Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | James C. L. Chow | -- | 2797 | 2022-08-23 14:56:25 | | | |
| 2 | Rita Xu | Meta information modification | 2797 | 2022-08-24 03:19:43 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Siddique, S.; Chow, J.C.L. Nanoparticles in Cancer Theranostics. Encyclopedia. Available online: https://encyclopedia.pub/entry/26398 (accessed on 31 May 2026).
Siddique S, Chow JCL. Nanoparticles in Cancer Theranostics. Encyclopedia. Available at: https://encyclopedia.pub/entry/26398. Accessed May 31, 2026.
Siddique, Sarkar, James C. L. Chow. "Nanoparticles in Cancer Theranostics" Encyclopedia, https://encyclopedia.pub/entry/26398 (accessed May 31, 2026).
Siddique, S., & Chow, J.C.L. (2022, August 23). Nanoparticles in Cancer Theranostics. In Encyclopedia. https://encyclopedia.pub/entry/26398
Siddique, Sarkar and James C. L. Chow. "Nanoparticles in Cancer Theranostics." Encyclopedia. Web. 23 August, 2022.
Copy Citation
Cancer theranostics is the combination of diagnosis and therapeutic approaches for cancer, which is essential in personalized cancer treatment. The aims of the theranostics application of nanoparticles in cancer detection and therapy are to reduce delays in treatment and hence improve patient care. Recently, it has been found that the functionalization of nanoparticles can improve the efficiency, performance, specificity and sensitivity of the structure, and increase stability in the body and acidic environment. Moreover, functionalized nanoparticles have been found to possess a remarkable theranostic ability and have revolutionized cancer treatment.
functionalized nanoparticles
MRI-guided therapy
molecular imaging
1. Introduction
Cancer treatment has gained considerable attention in biomedical research over the past few decades due to the serious threat it poses to human health. The mortality rate of cancer increases every year, which leads to the need for the development of more efficient cancer therapeutic strategies [1]. Even though there is a major advance in cancer therapy, it continues to be a significant challenge due to tolerability and adherence [2]. Theranostics is a term first used by John Funkhouser at the beginning of the 1990s. It is defined as a combination of diagnostic tools that are the most suitable for specific diseases [3]. Theranostics portrays a close connection between diagnostics and the consequent therapy, and the theranostic principle has attracted huge attention in personalized medicine, in particular oncology. This allowed tumours at the advanced stage to be treated accurately with fewer side effects. For decades theranostics have been used for the therapy of benign and malignant thyroid diseases; however, recently, theranostics have been applied to other malignancies [4]. Theranostics agents such as radioisotopes, liposomes, quantum dots and plasmonic nanobubbles can be attached to anticancer drugs, imaging agents and cancer cell markers with the support of imaging techniques, providing the potential to facilitate the diagnosis, treatment and management of cancer patients [5]. The development of highly sensitive imaging modalities such as SPECT and PET with the synthesis of novel radiolabelled molecules specific for different biochemical targets promoted nuclear medicine into a new era [6]. These molecular imaging modalities have been applied in cardiology, neuroscience, oncology, gene therapy and theranostics. Nanoparticles (NPs) have been used as therapeutic or imaging agents that enhance the efficacy and control biodistribution and reduce the toxicity of drugs. In 2014–2015, there were 51 FDA-approved nanomedicines that met the definition of nanomedicines as therapeutic or imaging agents, and 77 products in clinical trial [7]. One of the crucial characteristics of nanomaterials is their small size. Their high affinity, high specificity, high thermal stability, low off-target accumulation and good solubility are among many adventurous characteristics they possess in cancer therapy. They can penetrate dense tissues of the tumour very well [8]. Nanotechnology in medicine is currently developed for drug delivery, and many substances are under study for cancer therapy. Solid NPs can be used for drug targeting when they reach the intended diseased site in the body, and the toxicology of the drug nanocarriers has been evaluated [9]. Active targeting is accomplished by conjugating tumour-specific ligands to the NPs’ surface. It complements the enhanced permeability and retention effect (EPR). EPR is a universal pathophysiological phenomenon and mechanism where macromolecules with certain sizes above 40 kDa can progressively accumulate in the tumour vascularized area and achieve targeted delivery and retention of the anticancer compound into the solid tumour [10]. Some of the particles that are used to functionalize NPs are antibodies or antibody fragments, human transferrin protein, peptides, carbohydrates and vitamins. These biomarkers are recognized by their representative targeting ligands such as epidermal growth factor, human epidermal growth factor 2, Mucin-1, nucleolin, epithelial cell adhesion molecule and platelet-derived growth factor receptor 2. For anticancer drug delivery, Fu et al. [11] proposed to use aptamer-functionalized nanoparticles. This is because aptamers have favourable features such as a small size, very low immunogenicity, low cost of production and high affinity and specificity. The advantage of NPs as a theranostics agent is shown below in Figure 1 [12].
