Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Christian Bailly | -- | 2878 | 2022-08-25 11:14:06 | | | |
| 2 | Peter Tang | Meta information modification | 2878 | 2022-08-25 11:41:04 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Bailly, C. Pharmacological Properties of Alisols. Encyclopedia. Available online: https://encyclopedia.pub/entry/26490 (accessed on 06 June 2026).
Bailly C. Pharmacological Properties of Alisols. Encyclopedia. Available at: https://encyclopedia.pub/entry/26490. Accessed June 06, 2026.
Bailly, Christian. "Pharmacological Properties of Alisols" Encyclopedia, https://encyclopedia.pub/entry/26490 (accessed June 06, 2026).
Bailly, C. (2022, August 25). Pharmacological Properties of Alisols. In Encyclopedia. https://encyclopedia.pub/entry/26490
Bailly, Christian. "Pharmacological Properties of Alisols." Encyclopedia. Web. 25 August, 2022.
Copy Citation
More than 100 protostane triterpenoids have been isolated from the dried rhizomes of Alisma species, designated Alismatis rhizoma (AR), commonly used in Asian traditional medicine to treat inflammatory and vascular diseases. The main products are the alisols, with the lead compounds alisol-A/-B and their acetate derivatives being the most abundant products in the plant and the best-known bioactive products.
Alismatis rhizoma
alisol
cancer
inflammation
molecular targets
pharmacology
protostane triterpenoids
1. Alisols and Related Terpenoids
In the plant, protostanes are biosynthesized from squalene with the support of squalene epoxidases regulated by methyl jasmonate [1][2]. There are 22 alisol compounds, designated alisol A (Ali-A) to alisol V (Ali-V), and a multitude of related compounds bearing an acetate group at position 23 or 24 of the side chain appended to the tetracyclic protostane unit. There are also derivatives with a modified side chain at C-17, equipped with an open or epoxy aliphatic side chain, with a fused ring at C-16/C-17 or with a modified protostane unit (nor- or seco-protostane). The series includes numerous derivatives, such as the alismanols, alismanins, alisolides, and others. In 2020, Wang and co-workers had identified more than 115 protostane-type terpenoids from Alisma species [3]. Here, the researchers will essentially refer to Ali-A, -B, and -C, and occasionally to a few other compounds mentioned in Figure 1. Most of them preserve the C-3 carbonyl group of characteristics of Ali-A and a highly oxygenated C-17 side chain. New protostane terpenoids are regularly identified and isolated from AR, taking advantage of novel, highly efficient, and precise analytical methods. For example, the two new compounds, called Alisma A and Alisma B, have been recently discovered using a new analytical methodology called ‘force iteration molecular designing’. With this approach, the authors identified 473 protostane terpenoids from the two plant species of AR [4]. The different properties will be evocated in turn.
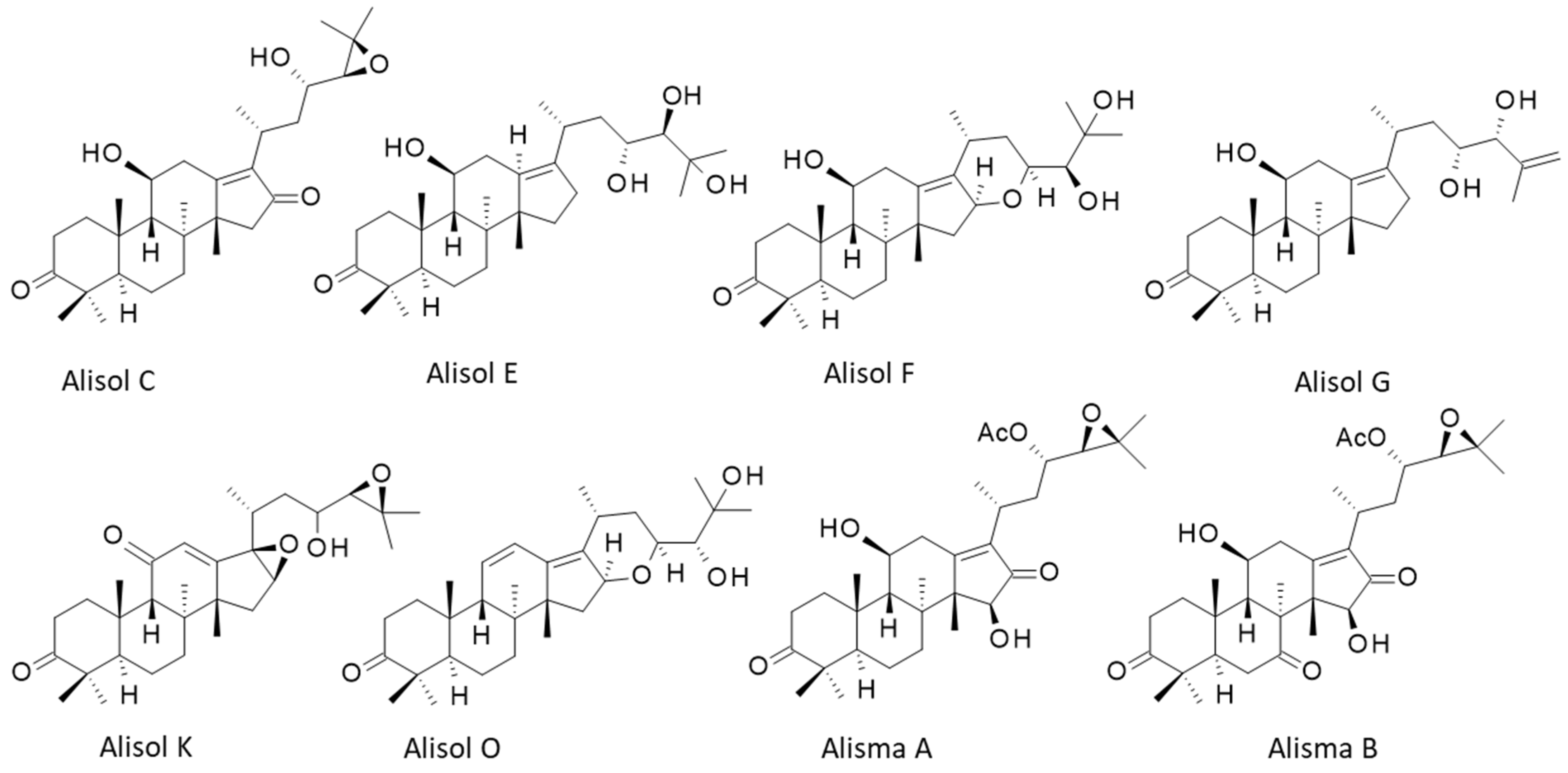
Figure 1. Structures of 8 selected alisol derivatives. The series includes 22 compounds (Ali-A to Ali-V) and 100 derivatives.
