Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Maryam Alsoufi | -- | 4194 | 2022-08-17 16:17:09 | | | |
| 2 | Catherine Yang | Meta information modification | 4194 | 2022-08-18 03:23:07 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Al-Soufi, M.H.; Alshwyeh, H.A.; Alqahtani, H.; Al-Zuwaid, S.K.; Al-Ahmed, F.O.; Al-Abdulaziz, F.T.; Raed, D.; Hellal, K.; Nani, N.H.M.; Zubaidi, S.N.; et al. Apricot. Encyclopedia. Available online: https://encyclopedia.pub/entry/26248 (accessed on 19 May 2026).
Al-Soufi MH, Alshwyeh HA, Alqahtani H, Al-Zuwaid SK, Al-Ahmed FO, Al-Abdulaziz FT, et al. Apricot. Encyclopedia. Available at: https://encyclopedia.pub/entry/26248. Accessed May 19, 2026.
Al-Soufi, Maryam Haroon, Hussah Abdullah Alshwyeh, Haifa Alqahtani, Safa Khalil Al-Zuwaid, Fatimah Othman Al-Ahmed, Fatima Taher Al-Abdulaziz, Daniya Raed, Khaoula Hellal, Nurul Hidayah Mohd Nani, Siti Norliyana Zubaidi, et al. "Apricot" Encyclopedia, https://encyclopedia.pub/entry/26248 (accessed May 19, 2026).
Al-Soufi, M.H., Alshwyeh, H.A., Alqahtani, H., Al-Zuwaid, S.K., Al-Ahmed, F.O., Al-Abdulaziz, F.T., Raed, D., Hellal, K., Nani, N.H.M., Zubaidi, S.N., Asni, N.S.M., Hamezah, H.S., Kamal, N., Al-Muzafar, H., & Mediani, A. (2022, August 17). Apricot. In Encyclopedia. https://encyclopedia.pub/entry/26248
Al-Soufi, Maryam Haroon, et al. "Apricot." Encyclopedia. Web. 17 August, 2022.
Copy Citation
Fruits maintain the image as the richest sources of vitamins. Focusing on apricots, utilization of apricot species for many applications is possible due to its various benefits. Many research studies demonstrated different perspectives of apricot, especially in medical used as it can act as antioxidant, anti-inflammatory, and antimicrobial agents. Moreover, in the industrial sectors, apricots can be used in the production of biofuels and batteries.
apricot
bioactive compounds
health benefit
industrial application
1. Nutritional Values of Apricots
Apricot has been phytochemically studied because of its nutritional value (sugars, organic acids, and minerals) and nutraceutical traits (total phenolic, total flavonoids, total carotenoid, and antioxidant activity) and because it is rich in bioactive phytochemicals that contribute to medicinal health benefits [1]. It also characterised by its morphological aspects (fruit weight, flesh/seed ratio, fruit firmness, and colour index). Apricot fruits are a rich source of fibres that prevents constipation and stimulates normal gastric motility [2]. Soluble fibre keeps blood sugar level stable by lowering blood cholesterol, and helps in reducing body weight [3][4][5][6][7][8].
Apricot fruits are a good source of sugars, including sucrose, glucose, fructose, and maltose. Vitamins C, E, B6, and A, as well as minerals and trace elements, are mostly represented by potassium, phosphorus, calcium, and magnesium with minor amounts of iron, sodium, and zinc. The most major organic acids found in apricots are malic acid (500–900 mg/100 g), citric acid (30–50 mg/100 g), and tartaric acid [9]. The kernel contains 14.1 to 45.3% protein, with the primary proteins being albumin, globulin, glutelin, and prolamin with 84.7, 7.65, 3.54, and 1.17%, respectively [10].
Apricots can be consumed fresh or as dried fruit, both of which contain nutritional values. Nutritional characteristics are highly dependent on the apricot cultivars, cultivation systems, and storage conditions [4]. Apart from their nutritional value, apricots contain phenolic compounds, such as chlorogenic acid, catechin, epicatechin, and rutin [10]. The importance of these compounds is to increase the antioxidant activity and are required in food because of the possible relationship between their content and the lower incidence rates of cancer and cardiovascular diseases [5].
The amount of organic acid is significantly different in Malatya apricots in which Iğdır and Bursa varieties has the highest amount of citric acid meanwhile malic acid content is high in Cataloğlu and Hasanbey [9]. Furthermore, among the Malatya apricot types studied, the Hasanbey variety exhibited the highest vitamin C content. In certain study, it reveals that Malatya apricot exhibit more vitamin C compared to other apricots varieties [11]. A study showed that the main sugar in apricot are glucose, sucrose and fructose in which all type of sugars is significantly different in various apricots [12]. Based on study, sucrose is found as predominant sugar in Malatya apricot. Hacıhaliloğlu and Tokaloğlu varieties contain the most amount of sucrose and the highest level of fructose can be found in Cöloğlu and Cataloğlu varieties. The higher sugar content and lower organic acid concentrations found in Malatya apricots provide the ideal sweetness and flavour [9]. Moreover, macro and micro elements of mineral also can be found in apricot varieties in which potassium is the most mineral that can be found and sodium is the least one for macro elements. Study has discovered that apricot is one the fruits with good source of minerals hence, it can achieve the demand of mineral needed by average adult [13].
Organic acids (OA) and sugars contribute greatly to fruit sensory consistency by adding a pleasant taste and scent [1]. The OA are involved in a variety of bio-logical processes. They inhibit the growth of bacteria, which aids in the preservation of fruits. Furthermore, OA can permeate across cell membranes and breakdown into subsequent ions and protons, hastening the onset of metabolic problems in cells caused by increasing intercellular acidity [14]. Furthermore, these OA can help to stabilise water-soluble vitamins B and C, as well as stimulate appetite and digestion, as well as absorption of minerals including potassium, copper, zinc, iron, and calcium. Additionally, due to their ability to chelate metals, OA may act as antioxidants, earning them the designation of synergistic or preventive. Antioxidants play a major role on the protection of many diseases including cardiovascular diseases, cancer, and inflammation [15].
To extract antioxidants from apricot, dried apricots were rehydrated at room temperature for 24 h [7][16]. To replicate the water content of fresh fruit, samples were rehydrated with the exact amount of water lost during the drying process. The significant loss of water during the drying process resulted in a concentration of the various apricot components, indicating the increase in ash content and acidity [6]. Prior to analysis, samples were homogenised for 2 min. The moisture (MC) and dry matter (DM) content of the obtained purees were determined. They were quantified in a vacuum oven set to 105 °C for 3 h (for fresh, dried, and rehydrated fruit), and the pH was determined (using a digital pH metre) according the previous studies methods [17][18] Then, titration with 0.1 N sodium hydroxide to an endpoint was used to determine the acidity (pH 8.3) and also the ash content was determined after five hours at 550 °C in a muffle furnace (%).
