
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kun Luo | -- | 546 | 2022-08-04 12:11:23 | | | |
| 2 | Kun Luo | + 4 word(s) | 608 | 2022-08-08 11:01:45 | | | | |
| 3 | Kun Luo | + 674 word(s) | 1282 | 2022-08-08 11:31:53 | | | | |
| 4 | Catherine Yang | -2 word(s) | 1280 | 2022-08-09 03:02:54 | | |
Video Upload Options
Carbon dots (CDs) have many advantages, such as tunable photoluminescence, large two-photon absorption cross-sections, easy functionalization, low toxicity, chemical inertness, good dispersion, and biocompatibility. Halogen doping further improves the optical and physicochemical properties of CDs, extending their applications in fluorescence sensors, biomedicine, photocatalysis, anti-counterfeiting encryption, and light-emitting diodes. The preparation of CDs via the “top-down” and “bottom-up”approaches and the preparation methods and applications of halogen (fluorine, chlorine, bromine, and iodine)-doped CDs were discussed here. The main challenges of CDs in the future are the elucidation of the luminescence mechanism, fine doping with elements (proportion, position, etc.), and their incorporation in practical devices.
1. Introduction
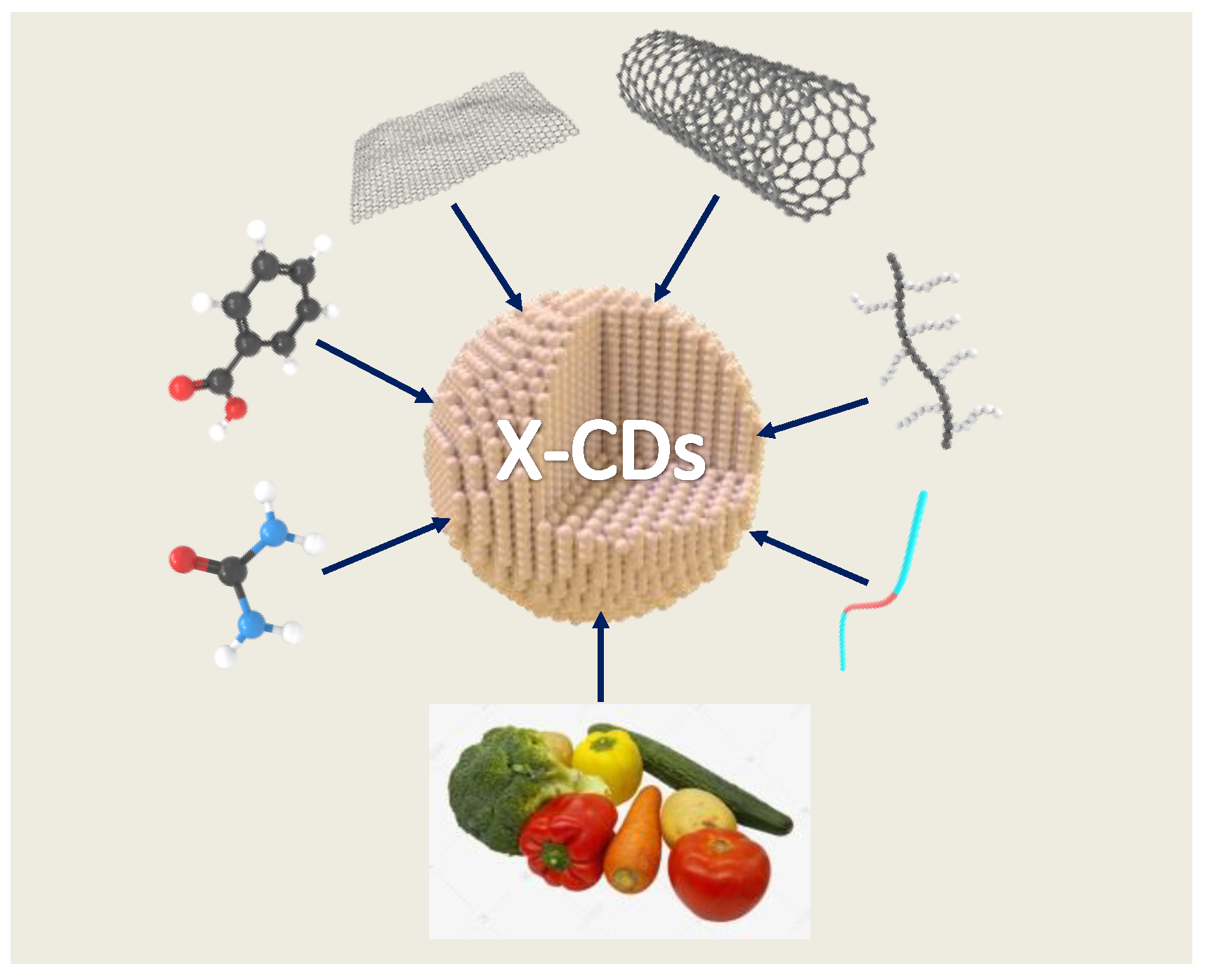
2. CDs Preparation Methods
2.1. Top-Down Approach
2.2. Bottom-Up Approach
3. Halogen-Doped CDs
4. Conclusions and Prospects
References
- Yiming Liu; Swagata Roy; Samrat Sarkar; Jiaqiang Xu; Yufeng Zhao; Jiujun Zhang; A review of carbon dots and their composite materials for electrochemical energy technologies. Carbon Energy 2021, 3, 795-826, 10.1002/cey2.134.
- Mohammad Jafar Molaei; A review on nanostructured carbon quantum dots and their applications in biotechnology, sensors, and chemiluminescence. Talanta 2018, 196, 456-478, 10.1016/j.talanta.2018.12.042.