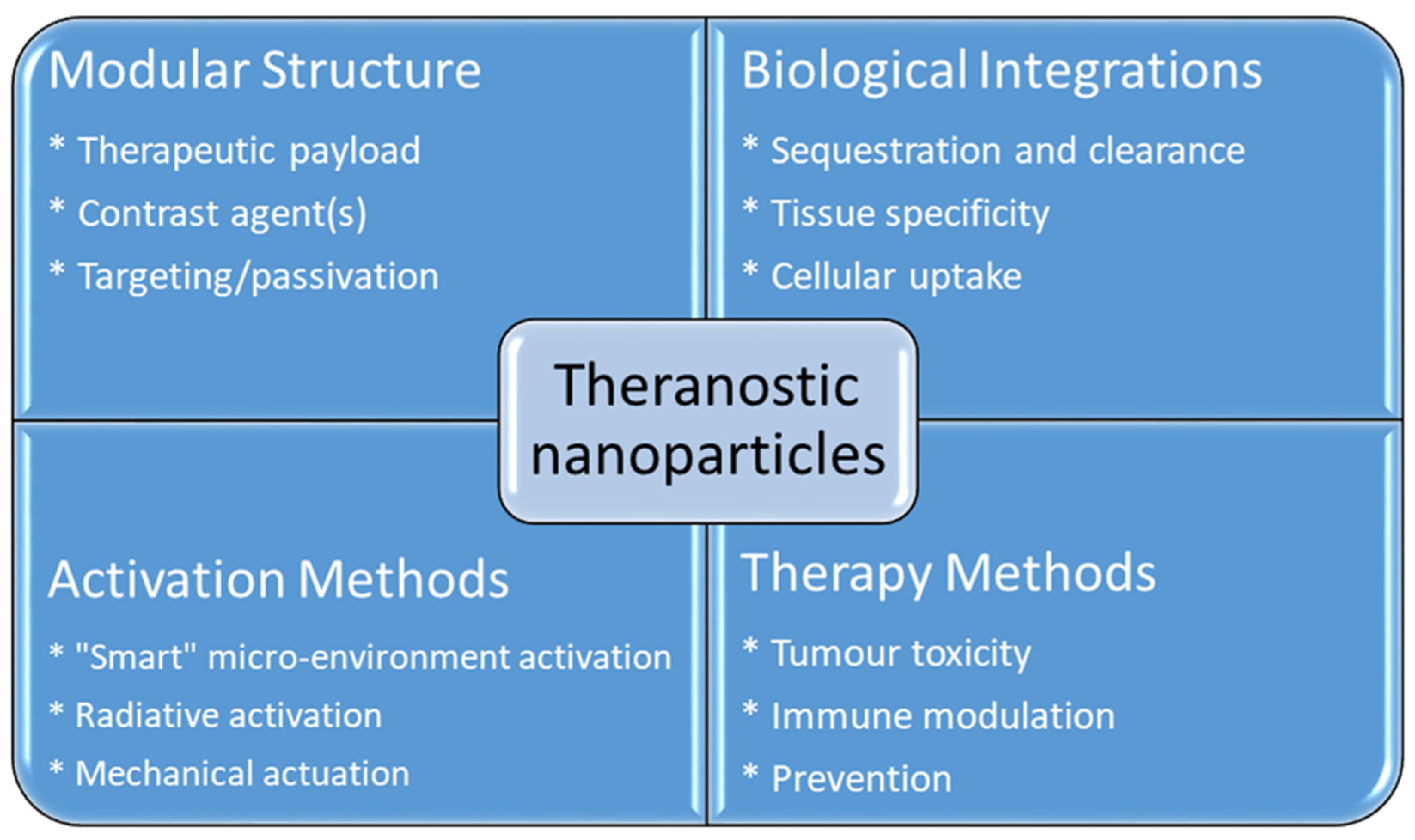
Figure 1. Advantages using nanoparticles in cancer theranostics.
2. Magnetic Resonance Imaging (MRI)
MRI is one of the most powerful means of clinical detection and prognosis observation [13]. MRI is an imaging modality that is non-invasive, and it provides comprehensive multi-parametric information generally used for brain imaging [14]. MRI benefits from the contrast agent that provides a more improved depiction of large and medium-sized vassals and can provide dynamic vascular/perfusional properties of tissues. Gadolinium (Gd)-based contrast agents are widely used in MRI [15][16]. MRI can be coupled with other therapy to provide image-guided therapy for better treatment outcomes and tumour-targeting ability [17]. A study synthesized a multifunctional Gd-DTPA-ONB lipid by adding the Gd-DTPA contrast agent to an o-nitro-benzyl ester lipid. It combines the MRI tracking ability with dual trigger release capabilities, which allow maximum sensitivity without reducing the drug encapsulation rate. It can be activated by both PH-trigger hydrolysis and photo treatment [18]. Another Gd nanocomposite was synthesized by decorating Gd NPs onto the graphene oxide, and then functionalized with polyethylene glycol and folic acid. It was used to load doxorubicin to accomplish targeted image-guided drug delivery with MRI [19]. Liposomes are a useful class of NPs due to their tunable properties and multiple liposomal drug formulation. They have been clinically approved for cancer treatment. A vast number of Gd-based liposomal MRI contrast agents have been developed that can be used for targeted image-guided drug delivery [20]. Chemical exchange saturation transfer MRI has important advantages such as its ability to detect diamagnetic compounds that are not detectable using conventional MRI. It makes a broad spectrum of bioorganic agents, nanocarriers and natural compounds directly MRI detectable with a high resolution. It is advantageous for image-guided drug delivery [21]. An in vivo study looked at amphiphilic polymer-coated magnetic iron oxide NPs that were conjugated with near-infrared (NIR) dye-labelled HER2 affibody and chemotherapy drugs. Cisplatin was the drug used as the chemotherapy drug. MRI-guided therapy and the optical imaging detection of the therapy-resistant tumour were examined in an orthotopic human ovarian cancer xenograft model with a high level of HER2 expression. The result shows it significant inhibited the primary tumour and peritoneal and lung metastases in the ovarian cancer model in mice [22]. Another study looked at the NP with a unique morphology, which consists of a superparamagnetic iron oxide core and star-shaped plasmonic shell with high aspect ratio branches. Its strong near-infrared responsive plasmonic properties and magnetic properties allow it to be used in multimodal quantitative imaging, which combines the advantageous functions of MRI, magnetic particle imaging (MPI) and photoacoustic imaging. It can be used for image-guided drug delivery with tunable drug release capacity [23]. Drug resistance in chemotherapy has been a challenge for a long time in pancreatic cancer due to the stomal barrier making it difficult to reach the tumour microenvironment. A study developed IGF1 receptor-directed multifunctional theragnostic NPs for the targeted delivery of Dos into IGF1R-expressing drug-resistant tumour cells and tumour-associated stromal cells. NPs were prepared by combining IGF1 with magnetic iron oxide NPs carrying dox. They provided an excellent theranostics platform and showed good tumour control in an in vivo study [24]. Superparamagnetic iron oxide NPs have also been widely used in MRI and nanotheranostics. They can be coated with a biocompatible polymer such as polyethylene glycol or dextran, which allows chemical conjugation. They have a very high potential in MRI-guided drug delivery [25]. Figure 2 shows superparamagnetic iron oxide NPs being used in liver imaging and lymph node imaging [26].