2. Biological Activities of Apricot
2.1. Antioxidants
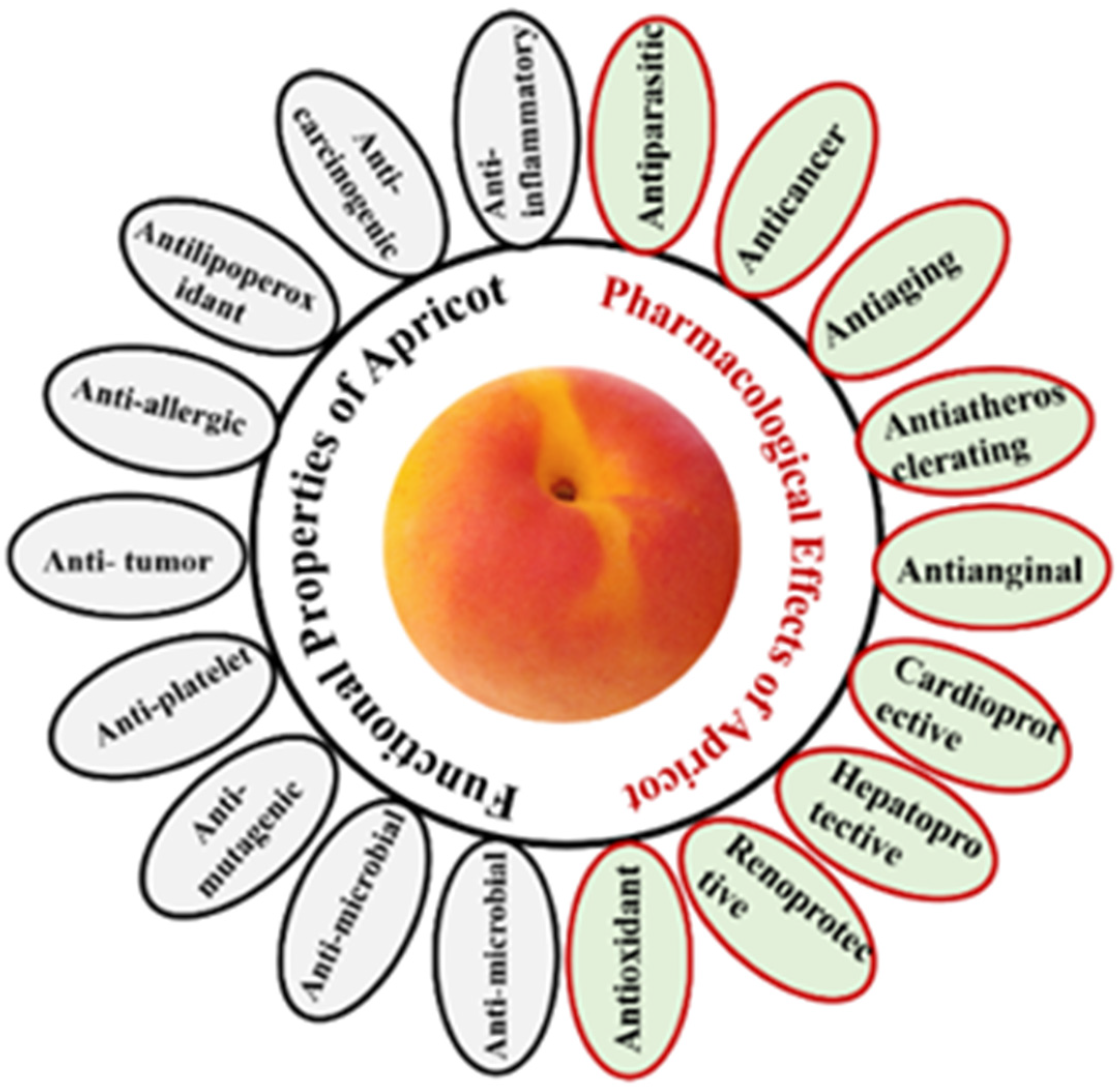
Cardiovascular diseases are the most common cause of death worldwide. Antioxidants components in apricots known for their effectivity in combating coronary heart diseases. Phenolic components in apricots combat the oxidation of low-density lipoprotein (LDL) and thus stimulate the antioxidative status of the body [3]. Figure 1 presents several functional properties and pharmacological effects of apricot as have been reported [2][8]. Apricot vitamins have a high capacity for scavenging free radicals and are found in all apricot species. Apricots’ antioxidant activity benefits a variety of medical conditions by enhancing the body’s defence mechanisms against free radicals and reducing the oxidative effect of free radicals [15][19]. Numerous studies reported shows the great potential of apricot for fighting off free radicals, and thus it considered as a functional food [3].
These conditions include chronic gastritis, oxidative intestinal damage, hepatic steatosis, atherosclerosis, coronary heart disease, and tumour formation. Additionally, apricots contain soluble dietary fibres that contribute to LDL cholesterol reduction [3], resulting in improved heart and liver health. Numerous apricot species are high in vitamins, which aid in the antioxidant pathways that help prevent a variety of illnesses and diseases including degenerative diseases, i.e., cancer, cardiovascular and haemostasis [2].
Flavonoids also have an effective function for fighting off free radicals. Being used as antioxidants for the benefit of living organisms. Measuring total flavonoids in apricot kernels showed promising results [5][20]. Several valuable effects of flavonoids in diseases including cardiovascular, some forms of cancer, Parkinson’s and Alzheimer’s diseases have been reported [21]. Carotenoids act as antioxidants and play a crucial role by fighting the reactive oxygen species that cause oxidative damage to living cells [3]. The total flavonoid content (TFC) of various apricot cultivars were tested and the cultivar Bora possessed the highest value of TFC with 153 mg CE/100 g dry weight while no flavonoid content was detected in the Congat and Velikyj cultivars [10].
DPPH radical scavenging activity of apricot varieties are significantly different. Study on 14 different types of apricot including Halma, Rakchekarpo, Khante, Shakanda, Viva Gold, CITH-1, CITH-2, Newcastle, Turkey, Nugget, Venatchaa, Shakarpara, Rakausilk, and Sterling showed that Rakausilk variety possesses high antioxidant activity followed by Viva Gold and Rkchekarpo. Furthermore, CITH-1 appears to have low activity of radical scavenging meanwhile Nugget and Shakarpa variety has no significant difference [22].
2.2. Anti-Inflammatory Activity
Apricot seeds and kernels play a major part as an anti-inflammatory. Apricots contain many significant compounds that reduce inflammation in diseased tissues of animals and humans. A study was made in 2018 for the anti-inflammatory effect of apricot 70% and 99.9% ethanolic extracts of apricot seeds in formalin induced paw edema in rats indicated significant reduction in the inflamed tissues (Figure 1) [23]. Studies support the presence and activity of fatty acids in apricot kernel oil with anti-inflammatory properties [5].
2.3. Anticancer Effect of Apricot
Cancer is currently the most prevalent kind of degenerative illness; it is a group of diseases characterized by unrestricted cellular growth and the spread of aberrant cells to other sections of the body. The cancer hallmarks are biological characteristics that evolve during tumour development [3]. These include insensitivity to anti-proliferative drugs, apoptosis avoidance, infinite reproduction, inducing angiogenesis, and beginning invasion of local tissues and metastasis to distant regions [24][25][26][27][28].