- XiaoYou Xu; Robert Ray; Yunlong Gu; Harry J. Ploehn; Latha Gearheart; Kyle Raker; Walter A. Scrivens; Electrophoretic Analysis and Purification of Fluorescent Single-Walled Carbon Nanotube Fragments. Journal of the American Chemical Society 2004, 126, 12736-12737, 10.1021/ja040082h.
- Ya-Ping Sun; Bing Zhou; Yi Lin; Wei Wang; K. A. Shiral Fernando; Pankaj Pathak; Mohammed Jaouad Meziani; Barbara A. Harruff; Xin Wang; Haifang Wang; et al.Pengju G. LuoHua YangMuhammet Erkan KoseBailin ChenL. Monica VecaSu-Yuan Xie Quantum-Sized Carbon Dots for Bright and Colorful Photoluminescence. Journal of the American Chemical Society 2006, 128, 7756-7757, 10.1021/ja062677d.
- Bin Bin Chen; Ze Xi Liu; Wen Chan Deng; Lei Zhan; Meng Li Liu; Cheng Zhi Huang; A large-scale synthesis of photoluminescent carbon quantum dots: a self-exothermic reaction driving the formation of the nanocrystalline core at room temperature. Green Chemistry 2016, 18, 5127-5132, 10.1039/c6gc01820c.
- Alexander P Demchenko; Mariia O Dekaliuk; Novel fluorescent carbonic nanomaterials for sensing and imaging. Methods and Applications in Fluorescence 2013, 1, 042001, 10.1088/2050-6120/1/4/042001.
- Y. Park; J. Yoo; B. Lim; W. Kwon; S.-W. Rhee; Improving the functionality of carbon nanodots: doping and surface functionalization. Journal of Materials Chemistry A 2016, 4, 11582-11603, 10.1039/c6ta04813g.
- Fanglong Yuan; Shuhua Li; Zetan Fan; Xiangyue Meng; Louzhen Fan; Shihe Yang; Shining carbon dots: Synthesis and biomedical and optoelectronic applications. Nano Today 2016, 11, 565-586, 10.1016/j.nantod.2016.08.006.
- Mingfei Pan; Xiaoqian Xie; Kaixin Liu; Jingying Yang; Liping Hong; Shuo Wang; Fluorescent Carbon Quantum Dots—Synthesis, Functionalization and Sensing Application in Food Analysis. Nanomaterials 2020, 10, 930, 10.3390/nano10050930.
