1. Zeolites in the Removal of Pharmaceuticals from the Environment
1.1. Where to Start?
Wide applications of pharmaceuticals, which are defined as substances used in the diagnosis, treatment, or prevention of disease and for restoring, correcting, or modifying organic functions, have contributed to the incredible improvement of human health and raised the quality and length of life to a great extent
[1]. The most recent example is the explosion of the use of pharmaceuticals during the SARS-CoV-2 pandemic, in the treatment, but also in the prevention and diagnosis of diseases on a global scale. Despite the many benefits of modern medicine, its usage is a double-edged sword, because of the fact that a significant percentage of drugs used will end their journey in the environment.
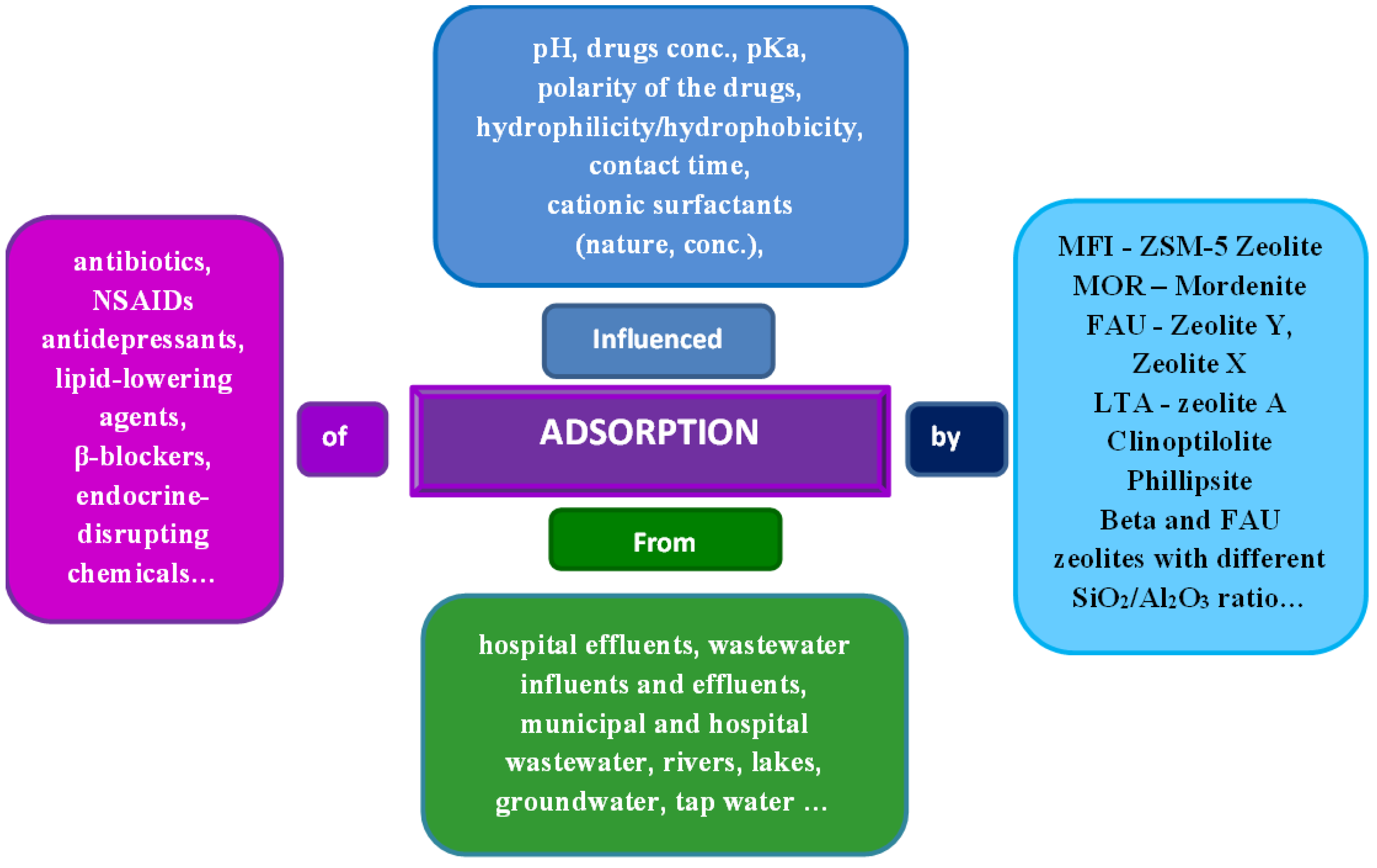
In addition to frequently administered drugs like antidepressants, lipid-lowering agents, β-blockers, etc., very often investigated the occurrence of antibiotics, and non-steroidal anti-inflammatory drugs-NSAIDs and their residues in wastewater
[2][3]. Although antibiotics levels are ng/L to µg/L, they are considered toxic compounds which contribute to the evolution of antibiotic-resistant bacteria and genes
[4][5].