The use of complementary and alternative medicine is becoming more popular. CAM has evolved gradually over the previous 15 years and has undeniably gained therapeutic, commercial, and sociological significance. Dietary supplements are the most popular CAM treatments. In apricot kernels the main component to have major effects on cancer is Amygdalin. It was used as an anticancer agent since the 1800s [2]. Amygdalin is a naturally occurring anti-cancer substance that is classified as a β-cyanogenic glycoside found in abundance in Rosaceae family members such as almonds, apricots, apples, and peaches. It is thought that amygdalin’s anticancer properties are attributable mostly to its active metabolite, hydrocyanic acid [29]. Amygdalin is being examined extensively for its potency in cancer cells. Human prostate cancer cells (DU145 and LNCap) exhibited multiple apoptotic markers after amygdalin treatment, including increased Bax generation and decreased Bcl-2 expression, leading to caspase-3 activation.
Another investigation found that amygdalin alone had suppressive properties and its components (activated with β-d-glucosidase) on proliferation rate of human hepatocellular carcinoma cells [30], i.e., HepG2. Its activated components had a higher apoptotic efficiency than amygdalin. Apoptosis’ specific mechanism is still being researched. This combo method may allow for the use of a low-dose medicine. The extract of Japanese apricots or Prunus mume, contained MK615, which inhibits the growth of cancer by dual inhibition of Aurora A and B kinases [31]. MK615 exerts anti-neoplastic effects on several known cancer growths including gastric, breast, and colon cancers and hepatocellular carcinoma. It is also known to induce apoptosis, programmed cell death, autophagy and suppresses A kinase production in cancer growth.
As a result of the anti-inflammatory and antioxidative actions of MK615, the hepatoprotective effects of MK615 was investigated [32]. Furthermore, the benefits of Misatol ME, an MK615-containing beverage approved as a health food product in Japan, were studied in patients with liver illnesses such as hepatitis C, chronic inflammation of the liver, and fatty liver disease, which is linked to oxidative stress.
An investigation had been carried out on 19 apricot varieties from northern Pakistan and Balaani variety has the highest amount of amygdalin meanwhile Staa Chuli contains the lowest content of amygdalin. Amygdalin content commonly appears high in less sweet cultivars [33].
2.4. Biological Activity of Apricot Leaves
Some of the leaves have been reported to be utilised as traditional treatments by various indigenous populations for the management of rheumatism, asthma, colds, diabetes and urinary tract inflammation. Certain studies stated that the leaves possessed property of antimicrobial, antidiabetic, anticancer, anti-inflammatory, antioxidant, and hypo-cholesterol activities.
3. Medicinal Properties of Japanese Apricot (Prunus mume)
The Japanese apricot (Prunus mume) enjoys great popularity in Japan. In addition, known as Chinese plum, belongs to Prunes Genus as does the peach, cherries, plum, almonds [34]. It is native to Japan and Korea and is widely planted throughout China. Prunus mume is a traditional medicinal herb and health food that has been utilised in Asian countries for over 2000 years because of its health benefits. P. mume’s fruit is used to make pickled plums, plum sauce, plum juice, and plum liquor, which can be eaten as a snack, condiment, or beverage. [35]. It is also economically valuable, as it can be processed into juice and wine. These products have been known to possess various medicinal benefits and have been frequently prescribed as a traditional folk remedy. It has been reported to possess such beneficial biological activities. This Japanese apricot is known to have anti-microbial properties and it is the only one well established for various medicinal purposes.
3.1. Phytochemical Constituents
The total chemical compounds have been identified in the P. mume was 192. Flower buds, fruit, wood, petal, and seed are all high in phenolic components. Phenylpropanoid sucrose esters, hydroxycinnamoylquinic acid derivatives, flavonoids, and other phenolics can all be subdivided. In general, organic acid, steroids, terpenes, lignans, furfurals, benzyl glycosides, cyanogenic glycosides, and alkaloids are among the medicinal chemical components found in the fruit [35].
3.2. Medicinal Usage of P. mume Phenolic Compound
3.2.1. Antidiabetic
Diabetes Mellitus (DM) is one of the most common metabolic illnesses in the world. Hyperglycemia is characterized by abnormal insulin production and insulin resistance. Obesity, which is generally accompanied with hyperglycemia and insulin resistance, is one of the most significant risk factors for DM. An aqueous extract of P. mume fruit combined with Lithospermum erythrorhizon root increased insulin sensitivity and reduced visceral obesity in rats fed a high-fat diet (HFD). Furthermore, a 70% ethanol extract of P. mume fruit and leaves has been shown to increase glucose uptake in C2C12 myotubes, improve fasting glucose levels and glucose intolerance, reduce body weight, and lower blood glucose levels in a dose-dependent manner; this activity is attributed to the phenolic compound. In addition, phenolic extracts of P. mume inhibited small intestine disaccharide activity and raised postprandial blood glucose levels in rats. The phenolic compounds extracted from the flower buds of P. mume show potential to prevent cataracts [35]. The phenolic extracts from apricot have cardioprotective activity in rats [2].
3.2.2. Helicobacter Pylori-Related Chronic Gastritis
Helicobacter pylori infection is linked to chronic gastritis. In vitro and in vivo, P. mume extract shows direct bactericidal activity against Helicobacter pylori [35]. H. pylori motility is required for migration and colonization. The active component syringaresinol isolated from P. mume inhibited H. pylori motility by 90% at a concentration of 500 mg/mL, with an IC50 value of 50 mg/mL, lowering its colonization in the stomach mucosa [36]. A study conducted on non-elderly individuals (65 years) with high-intake (X3 JA daily) significantly reduced H. pylori load, mononuclear, and neutrophil infiltration in the gastric mucosa, as well as the preventive effect of dried or pickled P. mume on chronic atrophic gastritis (CAG) by inhibiting the infection and reducing active mucosal inflammation. [37].
3.2.3. Antimicrobial and Antiviral Activity
Studies have shown that apricots possess a broad array of antibacterial activities. Consequently, two independent research groups have found that apricots prevent common periodontal bacterial infection, such as Aggregatibacter actinomycetemcomitans and Porphyromonas gingivalis as well as inhibit the growth of bacteria in the oral cavity [35]. Citric acid, chlorogenic acid, isoquercitrin asparagine, and epicatechin were isolated from ethanol extracts of P. mume [38]. These compounds exhibited antibiotic activities against five food-borne bacteria: Listeria monocytogenes, Salmonella enterica, Bacillus cereus, Staphylococcus aureus, and Escherichia coli. Compared to other compounds citric acid had higher-ranking activities against all five bacteria, chlorogenic acid inhibited four of the five bacteria, epicatechin inhibited Salmonella enterica and Listeria monocytogenes, while asparagine and isoquercitrin hindered Listeria monocytogenes only [39].
Moreover, P. mume with Schizandra chinenis H-20 show antibacterial activity against B. subtilis, S. aureus, E. coli KCCM 11591, and Pseudomonas aeruginosa KCTC 1750. The DNA of S. chinenis H-20 exhibits inhibition of B. subtilis, S. aureus, and E. coli KCCM 11591 at the beginning of cultivation and even after 5 h of initiating cultivation [36]. When P. mume, Schizandrae Fructus, and Coptidis Rhizoma combined, this led to the inhibition of some Escherichia coli and Salmonella strains. In addition, P. mume inhibits the release of toxins from some strains of E. coli. Furthermore, umesu phenolics obtained from P. mume revealed inhibition of the multiplication of herpes simplex virus (HSV) and might inhibit superficial HSV infections [35].