- Dibyendu Ghosh; Krishnendu Sarkar; Pooja Devi; Ki-Hyun Kim; Praveen Kumar; Current and future perspectives of carbon and graphene quantum dots: From synthesis to strategy for building optoelectronic and energy devices. Renewable and Sustainable Energy Reviews 2020, 135, 110391, 10.1016/j.rser.2020.110391.
- Wang,X; Zhang,T; Chen,J; Progress in preparation and application of doped carbon dots. Chem. Res 2019, 30, 13-33, 10.14002/j.hxya.2019.01.002.
- Na Gao; Wen Yang; Hailiang Nie; Yunqian Gong; Jing Jing; Loujun Gao; Xiaoling Zhang; Turn-on theranostic fluorescent nanoprobe by electrostatic self-assembly of carbon dots with doxorubicin for targeted cancer cell imaging, in vivo hyaluronidase analysis, and targeted drug delivery. Biosensors and Bioelectronics 2017, 96, 300-307, 10.1016/j.bios.2017.05.019.
- Wen-Sheng Zou; Cui-Hong Ye; Ya-Qin Wang; Wei-Hua Li; Xian-Huai Huang; A hybrid ratiometric probe for glucose detection based on synchronous responses to fluorescence quenching and resonance light scattering enhancement of boronic acid functionalized carbon dots. Sensors and Actuators B: Chemical 2018, 271, 54-63, 10.1016/j.snb.2018.05.115.
- Somayeh Hamd-Ghadareh; Abdollah Salimi; Fardin Fathi; Saman Bahrami; An amplified comparative fluorescence resonance energy transfer immunosensing of CA125 tumor marker and ovarian cancer cells using green and economic carbon dots for bio-applications in labeling, imaging and sensing. Biosensors and Bioelectronics 2017, 96, 308-316, 10.1016/j.bios.2017.05.003.
- Junjiao Deng; Yi You; Veena Sahajwalla; Rakesh K. Joshi; Transforming waste into carbon-based nanomaterials. Carbon 2016, 96, 105-115, 10.1016/j.carbon.2015.09.033.
- Hui Ding; Shang-Bo Yu; Ji-Shi Wei; Huan-Ming Xiong; Full-Color Light-Emitting Carbon Dots with a Surface-State-Controlled Luminescence Mechanism. ACS Nano 2015, 10, 484-491, 10.1021/acsnano.5b05406.
- Kai Jiang; Shan Sun; Ling Zhang; Yue Lu; Aiguo Wu; Congzhong Cai; Hengwei Lin; Red, Green, and Blue Luminescence by Carbon Dots: Full-Color Emission Tuning and Multicolor Cellular Imaging. Angewandte Chemie International Edition 2015, 54, 5360-5363, 10.1002/anie.201501193.
- Shi Ying Lim; Wei Shen; Zhiqiang Gao; Carbon quantum dots and their applications. Chemical Society Reviews 2014, 44, 362-381, 10.1039/c4cs00269e.
- Keenan J. Mintz; Yiqun Zhou; Roger M. Leblanc; Recent development of carbon quantum dots regarding their optical properties, photoluminescence mechanism, and core structure. Nanoscale 2019, 11, 4634-4652, 10.1039/c8nr10059d.
- Xiaoli Kou; Shicui Jiang; Soo-Jin Park; Long-Yue Meng; A review: recent advances in preparations and applications of heteroatom-doped carbon quantum dots. Dalton Transactions 2020, 49, 6915-6938, 10.1039/d0dt01004a.
- Shoujun Zhu; Junhu Zhang; Shijia Tang; Chunyan Qiao; Lei Wang; Haiyu Wang; Xue Liu; Bo Li; Yunfeng Li; Weili Yu; et al.Xingfeng WangHongchen SunBai Yang Surface Chemistry Routes to Modulate the Photoluminescence of Graphene Quantum Dots: From Fluorescence Mechanism to Up-Conversion Bioimaging Applications. Advanced Functional Materials 2012, 22, 4732-4740, 10.1002/adfm.201201499.