Ciprofloxacin, azithromycin and cephalexin, as frequently administered drugs, are suitable as markers of antibiotic pollution of the aquatic environment
[2]. Interestingly, the effects on aquatic systems of birth control pills or better defined, endocrine-disrupting chemicals, are the subject of numerous reported studies for more than 40 years
[6].
Excretion is the dominant way that drugs reach the environment, sometimes in their parent form or as metabolites. The environment’s physicochemical and bioconversion of excreted pharmaco-active compounds, which occurred after their biotransformation pathways, give as a result various and sometimes unpredictable products
[7]. The fate of pharmaceuticals in the aquatic environment is therefore a very attractive field of study
[8]. Although pharmaceuticals are generally considered as susceptible to diverse transformation reactions, the resulting products are often very stable. Such transformed hydrophilic compounds easily pass-through sewage treatment plants
[9]. Anyway, the drugs of even the same pharmacology active groups possess a variety of non-predictable excretion rates, with considerable impact to the aquatic environment
[10].
The importance of inadequate disposal of discarded drugs should not be neglected, although this area is officially regulated in most countries
[11].
Luckily, this topic is nowadays under the watchful eye of scientists, and water is an environment of interest for monitoring pharmaceutical levels, as well as the resulting impacts it leads to. Focus is placed on surface water samples, sewage treatment plants, and drinking water
[12][13][14].
1.2. Examples of Effective Removal of Pharmaceuticals with Zeolites
The procedures for removal of pharmaceuticals from the aquatic environment are numerous and more and more innovative. For example, pharmaceuticals can be removed from wastewater by biological processes, hydrodynamic cavitation and ultraviolet light treatment
[15]. Besides the general approach, it is usually necessary to create a specific action toward targeted compounds.
Although the conventional procedures, like an advanced oxidation, hydrolysis, and photo-degradation, are usually an efficient way for removal of pollutants, these strategies applied to pharmaceutical rich waters, such as hospital waste effluents or pharmaceutical industries are, can potentially cause environmental and health harmful products
[16][17], and must be considered in detail
[18].
Among numerous attempts, the use of zeolite for water purification from pollutants was highlighted as a simple, efficient, low-cost, and eco-friendly procedure. Zeolites (either in pristine or modified forms), are well-known materials for the adsorption of pesticides from wastewaters
[19][20][21][22][23].
The most important characteristic of zeolite for this application is its adsorption ability. The efforts of scientists in this field are focused on finding materials with improved adsorption capacities, appropriate kinetics, and examining the reversibility of the adsorption process, as well as the possibility of reuse.
First, investigations often start with neat structures, to elucidate key parameters, such as Si/Al ratio, surface area and extra-framework ions. The experimental design relies on sample loading optimization; however, the practical approach imposes the use of low loadings to boost adsorption capacity. To participate in this quest, researchers sometimes report loading that exceeds the starting adsorbent amount several times, although this has no physical meaning. Another challenge is to detect important parameters for efficient removal of environmentally significant concentrations, which are often for pharmaceuticals at the ng/mL level. Such a low concentration masks real adsorbent performance, and lab research usually employs mg/L concentration to boost experimental sensitivity
[24].
A search of the literature can reveal the published results of testing a large number of zeolite structures with the aim of testing their ability to remove drugs, i.e., their adsorption from wastewater. Table 1. gives the most explored types of zeolites for this purpose.