2. Pharmacological Properties of Alisols
2.1. Lipid-Lowering Effects and Hepatoprotection
Historically, in Asia, AR has been known for its diuretic action. It was traditionally used to treat diabetes and swelling. A search of AR extracts for compounds with a hypocholesterolemic action was initiated in the late 1960s, leading to the identification of active fractions and then pure compounds. Ali-A/-B/-C, along with their monoacetates, were discovered to be capable of reducing plasma and liver cholesterol levels in rats after oral administration [5]. The mode of action for these compounds has been investigated much more deeply now, leading to a better understanding of the pathways implicated in the capacity of Ali-A/-B to reduce lipid accumulation and lipotoxicity. A recent work demonstrated how Ali-B regulates hepatic gene expression via the RARα-HNF4α-PPARγ cascade in a murine model of non-alcoholic steatohepatitis (NASH). Specifically, Ali-B enhances RARα gene expression, thereby reducing expression of HNF4α and PPARγ, leading to a suppressed expression of translocase fatty acid CD36 [6]. The lipid-lowering components of AR includes Ali-B, Ali-B 23-acetate, and Ali-C 23-acetate, which are believed to bind to the farnesoid X receptor (FXR) protein and to behave as agonists, according to a molecular docking study [7]. The hypothesis is entirely plausible because Ali-B 23-acetate has been shown to increase the expression of FXR in cultured hepatocyte L02 cells and to activate hepatic BSEP (bile salt export pump) signaling [8]. The capacity of Ali-B 23-acetate to function as a FXR agonist has been demonstrated in a model of renal ischemia-reperfusion injury. The compound was found to activate renal FXR and to induce FXR downstream gene expression in mouse kidneys [9], and also in a model of non-alcoholic steatohepatitis (NASH) [10]. It is a potent agonist of several pregnane X receptors (PXRs), including FXR, but also LXR and PPARα/δ/γ [11]. This compound displays marked hepato-protective functions, due to FXR-mediated gene regulation [12][13][14][15]. The related compound, Ali-A 24-acetate, is also able to reduce liver lipid deposition in hyperlipidemic mice via a blockade of AMPK activation to promote glucose metabolism and by promoting expression of ATP binding cassette transporters ABCG1 and ABCA1 [16][17]. The compound stimulates lipolysis of adipocytes by activating protein kinase A (PKA)-mediated phosphorylation of hormone-sensitive lipase [18]. These observations make Ali-A 24-acetate and Ali-B 23-acetate good candidates for the treatment of nonalcoholic fatty liver disease and its comorbidities [19]. They are hepatoprotective agents that are also useful to prevent or treat hepatic damages induced by chemicals [15].
2.2. Treatment of Atherosclerosis
Anti-atherosclerotic actions have been reported with Ali-A, its 24-acetate derivative and Ali-B 23-acetate. For the latter compound, the cholesterol-lowering effect is associated with an increase in FXR-BSEP signaling which leads to a reduction in liver cholesterol, hepatic lipolysis, and bile acids efflux. These cumulated effects concur in reducing damages of atherosclerosis [8]. In addition, the compound exerts anti-inflammatory effects, notably an inhibition of the production of inflammatory cytokines IL-12 (interleukin-12) and IFN-γ (interferon-γ), also contributing to the anti-atherosclerotic effect [20]. The capacity of Ali-B 23-acetate to markedly reduce the atherosclerotic plaque area and lipid accumulation is most likely the resultant of a cumulative effect implicating FXR agonism, anti-inflammatory effects and regulation of cholesterol synthesis and export (Figure 2). Indeed, Ali-B 23-acetate negatively regulates Acyl-CoA cholesterol acyltransferase 2 (ACAT2) in cultured Caco-2 cells and increases expression of ATP-binding cassette transfer proteins G5/G8 (ABCG5/G8) [21]. Multiple receptors and pathways contribute to the capacity of Ali-B 23-acetate to reduce accumulation of triglycerides and cholesterol, including the estrogen receptor α (ERα) to which the compound may bind directly, according to a molecular docking analysis [22]. However, other receptors have been proposed also. For example, the related compounds 16-hydroxy-Ali B 23-acetate and Ali-M 23-acetate have been predicted to bind, with a high affinity, to the liver X receptor β (LXRβ) on the basis of molecular docking and molecular dynamic simulations and to exert an agonist effect [23].
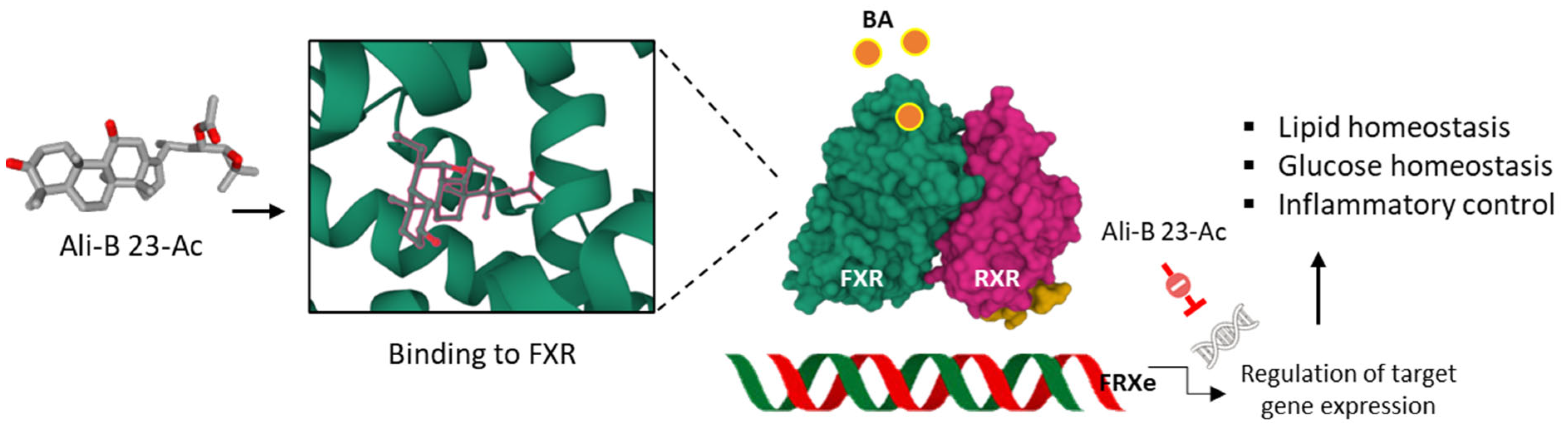
Figure 2. Anti-atherosclerosis activity of Ali-B 23-acetate. The compound binds to the FRX protein and displays an agonist activity which leads to the regulation of the expression of genes implicated in the control of inflammation, glucose, and lipid levels. Via this mechanism, Ali-B 23-acetate reduces atherosclerotic plaque.
2.3. Anti-Inflammatory Activity
Soluble epoxide hydrolase (sEH) is a therapeutic target considered for the treatment of inflammation-based diseases, and various pathologies, including cardiovascular diseases and neuropsychiatric disorders [24][25]. A methanol extract of AR was shown to attenuate the decrease in epoxide hydrolase induced in rats intoxicated with bromobenzene [26]. The extracts contained protostane triterpenoids acting as inhibitors of sEH, such as alismanin B, 11-deoxy-25-anhydro-Ali-E (Figure 3), and 11-deoxy-Ali-B with IC50 values of 7.15, 3.40, and 5.94 μM, respectively. The compounds were predicted to bind to the active site of she, acting as competitive inhibitors [27]. More recently, two other compounds in the series were described shesEH inhibitors: 3β-hydroxy-25-anhydro-Ali-F and 3β-hydroxy-Ali-G (2), with IC50 values of 10.06 and 30.45 μM, respectively [28]. In contrast to Ali-F, Ali-A and Ali-B have not been described as sEH inhibitors. Ali-F and 25-anhydro–Ali-F are interesting compounds because they both behave as inhibitors of lipopolysaccharide-induced NO production in macrophage RAW 264.7 macrophages [29] and regulate the production of inflammatory cytokines, such as TNF-α, IL-1β, and IL-6, via the MAPK/STAT3/NFκB pathway [30]. sEH contributes to multiple human pathologies, chiefly inflammatory diseases but also cancer. Compounds such as Ali-E and Ali-F could be further exploited to design sEH-targeted drugs for the treatment of advanced liver cancers and other liver diseases.