- Liping Lin; Yaxin Luo; Peiyu Tsai; Jiajing Wang; Xi Chen; Metal ions doped carbon quantum dots: Synthesis, physicochemical properties, and their applications. TrAC Trends in Analytical Chemistry 2018, 103, 87-101, 10.1016/j.trac.2018.03.015.
- Manpreet Kaur; Manpreet Kaur Ubhi; Jaspreet Kaur Grewal; Virender K. Sharma; Boron- and phosphorous-doped graphene nanosheets and quantum dots as sensors and catalysts in environmental applications: a review. Environmental Chemistry Letters 2021, 19, 4375-4392, 10.1007/s10311-021-01281-0.
- Haifang Liu; Zhaohui Li; Yuanqiang Sun; Xin Geng; Yalei Hu; Hongmin Meng; Jia Ge; Lingbo Qu; Synthesis of Luminescent Carbon Dots with Ultrahigh Quantum Yield and Inherent Folate Receptor-Positive Cancer Cell Targetability. Scientific Reports 2018, 8, 1-8, 10.1038/s41598-018-19373-3.
- Fan Yang; Weijie Bao; Tianxing Liu; Bing Zhang; Shuo Huang; Wang Yang; Yun Li; Na Li; Chunxia Wang; Caiwen Pan; et al.Yongfeng Li Nitrogen-doped graphene quantum dots prepared by electrolysis of nitrogen-doped nanomesh graphene for the fluorometric determination of ferric ions. Microchimica Acta 2020, 187, 1-10, 10.1007/s00604-020-04294-8.
- Rabia Riaz; Mumtaz Ali; T. Maiyalagan; Aima Sameen Anjum; Seoyun Lee; Min Jae Ko; Sung Hoon Jeong; Dye-sensitized solar cell (DSSC) coated with energy down shift layer of nitrogen-doped carbon quantum dots (N-CQDs) for enhanced current density and stability. Applied Surface Science 2019, 483, 425-431, 10.1016/j.apsusc.2019.03.236.
- Yongsheng Yang; BingLi Gu; Zhiduo Liu; Da Chen; Yun Zhao; Qinglei Guo; Gang Wang; Hydrothermal synthesis of N, P co-doped graphene quantum dots for high-performance Fe3+ detection and bioimaging. Journal of Nanoparticle Research 2021, 23, 1-12, 10.1007/s11051-021-05154-z.
- Ying Li; HeChun Lin; Chunhua Luo; Yunqiu Wang; Chunli Jiang; Ruijuan Qi; Rong Huang; Jadranka Travas-Sejdic; Hui Peng; Aggregation induced red shift emission of phosphorus doped carbon dots. RSC Advances 2017, 7, 32225-32228, 10.1039/c7ra04781a.
- Jun-Won Kang; Dong-Hyun Kang; Effect of amino acid-derived nitrogen and/or sulfur doping on the visible-light-driven antimicrobial activity of carbon quantum dots: A comparative study. Chemical Engineering Journal 2021, 420, 129990, 10.1016/j.cej.2021.129990.
- Shiyue Bian; Chao Shen; Yuting Qian; Jiyang Liu; Fengna Xi; Xiaoping Dong; Facile synthesis of sulfur-doped graphene quantum dots as fluorescent sensing probes for Ag+ ions detection. Sensors and Actuators B: Chemical 2017, 242, 231-237, 10.1016/j.snb.2016.11.044.
- Ge Gao; Yao-Wen Jiang; Hao-Ran Jia; Jingjing Yang; Fu-Gen Wu; On-off-on fluorescent nanosensor for Fe3+ detection and cancer/normal cell differentiation via silicon-doped carbon quantum dots. Carbon 2018, 134, 232-243, 10.1016/j.carbon.2018.02.063.
- Mohammad Amjadi; Tooba Hallaj; Jamshid L. Manzoori; Tahmineh Shahbazsaghir; An amplified chemiluminescence system based on Si-doped carbon dots for detection of catecholamines. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 2018, 201, 223-228, 10.1016/j.saa.2018.04.058.
- Meng-Jey Youh; Meng-Chih Chung; Hung-Chun Tai; Ching-Yi Chen; Yuan-Yao Li; Fabrication of carbon quantum dots via ball milling and their application to bioimaging. Mendeleev Communications 2021, 31, 647-650, 10.1016/j.mencom.2021.09.018.