4. Industrial Utilization of Apricot Seed Waste and Kernels
4.1. Applications in Food Industry
Agricultural waste is generally underutilized, and their accumulation in the environment could lead to severe pollution if treated improperly. Apricot (Prunus armeniaca) is one of the world’s most extensively cultivated crops due to its nutritional value. One of the options to be explored is apricot stone, seed/kernel and shell. Food sources of protein, fat, and fibre can be found in apricot kernels. All parts of apricot are renowned for its numerous industrial applications, including food, cosmetic, and pharmacy as well as the thermal energy storage. Most critically, it appears that applications for apricot seeds and kernels in the food industries have been not well studied. The apricot kernel is very useful in the food industry for making low-fat biscuits, cakes, pastries, and antimicrobial films. Apricot kernel flour has been claimed to be a good source of minerals, protein, bioactive substances, and fibre, and it has mostly been utilized for product protection in the bakery business. Apricot kernels are primarily utilized in the manufacturing of oils and benzaldehyde; nevertheless, the kernels are also used to baked items, either whole or crushed, and eaten as an appetizer [40]. The kernel powder was defatted and employed as a protein source in yoghurt and ice cream in the proportions of 10–40% and 10–50%, respectively [41]. Similarly, the addition of apricot kernel powder to yoghurt reduced the lactic acid bacteria count, pH, and acetaldehyde value [42]. It was determined that skim milk could be substituted in yoghurt up to a 20% level [43]. Apricot kernel powder was found to improve the quality of stirred yoghurt in a study. Cow milk was replaced with one percent apricot kernel powder. The inclusion of apricot kernel powder enhanced the titratable acidity, ash content, total solids, and protein content [44]. To maintain the quality of the fortified products that are customised for consumers, further studies should also take optimization and the ideal ratio of each waste into account. It can be suggest using apricot seeds in the production of innovative juices rich in bioactive peptides, baked goods, veggie-meat-based products, and industrial fermentation to manufacture various food additives such as enzymes, proteins, and flavours.
Apricot kernels are a by-product of the food canning industry in vast numbers. The kernels are viewed as an unconventional source of oil due to their potential and are also rich in unsaturated fatty acids, vitamin E active compounds, triterpenoids, carotenoids, phytosterols, and polyphenols. The strong demand for and consumption of apricots are discarded yearly at processing plants [38] has helped alleviate the environmental problem caused by residue disposal or incineration. Additionally, food sustainability is a critical objective that must be achieved to avoid wasting natural resources and mitigate future effects on climate change and global economic shifts. Utilising apricot oils from seed waste and kernels has become economically viable due to the advent of an issue linked to a massive waste disposal problem of potentially valuable resources. This type of waste showed promising results in an industrial setting, and was utilized by its addition to certain materials, such as: food packaging, polyurethane foams and even batteries, as well as being used as potential feedstock to produce biofuels and activated carbon.
4.2. Apricot Seed Waste in Polymer Production
4.2.1. Food Packaging
Kernels of bitter apricots—a major agricultural waste—were used to extract apricot kernel essential oil (AKEO). One important component of which is N-methyl-2-pyrrolidone (NMP), a powerful antioxidant, antimicrobial agent, present in a significant quantity.
AKEO was incorporated with chitosan with the intent of producing food packaging films. Chitosan is an abundant biopolymer that exhibits non-toxic, antimicrobial and biodegradable properties [45], the extracted kernel oil was added to examine the extent of which these films could be improved. The films were prepared with the solvent casting method, and the incorporation of AKEO was verified by Fourier Transform-Infra Red (FT-IR) spectra, as well as Field Emission Scanning Electron Microscope (FE-SEM) images.
The revised films demonstrated better water resistance and 41% improved water vapor barrier quality, when chitosan to AKEO ratio is 1:1. The elongation percentage value considerably increased, with oil ratio of 0.125 with respect to chitosan, however, a significant drop resulted with further addition. Despite that, a continuous increase in tensile strength value with increasing oil concentrations and a 94% improvement were noticed in the film with equal AEKO to chitosan ratio. antimicrobial and antioxidant properties were noticeably greater in films embedded with AKEO compared to regular ones made without its addition, as they were successful in the inhibition of fungal growth on packaged bread pieces (Figure 2) [46].
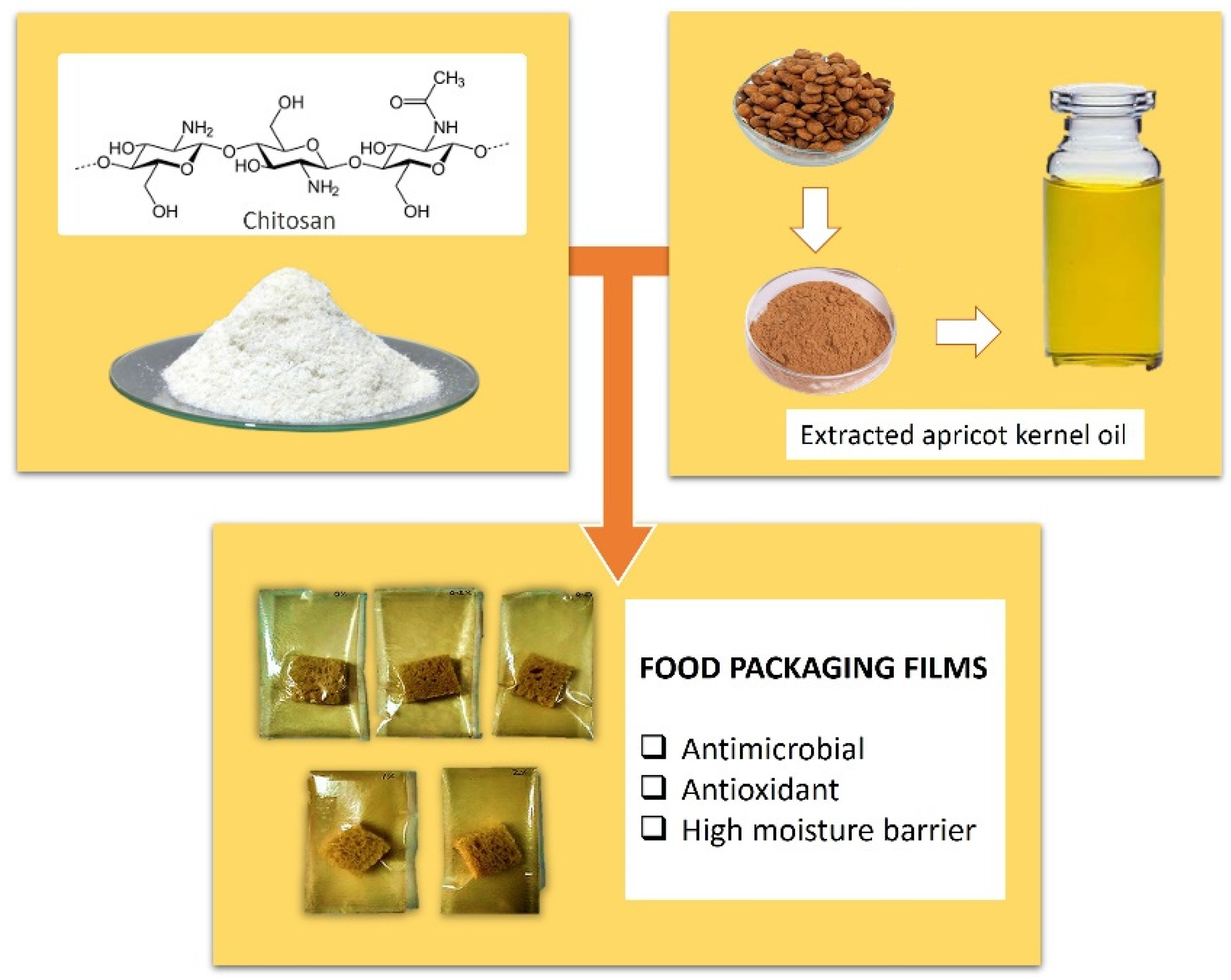
Figure 2. Chitosan films incorporated with AKEO [46].