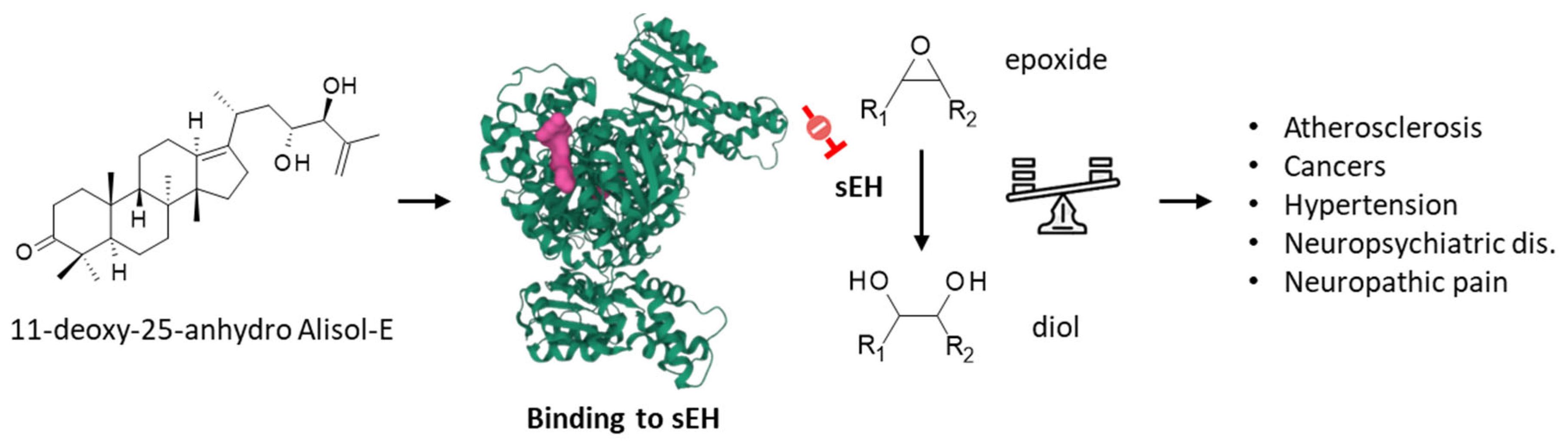
Figure 3. Binding of 11-deoxy-25-anhydro alisol-E to soluble epoxide hydrolase (sEH), the enzyme which converts epoxide molecules into diol molecules, as represented. The modulation of the epoxide/diol ratio is a mechanism by which the compound exerts activities against multiple pathologies, as indicated.
Ali-B 23-acetate displays anti-inflammatory effects, apparently not via sEH inhibition, but through inhibition of the TLR4-NOX1/ROS signaling pathway. It also reduces production and infiltration of the same inflammatory cytokines as 25-anhydro-Ali-F. It suppressed expression of TLR4 and NOX2 and reduced the production of ROS [31]. Via these signaling pathways, the compound provides an anti-inflammatory action and maintains the integrity of the intestinal barrier as well [32][33].
2.4. Protection against Fibrosis and Kidney Injuries
Renal fibrosis is an important component of chronic kidney disease (CKD), which affects more than 10% of the world’s population [34]. Ali-B 23-acetate can attenuate the progression of CKD, at least in a rat model. An oral treatment with the compound (5–10 mg/kg) has been shown to inhibit expression of mRNA and proteins implicated in CKD pathogenesis, such as collagen I, fibronectin, vimentin, α-smooth muscle actin, and fibroblast-specific protein-1. Interestingly, the compound protected against renal fibrosis in part by re-establishing dysbiosis of the gut microbiome and regulating blood pressure (reducing hypertension). It did so by repressing the Wnt/β-catenin signaling pathway implicated in the activation and proliferation of renal fibroblasts [35]. The renin–angiotensin system and the Wnt/β-catenin axis play a role in kidney inflammation and fibrosis, and thus contribute to CKD. A treatment with Ali-B 23-acetate could reduce disease progression [36]. The activity is not specific to Ali-B 23-acetate, as the related product Ali-F 25-O-methyl has been shown also to attenuate tubulo-interstitial fibrosis, even if a distinct biochemical pathway (inhibition of TGF-β-mediated Smad3 phosphorylation) was invoked in that case [37].
The capacity of Ali-B 23-acetate to regulate the gut microbiome is beneficial not only for kidneys, but also for the gastro-intestinal tract and for immune regulation in general. The gut microbiota plays a role in metabolic diseases, cancers, and other pathologies. The probiotic regulation with Ali-B 23-acetate can reduce organ fibrosis (such as hepatic and renal fibrosis) but also colitis (associated with colon cancer) [32][38][39]. Ali-B 23-acetate and the other protostane triterpenoids of AR behave as regulators of the gut microecology. They can equilibrate the gut microflora, notably in cases of metabolic diseases and atherosclerosis [40][41].
2.5. Antioxidant Activity and Neuro-Protection
In the alisol series, the derivative Ali-A 24-acetate is an interesting cell-protective compound that is useful to limit brain damages. Both Ali-A and -B can cross the blood–brain barrier. Their acetate derivatives can also distribute in the brain tissue, although they preferentially accumulate in the intestine, stomach, liver, and kidneys, after oral administration [42]. Nevertheless, Ali-A 24-acetate has been shown to protect brain microvascular endothelial cells (BMEC) from damages induced by oxygen-glucose deprivation [43]. It exerts anti-apoptotic effects in this cell system [44] and also reduces apoptosis of neurons, via up-regulation of the expression of phosphorylated PI3K and AKT [45]. The blockade of thee PI3K/Akt/mTOR signaling pathway is important to the mechanism of action of AR and specifically to Ali-A 24-acetate, not only providing a protection against neuronal damages but also essential to the anticancer action of the extract and the product, as discussed thereafter [46].
2.6. Bone Preservation and Prevention of Osteoporosis
The acetate derivatives of Ali-A and Ali-B have been shown to protect against a decrease in bone mass. As a matter of fact, AR and different TCM preparations containing Alisma species (A. orientale, A. plantago-aquatica, A. canaliculatum) are used to regulate bone resorption, such as Yukmi-jihang-tang-Jahage [47] and Liuwei Dihuang [48]. Both Ali-A 24-acetate and Ali-C 23-acetate display anti-osteoporotic effects [49]. The latter compound is a potent inhibitor of osteoclastogenesis, acting via a blockade of RANKL-induced osteoclast differentiation and function (Figure 4) [50].