- Haitao Li; Xiaodie He; Zhenhui Kang; Hui Huang; Yang Liu; Jinglin Liu; Suoyuan Lian; Alpha Chi Him Tsang; Xiaobao Yang; Shuit-Tong Lee; et al. Water-Soluble Fluorescent Carbon Quantum Dots and Photocatalyst Design. Angewandte Chemie International Edition 2010, 49, 4430-4434, 10.1002/anie.200906154.
- Arvind Singh; Pranab Kishore Mohapatra; Dinesh Kalyanasundaram; Sunil Kumar; Self-functionalized ultrastable water suspension of luminescent carbon quantum dots. Materials Chemistry and Physics 2018, 225, 23-27, 10.1016/j.matchemphys.2018.12.031.
- Juan Peng; Wei Gao; Bipin Kumar Gupta; Zheng Liu; Rebeca Romero-Aburto; Liehui Ge; Li Song; Lawrence B. Alemany; Xiaobo Zhan; Guanhui Gao; et al.Sajna Antony VithayathilBenny Abraham KaipparettuAngel A. MartiTakuya HayashiJun-Jie ZhuPulickel M. Ajayan Graphene Quantum Dots Derived from Carbon Fibers. Nano Letters 2012, 12, 844-849, 10.1021/nl2038979.
- Jing Wang; Cai-Feng Wang; Su Chen; Amphiphilic Egg-Derived Carbon Dots: Rapid Plasma Fabrication, Pyrolysis Process, and Multicolor Printing Patterns. Angewandte Chemie International Edition 2012, 51, 9297-9301, 10.1002/anie.201204381.
- Tong-Yang Shen; Pei-Yun Jia; Da-Shu Chen; Li-Na Wang; Hydrothermal synthesis of N-doped carbon quantum dots and their application in ion-detection and cell-imaging. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 2020, 248, 119282, 10.1016/j.saa.2020.119282.
- Nargish Parvin; Tapas K. Mandal; Dually emissive P,N-co-doped carbon dots for fluorescent and photoacoustic tissue imaging in living mice. Microchimica Acta 2017, 184, 1117-1125, 10.1007/s00604-017-2108-4.
- Taher Alizadeh; Mahrokh Shokri; A new humidity sensor based upon graphene quantum dots prepared via carbonization of citric acid. Sensors and Actuators B: Chemical 2016, 222, 728-734, 10.1016/j.snb.2015.08.122.
- Yunyang Zhao; Songlin Zuo; Meng Miao; The effect of oxygen on the microwave-assisted synthesis of carbon quantum dots from polyethylene glycol. RSC Advances 2017, 7, 16637-16643, 10.1039/C7RA01804E.
- Zhan Zhou; Zhuosen Wang; Yiping Tang; Yuhui Zheng; Qianming Wang; Optical detection of anthrax biomarkers in an aqueous medium: the combination of carbon quantum dots and europium ions within alginate hydrogels. Journal of Materials Science 2018, 54, 2526-2534, 10.1007/s10853-018-2955-3.
- Haitao Li; Chenghua Sun; R. Vijayaraghavan; Fengling Zhou; Xinyi Zhang; Douglas R. MacFarlane; Long lifetime photoluminescence in N, S co-doped carbon quantum dots from an ionic liquid and their applications in ultrasensitive detection of pesticides. Carbon 2016, 104, 33-39, 10.1016/j.carbon.2016.03.040.
- Vaibhavkumar N. Mehta; Sanjay Jha; Suresh Kumar Kailasa; One-pot green synthesis of carbon dots by using Saccharum officinarum juice for fluorescent imaging of bacteria (Escherichia coli) and yeast (Saccharomyces cerevisiae) cells. Materials Science and Engineering: C 2014, 38, 20-27, 10.1016/j.msec.2014.01.038.
- Meiqin He; Jin Zhang; Hai Wang; Yanrong Kong; Yiming Xiao; Wen Xu; Material and Optical Properties of Fluorescent Carbon Quantum Dots Fabricated from Lemon Juice via Hydrothermal Reaction. Nanoscale Research Letters 2018, 13, 175, 10.1186/s11671-018-2581-7.