4.2.2. Polyurethane Composites
Polyurethane (PUR) composites are broadly used in a variety of applications, which could be contributed to the various raw materials from which they could be acquired, such as: polyols and poly-isocyanates, with the addition of certain modifiers such as catalysts, fillers, flame retardants and more [47]. With the majority of the basic ingredients being acquired from non-renewable petrochemical raw materials. PUR foams are usually obtained using the one step method, two-component system; component A is a polyol system with the addition of the aforementioned modifiers, component B is an isocyanate system. Isocyanates are highly reactive molecules with low molecular weight, that are usually associated with health issues [48]. They are primarily used as fillers in PUR foams, acting similar to a binder. Moreover, polyols and isocyanates could be obtained from natural waste, and the fatty acid rich composition of the apricot stone allows it, thus a natural renewable source is accessible. The filler considered was a combination of apricot stones—a source of bio-polyols and isocyanates—and casein which is a natural flame-retardant; due to presence of substantial amounts of nitrogen and phosphorus, which use proved effective in previous studies. This particular mixture was considered to investigate the possibility of simultaneously improving the mechanical properties and the reduction of flammability. The filler was prepared with mechanically ground apricot stones, that later were sieved and mixed with casein powder with 1:2 ratio—by weight [49]. Additionally, the filler underwent a physical modification. It was used as the reinforcing filler in the making of PUR composites (rigid PUR foams) and was incorporated at different weight percentages (1, 2 and 5 wt.%), the other components were also added by weight (Figure 3).

Figure 3. Making of PUR composites reinforced with apricot/casein filler [49].
4.3. Apricot Seed Waste in Biofuel and Activated Carbon Production
Oil was extracted from apricot seed kernel, with a yield of 49.44% w/w of kernels. The selection of potassium hydroxide (KOH) could be contributed to its properties as it has low activation temperatures and produces higher yields [50]. KOH was used as the catalyst for the transesterification—displacement of alcohol from an ester with apricot seed kernel oil with methanol and ethanol to produce methyl and ethyl, respectively. The characteristics of the acquired biodiesels were evaluated and found to comply with the American Society for Testing and Materials (ASTM) D6751 limits. Pyrolysis of the de-oiled seed kernel occurred in a semi-batch reactor for bio-oil production. Various parameters affecting the yield of bio-oil were studied: pyrolysis temperatures, pyrolysis time, feed particles size. The greatest production of bio-oil (43.66% w/w) was accomplished at: pyrolysis temperature 450 °C, pyrolysis time 60 min, feed particles size 0.25 mm [51].
The bio-oil acquired under the optimal conditions was characterised by elemental analysis, FTIR spectroscopy, as well as column chromatography. The FTIR analysis of the biofuel showed that it is composed mostly of alkanes, alkenes, ketones, carboxylic acids, and amines. The characteristics of the bio-oil were analysed with reference to calorific value, density, flash point, pH, acid value, pour point, and refractive index [51][52]. The characteristics were similar to those of petroleum fractions and comparable to those of different bio-oils published in previous literature. The resulting bio-oil can be employed to be used in place of traditional, commonly used fuel and chemicals.
The chemical activation method, using KOH as the activating agent was used to convert the biochar into activated carbon. The effect of the process parameters; the activation temperatures, activation time, and feed particles size on the yield, Iodine adsorption, as well as the surface area of the acquired activated carbon were studied. The better activated carbon sample was acquired at activation temperature 600 °C, activation time 90 min, feed size 60 mesh. The obtained activated carbon demonstrated good characteristics for the potential use in the removal of organic micro-pollutants from wastewater, as well as heavy metal adsorption, as demonstrated by scanning electron microscopy (SEM), as well as FTIR spectroscopy (Figure 4) [51][52][53].
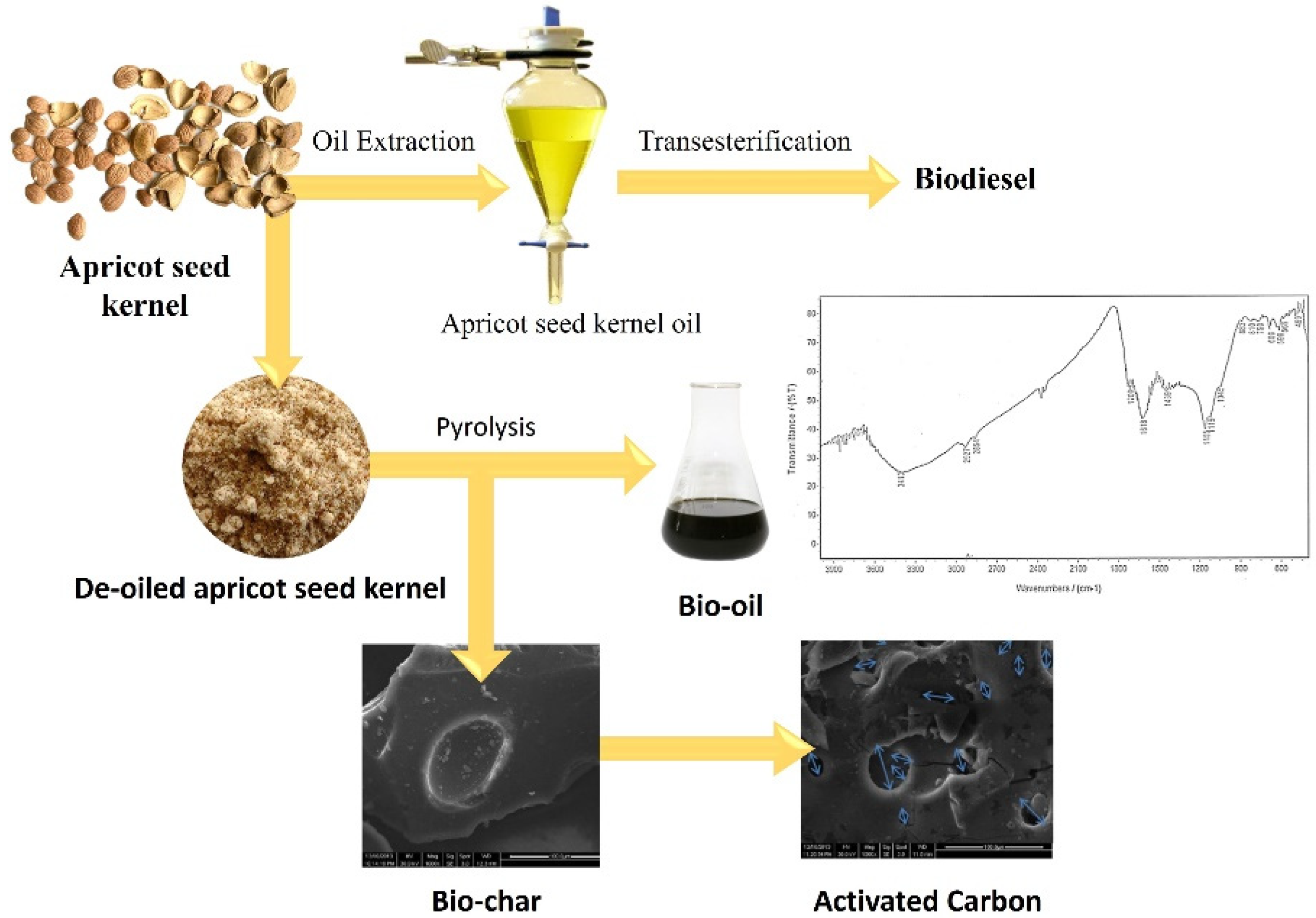
Figure 4. Production process of biofuel and activated carbon from apricot seed kernel [51].