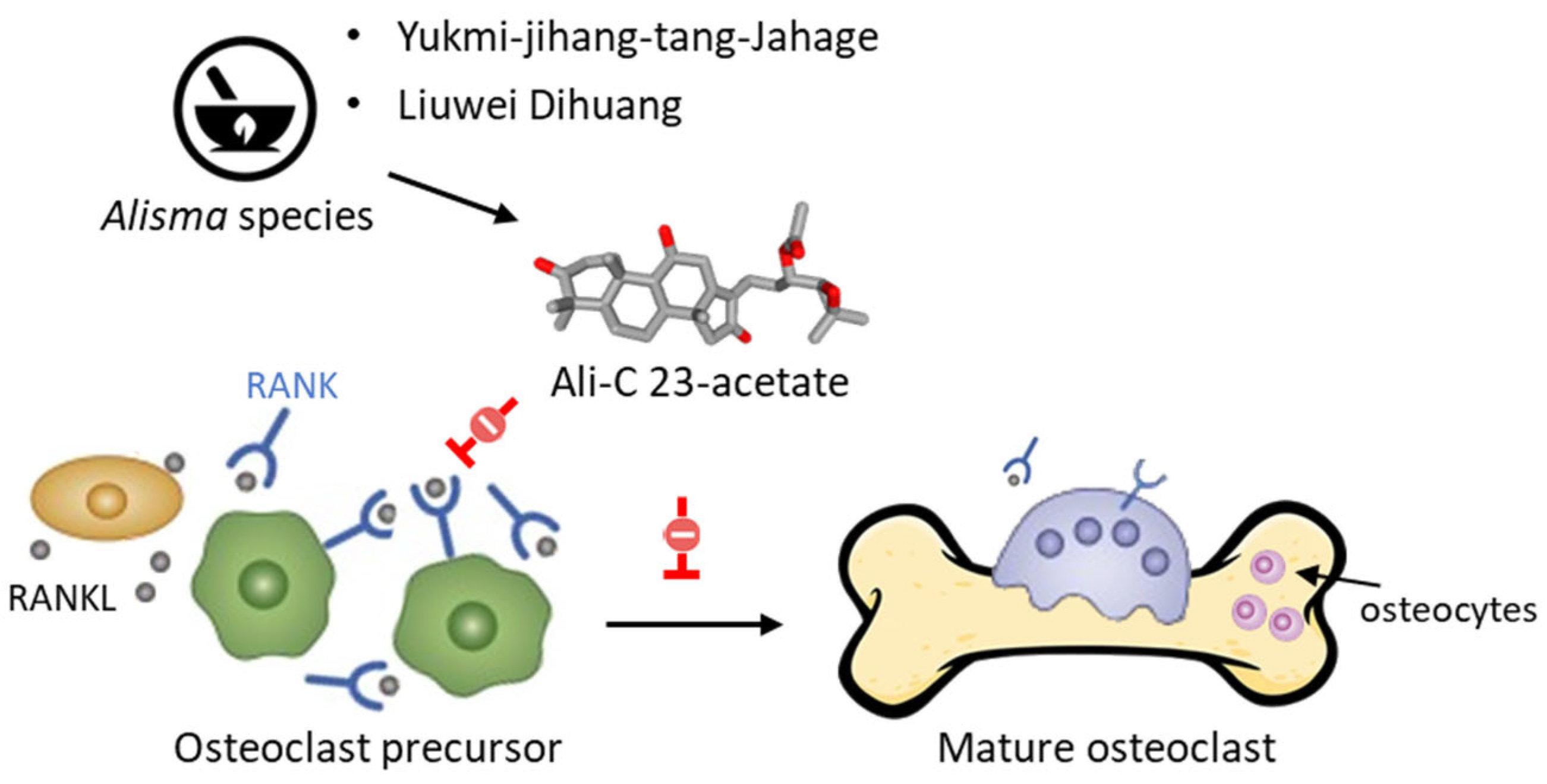
Figure 4. Inhibition of RANKL-induced osteoclast differentiation and function by Ali-C 23-acetate (AC23A). The compound would act selectively on osteoblast precursors, rather than mature osteoblasts, by inhibiting RANKL-induced osteoclastogenesis. The compound blocks phosphorylation of JNK and reduces expression of osteoclastogenic mediators, such as TRAP, c-Fos, MMP9, NFATc1, and cathepsin K [50]. AC23A can be found in phytomedicines made from Alisma rhizoma, such as Liuwei Dihuang and Yukmi-jihang-tang-Jahage, both used to treat osteoporosis.
Ali-A 24-acetate also functions as an inhibitor of osteoclastogenesis. It inhibits RANKL-mediated osteoclast differentiation by downregulating the expression of nuclear factor of activated T cell 1 (NFATc1), which is an important regulator of osteolysis [51]. In an animal model of postmenopausal osteoporosis (ovariectomized mice), a daily administration of Ali-A 24-acetate was shown to prevent bone loss, inhibiting activity of the bone resorption marker TRAP and restoring the proportion of Treg and Th17 immune cells [52]. RANKL-mediated effects have been also reported with Ali-B, e.g., inhibition of osteoclast formation and suppression of hypercalcemia [53].
2.7. Anticancer Activity
There is ample experimental evidence to support the activity of alisol derivatives against cancer cell proliferation and tumor progression. The main compounds Ali-A/-B and their acetate derivatives have been used in different tumor models, the other alisol derivatives have been used less frequently. However, Ali-F 24-acetate was shown to reverse multidrug resistance, thereby promoting nuclear accumulation of the cytotoxic drug doxorubicin and apoptosis of MCF-7 breast cancer cells [54]. A comparable reversal of P-glycoprotein-mediated multidrug resistance was evidenced with Ali-B 23-acetate [55]. There are not many studies comparing the anticancer efficacy of the different products, and the cell models used vary significantly from one study to another. Ali-A has revealed a marked capacity to reduce proliferation and invasion/migration of breast cancer cells [56][57], whereas Ali-B has shown a potent activity against hepatocellular carcinoma cells [58][59] and other models. It is not easy to compare the potency of the products. However, there are common characteristics between the compounds, such as their capacity to induce G0/G1 cell cycle arrest and to trigger apoptosis via the mitochondrial pathway.
The PI3K/Akt/mTOR pathway is central to the mechanism of action of Ali-A/-B [46] but it is not the sole pathway implicated in the anti-proliferative and antimetastatic action of the natural products. A recent study pointed out the major implication of the Wnt/β-catenin axis in the mechanism of action of Ali-B 23-acetate in liver cancer [60]. The compound functioned as an inhibitor in SMMC-7721 and MHCC97 cancer cells to promote the anticancer action of the cardiac glycoside bufalin (an active ingredient of the traditional Chinese medicine Chansu) [60]. The same pathway has been invoked to explain the anti-fibrotic action of the product [36]. The complexity of the signaling pathways involved in the mechanism of action of natural products is not surprising, it is frequently the case with terpenoids [61].
At the molecular level, several protein targets have been evoked to account for the anticancer activity of Ali-A/-B, essentially on the basis of molecular modeling studies, such as the transcriptional coactivator YAP which could interact with Ali-A [62], the sarcoplasmic/endoplasmic reticulum Ca2+ ATPase which can bind Ali-B [63], the soluble epoxide hydrolase [27], the farnesoid X receptor protein [7], human liver carboxylesterase 1 (HCE-2) [64], and hormone receptors [65] (Figure 5). Occasionally, non-protein targets have been mentioned as well, such as DNA. Both Ali-A and Ali-B have been docked onto DNA and shown to compete with DNA intercalation of the fluorescent dye ethidium bromide [66]. The c-myc DNA sequence has been considered as a potential target for Ali-A/-B acetates and the compounds’ mixture [67]. The hypothesis has not been validated experimentally at present. These terpenoids are multi-targeted products.