- Fu Wang; Shuping Pang; Long Wang; Qin Li; Maximilian Kreiter; Chun-Yan Liu; One-Step Synthesis of Highly Luminescent Carbon Dots in Noncoordinating Solvents. Chemistry of Materials 2010, 22, 4528-4530, 10.1021/cm101350u.
- Danyang Zhang; Daiyong Chao; Chunyu Yu; Qi Zhu; Shihong Zhou; Long Tian; Liang Zhou; One-Step Green Solvothermal Synthesis of Full-Color Carbon Quantum Dots Based on a Doping Strategy. The Journal of Physical Chemistry Letters 2021, 12, 8939-8946, 10.1021/acs.jpclett.1c02475.
- Mohamed E. Mahmoud; Nesma A. Fekry; Amir M. Abdelfattah; Removal of uranium (VI) from water by the action of microwave-rapid green synthesized carbon quantum dots from starch-water system and supported onto polymeric matrix. Journal of Hazardous Materials 2020, 397, 122770, 10.1016/j.jhazmat.2020.122770.
- Haitao Li; Xiaodie He; Yang Liu; Hang Yu; Zhenhui Kang; Shuit-Tong Lee; Synthesis of fluorescent carbon nanoparticles directly from active carbon via a one-step ultrasonic treatment. Materials Research Bulletin 2011, 46, 147-151, 10.1016/j.materresbull.2010.10.013.
- Yiyang Wu; Youdi Liu; Jingya Yin; Haitao Li; Jun Huang; Facile ultrasonic synthesized NH2-carbon quantum dots for ultrasensitive Co2+ ion detection and cell imaging. Talanta 2019, 205, 120121, 10.1016/j.talanta.2019.120121.
- Subodh Kumar; Sk Tarik Aziz; Olga Girshevitz; Gilbert Daniel Nessim; One-Step Synthesis of N-Doped Graphene Quantum Dots from Chitosan as a Sole Precursor Using Chemical Vapor Deposition. The Journal of Physical Chemistry C 2018, 122, 2343-2349, 10.1021/acs.jpcc.7b05494.
- Donghua Liu; Xiaosong Chen; Yibin Hu; Tai Sun; Zhibo Song; Yujie Zheng; Yongbin Cao; Zhi Cai; Min Cao; Lan Peng; et al.Yuli HuangLei DuWuli YangGang ChenDapeng WeiAndrew Thye Shen WeeDacheng Wei Raman enhancement on ultra-clean graphene quantum dots produced by quasi-equilibrium plasma-enhanced chemical vapor deposition. Nature Communications 2018, 9, 193, 10.1038/s41467-017-02627-5.
- Lingpeng Yan; Yongzhen Yang; Chang-Qi Ma; Xuguang Liu; Hua Wang; Bingshe Xu; Synthesis of carbon quantum dots by chemical vapor deposition approach for use in polymer solar cell as the electrode buffer layer. Carbon 2016, 109, 598-607, 10.1016/j.carbon.2016.08.058.
- Ruili Liu; Dongqing Wu; Shuhua Liu; Kaloian Koynov; Wolfgang Knoll; Qin Li; An Aqueous Route to Multicolor Photoluminescent Carbon Dots Using Silica Spheres as Carriers. Angewandte Chemie International Edition 2009, 48, 4598-4601, 10.1002/anie.200900652.
- Jin Zhou; Pei Lin; Juanjuan Ma; Xiaoyue Shan; Hui Feng; Congcong Chen; Jianrong Chen; Zhaosheng Qian; Facile synthesis of halogenated carbon quantum dots as an important intermediate for surface modification. RSC Advances 2013, 3, 9625-9628, 10.1039/c3ra41243a.
- Haoqiang Song; Jingkun Yu; Zhiyong Tang; Bai Yang; Siyu Lu; Halogen‐Doped Carbon Dots on Amorphous Cobalt Phosphide as Robust Electrocatalysts for Overall Water Splitting. Advanced Energy Materials 2022, 12, 2102573, 10.1002/aenm.202102573.