Among different zeolites, the FAU framework is especially present for wastewater treatment due to its substantial adsorption capacity toward different molecules. Additionally, in some cases there is no need for functionalization, and commercially available zeolite can be readily employed. Following its beneficial adsorption behaviour, FAU zeolite has been so far employed for the removal of sulfonamide
[25][26] and fluoroquinolone antibiotics
[27][28]. A special study was dedicated to the macrolide class representative, azithromycin removal by FAU zeolite
[29]. Another reason for FAU selection lies in its fast kinetics, which takes up to several minutes for half an hour, which is of utmost importance for environmental application. Antibiotics are often oxygen and nitrogen-rich compounds whose main mechanism of interaction with the zeolite surface is hydrogen bonding. If this is the case, it is necessary to examine pH effects on the adsorption process
[29].
Scheme 1 illustrates the drug removal by zeolites.
Scheme 1. Pharmaceutical adsorption by zeolites from aquatic media.
Zeolites sometimes require functionalization/composite preparation to establish new and improved features. MFI zeolite, for example, suffers from low adsorption capacity, and combination with excellent adsorbents enabled the removal technique to be extended from adsorption to catalytic degradation in the presence of suitable oxidants. For instance, composite prepared of MFI zeolite and carbon adsorbent is proposed for 150 ppm ciprofloxacin adsorption
[30].
These procedures, however, sometimes tend to be costly and complicated or even pose a bigger environmental threat, due to hazardous solvents or higher toxicity, leaving of constituents, etc.
The adsorption conditions of antibiotics on zeolite are often examined, like in the research of chlortetracycline, oxytetracycline (OTC), ofloxacin, and enrofloxacin adsorption on natural zeolite
[31]. The Langmuir-Freundlich sorption model was employed to estimate the maximum sorption capacity, and it was found that the capacity increased if the solution pH decreased. The presence of natural organic matter reduced the sorption of OTC but improved the sorption of the remaining antibiotics. An ofloxacin removal was additionally tested on LTA zeolite prepared from red mud/fly ash/spinel iron oxide nanoparticles
[32]. Tested adsorption was investigated in different matrices—tap and river water—by spiking the samples with 10 µg/L antibiotics.
As another example, erythromycin (ERY) and levofloxacin (FLX) were almost completely adsorbed by Y zeolite (as an example of organophilic zeolite) from water samples collected at the outlet of a wastewater treatment plant
[27].
The “2-in-1” idea to lower the overall costs and to use environmentally undesirable materials is reported in the research where coal fly ash (CFA) driven zeolites were applied for the adsorptive removal of ceftazidime, a broad-spectrum antibiotic
[33], where CFA is a leftover product of burned coal, very harmful for the environment.
Sometimes reviews about zeolites and mesoporous silica materials as effective adsorbents of drugs, besides antibiotics, are focused on the removal of NSAIDs, as another most frequently used pharmaceuticals. The review of Grela et al. summarized available literature data and concluded that the highest concentrations of diclofenac, ibuprofen, and ketoprofen were found in wastewater influents, municipal wastewater, and hospital effluents, and gave the order of NSAIDs and antibiotics concentrations in different types of water samples: hospital effluents > wastewater influents > municipal wastewater > secondary wastewater > river water > wastewater effluents > groundwater > surface water > seawater > tap water > hospital wastewater > surface water (lakes)
[34].
High concentrations of pollutants in simulations of real water samples are necessary to elucidate key parameters for adsorption and to select the best adsorbent. But, directing further research towards lower concentrations in environmental real water samples (real effluents in environmentally relevant concentrations) and information on the practical implementation of these materials in real-life wastewater treatment is of extreme importance. Recently, Ajo et al. showed that the actual removal rates of ibuprofen cannot be accurately estimated in the context of real wastewaters without negative bias from simultaneous reformation
[35]. The review by Shearer et al. focused on metformin and macrolides and assesses isotherm, kinetic and thermodynamic studies, as well as the adsorption mechanisms, with discussion on some identified mistakes and inconsistencies. That review also sought to identify gaps in knowledge, particularly real-world applications, which should be priorities for future investigations
[36]. The removal of pharmaceuticals from wastewater can be challenging due to its complex matrix. The examination of removal efficiency of fluoroquinolone antibiotics, norfloxacin and ofloxacin, by using nanoscale zero-valent iron loaded zeolites modified with polyethylene glycol surfactant in samples taken from the Yellow River is directed towards such applications
[37]. A similar investigation, considering real river samples, was conducted for the Photo-Fenton treatment of water sample from the Meurthe River in France for the removal of 21 different pollutants, including 17 pharmaceutical compounds such as diclofenac, erythromycin, ibuprofen, ketoprofen, and lidocaine using Faujasite Y zeolite containing iron as catalyst
[38]. In an interesting study, De Sousa et al. studied wastewater effluent samples from Girona wastewater treatment plants in Spain, which included wastewater from hospitals, homes, and urban areas. Two FAU zeolites with different SiO2/Al2O3 ratios were used as adsorbents for determining the concentration of azithromycin, ofloxacin, and sulfamethoxazole
[29]. However, a larger number of studies refer to the analysis of samples that do not actually contain pollutants, but real matrix (for example, river water) spiked with pollutants.