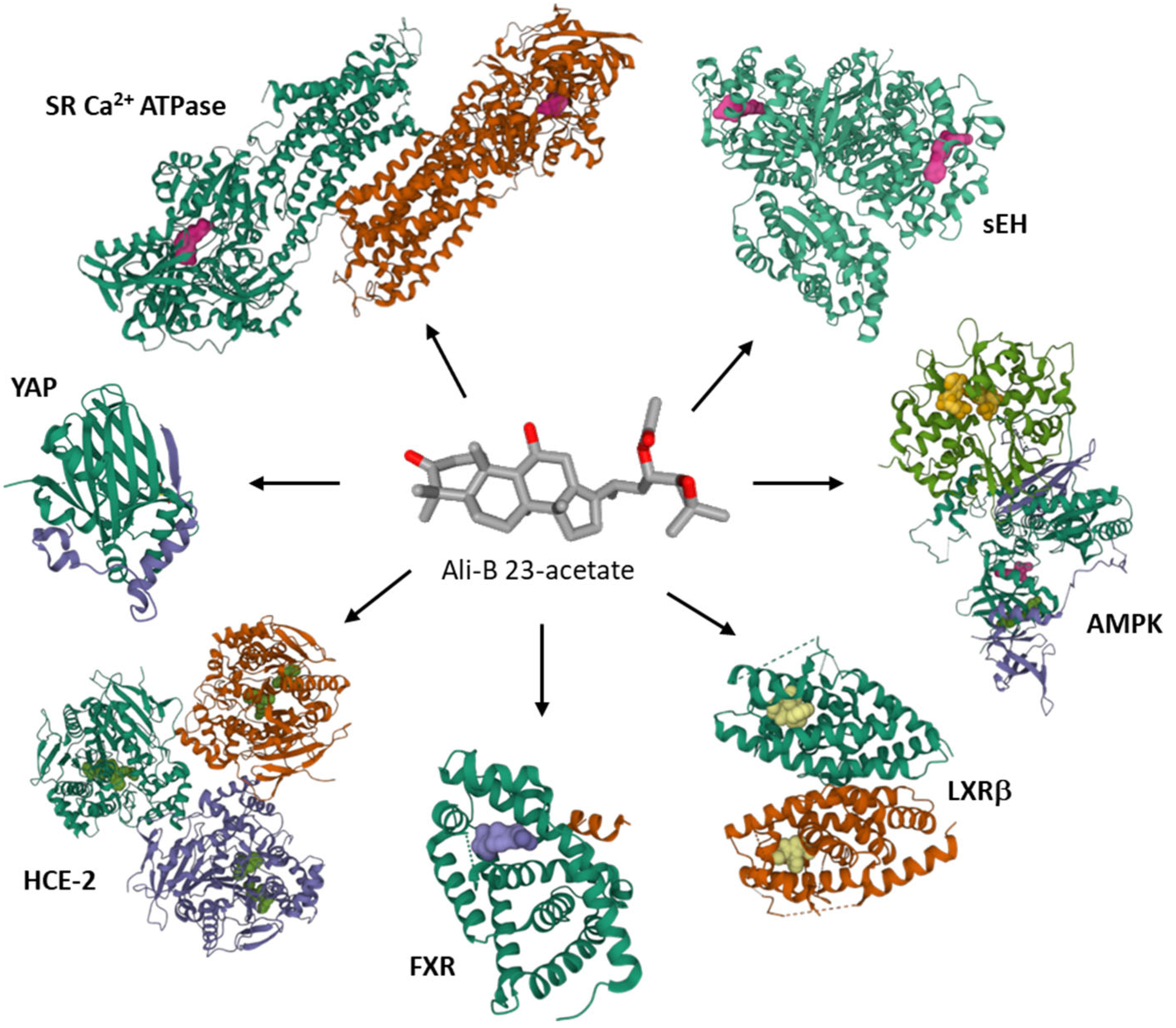
Figure 5. Proposed protein targets for Ali-B 23-acetate. Molecular modeling studies have evaluated binding of the compound to soluble epoxide hydrolase (sEH) [27][28], 5’-adenosine monophosphate-activated protein kinase (AMPK) [68], liver X receptor β (LXRβ) [23], farnesoid X receptor (FXR) [7], human liver carboxylesterase 1 (HCE-2) [64], Yes-associated protein (YAP) [62], sarcoplasmic/endoplasmic reticulum Ca2+ ATPase (SR Ca2+-ATPase) [63]. The protein models shown correspond to the PDB structures 4HAI for sEH, 7MYJ for AMPK, 6K9H for LXRβ, 6HL1 for FXR, YA4 for HCE-1, 3KYS for YAP, 1VFP for SR Ca2+-ATPase.
There are many published in vitro studies with Ali-A/-B using different cell lines, but relatively little evidence of antitumor activity in vivo. A study reported the capacity of Ali-B 23-acetate to reduce growth of a subcutaneously implanted hepatocellular carcinoma tumor in mice, but it was a tumorigenesis experiments with tumor cells treated with the compound (at 30 μM for 24 h) prior to grafting the tumor cells to mice. The compound delayed the growth of the tumor in mice [69]. It is not an anticancer treatment sensu stricto. There is a lack of robust evidence of an antitumor action of alisols in xenograft models.
2.8. Anti-Allergic Activity
In traditional medicine, the dried rhizomes of Alisma orientale are used to treat allergic reactions. The capacity of AR extracts to inhibit antibody-mediated allergic reactions has been recognized more than 25 years ago and, at that time, the anti-allergic contribution of compounds such as Ali-A and Ali-B was already mentioned [70]. Ali-B 23-acetate has been characterized as a major inhibitor of 5-lipoxygenase-catalyzed leukotriene C4 production in rat basophilic leukemia RBL-1 cells [71]. This compound reduces IgE/antigen-induced degranulation of mast cells and ameliorates allergic reaction [72].
2.9. Antibacterial and Antiviral Effects
Ali-A/-B are not highly potent antibacterial agents per se. They showed little, if any, antimicrobial effects against pathogenic bacteria, such as Bacillus subtilis, Escherichia coli, and Staphylococcus aureus. Only minor effects have been reported with the derivatives such as Ali-G and 25-anhydro-Ali-F [29]. Both Ali-A 24-acetate and Ali-B 23-acetate have been found to be active against antibiotic-resistant strains of S. aureus and E. coli [73] but their antibacterial effects have not been further investigated. However, Ali-B 23-acetate is a useful regulator of gut microbiota, capable of reducing the expansion of pathogenic bacteria (such as Klebsiella, and Citrobacter) and increasing growth of beneficial bacteria (such as Bacteroides, and Lactobacillus) [39].
Similarly, Ali-A/-B are not highly effective antiviral compounds, although there are occasional reports of alisol derivatives being applied effectively against specific viruses, notably such as limiting the secretion of hepatitis B virus (HBV) surface antigen with a tri-acetate derivative of Ali-A and some derivatives [74][75]. Based on this observation, acylated derivatives of Ali-A have been designed, leading to the identification of tetra-acetyl Ali-A as an orally bioavailable derivative with satisfactory pharmacokinetic properties, such as being active against HBV in cultured HepG 2.2.15 cells [76]. However, no further development has been reported for this derivative.
References
- Rong, T.; Chunchun, Z.; Wei, G.; Yuchen, G.; Fei, X.; Tao, L.; Yuanyuan, J.; Chenbin, W.; Wenda, X.; Wenqing, W. Proteomic insights into protostane triterpene biosynthesis regulatory mechanism after MeJA treatment in Alisma orientale (Sam.) Juz. Biochim. Biophys. Acta Proteins Proteom. 2021, 1869, 140671.
- Tian, R.; Gu, W.; Gu, Y.; Geng, C.; Xu, F.; Wu, Q.; Chao, J.; Xue, W.; Zhou, C.; Wang, F. Methyl jasmonate promote protostane triterpenes accumulation by up-regulating the expression of squalene epoxidases in Alisma orientale. Sci. Rep. 2019, 9, 18139.
- Wang, P.; Song, T.; Shi, R.; He, M.; Wang, R.; Lv, J.; Jiang, M. Triterpenoids From Alisma Species: Phytochemistry, Structure Modification, and Bioactivities. Front. Chem. 2020, 8, 363.
- Zhang, J.; Jin, Q.; Wu, W.; Jin, X.; An, Y.; Liu, C.; Wei, W.; Li, Z.; Yao, C.; Yao, S.; et al. “Force iteration molecular designing” strategy for the systematic characterization and discovery of new protostane triterpenoids from Alisma Rhizoma by UHPLC/LTQ-Orbitrap-MS. Anal. Bioanal. Chem. 2021, 413, 1749–1764.
- Imai, Y.; Matsumura, H.; Aramaki, Y. Hypocholesterolemic effect of alisol A-24-monoacetate and its related compounds in rats. Jpn. J. Pharmacol. 1970, 20, 222–228.
- Zhao, Z.; Deng, Z.T.; Huang, S.; Ning, M.; Feng, Y.; Shen, Y.; Zhao, Q.S.; Leng, Y. Alisol B Alleviates Hepatocyte Lipid Accumulation and Lipotoxicity via Regulating RARα-PPARγ-CD36 Cascade and Attenuates Non-Alcoholic Steatohepatitis in Mice. Nutrients 2022, 14, 2411.
- Chang, X.Y.; Wu, J.S.; Zhang, F.Q.; Li, Z.Z.; Jin, W.Y.; Wang, J.X.; Wang, W.H.; Shi, Y. A Strategy for Screening the Lipid-Lowering Components in Alismatis Rhizoma Decoction Based on Spectrum-Effect Analysis. J. Anal. Methods Chem. 2022, 2022, 2363242.
- Fu, Y.; Feng, H.; Ding, X.; Meng, Q.H.; Zhang, S.R.; Li, J.; Chao, Y.; Ji, T.T.; Bi, Y.H.; Zhang, W.W.; et al. Alisol B 23-acetate adjusts bile acid metabolisim via hepatic FXR-BSEP signaling activation to alleviate atherosclerosis. Phytomedicine 2022, 101, 154120.