4.4. Apricot Seed Waste in Batteries
Apricot shell waste was employed in this application and was fed to sodium ion batteries. Hydro-thermal carbonization was used to create hard carbon anode materials from apricot shell waste (HTC) [54][55]. Additional pyrolysis at various temperatures was used to achieve acceptable conductivity and a larger surface area, both of which are important for battery applications. Their goal was to have a surplus of sodium storage capacity values. Under HTC circumstances, SnO2 nanoparticles are also added to the generated carbons obtained from apricot shell waste. Over 250 cycles, maximum capacity values were obtained using 1000 °C treated hard carbon anode materials with 184 mAh/g of capacity. The SnO2 hard carbon anode preparation method results in significantly improved electrochemical characteristics. Mechanically combining SnO2 with hard carbon, on the other hand, results in quick fading capacity (Figure 5) [54].
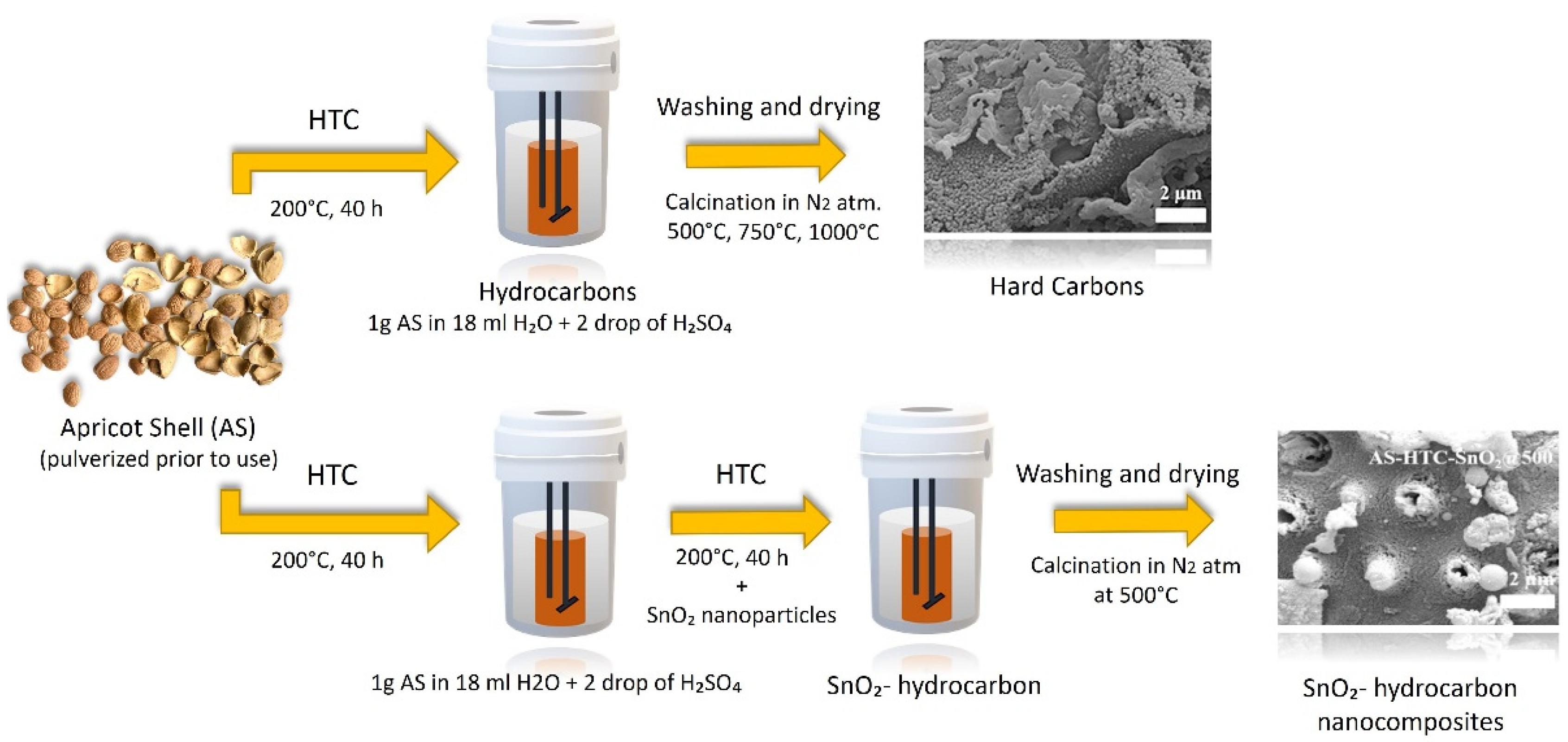
Figure 5. Production of anode materials for sodium ion batteries from apricot shell [54].
The use of agricultural waste such as seeds and kernels are a smart move, as they are frequently overlooked by-products despite their numerous characteristics. For example, they are non-edible and thus do not compete with the demand for food supply; they are also a renewable, cost-effective, and environmentally friendly source of energy. Rather than being disposed of, they could be repurposed to benefit humanity and alleviate some of the environmental burden. The use of such waste in food packaging is an excellent idea, as it comes from a renewable, biodegradable source that poses fewer environmental risks than conventional plastic. Additionally, its use as a filler in polyurethane materials would result in a more thermally stable and environmentally friendly product. Utilizing it as a feedstock to produce liquid biofuels may be a more accessible and cost-effective option. Incorporating apricot seed waste and utilizing it proved effective and with such promising results in each application, it would open doors for more in the future, and with that perhaps the environment would recover.