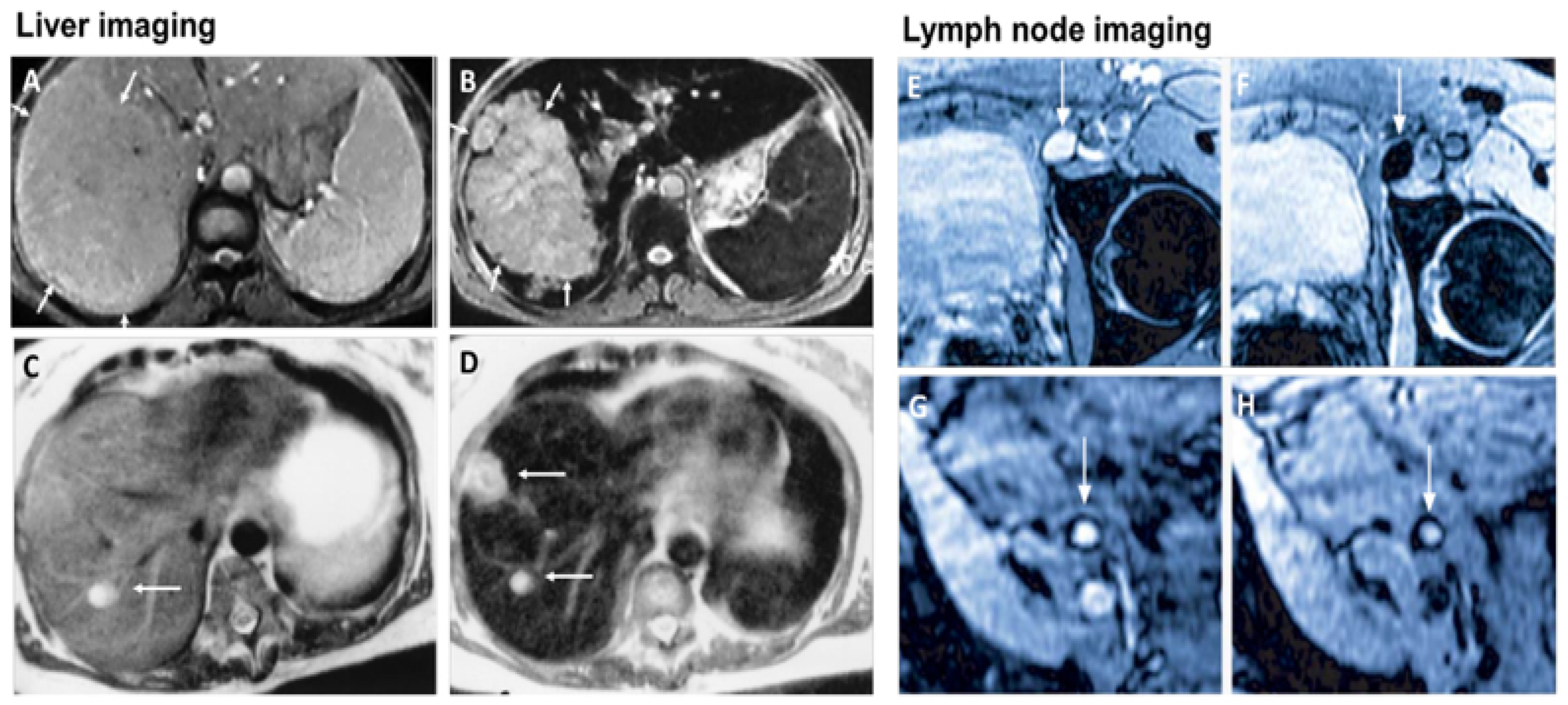
Figure 2. Superparamagnetic iron oxide NPs being used in liver imaging and lymph node imaging. (A,B): T2-weighted MR image of a liver with a large hepatocellular carcinoma before (A) and after (B) the administration of SPION. The lesion is demarcated with arrows. (C,D): Standard (C) and SPION-based contrast-enhanced (D) MR imaging of liver metastasis in a patient with colorectal cancer. After administration of ferumoxide SPION, a second metastasis becomes visible on T2-weighted MR image. (E,H): Lymph node in left iliac region (arrow), with and without metastatic infiltration. T2-weighted images before (E,G) and 24 h after (F,H) administration of ferumoxtran. Lymph node (arrow) appears bright before injection of UPIO (E,G). One day after injection, a signal loss in the lymph node (arrow) due to high UPIO macrophage uptake can be observed, thus indicating functionality and no metastasis (F). Conversely, in the lower panel, the lymph node (arrow) stays bright, indicating no trafficking of USPIO and thus metastatic colonization (H). Reprinted with permission from Ref. [26]. Copyright 2020 Elsevier.
2.1. MRI-Guided NPs for Gene Therapy
Gene therapy has gained considerable attention over the years and the health community has gained much more new information and knowledge regarding gene therapy [27]. Gene therapy is a form of engineered viruses carrying a therapeutic agent or containing genetically modified cells such as when chimeric antigen receptors are introduced to the T lymphocytes for cancer therapy such as for leukemia [28]. New gene therapy has shown its potential to significantly improve the survival rate of cancer patients [29]. For cancer gene therapy, the therapeutic agent generally requires a carrier such as an NP. MRI allows the tracking of that carrier and allows image-guided therapy, which can significantly improve the outcome [30]. A study looked at low molecular weight poly (ethylenimine)-poly (ethylene glycol) nanogels loaded with transforming growth factor -β1 siRNA and ultra-small iron oxide NPs for gene therapy and a T1-weighted MRI of tumour and tumour metastasis in a mouse sarcoma model. The study result shows it enhances the MRI image and effectively delivers the siRNA and inhibits tumour growth in the subcutaneous sarcoma tumour model and lung metastasis by silencing the TGF-β1 gene [31]. Another study investigated shaped, controlled magnetic mesoporous silica NPs and their performances in magnetic resonance image-guided targeted hyperthermia-enhanced suicide gene therapy of hepatocellular carcinoma. They had a higher loading capacity and better magnetic hyperthermia properties. They also had decreased cytotoxicity [32]. A bowl-shaped Fe3O4 NP with a self-assembly concept and appropriately surface-functionalized was studied with the aim for it to be used as a multifunctional carrier in combination therapy and gene therapy. The in vivo result shows promising results in the mouse breast cancer model [33]. The catalytic deoxy ribozyme has great potential in gene therapy via gene regulation but requires the carrier to reach the tumour target. A study showed polydopamine-Mn2+ NPs to be effective carriers and together they can be used as a photothermal agent and contrast agent for photoacoustic and magnetic resonance imaging [34]. Another study developed Fe3O4@PDA NPs to transport siRNA for gene therapy. The NPs were coated with mesenchymal stem cells to form a membrane. The overall complex showed good transport ability and photothermal functionality, and enhanced MRI capability [35].