- Luan, Z.L.; Ming, W.H.; Sun, X.W.; Zhang, C.; Zhou, Y.; Zheng, F.; Yang, Y.L.; Guan, Y.F.; Zhang, X.Y. A naturally occurring FXR agonist, alisol B 23-acetate, protects against renal ischemia-reperfusion injury. Am. J. Physiol. Renal Physiol. 2021, 321, F617–F628.
- Meng, Q.; Duan, X.P.; Wang, C.Y.; Liu, Z.H.; Sun, P.Y.; Huo, X.K.; Sun, H.J.; Peng, J.Y.; Liu, K.X. Alisol B 23-acetate protects against non-alcoholic steatohepatitis in mice via farnesoid X receptor activation. Acta Pharmacol. Sin. 2017, 38, 69–79.
- Kanno, Y.; Yatsu, T.; Yamashita, N.; Zhao, S.; Li, W.; Imai, M.; Kashima, M.; Inouye, Y.; Nemoto, K.; Koike, K. Alisol B 23-acetate from the rhizomes of Alisma orientale is a natural agonist of the human pregnane X receptor. Phytomedicine 2017, 26, 22–27.
- Meng, Q.; Chen, X.; Wang, C.; Liu, Q.; Sun, H.; Sun, P.; Peng, J.; Liu, K. Alisol B 23-acetate promotes liver regeneration in mice after partial hepatectomy via activating farnesoid X receptor. Biochem. Pharmacol. 2014, 92, 289–298.
- Meng, Q.; Chen, X.; Wang, C.; Liu, Q.; Sun, H.; Sun, P.; Huo, X.; Liu, Z.; Yao, J.; Liu, K. Protective Effects of Alisol B 23-Acetate Via Farnesoid X Receptor-Mediated Regulation of Transporters and Enzymes in Estrogen-Induced Cholestatic Liver Injury in Mice. Pharm. Res. 2015, 32, 3688–3698.
- Meng, Q.; Chen, X.L.; Wang, C.Y.; Liu, Q.; Sun, H.J.; Sun, P.Y.; Huo, X.K.; Liu, Z.H.; Yao, J.H.; Liu, K.X. Alisol B 23-acetate protects against ANIT-induced hepatotoxity and cholestasis, due to FXR-mediated regulation of transporters and enzymes involved in bile acid homeostasis. Toxicol. Appl. Pharmacol. 2015, 283, 178–186.
- Meng, Q.; Chen, X.; Wang, C.; Liu, Q.; Sun, H.; Sun, P.; Huo, X.; Liu, Z.; Liu, K. Protective effects of alisol B 23-acetate from edible botanical Rhizoma alismatis against carbon tetrachloride-induced hepatotoxicity in mice. Food Funct. 2015, 6, 1241–1250.
- Zhou, X.; Ren, Q.; Wang, B.; Fang, G.; Ling, Y.; Li, X. Alisol A 24-Acetate Isolated from the Alismatis Rhizoma Improves Hepatic Lipid Deposition in Hyperlipidemic Mice by ABCA1/ABCG1 Pathway. J. Nanosci. Nanotechnol. 2019, 19, 5496–5502.
- Chen, J.X.; Li, H.Y.; Li, T.T.; Fu, W.C.; Du, X.; Liu, C.H.; Zhang, W. Alisol A-24-acetate promotes glucose uptake via activation of AMPK in C2C12 myotubes. BMC Complement. Med. Ther. 2020, 20, 22.
- Lou, H.X.; Fu, W.C.; Chen, J.X.; Li, T.T.; Jiang, Y.Y.; Liu, C.H.; Zhang, W. Alisol A 24-acetate stimulates lipolysis in 3 T3-L1 adipocytes. BMC Complement. Med. Ther. 2021, 21, 128.
- Choi, E.; Jang, E.; Lee, J.H. Pharmacological Activities of Alisma orientale against Nonalcoholic Fatty Liver Disease and Metabolic Syndrome: Literature Review. Evid. Based Complement. Alternat. Med. 2019, 2019, 2943162.
- Sun, Y.; Long, J.; Chen, W.; Sun, Y.; Zhou, L.; Zhang, L.; Zeng, H.; Yuan, D. Alisol B 23-acetate, a new promoter for cholesterol efflux from dendritic cells, alleviates dyslipidemia and inflammation in advanced atherosclerotic mice. Int. Immunopharmacol. 2021, 99, 107956.
- Yu, X.C.; Fu, Y.; Bi, Y.H.; Zhang, W.W.; Li, J.; Ji, T.; Chao, Y.; Meng, Q.H.; Chen, Q.; Ma, M.H.; et al. Alisol B 23-acetate activates ABCG5/G8 in the jejunum via the LXRα/ACAT2 pathway to relieve atherosclerosis in ovariectomized ApoE-/- mice. Aging 2020, 12, 25744–25766.
- Chen, Q.; Chao, Y.; Zhang, W.; Zhang, Y.; Bi, Y.; Fu, Y.; Cai, D.; Meng, Q.; Li, Y.; Bian, H. Activation of estrogen receptor α (ERα) is required for Alisol B23-acetate to prevent post-menopausal atherosclerosis and reduced lipid accumulation. Life Sci. 2020, 258, 118030.
- Lin, C.; Li, J.; Wu, C.; Bao, J. Identifying selective agonists targeting LXRβ from terpene compounds of alismatis rhizoma. J. Mol. Model. 2021, 27, 91.
- Charles, R.; Eaton, P. Redox Regulation of Soluble Epoxide Hydrolase-Implications for Cardiovascular Health and Disease. Cells 2022, 11, 1932.
- Shan, J.; Hashimoto, K. Soluble Epoxide Hydrolase as a Therapeutic Target for Neuropsychiatric Disorders. Int. J. Mol. Sci. 2022, 23, 4951.
- Hur, J.M.; Choi, J.W.; Park, J.C. Effects of methanol extract of Alisma orientale rhizome and its major component, alisol B 23-acetate, on hepatic drug metabolizing enzymes in rats treated with bromobenzene. Arch. Pharm. Res. 2007, 30, 1543–1549.
- Sun, C.P.; Zhang, J.; Zhao, W.Y.; Yi, J.; Yan, J.K.; Wang, Y.L.; Morisseau, C.; Liu, Z.B.; Hammock, B.D.; Ma, X.C. Protostane-type triterpenoids as natural soluble epoxide hydrolase inhibitors: Inhibition potentials and molecular dynamics. Bioorg. Chem. 2020, 96, 103637.
- Zhao, W.Y.; Zhang, X.Y.; Zhou, M.R.; Tian, X.G.; Lv, X.; Zhang, H.L.; Deng, S.; Zhang, B.J.; Sun, C.P.; Ma, X.C. Natural soluble epoxide hydrolase inhibitors from Alisma orientale and their potential mechanism with soluble epoxide hydrolase. Int. J. Biol. Macromol. 2021, 183, 811–817.
- Ma, Q.; Han, L.; Bi, X.; Wang, X.; Mu, Y.; Guan, P.; Li, L.; Huang, X. Structures and biological activities of the triterpenoids and sesquiterpenoids from Alisma orientale. Phytochemistry 2016, 131, 150–157.
- Bi, X.; Wang, P.; Ma, Q.; Han, L.; Wang, X.; Mu, Y.; Guan, P.; Qu, X.; Wang, Z.; Huang, X. Anti-Inflammatory Activities and Liver Protection of Alisol F and 25-Anhydroalisol F through the Inhibition of MAPK, STAT3, and NF-κB Activation In Vitro and In Vivo. Molecules 2017, 22, 951.
- Wang, B.; Chen, L.; Dai, L.; Fang, W.; Wang, H. Alisol B 23-Acetate Ameliorates Lipopolysaccharide-Induced Cardiac Dysfunction by Suppressing Toll-Like Receptor 4 (TLR4)/NADPH Oxidase 2 (NOX2) Signaling Pathway. Med. Sci. Monit. 2019, 25, 8472–8481.