References
- Milošević, T.; Milošević, N.; Mladenović, J. Soluble solids, acidity, phenolic content and antioxidant capacity of fruits and berries cultivated in Serbia. Fruits 2016, 71, 239–248.
- Jaafar, H.J. Effects of Apricot and Apricot Kernels on Human Health and Nutrition: A Review of Recent Human Research. Tech. Biochem. 2021, 2, 139–162.
- Fatima, T.; Bashir, O.; Gani, G.; Bhat, T.; Jan, N. Nutritional and Health Benefits of Apricots. Int. J. Unani Integr. Med. 2018, 2, 5–9.
- Moustafa, K.; Cross, J. Production, Pomological and Nutraceutical Properties of Apricot. J. Food Sci. Technol. 2019, 56, 12–23.
- Göttingerová, M.; Kumšta, M.; Rampáčková, E.; Kiss, T.; Nečas, T. Analysis of Phenolic Compounds and Some Important Analytical Properties in Selected Apricot Genotypes. HortScience 2021, 56, 1446–1452.
- eddine Derardja, A.; Barkat, M. Effect of Traditional Sun-Drying and Oven-Drying on Carotenoids and Phenolic Compounds of Apricot (Prunus armeniaca L.). N. Afr. J. Food Nutr. Res. 2019, 3, 186–194.
- Hasan, M.U.; Malik, A.U.; Ali, S.; Imtiaz, A.; Munir, A.; Amjad, W.; Anwar, R. Modern Drying Techniques in Fruits and Vegetables to Overcome Postharvest Losses: A Review. J. Food Process. Preserv. 2019, 43, 12.
- Gupta, S.; Chhajed, M.; Arora, S.; Thakur, G.; Gupta, R. Medicinal Value of Apricot: A Review. Indian J. Pharm. Sci. 2018, 80, 790–794.
- Akin, E.B.; Karabulut, I.; Topcu, A. Some Compositional Properties of Main Malatya Apricot (Prunus armeniaca L.) Varieties. Food Chem. 2008, 107, 939–948.
- Rampáčková, E.; Göttingerová, M.; Gála, P.; Kiss, T.; Ercişli, S.; Nečas, T. Evaluation of Protein and Antioxidant Content in Apricot Kernels as a Sustainable Additional Source of Nutrition. Sustainability 2021, 13, 4742.
- Thompson, C.O.; Trenerry, V.C. A Rapid Method for the Determination of Total L-Ascorbic Acid in Fruits and Vegetables by Micellar Electrokinetic Capillary Chromatography. Food Chem. 1995, 53, 43–50.
- Asma, B.M.; Kan, T.; Birhanlı, O. Characterization of Promising Apricot (Prunus armeniaca L.) Genetic Resources in Malatya, Turkey. Genet. Resour. Crop Evol. 2007, 54, 205–212.
- Munzuroglu, O.; Karatas, F.; Geckil, H. The Vitamin and Selenium Contents of Apricot Fruit of Different Varieties Cultivated in Different Geographical Regions. Food Chem. 2003, 83, 205–212.
- Theron, M.M.; Lues, J.F.R. Organic Acids and Meat Preservation: A Review. Food Rev. Int. 2007, 23, 141–158.
- Karataş, N.; Şengül, M. Some Important Physicochemical and Bioactive Characteristics of the Main Apricot Cultivars from Turkey. Turkish J. Agric. For. 2020, 44, 651–661.
- Siddiq, M. Handbook of Fruits and Fruit Processing; John Wiley & Sons: New York, NY, USA, 2006; Volume 697, pp. 279–292.
- Muratore, G.; Rizzo, V.; Licciardello, F.; Maccarone, E. Partial Dehydration of Cherry Tomato at Different Temperature, and Nutritional Quality of the Products. Food Chem. 2008, 111, 887–891.
- Leiva Díaz, E.; Giannuzzi, L.; Giner, S.A. Apple Pectic Gel Produced by Dehydration. Food Bioprocess Technol. 2009, 2, 194–207.
- Hamid, A.A.; Aiyelaagbe, O.O.; Usman, L.A.; Ameen, O.M.; Lawal, A. Antioxidants: Its Medicinal and Pharmacological Applications. Afr. J. Pure Appl. Chem. 2010, 4, 142–151.
- Ozsahin, A.D.; Yilmaz, O. Fruit Sugar, Flavonoid and Phytosterol Contents of Apricot Fruits (Prunus armeniaca L. cv. Kabaasi) and Antioxidant Effects in the Free Radicals Environment. Asian J. Chem. 2010, 22, 6403–6412.
- Yilmaz, I. The Biological and Pharmacological İmportance of Apricot. SOJ Pharm. Pharm. Sci. 2018, 5, 1–4.
- Mohd Wani, S.; Rashid Hussain, P.; Ahmad Masoodi, F.; Ahmad, M.; Ahmad Wani, T.; Gani, A.; Ahmed Rather, S.; Suradkar, P. Evaluation of the Composition of Bioactive Compounds and Antioxidant Activity in Fourteen Apricot Varieties of North India. J. Agric. Sci. 2017, 9, 66–82.
- Ramadan, A.; Kamel, G.; Awad, N.E.; Shokry, A.A.; Fayed, H.M. The Pharmacological Effect of Apricot Seeds Extracts and Amygdalin in Experimentally Induced Liver Damage and Hepatocellular Carcinoma. J. Herbmed Pharmacol. 2020, 9, 400–407.
- Miyazawa, M.; Utsunomiya, H.; Inada, K.; Yamada, T.; Okuno, Y.; Tanaka, H.; Tatematsu, M. Inhibition of Helicobacter Pylori Motility by (+)-Syringaresinol from Unripe Japanese Apricot. Biol. Pharm. Bull. 2006, 29, 172–173.
- Mori, S.; Sawada, T.; Okada, T.; Ohsawa, T.; Adachi, M.; Keiichi, K. New Anti-Proliferative Agent, MK615, from Japanese Apricot “ Prunus Mume ” Induces Striking Autophagy in Colon Cancer Cells in Vitro. World J. Gastroenterol. 2007, 13, 6512.
- Okada, T.; Sawada, T.; Osawa, T.; Adachi, M.; Kubota, K. MK615 Inhibits Pancreatic Cancer Cell Growth by Dual Inhibition of Aurora A and B Kinases. World J. Gastroenterol. 2008, 14, 1378–1382.
- Matsushita, S.; Tada, K.; Kawahara, K.; Kawai, K.; Hashiguchi, T.; Maruyama, I.; Kanekura, T. Advanced Malignant Melanoma Responds to Prunus Mume Sieb. Et Zucc (Ume) extract: Case report and in vitro study. Exp. Ther. Med. 2010, 1, 569–574.
- Saleem, M.; Asif, J.; Asif, M.; Saleem, U. Amygdalin from Apricot Kernels Induces Apoptosis and Causes Cell Cycle Arrest in Cancer Cells: An Updated Review. Anticancer. Agents Med. Chem. 2018, 18, 1650–1655.