2.2. MRI-Guided NPs for Thermal Therapy
Light-activated therapies have been introduced for cancer treatment for numerous cancers. Two of the main methods are localizing chemical exchange on the tumour known as photodynamic therapy (PDT) and localized thermal damage to the tumour, also known as photothermal therapy (PTT) [36]. Inorganic NPs have gained significant attention in image-guided thermal therapy in recent years, and the applications of inorganic NPs in tumour imaging and therapy are shown in Figure 3. The NPs contain metal, a semiconductor, metal oxide, nanocrystal and lanthanide-doped up conversion NPs. They can generate heat and reactive oxygen species, so they are ideal for image-guided PTT [37]. The thermal energy also promotes the gasification of perfluoropentane to enable the visualization of cancer tissue in ultrasound imaging, as well as enhances MRI imaging, and makes it ideal for dual MRI ultrasound imaging [38]. Core/shell nanoparticles were investigated for MRI imaging, magnetic hyperthermia and PTT due to their surface being coated with a porous shell. It can entrap large quantities of water around the nanoparticles and allows enhanced and efficient water exchange, which provides an improved magnetic resonance contrast signal. It also helps with NIR absorbance of the core and can have an enhanced thermal effect via synergistic PTT and magnetic hyperthermia. The nanoparticles investigated for this purpose were MnFe2O4/PB [39]. Another study developed temperature-activated engineered neutrophils by combining indocyanine green-loaded magnetic silica NIR sensitive nanoparticles. It provides a platform for dual-targeted PTT. The combination of magnetic targeting and neutrophil targeting provides an enhanced accumulation of the photothermal agent at the tumour site [40]. A study wrapped together gadolinium-DTPA, indocyanine green and perfluoropentane in a poly (lactic-co-glycolic) acid shell membrane by a double emulsion approach. Under NIR the indocyanine green converts the optic energy into thermal energy and converts oxygen to singlet oxygen, which destroys cancer cells through PTT and PDT. Another nanotheranostics agent was prepared via the participation of hydrophilic CuS nanoparticles, styrene, methacrylic acid, N-isopropylacrylamide and a polymerizable rare earth complex. It had good biocompatibility with a high loading capacity for DOX-HCI. Drug release can be activated via PH or high temperature. All these properties make it ideal for PTT and chemotherapy. MRI can also be used on it for image-guided drug delivery [41]. CuS material shows poor MRI ability but excellent photo absorption ability, whereas Fe-based materials have good MRI ability. A study combined the two and made a CuxFeySz sample that includes CuFeS2, FeS2 and Cu5FeS4 nanomaterials. The study result shows it to have high potential in MRI-guided photothermal enhanced chemo dynamic therapy [42].
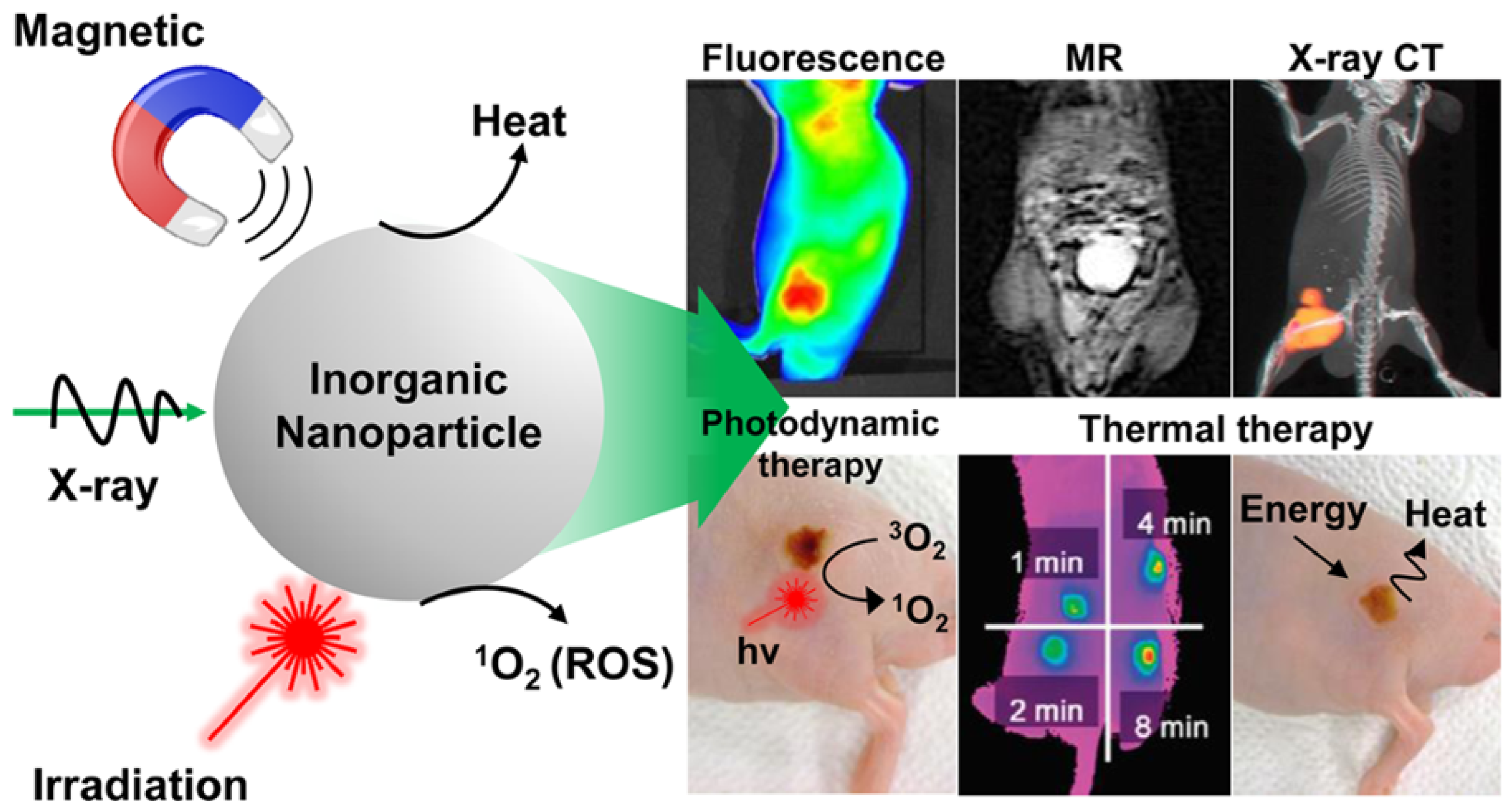
Figure 3. Applications of inorganic NPs for cancer therapy and imaging. Reproduced with permission from [36]. Copyright 2017 ACS Publications.