- Xia, F.; Xiang, S.; Chen, Z.; Song, L.; Li, Y.; Liao, Z.; Ge, B.; Zhou, B. The probiotic effects of AB23A on high-fat-diet-induced non-alcoholic fatty liver disease in mice may be associated with suppressing the serum levels of lipopolysaccharides and branched-chain amino acids. Arch. Biochem. Biophys. 2021, 714, 109080.
- Xia, F.; Li, Y.; Deng, L.; Ren, R.; Ge, B.; Liao, Z.; Xiang, S.; Zhou, B. Alisol B 23-Acetate Ameliorates Lipopolysaccharide-Induced Intestinal Barrier Dysfunction by Inhibiting TLR4-NOX1/ROS Signaling Pathway in Caco-2 Cells. Front. Pharmacol. 2022, 13, 911196.
- Moeller, M.J.; Kramann, R.; Lammers, T.; Hoppe, B.; Latz, E.; Ludwig-Portugall, I.; Boor, P.; Floege, J.; Kurts, C.; Weiskirchen, R.; et al. New Aspects of Kidney Fibrosis-From Mechanisms of Injury to Modulation of Disease. Front. Med. 2022, 8, 814497.
- Chen, H.; Wang, M.C.; Chen, Y.Y.; Chen, L.; Wang, Y.N.; Vaziri, N.D.; Miao, H.; Zhao, Y.Y. Alisol B 23-acetate attenuates CKD progression by regulating the renin-angiotensin system and gut-kidney axis. Ther. Adv. Chronic Dis. 2020, 11, 2040622320920025.
- Chen, L.; Chen, D.Q.; Wang, M.; Liu, D.; Chen, H.; Dou, F.; Vaziri, N.D.; Zhao, Y.Y. Role of RAS/Wnt/β-catenin axis activation in the pathogenesis of podocyte injury and tubulo-interstitial nephropathy. Chem. Biol. Interact. 2017, 273, 56–72.
- Chen, H.; Yang, T.; Wang, M.C.; Chen, D.Q.; Yang, Y.; Zhao, Y.Y. Novel RAS inhibitor 25-O-methylalisol F attenuates epithelial-to-mesenchymal transition and tubulo-interstitial fibrosis by selectively inhibiting TGF-β-mediated Smad3 phosphorylation. Phytomedicine 2018, 42, 207–218.
- Xue, X.H.; Zhou, X.M.; Wei, W.; Chen, T.; Su, Q.P.; Tao, J.; Chen, L.D. Alisol A 24-Acetate, a Triterpenoid Derived from Alisma orientale, Inhibits Ox-LDL-Induced Phenotypic Transformation and Migration of Rat Vascular Smooth Muscle Cells through Suppressing ERK1/2 Signaling. J. Vasc. Res. 2016, 53, 291–300.
- Zhu, H.C.; Jia, X.K.; Fan, Y.; Xu, S.H.; Li, X.Y.; Huang, M.Q.; Lan, M.L.; Xu, W.; Wu, S.S. Alisol B 23-Acetate Ameliorates Azoxymethane/Dextran Sodium Sulfate-Induced Male Murine Colitis-Associated Colorectal Cancer via Modulating the Composition of Gut Microbiota and Improving Intestinal Barrier. Front. Cell. Infect. Microbiol. 2021, 11, 640225.
- Xu, X.; Li, L.; Zhang, Y.; Lu, X.; Lin, W.; Wu, S.; Qin, X.; Xu, R.; Lin, W. Hypolipidemic effect of Alisma orientale (Sam.) Juzep on gut microecology and liver transcriptome in diabetic rats. PLoS ONE 2020, 15, e0240616.
- Zhu, B.; Zhai, Y.; Ji, M.; Wei, Y.; Wu, J.; Xue, W.; Tao, W.W.; Wu, H. Alisma orientalis Beverage Treats Atherosclerosis by Regulating Gut Microbiota in ApoE-/- Mice. Front. Pharmacol. 2020, 11, 570555.
- Xu, W.; Li, X.; Lin, N.; Zhang, X.; Huang, X.; Wu, T.; Tai, Y.; Chen, S.; Wu, C.H.; Huang, M.; et al. Pharmacokinetics and tissue distribution of five major triterpenoids after oral administration of Rhizoma Alismatis extract to rats using ultra high-performance liquid chromatography-tandem mass spectrometry. J. Pharm. Biomed. Anal. 2017, 146, 314–323.
- Lu, L.; Lu, T.; Shen, J.; Lv, X.; Wei, W.; Wang, H.; Xue, X. Alisol A 24-acetate protects against brain microvascular endothelial cells injury through inhibiting miR-92a-3p/tight junctions axis. Aging 2021, 13, 15353–15365.
- Zhou, Y.; Wei, W.; Shen, J.; Lu, L.; Lu, T.; Wang, H.; Xue, X. Alisol A 24-acetate protects oxygen-glucose deprivation-induced brain microvascular endothelial cells against apoptosis through miR-92a-3p inhibition by targeting the B-cell lymphoma-2 gene. Pharm. Biol. 2021, 59, 513–524.
- Lu, T.; Li, H.; Zhou, Y.; Wei, W.; Ding, L.; Zhan, Z.; Liu, W.; Tao, J.; Xue, X. Neuroprotective effects of alisol A 24-acetate on cerebral ischaemia-reperfusion injury are mediated by regulating the PI3K/AKT pathway. J. Neuroinflammation 2022, 19, 37.
- Jang, E.; Lee, J.H. Promising Anticancer Activities of Alismatis rhizome and Its Triterpenes via p38 and PI3K/Akt/mTOR Signaling Pathways. Nutrients 2021, 13, 2455.
- Jin, U.H.; Kim, D.I.; Lee, T.K.; Lee, D.N.; Kim, J.K.; Lee, I.S.; Kim, C.H. Herbal formulation, Yukmi-jihang-tang-Jahage, regulates bone resorption by inhibition of phosphorylation mediated by tyrosine kinase Src and cyclooxygenase expression. J. Ethnopharmacol. 2006, 106, 333–343.
- Xia, B.; Xu, B.; Sun, Y.; Xiao, L.; Pan, J.; Jin, H.; Tong, P. The effects of Liuwei Dihuang on canonical Wnt/β-catenin signaling pathway in osteoporosis. J. Ethnopharmacol. 2014, 153, 133–141.
- An, J.; Hao, D.; Zhang, Q.; Chen, B.; Zhang, R.; Wang, Y.; Yang, H. Natural products for treatment of bone erosive diseases: The effects and mechanisms on inhibiting osteoclastogenesis and bone resorption. Int. Immunopharmacol. 2016, 36, 118–131.
- Jia, X.; Zhu, H.; Li, G.; Lan, M.; Li, X.; Huang, M.; Xu, W.; Wu, S. Anti-osteoporotic effects of alisol C 23-acetate via osteoclastogenesis inhibition. Biomed. Pharmacother. 2021, 137, 111321.
- Kim, K.J.; Leutou, A.S.; Yeon, J.T.; Choi, S.W.; Kim, S.H.; Yee, S.T.; Choi, K.H.; Nam, S.J.; Son, Y.J. The Inhibitory Effect of Alisol A 24-Acetate from Alisma canaliculatum on Osteoclastogenesis. Int. J. Endocrinol. 2015, 2015, 132436.
- Hwang, Y.H.; Kang, K.Y.; Lee, S.J.; Nam, S.J.; Son, Y.J.; Yee, S.T. The Protective Effects of Alisol A 24-Acetate from Alisma canaliculatum on Ovariectomy Induced Bone Loss in Vivo. Molecules 2016, 21, 74.
- Lee, J.W.; Kobayashi, Y.; Nakamichi, Y.; Udagawa, N.; Takahashi, N.; Im, N.K.; Seo, H.J.; Jeon, W.B.; Yonezawa, T.; Cha, B.Y.; et al. Alisol-B, a novel phyto-steroid, suppresses the RANKL-induced osteoclast formation and prevents bone loss in mice. Biochem. Pharmacol. 2010, 80, 352–361.