Natural zeolite, like Jordanian zeolite (Intermediate silica), was successfully applied as an adsorbent for the removal of several frequently used pharmaceuticals such as ibuprofen, diclofenac sodium, indomethacin, chlorpheniramine maleate, and paracetamol from water
[39]. The research showed the optimal was pH 2 for the removal of all tested compounds, except for diclofenac sodium it was pH 6, with 80 min as the optimum adsorption time. After optimization, the highest removal was found to be 88.3% for NSAID ibuprofen, and 85.8% for antihistaminic chlorpheniramine maleate. The adsorption efficiencies were evaluated, and it turned out that Freundlich isotherm fits the experimental data for both ibuprofen and chlorpheniramine maleate. The results of a continuous flow experiment performed on ibuprofen under constant influent concentration and fixed flow rate indicated that the percentage removal of ibuprofen on zeolite was the highest after fraction 9 with 78% removal.
The introduction of cationic surfactants (cetylpyridinium chloride and Arquad
® 2HT-75) into natural zeolites, such as clinoptilolite (CLI) and phillipsite, leads to a better effect of the obtained composites in the removal of ibuprofen and naproxen
[40]. The zeolitic surfaces were prepared as monolayer and bilayer surfactant coverage. The Langmuir model gives the conclusion that the highest adsorption capacity for the composite characterized by a bilayered surfactant at the clinoptilolite surface was 19.7 mg/g for ibuprofen and 16.1 mg/g for naproxen. The influence of the initial drug concentrations and contact time on adsorption of ibuprofen and naproxen and zeolite clinoptilolite and phillipsite, are surely very important factors for the process of drug adsorption from buffer solutions
[40].
Simply discovering that a certain zeolite has the ability to adsorb a pharmaceutical, so it can be used for wastewater treatment, can in no way satisfy the scientific public. It is extremely important to shed light on the adsorption process itself, e.g., to determine the factors that affect the capacity, dynamics and reversibility of this process. For this purpose, a multidisciplinary approach and a number of modern instrumental methods are used. This approach will be illustrated by citing some examples from the literature.
Adsorption isotherms and thermogravimetric analysis show that ERY, FLX and carbamazepine (CBZ) are adsorbed in remarkable amounts by Y zeolite. X-ray structure analyses carried out on zeolite after adsorption revealed the selected drugs inside the Y cage. The research indicates that the adsorption properties of zeolitic materials do not only depend on micropore size, and that zeolite shape selectivity also depends on structural features
[27].
The beta zeolites with different SiO
2/Al
2O
3 ratio (i.e., 25, 38 and 360) were tested for adsorption of ketoprofen, hydrochlorothiazide and atenolol from diluted aqueous solutions, with changing the ionic strength and the pH, before and after thermal treatment of the adsorbents
[41]. The processes were followed by thermogravimetry and X-ray diffraction. The research confirmed that the adsorption capacity was dependent on both the solution pH and the alumina content of the beta zeolites. The noticed difference was explained as a function of the interactions between drug molecules and zeolite surface functional groups. Atenolol was adsorbed on the less hydrophobic zeolite, under pH conditions in which electrostatic interactions were predominant, while ketoprofen adsorption was mainly determined by hydrophobic interactions. The adsorption capability for undissociated molecules increased with the increase of hydrophobicity.
Certain studies were conducted with the aim to better understand the interaction between the natural zeolite clinoptilolite and antibiotics that caused gastric side effects, such as metronidazole and sulfamethoxazole
[42]. Beside the considerable importance of pH on the adsorption process, the research reported that interaction of metronidazole and sulfamethoxazole with the clinoptilolite and its forms is fundamentally related with the polarity of the molecules and the nature of the zeolitic material.