2.3. Magnetic Hyperthermia Treatment (MHT)
In cancer treatment, the use of a magnetic implant as a thermal seed exposed to the alternating magnetic field is the primary principle behind magnetic hypothermia. Magnetic hypothermia has been used for cancer treatment since the 1950s [43]. Traditionally, deep tumour treatment via magnetic fluid hyperthermia was not possible due to the very low-frequency excitation field being no longer than 100 m in vivo. Now it is possible due to NPs and magnetic particle imaging [44]. In magnetic hyperthermia, the tumour is heated to a moderate temperature of 40–30 °C to destroy cancer cells without the side effects associated with conventional treatment. It can also be co-administered with conventional treatment for better outcomes [45]. Iron oxide NPs have been employed as intra-tumour MTH agents in brain and prostate tumour clinical trials [46]. A study looked at encapsulating produced magnetic iron oxide nanocomposites due to their excellent magnetic saturation and superior magnetic to thermal conversion efficiency with a specific absorption range. It shows the good potential for magnetic hyperthermia therapy [47]. A side effect of magnetic hypothermia is heating of the tumour’s surrounding tissue, which is aimed to be minimized as much as possible [48]. Using NPs can localize the heat and minimizes the damage to the tissue. One example is the release of heat due to the transfer of magnetic field energy into heat by adding magnetic NPs to the tumour in a time-varying magnetic field. This heats the cancer cells, whereas surrounding non-malignant tissues can be spared [49].
2.4. MRI-Guided Chemotherapy
NPs with magnetite composition and polymer encapsulation are used in many applications as theranostic agents for drug delivery and MRI [50]. MRI provides a high-resolution image of structures in the body, and when combined with other imaging modalities, together they can provide complementary diagnostic information for more accurate tumour characteristics identification and the precise guidance of anticancer therapy [51]. The applications of functionalized magnetic NPs in cancer nanotheranostics are shown in Figure 4. Magnetic NPs can be functionalized and guided by a magnetic field. They allow advanced MRI-guided gene and drug delivery, magnetic hyperthermia cancer therapy, cell tracking and bioseparation and tissue engineering [52]. Iron oxide NPs can be used in the diagnosis of liver, inflammation and liver and vascular imaging via MRI. They are also used for therapeutic applications such as iron supplementation in anaemia, macrophage polarization, magnetic drug targeting and magnetic fluid hyperthermia. Due to these properties, they are very useful in theranostic applications [26]. A multifunctional theranostic platform was developed based on amphiphilic hyaluronan/poly-(N-ε-Carbobenzyloxy-L-lysine) derivative (HA-g-PZLL) superparamagnetic iron oxide and aggregation-induced emission (AIR) NPs for magnetic resonance and fluorescence dual-modal image-guided PDT [53]. Gadolinium-based NPs have high relaxivity, passive uptake in the tumour due to an enhanced permeability and retention effect, and adapted biodistribution. These properties make them ideal contrast agents for positive MRI imaging. They can also act as an effective radiosensitizer in radiotherapy, neutron therapy and hadron therapy [54]. Ultra-small gold NPs have low toxicity, and they are non-immunogenic by nature. They have fast kidney clearance and can be used in NIR resonant biomedical imaging modalities. They can be used as an enhancer in MRI, photoacoustic imaging, X-ray and fluorescence imaging. They can also be used to generate heat and local hyperthermia of cancer tissue in PTT. They can also be functionalized to deliver the drug to the cancer cells. All these properties make them ideal for theranostic applications [55]. Another study synthesized a polydopamine-coated manganese oxide NP (FA-Mn3O4@PDA@PEG) conjugate for MRI-guided chemo (PTT). It has a relaxivity of 14.47 mM−1 s−1, which makes it an excellent contrast agent for MRI [56].
