Adult Height in Girls With Turner Syndrome Following IGF-1 Titrated Growth Hormone Treatment. J. Clin. Endocrinol. Metab. 2020, 105, 2566–2574.
- Fujita, K.; Yokoya, S.; Fujieda, K.; Shimatsu, A.; Tachibana, K.; Tanaka, H.; Tanizawa, T.; Teramoto, A.; Nishi, Y.; Hasegawa, Y.; et al. Adult heights of 258 girls with turner syndrome on low dose of growth hormone therapy in Japan. Clin. Pediatr. Endocrinol. 2010, 19, 63–68.
- Yang, H.; Yan, K.; Yuping, X.; Zhang, Q.; Wang, L.; Gong, F.; Zhu, H.; Xia, W.; Pan, H. Bone microarchitecture and volumetric bone density impairment in young male adults with childhood-onset growth hormone deficiency. Eur. J. Endocrinol. 2019, 180, 145–153.
- Tritos, N.A.; Johannsson, G.; Korbonits, M.; Miller, K.K.; Feldt-Rasmussen, U.; Yuen, K.C.; King, D.; Mattsson, A.F.; Jonsson, P.J.; Koltowska-Haggstrom, M.; et al. Effects of long-term growth hormone replacement in adults with growth hormone deficiency following cure of acromegaly: A KIMS analysis. J. Clin. Endocrinol. Metab. 2014, 99, 2018–2029.
- Barake, M.; Klibanski, A.; Tritos, N.A. Effects of recombinant human growth hormone therapy on bone mineral density in adults with growth hormone deficiency: A meta-analysis. J. Clin. Endocrinol. Metab. 2014, 99, 852–860.
- Willemsen, R.H.; Arends, N.J.; Bakker-van Waarde, W.M.; Jansen, M.; van Mil, E.G.; Mulder, J.; Odink, R.J.; Reeser, M.; Rongen-Westerlaken, C.; Stokvis-Brantsma, W.H.; et al. Long-term effects of growth hormone (GH) treatment on body composition and bone mineral density in short children born small-for-gestational-age: Six-year follow-up of a randomized controlled GH trial. Clin. Endocrinol. 2007, 67, 485–492.
- Sas, T.C.; de Muinck Keizer-Schrama, S.M.; Stijnen, T.; van Teunenbroek, A.; van Leeuwen, W.J.; Asarfi, A.; van Rijn, R.R.; Drop, S.L.; Dutch Advisory Group on Growth Hormone. Bone mineral density assessed by phalangeal radiographic absorptiometry before and during long-term growth hormone treatment in girls with Turner’s syndrome participating in a randomized dose-response study. Pediatr. Res. 2001, 50, 417–422.
- Ari, M.; Bakalov, V.K.; Hill, S.; Bondy, C.A. The effects of growth hormone treatment on bone mineral density and body composition in girls with turner syndrome. J. Clin. Endocrinol. Metab. 2006, 91, 4302–4305.
- Aycan, Z.; Cetinkaya, E.; Darendeliler, F.; Vidinlisan, S.; Bas, F.; Bideci, A.; Demirel, F.; Darcan, S.; Buyukgebiz, A.; Yildiz, M.; et al. The effect of growth hormone treatment on bone mineral density in prepubertal girls with Turner syndrome: A multicentre prospective clinical trial. Clin. Endocrinol. 2008, 68, 769–772.
- Carrascosa, A.; Gussinye, M.; Terradas, P.; Yeste, D.; Audi, L.; Vicens-Calvet, E. Spontaneous, but not induced, puberty permits adequate bone mass acquisition in adolescent Turner syndrome patients. J. Bone Miner. Res. 2000, 15, 2005–2010.
- Bakalov, V.K.; Van, P.L.; Baron, J.; Reynolds, J.C.; Bondy, C.A. Growth hormone therapy and bone mineral density in Turner syndrome. J. Clin. Endocrinol. Metab. 2004, 89, 4886–4889.
- Bertelloni, S.; Cinquanta, L.; Baroncelli, G.I.; Simi, P.; Rossi, S.; Saggese, G. Volumetric bone mineral density in young women with Turner’s syndrome treated with estrogens or estrogens plus growth hormone. Horm. Res. 2000, 53, 72–76.
- Nour, M.A.; Burt, L.A.; Perry, R.J.; Stephure, D.K.; Hanley, D.A.; Boyd, S.K. Impact of Growth Hormone on Adult Bone Quality in Turner Syndrome: A HR-pQCT Study. Calcif. Tissue Int. 2016, 98, 49–59.
- Barake, M.; Arabi, A.; Nakhoul, N.; El-Hajj Fuleihan, G.; El Ghandour, S.; Klibanski, A.; Tritos, N.A. Effects of growth hormone therapy on bone density and fracture risk in age-related osteoporosis in the absence of growth hormone deficiency: A systematic review and meta-analysis. Endocrine 2018, 59, 39–49.
- Benetti-Pinto, C.L.; Bedone, A.; Magna, L.A.; Marques-Neto, J.F. Factors associated with the reduction of bone density in patients with gonadal dysgenesis. Fertil. Steril. 2002, 77, 571–575.
- Silva, B.C.; Broy, S.B.; Boutroy, S.; Schousboe, J.T.; Shepherd, J.A.; Leslie, W.D. Fracture Risk Prediction by Non-BMD DXA Measures: The 2015 ISCD Official Positions Part 2: Trabecular Bone Score. J. Clin. Densitom. 2015, 18, 309–330.
- Nakamura, T.; Tsuburai, T.; Tokinaga, A.; Nakajima, I.; Kitayama, R.; Imai, Y.; Nagata, T.; Yoshida, H.; Hirahara, F.; Sakakibara, H. Efficacy of estrogen replacement therapy (ERT) on uterine growth and acquisition of bone mass in patients with Turner syndrome. Endocr. J. 2015, 62, 965–970.
- Hernandez, C.J.; Beaupre, G.S.; Carter, D.R. A theoretical analysis of the relative influences of peak BMD, age-related bone loss and menopause on the development of osteoporosis. Osteoporos. Int. 2003, 14, 843–847.
- Rizzoli, R.; Bianchi, M.L.; Garabedian, M.; McKay, H.A.; Moreno, L.A. Maximizing bone mineral mass gain during growth for the prevention of fractures in the adolescents and the elderly. Bone 2010, 46, 294–305.
- Weaver, C.M.; Gordon, C.M.; Janz, K.F.; Kalkwarf, H.J.; Lappe, J.M.; Lewis, R.; O’Karma, M.; Wallace, T.C.; Zemel, B.S. The National Osteoporosis Foundation’s position statement on peak bone mass development and lifestyle factors: A systematic review and implementation recommendations. Osteoporos. Int. 2016, 27, 1281–1386.
- Berger, C.; Goltzman, D.; Langsetmo, L.; Joseph, L.; Jackson, S.; Kreiger, N.; Tenenhouse, A.; Davison, K.S.; Josse, R.G.; Prior, J.C.; et al. Peak bone mass from longitudinal data: Implications for the prevalence, pathophysiology, and diagnosis of osteoporosis. J. Bone Miner. Res. 2010, 25, 1948–1957.
More
Information
Subjects:
Others
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
876
Revisions:
4 times
(View History)
Update Date:
29 Aug 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