- Pan, G.; Li, T.; Zeng, Q.; Wang, X.; Zhu, Y. Alisol F 24 Acetate Enhances Chemosensitivity and Apoptosis of MCF-7/DOX Cells by Inhibiting P-Glycoprotein-Mediated Drug Efflux. Molecules 2016, 21, 183.
- Wang, C.; Zhang, J.X.; Shen, X.L.; Wan, C.K.; Tse, A.K.; Fong, W.F. Reversal of P-glycoprotein-mediated multidrug resistance by Alisol B 23-acetate. Biochem. Pharmacol. 2004, 68, 843–855.
- Lou, C.; Xu, X.; Chen, Y.; Zhao, H. Alisol A Suppresses Proliferation, Migration, and Invasion in Human Breast Cancer MDA-MB-231 Cells. Molecules 2019, 24, 3651.
- Shi, Y.; Wang, M.; Wang, P.; Zhang, T.; Yu, J.; Shi, L.; Li, M.; Wang, H.; Zhang, Q.; Zhao, H. Alisol A is potentially therapeutic in human breast cancer cells. Oncol. Rep. 2020, 44, 1266–1274.
- Xia, J.; Luo, Q.; Huang, S.; Jiang, F.; Wang, L.; Wang, G.; Xie, J.; Liu, J.; Xu, Y. Alisol B 23-acetate-induced HepG2 hepatoma cell death through mTOR signaling-initiated G1 cell cycle arrest and apoptosis: A quantitative proteomic study. Chin. J. Cancer Res. 2019, 31, 375–388.
- Li, L.; Cheng, J.; Zhu, D.; Shi, X.; Wei, Y.; Chen, S.; Wang, Z.; Yuan, D. The effects of Alisol B 23-acetate in hepatocellular carcinoma via inducing cell apoptosis and inhibiting cell migration and invasion. Gen. Physiol. Biophys. 2020, 39, 219–228.
- Ye, M.; Tang, Y.; He, J.; Cao, X.; Liu, J.; Kou, S.; Yang, Y.; Xue, J.; Li, F. Alisol B 23-Acetate Increases the Antitumor Effect of Bufalin on Liver Cancer through Inactivating Wnt/β-Catenin Axis. Comput. Math. Methods Med. 2022, 2022, 6249534.
- El-Baba, C.; Baassiri, A.; Kiriako, G.; Dia, B.; Fadlallah, S.; Moodad, S.; Darwiche, N. Terpenoids’ anti-cancer effects: Focus on autophagy. Apoptosis 2021, 26, 491–511.
- Chen, X.; Liu, H. Alisol A Inhibited the Proliferation, Migration, and Invasion of Nasopharyngeal Carcinoma Cells by Inhibiting the Hippo Signaling Pathway. Yonsei Med. J. 2021, 62, 895–902.
- Law, B.Y.; Wang, M.; Ma, D.L.; Al-Mousa, F.; Michelangeli, F.; Cheng, S.H.; Ng, M.H.; To, K.F.; Mok, A.Y.; Ko, R.Y.; et al. Alisol B, a novel inhibitor of the sarcoplasmic/endoplasmic reticulum Ca(2+) ATPase pump, induces autophagy, endoplasmic reticulum stress, and apoptosis. Mol. Cancer Ther. 2010, 9, 718–730.
- Wang, Y.L.; Zhao, J.C.; Liang, J.H.; Tian, X.G.; Huo, X.K.; Feng, L.; Ning, J.; Wang, C.; Zhang, B.J.; Chen, G.; et al. A bioactive new protostane-type triterpenoid from Alisma plantago-aquatica subsp. orientale (Sam.) Sam. Nat. Prod. Res. 2019, 33, 776–781.
- Lin, H.R. Triterpenes from Alisma orientalis act as androgen receptor agonists, progesterone receptor antagonists, and glucocorticoid receptor antagonists. Bioorg. Med. Chem. Lett. 2014, 24, 3626–3632.
- Xu, F.; Lu, C.; Wu, Q.; Chen, J.; Gu, W.; Du, W.; You, M. Study on antitumor molecular mechanism of Alisols based on p53DNA. Int. J. Biol. Macromol. 2018, 116, 1163–1174.
- Xu, F.; Chen, J.; Wu, Q.; Gu, W.; Shen, Y.; Lu, C.; Zhang, Y.; Liu, S.; Liao, H. The antitumor molecular mechanism of Alisma orientalis with c-myc DNA: Multi-spectroscopic analysis and molecular simulation. J. Biomol. Struct. Dyn. 2020, 38, 4189–4209.
- Ho, C.; Gao, Y.; Zheng, D.; Liu, Y.; Shan, S.; Fang, B.; Zhao, Y.; Song, D.; Zhang, Y.; Li, Q. Alisol A attenuates high-fat-diet-induced obesity and metabolic disorders via the AMPK/ACC/SREBP-1c pathway. J. Cell. Mol. Med. 2019, 23, 5108–5118.
- Zhang, A.; Sheng, Y.; Zou, M. Antiproliferative activity of Alisol B in MDA-MB-231 cells is mediated by apoptosis, dysregulation of mitochondrial functions, cell cycle arrest and generation of reactive oxygen species. Biomed. Pharmacother. 2017, 87, 110–117.
- Kubo, M.; Matsuda, H.; Tomohiro, N.; Yoshikawa, M. Studies on Alismatis rhizoma. I. Anti-allergic effects of methanol extract and six terpene components from Alismatis rhizoma (dried rhizome of Alisma orientale). Biol. Pharm. Bull. 1997, 20, 511–516.
- Lee, J.H.; Kwon, O.S.; Jin, H.G.; Woo, E.R.; Kim, Y.S.; Kim, H.P. The rhizomes of Alisma orientale and alisol derivatives inhibit allergic response and experimental atopic dermatitis. Biol. Pharm. Bull. 2012, 35, 1581–1587.
- Shao, C.; Fu, B.; Ji, N.; Pan, S.; Zhao, X.; Zhang, Z.; Qiu, Y.; Wang, R.; Jin, M.; Wen, K.; et al. Alisol B 23-Acetate Inhibits IgE/Ag-Mediated Mast Cell Activation and Allergic Reaction. Int. J. Mol. Sci. 2018, 19, 4092.
- Jin, H.G.; Jin, Q.; Ryun Kim, A.; Choi, H.; Lee, J.H.; Kim, Y.S.; Lee, D.G.; Woo, E.R. A new triterpenoid from Alisma orientale and their antibacterial effect. Arch. Pharm. Res. 2012, 35, 1919–1926.
- Zhang, Q.; Jiang, Z.Y.; Luo, J.; Cheng, P.; Ma, Y.B.; Zhang, X.M.; Zhang, F.X.; Zhou, J.; Chen, J.J. Anti-HBV agents. Part 1: Synthesis of alisol A derivatives: A new class of hepatitis B virus inhibitors. Bioorg. Med. Chem. Lett. 2008, 18, 4647–4650.
- Zhang, Q.; Jiang, Z.Y.; Luo, J.; Liu, J.F.; Ma, Y.B.; Guo, R.H.; Zhang, X.M.; Zhou, J.; Chen, J.J. Anti-HBV agents. Part 2: Synthesis and in vitro anti-hepatitis B virus activities of alisol A derivatives. Bioorg. Med. Chem. Lett. 2009, 19, 2148–2153.
- Zhang, Q.; Jiang, Z.Y.; Luo, J.; Ma, Y.B.; Liu, J.F.; Guo, R.H.; Zhang, X.M.; Zhou, J.; Niu, W.; Du, F.F.; et al. Anti-HBV agents. Part 3: Preliminary structure-activity relationships of tetra-acylalisol A derivatives as potent hepatitis B virus inhibitors. Bioorg. Med. Chem. Lett. 2009, 19, 6659–6665.
More
Information
Subjects:
Integrative & Complementary Medicine
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.2K
Revisions:
2 times
(View History)
Update Date:
25 Aug 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