Other agents that can modify the properties of the solid surface of zeolites and improve the adsorption of some pharmaceutics are surfactants. The study of Lam et al. reported the results of semiempirical calculations applied on the systems formed by surfactants, drugs, water and a clinoptilolite channel model
[43]. Special attention was paid to the interaction of each drug molecule with the external surface of the clinoptilolite model. The cationic surfactant seems to be well adsorbed on the clinoptilolite model, contrary to the anionic surfactant. The polarity of the drugs plays a very important role in the adsorption process from the solution: the most polar studied drug, metronidazole, was best adsorbed on the zeolite model, followed by acetylsalicylic acid and sulfamethoxazole. If the same system contains the cationic surfactant, the order of the drug adsorption is opposite: the adsorption of sulfamethoxazole as a hydrophobic molecule is more pronounced. Those conclusions can help in adjusting the adsorption of certain drugs on clinoptilolite in the desired direction by the presence of surfactant on the zeolite external surface.
Zeolitic imidazolate metal organic framework group of compounds, such as ZIF-8, belongs to metal-organic frameworks (MOFs), and possess similar characteristics to zeolite, including the high adsorption capability, and therefore can be a great candidate for the testing of removal of the pharmaceuticals from wastewater. As an example, ZIF-8 exhibits ultra-high adsorption capacity to tetracycline from aquatic media
[44].
2. Zeolite-Based Biomaterials for Biomedical Application
The biocompatibility and mechanical strengths of zeolite make them suitable for biomedical application as pharmaco active compounds or as biomaterial used for dental fillers, bone grafts, implant coating, or as a drug carrier agent
[45].
An illustration of the positive example of application of zeolites themselves in pharmacotherapy is clinoptilolite which can stand out as a potent detoxifying, antioxidant, and anti-inflammatory agent
[46][47]. Zeolites are currently regarded as dietary super-materials
[46], and this, sometimes, is an overstatement. Every drugstore is selling zeolite-based nutritional formulations enhanced with vitamins, enzymes, etc., often without any scientific basis to support the claims of high nutritional benefits. Antioxidant, antimicrobial, detoxifying, and anticancer activity is often attributed to both synthetic and natural zeolites. However, a strict survey of the available literature gives somewhat controversial findings. For instance, zeolite’s ability for trapping radical species in order for them to be safely removed from the body is a cornerstone of many studies
[48]. A part of this is actually true, zeolite structure does enable radical species removal
[49][50], although they shouldn’t be a part of a human diet in spite of being used for cattle because tightly bind mycotoxins from animal feed in the gastrointestinal tract and thereby decrease their bioavailability
[51]. Concerning this, Ipek et al. assessed the effect of natural zeolite, clinoptilolite supplementation on the oxidative status in cows and concluded that it did not support cow’s systems against oxidative stress
[52].
2.1. Zeolites for Dental Applications
In addition to their wide application in medicine and other sciences, zeolites have also displayed a significant potential for use in the field of dentistry. Zeolites may be applied in root canal therapy, periodontics, implant and restorative dentistry, and tissue engineering
[53]. Zeolites were introduced into dental practice mainly as root filler materials, based on their hemolytic and cytotoxic properties
[54], or their antimicrobial and mechanical characteristics
[53]. Remineralizing the ability of calcium-rich zeolite makes it a promising candidate as a dental composite filler
[55]. It can be said that a wide application of zeolite in dentistry is not primarily based on adsorption as a phenomenon, but certain functions of zeolite are significantly improved thanks to the adsorption effect, which will be illustrated by examples.
In general, zeolites alone have little or no effect on antimicrobial properties, unless the zeolites are doped with ions, such as silver or zinc. The ability of zeolites to uptake and release ions, combined with their exceptional biocompatibility and long-lasting effects, has been used for the antimicrobial treatment against pathogenic oral microorganisms. Regarding dental restorative materials, zeolites are generally combined with glass ionomer cements (GIC), resin cements, or bonding agents. The antibacterial effects of GIC containing silver-zeolite (AgZ) were demonstrated on
Streptococcus mutans in vitro
[56]. Similar antimicrobial results can also be found in a zinc-doped zeolite (ZnZ) against
Escherichia coli,
Staphylococcus aureus,
Pseudomonas aeruginosa,
Bacillus subtilis, and
Candida albicans [57]. A functional dental restorative platform consisting of zeolite nanoparticles as a drug delivery carrier loaded with chlorhexidine (CHX), was incorporated into commercial dental GIC, demonstrating a stronger inhibitory effect on
S. mutans compared to GIC alone
[58]. AgZ combined with GIC sealer showed a stronger antimicrobial effect towards
E. faecalis compared to GIC sealer alone, which was concentration- and time-independent
[59].