- Inoue, K.; Kawahara, K.I.; Biswas, K.K.; Ando, K.; Mitsudo, K.; Nobuyoshi, M.; Maruyama, I. HMGB1 Expression by Activated Vascular Smooth Muscle Cells in Advanced Human Atherosclerosis Plaques. Cardiovasc. Pathol. 2007, 16, 136–143.
- Chauhan, S.K.; Tyagi, S.M.; Singh, D. Pectinolytic Liquefaction of Apricot, Plum, and Mango Pulps for Juice Extraction. Int. J. Food Prop. 2001, 4, 103–109.
- Adachi, M.; Suzuki, Y.; Mizuta, T.; Osawa, T.; Suzuki, K.; Shiojima, K.; Arai, Y.; Masuda, K.; Uchiyama, M.; Oyamada, T. The Japanese Apricot ‘Prunus Mume Sieb. Zucc’ is a rich Natural Source of Novel Anti-Cancer Substance. Int. J. Food Prop. 2007, 10, 375–384.
- Houghton, A.; Polsky, D.F. Focus on Melanoma. Cancer Cell 2002, 2, 275–278.
- Chen, Y.; Al-Ghamdi, A.A.; Elshikh, M.S.; Shah, M.H.; Al-Dosary, M.A.; Abbasi, A.M. Phytochemical Profiling, Antioxidant and HepG2 Cancer Cells’ Antiproliferation Potential in the Kernels of Apricot Cultivars. Saudi J. Biol. Sci. 2020, 27, 163–172.
- Bortiri, E.; Sang-Hun, O.H.; Jiang, U.O.; Baggett, S.; Granger, A.; Weeks, C.; Buckingham, M.; Potter, D.; Parfitt, D.E. Phylogeny and Systematics of Prunus (Rosaceae) as Determined by Sequence Analysis of ITS and the Chloroplast TrnL-TrnF Spacer DNA. Syst. Bot. 2001, 26, 797–807.
- Gong, X.P.; Tang, Y.; Song, Y.Y.; Du, G.; Li, J. Comprehensive Review of Phytochemical Constituents, Pharmacological Properties, and Clinical Applications of Prunus Mume. Front. Pharmacol. 2021, 12, 679378.
- Kim, Y.-S.P. Antimicrobial Activity of Prunus Mume and Schizandra Chinenis H-20 Extracts and Their Effects on Quality of Functional Kochujang. Korean J. Food Sci. Technol. 2003, 35, 893–897.
- Enomoto, S.; Yanaoka, K.; Utsunomiya, H.; Niwa, T.; Inada, K.; Deguchi, H.; Ueda, K.; Mukoubayashi, C.; Inoue, I.; Maekita, T.; et al. Inhibitory Effects of Japanese Apricot (Prunus Mume Siebold et Zucc.; UME) on Helicobacter Pylori-Related Chronic Gastritis. Nat. News 2010, 64, 714–719.
- Femenia, A.; Rosselló, C.; Mulet, A.; Cañellas, J. Chemical Composition of Bitter and Sweet Apricot Kernels. J. Agric. Food Chem. 1995, 43, 356–361.
- Layne, R.E.C.; Bailey, C.H.; Hough, L.F. Apricots. In Fruit Breeding; Janick, J., Moore, J.N., Eds.; John Wiley & Sons, Inc.: New York, NY, USA, 1996; Volume 1, pp. 79–111.
- Fogarasi, S. Apricot Kernel: Bioactivity, Characterization, Applications, and Health Attributes. Foods 2022, 11, 2184.
- Hyun, S.-W.; Kim, J.; Park, B.; Jo, K.; Lee, T.G.; Kim, J.S.; Kim, C.-S. Molecules Apricot Kernel Extract and Amygdalin Inhibit Urban Particulate Matter-Induced Keratoconjunctivitis Sicca. Molecules 2019, 24, 650.
- Saini, D.; Rawat, N.; Negi, T.; Barthwal, R.; Sharma, S. Utilization, Valorization and Functional Properties of Wild Apricot Kernels. J. Pharmacogn. Phytochem. 2021, 10, 119–126.
- Raj, V.; Mishra, A.K.; Mishra, A.; Khan, N.A. Hepatoprotective Effect of Prunus armeniaca L. (Apricot) Leaf Extracts on Paracetamol Induced Liver Damage in Wistar Rats. Pharmacogn. J. 2016, 8, 154–158.
- Huang, C.; Tang, X.; Liu, Z.; Huang, W.; Ye, Y. Enzymes-Dependent Antioxidant Activity of Sweet Apricot Kernel Protein Hydrolysates. LWT 2022, 154, 112825.
- Aizat, M.; Aziz, F. Nanotechnology in Water and Wastewater Treatment; Elsevier: Amsterdam, The Netherlands, 2019; pp. 243–265.
- Priyadarshi, R.; Kumar, B.; Deeba, F.; Kulshreshtha, A.; Negi, Y. Chitosan Films Incorporated with Apricot (Prunus armeniaca) Kernel Essential Oil as Active Food Packaging Material. Food Hydrocoll. 2018, 85, 158–166.
- Sharmin, E.; Zafar, F. Polyurethane: An Introduction. In Polyurethane; Zafar, F., Sharmin, E., Eds.; IntechOpen: London, UK, 2012.
- Heath, R. Isocyanate-Based Polymers: Polyurethanes, Polyureas, Polyisocyanurates, and Their Copolymers; Elsevier Ltd.: Amsterdam, The Netherlands, 2017; ISBN 9780323358248.
- Członka, S.; Kairytė, A.; Miedzińska, K.; Strąkowska, A. Casein/Apricot Filler in the Production of Flame-Retardant Polyurethane Composites. Materials 2021, 14, 3620.
- Wang, J.; Kaskel, S. KOH Activation of Carbon-Based Materials for Energy Storage. J. Mater. Chem. 2012, 22, 23710–23725.
- Fadhil, A. Evaluation of Apricot (Prunus armeniaca L.) Seed Kernel as a Potential Feedstock for the Production of Liquid Bio-Fuels and Activated Carbons. Energy Convers. Manag. 2017, 133, 307–317.
- Lang, X.; Dalai, A.; Bakhshi, N.; Reaney, M.; Hertz, P. Preparation and Characterization of Bio-Diesels from Various Bio-Oils. Bioresour. Technol. 2001, 80, 53–62.
- Janković, B.; Manić, N.; Dodevski, V.; Radović, I.; Pijović, M.; Katnić, Đ.; Tasić, G. Physico-Chemical Characterization of Carbonized Apricot Kernel Shell as Precursor for Activated Carbon Preparation in Clean Technology Utilization. J. Clean. Prod. 2019, 236, 117614.
- Demir, E.; Aydin, M.; Arie, A.A.; Demir-Cakan, R. Apricot Shell Derived Hard Carbons and Their Tin Oxide Composites as Anode Materials for Sodium-Ion Batteries. J. Alloys Compd. 2019, 788, 1093–1102.
- Hu, B.; Wang, K.; Wu, L.; Yu, S.; Antonietti, M.; Titirici, M. Engineering Carbon Materials from the Hydrothermal Carbonization Process of Biomass. Adv. Mater. 2010, 22, 813–828.
More
Information
Subjects:
Others
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
2.6K
Revisions:
2 times
(View History)
Update Date:
18 Aug 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No