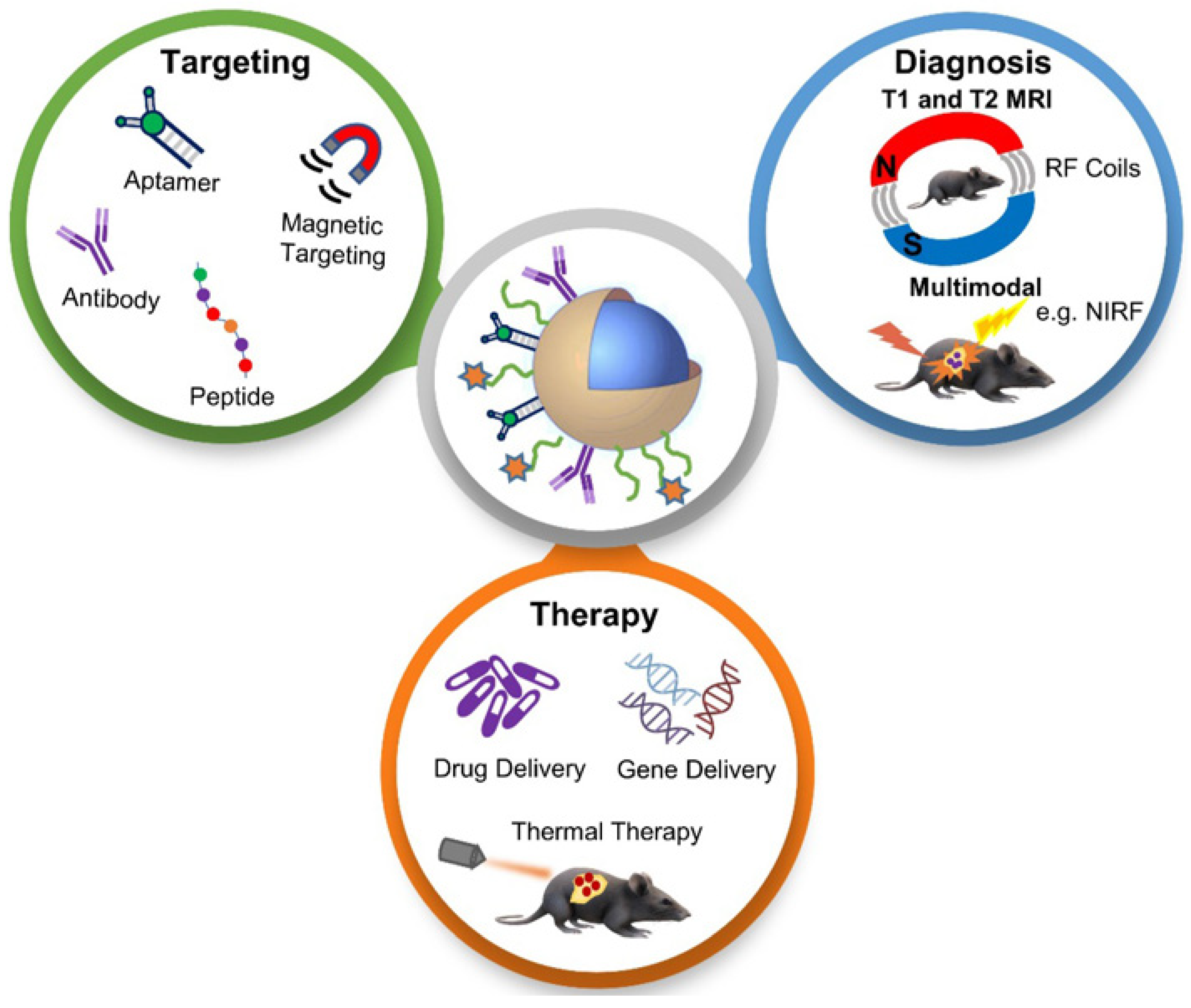
Figure 4. Schematic diagram showing applications of functionalized magnetic NPs in MRI-based diagnosis and anticancer therapy. Reproduced with permission from [51]. Copyright Anani et al. 2020.
References
- Xin, J.; Deng, C.; Aras, O.; Zhou, M.; Wu, C.; An, F. Chemodynamic nanomaterials for cancer theranostics. J. Nanobiotechnol. 2021, 19, 192.
- Mun, E.J.; Babiker, H.M.; Weinberg, U.; Kirson, E.D.; Von Hoff, D.D. Tumor-Treating Fields: A Fourth Modality in Cancer Treatment. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2018, 24, 266–275.
- Choudhury, P.S.; Gupta, M. Theranostics and precision medicine special feature: Review Article Differentiated thyroid cancer theranostics: Radioiodine and beyond. Br. J. Radiol. 2018, 91, 20189004.
- Langbein, T.; Weber, W.A.; Eiber, M. Future of Theranostics: An Outlook on Precision Oncology in Nuclear Medicine. J. Nucl. Med. 2019, 60, 13S–19S.
- Jeyamogan, S.; Khan, N.A.; Siddiqui, R. Application and Importance of Theranostics in the Diagnosis and Treatment of Cancer. Arch. Med. Res. 2021, 52, 131–142.
- James, M.L.; Gambhir, S.S. A Molecular Imaging Primer: Modalities, Imaging Agents, and Applications. Physiol. Rev. 2012, 92, 897–965.
- Bobo, D.; Robinson, K.J.; Islam, J.; Thurecht, K.J.; Corrie, S.R. Nanoparticle-Based Medicines: A Review of FDA-Approved Materials and Clinical Trials to Date. Pharm. Res. 2016, 33, 2373–2387.
- Lecocq, Q.; De Vlaeminck, Y.; Hanssens, H.; D’Huyvetter, M.; Raes, G.; Goyvaerts, C.; Keyaerts, M.; Devoogdt, N.; Breckpot, K. Theranostics in immuno-oncology using nanobody derivatives. Theranostics 2019, 9, 7772–7791.
- Jong, W.H.; Borm, P.J.A. Drug delivery and nanoparticles: Applications and hazards. Int. J. Nanomed. 2008, 3, 133–149.
- Wu, J. The Enhanced Permeability and Retention (EPR) Effect: The Significance of the Concept and Methods to Enhance Its Application. J. Pers. Med. 2021, 11, 771.
- Fu, Z.; Xiang, J. Aptamer-Functionalized Nanoparticles in Targeted Delivery and Cancer Therapy. Int. J. Mol. Sci. 2020, 21, 9123.
- Andreou, C.; Pal, S.; Rotter, L.; Yang, J.; Kircher, M.F. Molecular Imaging in Nanotechnology and Theranostics. Mol. Imaging Biol. 2017, 19, 363–372.
- Cai, X.; Zhu, Q.; Zeng, Y.; Zeng, Q.; Chen, X.; Zhan, Y. Manganese Oxide Nanoparticles As MRI Contrast Agents In Tumor Multimodal Imaging And Therapy. Int. J. Nanomed. 2019, 14, 8321–8344.
- Yousaf, T.; Dervenoulas, G.; Politis, M. Advances in MRI Methodology. Int. Rev. Neurobiol. 2018, 141, 31–76.
- Bashir, M.R.; Bhatti, L.; Marin, D.; Nelson, R.C. Emerging applications for ferumoxytol as a contrast agent in MRI. J. Magn. Reson. Imaging 2014, 41, 884–898.
- Lux, J.; Sherry, A.D. Advances in gadolinium-based MRI contrast agent designs for monitoring biological processes in vivo. Curr. Opin. Chem. Biol. 2018, 45, 121–130.