The root canal treatment has been related to persistent periradicular lesions, including primary endodontic infections and persistent infectious progression with the
E. faecalis as the main etiological factor in these diseases. Zeolites in endodontics were generally added to calcium hydroxide and mineral trioxide aggregate (MTA) and used as root canal irrigants to enhance their antimicrobial properties. Ghatole et al. showed that adding AgZ to calcium hydroxide enhanced the antimicrobial effect against
E. faecalis compared to the control or when chlorhexidine is added
[60]. Among the root end filling materials, MTA is regarded as biocompatible and is most commonly used in clinical applications, but with limited antimicrobial activity. The addition of AgZ to MTA enhanced antimicrobial effects toward selected oral microflora such as
E. faecalis,
S. aureus, and
Candida albicans in a concentration-dependent manner throughout 72 h
[61].
Two types of zeolites, Zeolite A and ZSM-5 were investigated for their potential to adsorb volatile sulphide compounds (VSC), which are produced in the oral environment by Gram-negative bacteria and cause periodontal disease and mouth odour. The amount of H
2S adsorbed on zeolite A was found to be larger than that on ZSM-5, suggesting that the adsorptive property of zeolites depends on their Si/Al ratio. By optimising Si/Al ratio, it is expected to develop an adsorbent material, which highly adsorbs VSC, and may contribute to oral health
[62].
Zeolite in prosthesis can be added to both acrylic resin and non-acrylic materials, such as ceramic. The non-acrylic materials that were tested with zeolite demonstrated that adding AgZ to soft liners enhanced its antimicrobial properties against
C. albicans and gram-negative bacteria while also maintaining its viscoelastic properties
[63]. Sodalite zeolite is a subtype of zeolite that can easily infiltrate other materials due to its selectivity and strong catalytic activity and has been often applied to ceramic prostheses to improve their mechanical properties
[64]. Incorporation of Ag-Zn zeolite with acrylic resin materials showed beneficial effects by improving their surface finish and resistance to surface damage by increasing hardness
[65].
Application of zeolites in dentistry can also extend to antibacterial coatings on implants since coating titanium implants with AgZ was effective in inhibiting methicillin-resistant
S. aureus growth
[66].
Zeolites are known for their applicability in different composite materials, including dental materials. Zeolite fillers associated with phenol−formaldehyde resins and poly-(vinylidene fluoride) are often used
[67]. An important fact is that zeolites have a positive impact on the mechanical properties of composites, which is combined with their ability to deliver the calcium ions (Ca
2+) to the tooth surface, showing the remineralizing potential of the hydroxyapatite structure of dentin and enamel
[68]. FAU zeolites (X and Y type) have the highest remineralisation potential
[69] and can be also used as scaffolds because they do not affect cell viability
[55]. Since Gram-negative bacteria do not adhere to the type X zeolite, composite materials with zeolite X will not be sensitive to secondary caries
[55].
Dental composites consist of inorganic fillers and organic resin matrix with some amounts of additives
[70]. Due to the low adhesion of inorganic fillers to resins, it is often necessary to modify their surface before mixing these components. One of the main types of modifications that is used in dental fillings is silanization. This modification empowers bringing into contact the groups that are involved in cross-linking of the composite to a filler’s surface
[71]. Silane molecules that ensure the binding between the organic matrix and inorganic filler make the composite have a more rigid structure and improve the mechanical features of the composite, such as compressive and flexural strength, to obtain similar mechanical characteristics of the tooth structure
[72]. Application of calcium-rich 13X zeolites as active fillers improved the remineralizing effect of examined composites by providing sustained release of calcium ions in conditions simulating a natural oral environment. Furthermore, the silanization of these composites significantly improved flexural strength and compressive strength values. The beneficial effects of silanization are the consequence of a stronger bond between fillers particles and resins from the organic matrix due to the introduction of methacrylic groups to the fillers’ surface, which form covalent bonds between the resin and the filler
[55].
Modification of the zeolite surface by a 4-(dimethylamino)benzenediazonium cation to acquire an active filler in methacrylic-resin-based composites was performed to verify some mechanical properties, as well as crosslinking ability
[73]. All conducted tests proved that the addition of modified zeolite improved the compressive and flexural strength of the composite. The modification process, as well, has a crucial impact on these values. The results also show that it affected the crosslinking properties of the resin.
Soft and hard tissue engineering of the oral cavity represents the therapeutic approach with great potential. Zeolites, due to their favourable properties, have appeared promising as scaffolds in bone- and tooth-tissue engineering. The clinoptilolite-composite scaffolds enhanced mechanical, physical, and biological properties of polymer-based scaffolds, increased in vitro protein adsorption capacity of the scaffold and led to higher osteoinductivity and intracellular calcium deposition
[74].
It is expected that the further research effort should focus on zeolite-based materials and concentrate on zeolite effects in response to microbial challenges in vivo, but also to determine the proper concentration of zeolite that may be incorporated into various dental materials and establish zeolite’s impact on their mechanical properties. The possibilities and beneficial effects of various surface modification methods should also be involved. Zeolite’s favourable properties and wide range of the composition and hierarchical pore structure makes the zeolitic materials convenient for tissue engineering purposes.
2.2. Zeolites as Drug Carriers in Pharmacotherapy
The next logical step in the medical application of zeolites was their usage as drug carriers. That idea was surely provoked by two main groups of reasons. The first one is the reversibility of the adsorption of pharmaceuticals by zeolites, and which requires thorough studying of many important factors which influence this process. The focus, for sure, must be put on the ability to control and prolong drug release.
At the same time, scientists have made great efforts to exploit the possible synergistic bioactivities of zeolites with applied drugs. The resulting effects are often unpredictable and require detailed study, especially when cytotoxic effects on fibroblasts and tumour cells are performed in parallel.
The application of zeolites in biomedicine, as drug carriers, is based on their large specific surface area, high adsorption capacity
[75], biocompactness, low toxicity
[76], and microporous structure, which allows drug encapsulation within the zeolite and the ability to control and prolong drug release. Drug release is monitored in liquids with different pH values that correspond to the pH of the regions through which the drug-loaded zeolite carrier passes
[77]. Drug encapsulation in drug delivery systems (DDS) eliminates side effects while maintaining treatment effectiveness
[78], reduces drug concentration, and provides targeted delivery
[79].
Zeolite nanocarriers enable drug delivery to a specific target and drug release without affecting surrounding healthy cells
[80], and they display enhanced permeability, controlled drug distribution, and prolonged life in the blood system
[81]. Synergistic effects of zeolites, such as gastroprotective influence, especially in the case of clinoptilolite DDS, could be ascribed to zeolites ability to attach to hydrogen ions and biologically active amines and nitrates
[82].
The differences between the zeolite pore size and the targeted drug, and in hydrophilicity between zeolites and drugs, which are usually considered as limitation in their loading capacity, can be improved by surface modification of the zeolite
[83], with the aim to adjust the surface of a zeolite depending on the delivered drug characteristics.
The efficiency of zeolites as DDS can be improved by the adsorption of cationic surfactants on the surface of zeolites through ion exchange and hydrophobic interactions, leading to the formation of monolayers or bilayers, depending on their concentration
[84]. It looks that very important role here concerns the concentration of the cationic surfactant in relation to external cation exchange capacity—ECEC value, confirmed by the research performed on the natural zeolite with high clinoptilolite content with different levels of cetylpyridinium chloride (CPC)
[85]. If the surfactant loading level was equal to ECEC, a monolayer of the organic phase is present at the zeolitic surface. When the amount of the CPC was above the ECEC value, a less extended bilayer is formed, while the sample with the highest surfactant content ordered bilayer or admicelles exist at the zeolitic surface. Similar results, obtained for the cationic surfactant-hexadecyltrimethylammonium bromide (HB) showed proportional increase of drug adsorption by increasing the amount of surfactant used for zeolite modification, leading to interactions between DS and HB at the zeolitic surface
[86].