- Yang, X.; Atalar, E. MRI-guided gene therapy. FEBS Lett. 2006, 580, 2958–2961.
- Liu, C.; Ewert, K.K.; Wang, N.; Li, Y.; Safinya, C.R.; Qiao, W. A multifunctional lipid that forms contrast-agent liposomes with dual-control release capabilities for precise MRI-guided drug delivery. Biomaterials 2019, 221, 119412.
- Shi, J.; Wang, B.; Chen, Z.; Liu, W.; Pan, J.; Hou, L.; Zhang, Z. A Multi-Functional Tumor Theranostic Nanoplatform for MRI Guided Photothermal-Chemotherapy. Pharm. Res. 2016, 33, 1472–1485.
- Langereis, S.; Geelen, T.; Grüll, H.; Strijkers, G.; Nicolay, K. Paramagnetic liposomes for molecular MRI and MRI-guided drug delivery. NMR Biomed. 2013, 26, 728–744.
- Han, Z.; Liu, G. CEST MRI trackable nanoparticle drug delivery systems. Biomed. Mater. 2021, 16, 024103.
- Satpathy, M.; Wang, L.; Zielinski, R.J.; Qian, W.; Wang, Y.A.; Mohs, A.; Kairdolf, B.A.; Ji, X.; Capala, J.; Lipowska, M.; et al. Targeted Drug Delivery and Image-Guided Therapy of Heterogeneous Ovarian Cancer Using HER2-Targeted Theranostic Nanoparticles. Theranostics 2019, 9, 778–795.
- Tomitaka, A.; Arami, H.; Ahmadivand, A.; Pala, N.; McGoron, A.J.; Takemura, Y.; Febo, M.; Nair, M. Magneto-plasmonic nanostars for image-guided and NIR-triggered drug delivery. Sci. Rep. 2020, 10, 10115.
- Zhou, H.; Qian, W.; Uckun, F.M.; Wang, L.; Wang, Y.A.; Chen, H.; Kooby, D.; Yu, Q.; Lipowska, M.; Staley, C.A.; et al. IGF1 receptor targeted theranostic nanoparticles for targeted and image-guided therapy of pancreatic cancer. ACS Nano 2015, 9, 7976–7991.
- Wahajuddin; Arora, S. Superparamagnetic iron oxide nanoparticles: Magnetic nanoplatforms as drug carriers. Int. J. Nanomed. 2012, 7, 3445–3471.
- Dadfar, S.M.; Roemhild, K.; Drude, N.I.; Von, S. Europe PMC Funders Group Iron Oxide Nanoparticles: Diagnostic, Therapeutic and Theranostic Applications. Gene 2020, 138, 302–325.
- Wirth, T.; Parker, N.; Ylä-Herttuala, S. History of gene therapy. Gene 2013, 525, 162–169.
- Smith, E.; Blomberg, P. Gene therapy—From idea to reality. Lakartidningen 2017, 114, EWYL. (In Swedish)
- Sun, W.; Shi, Q.; Zhang, H.; Yang, K.; Ke, Y.; Wang, Y.; Qiao, L. Advances in the techniques and methodologies of cancer gene therapy. Discov. Med. 2019, 27, 45–55.
- Mohammadinejad, R.; Dadashzadeh, A.; Moghassemi, S.; Ashrafizadeh, M.; Dehshahri, A.; Pardakhty, A.; Sassan, H.; Sohrevardi, S.-M.; Mandegary, A. Shedding light on gene therapy: Carbon dots for the minimally invasive image-guided delivery of plasmids and noncoding RNAs—A review. J. Adv. Res. 2019, 18, 81–93.
- Peng, Y.; Gao, Y.; Yang, C.; Guo, R.; Shi, X.; Cao, X. Low-Molecular-Weight Poly(ethylenimine) Nanogels Loaded with Ultrasmall Iron Oxide Nanoparticles for T1-Weighted MR Imaging-Guided Gene Therapy of Sarcoma. ACS Appl. Mater. Interfaces 2021, 13, 27806–27813.
- Wang, Z.; Chang, Z.; Lu, M.; Shao, D.; Yue, J.; Yang, D.; Zheng, X.; Li, M.; He, K.; Zhang, M.; et al. Shape-controlled magnetic mesoporous silica nanoparticles for magnetically-mediated suicide gene therapy of hepatocellular carcinoma. Biomaterials 2017, 154, 147–157.
- Wang, R.; Dai, X.; Duan, S.; Zhao, N.; Xu, F.-J. A flexible bowl-shaped magnetic assembly for multifunctional gene delivery systems. Nanoscale 2019, 11, 16463–16475.
- Feng, J.; Xu, Z.; Liu, F.; Zhao, Y.; Yu, W.; Pan, M.; Wang, F.; Liu, X. Versatile Catalytic Deoxyribozyme Vehicles for Multimodal Imaging-Guided Efficient Gene Regulation and Photothermal Therapy. ACS Nano 2018, 12, 12888–12901.
- Mu, X.; Li, J.; Yan, S.; Zhang, H.; Zhang, W.; Zhang, F.; Jiang, J. siRNA Delivery with Stem Cell Membrane-Coated Magnetic Nanoparticles for Imaging-Guided Photothermal Therapy and Gene Therapy. ACS Biomater. Sci. Eng. 2018, 4, 3895–3905.
- Li, X.; Lovell, J.F.; Yoon, J.; Chen, X. Clinical development and potential of photothermal and photodynamic therapies for cancer. Nat. Rev. Clin. Oncol. 2020, 17, 657–674.
- Yoon, H.Y.; Jeon, S.; You, D.G.; Park, J.H.; Kwon, I.C.; Koo, H.; Kim, K. Inorganic Nanoparticles for Image-Guided Therapy. Bioconjug. Chem. 2017, 28, 124–134.
- Shi, M.; Zuo, F.; Taoa, Y.; Liua, Y.; Lua, J.; Zhenga, S.; Lub, J.; Houcd, P.; Liab, J.; Xuab, K. Near-infrared laser-induced phase-shifted nanoparticles for US/MRI-guided therapy for breast cancer. Colloids Surf. B Biointerfaces 2020, 196, 111278.
- Zhou, X.; Lv, X.; Zhao, W.; Zhou, T.; Zhang, S.; Shi, Z.; Ye, S.; Ren, L.; Chen, Z. Porous MnFe2O4-decorated PB nanocomposites: A new theranostic agent for boosted T1/T2 MRI-guided synergistic photothermal/magnetic hyperthermia. RSC Adv. 2018, 8, 18647–18655.
- Wang, J.; Mei, T.; Liu, Y.; Zhang, Y.; Zhang, Z.; Hu, Y.; Wang, Y.; Wu, M.; Yang, C.; Zhong, X.; et al. Dual-targeted and MRI-guided photothermal therapy via iron-based nanoparticles-incorporated neutrophils. Biomater. Sci. 2021, 9, 3968–3978.
- Zhang, L.; Yang, Z.; Zhu, W.; Ye, Z.; Yu, Y.; Xu, Z.; Ren, J.; Li, P. Dual-Stimuli-Responsive, Polymer-Microsphere-Encapsulated CuS Nanoparticles for Magnetic Resonance Imaging Guided Synergistic Chemo-Photothermal Therapy. ACS Biomater. Sci. Eng. 2017, 3, 1690–1701.
- Wang, Z.; Wang, Y.; Guo, H.; Yu, N.; Ren, Q.; Jiang, Q.; Xia, J.; Peng, C.; Zhang, H.; Chen, Z. Synthesis of one-for-all type Cu5FeS4 nanocrystals with improved near infrared photothermal and Fenton effects for simultaneous imaging and therapy of tumor. J. Colloid Interface Sci. 2021, 592, 116–126.
- Vilas-Boas, V.; Carvalho, F.; Espiña, B. Magnetic Hyperthermia for Cancer Treatment: Main Parameters Affecting the Outcome of In Vitro and In Vivo Studies. Molecules 2020, 25, 2874.
- Lu, Y.; Rivera-Rodriguez, A.; Tay, Z.W.; Hensley, D.; Fung, K.B.; Colson, C.; Saayujya, C.; Huynh, Q.; Kabuli, L.; Fellows, B.; et al. Combining magnetic particle imaging and magnetic fluid hyperthermia for localized and image-guided treatment. Int. J. Hyperth. 2020, 37, 141–154.
- Jose, J.; Kumar, R.; Harilal, S.; Mathew, G.E.; Parambi, D.G.T.; Prabhu, A.; Uddin, S.; Aleya, L.; Kim, H.; Mathew, B. Magnetic nanoparticles for hyperthermia in cancer treatment: An emerging tool. Environ. Sci. Pollut. Res. 2019, 27, 19214–19225.
- Gavilán, H.; Avugadda, S.K.; Fernández-Cabada, T.; Soni, N.; Cassani, M.; Mai, B.T.; Chantrell, R.; Pellegrino, T. Magnetic nanoparticles and clusters for magnetic hyperthermia: Optimizing their heat performance and developing combinatorial therapies to tackle cancer. Chem. Soc. Rev. 2021, 50, 11614–11667.
- Zhang, Y.; Wang, X.; Chu, C.; Zhou, Z.; Chen, B.; Pang, X.; Lin, G.; Lin, H.; Guo, Y.; Ren, E.; et al. Genetically engineered magnetic nanocages for cancer magneto-catalytic theranostics. Nat. Commun. 2020, 11, 5421.
- Tsiapla, A.-R.; Kalimeri, A.-A.; Maniotis, N.; Myrovali, E.; Samaras, T.; Angelakeris, M.; Kalogirou, O. Mitigation of magnetic particle hyperthermia side effects by magnetic field controls. Int. J. Hyperth. 2021, 38, 511–522.
- Tishin, A.M.; Shtil, A.A.; Pyatakov, A.P.; Zverev, V.I. Developing Antitumor Magnetic Hyperthermia: Principles, Materials and Devices. Recent Pat. Anti-Cancer Drug Discov. 2016, 11, 360–375.
- Perecin, C.J.; Gratens, X.P.M.; Chitta, V.A.; Leo, P.; de Oliveira, A.M.; Yoshioka, S.A.; Cerize, N.N.P. Synthesis and Characterization of Magnetic Composite Theragnostics by Nano Spray Drying. Materials 2022, 15, 1755.
- Anani, T.; Rahmati, S.; Sultana, N.; David, A.E. MRI-traceable theranostic nanoparticles for targeted cancer treatment. Theranostics 2021, 11, 579–601.
- Morris, B.J.; Willcox, D.C.; Donlon, T.A.; Willcox, B.J. FOXO3: A major gene for human longevity-a mini-review. Gerontology 2015, 61, 515–525.
- Yang, H.; He, Y.; Wang, Y.; Yang, R.; Wang, N.; Zhang, L.-M.; Gao, M.; Jiang, X. Theranostic Nanoparticles with Aggregation-Induced Emission and MRI Contrast Enhancement Characteristics as a Dual-Modal Imaging Platform for Image-Guided Tumor Photodynamic Therapy. Int. J. Nanomed. 2020, 15, 3023–3038.
- Lux, F.; Sancey, L.; Bianchi, A.; Crémillieux, Y.; Roux, S.; Tillement, O. Gadolinium-based nanoparticles for theranostic MRI-radiosensitization. Nanomedicine 2015, 10, 1801–1815.
- Fan, M.; Han, Y.; Gao, S.; Yan, H.; Cao, L.; Li, Z.; Liang, X.-J.; Zhang, J. Ultrasmall gold nanoparticles in cancer diagnosis and therapy. Theranostics 2020, 10, 4944–4957.
- Ding, X.; Liu, J.; Li, J.; Wang, F.; Wang, Y.; Song, S.; Zhang, H. Polydopamine coated manganese oxide nanoparticles with ultrahigh relaxivity as nanotheranostic agents for magnetic resonance imaging guided synergetic chemo-/photothermal therapy. Chem. Sci. 2016, 7, 6695–6700.
More
Information
Subjects:
Oncology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Entry Collection:
Biopharmaceuticals Technology
Revisions:
2 times
(View History)
Update Date:
24 Aug 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





