
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Nabti Nabti | + 2926 word(s) | 2926 | 2020-10-12 05:06:40 | | | |
| 2 | Dean Liu | -1430 word(s) | 1496 | 2020-10-13 07:52:09 | | | | |
| 3 | Dean Liu | Meta information modification | 1496 | 2020-10-27 07:58:06 | | |
Video Upload Options
The salinization of soils is the main threat of agriculture, it affects a large area every year. thus, instead of chemical buffers, bacteria are suggested as an effective solution. In addition, bacteria promoting plant growth are in fact known as a potential alternative to restore, promote and protect crops.
1. Introudction
The worldwide enhancement of both human population and the associated environmental deterioration has the unfortunate influence that global food manufacture might soon become unsatisfactory to nourish all of the world’s people. It is therefore crucial to increase farming productivity within the next few decades[1]. On the other hand, in the natural environment, plants are constantly exposed to various abiotic and biotic factors, which may affect their increase and yield[2]. Stress is described as any external abiotic (salinity, water, heat) or biotic (herbivore) constraint that restricts the level of photosynthesis and decreases a plant’s capacity to transform energy to biomass[3]. Especially, abiotic factors are main constraints for global food security, food quality, and crop productivity[4].
Salinity is recognized to affect almost one fourth of world cultivable land, a main cause of desertification[5]. It is responsible for the degradation of 10 million ha of world farming per year[6], influencing about 1 million ha in the European Union, mainly in the Mediterranean countries. Various salt ions in the soil, that are dissoluble in water like chloride (Cl−), sodium (Na+), magnesium (Mg2+), potassium (K+), bicarbonate (HCO3−), sulfate (SO42−), carbonate (CO32−), and calcium (Ca2+) may cause salinization[7]. Salinization is a natural process in semiarid and arid zones and may additionally be stimulated by anthropogenic practices. It significantly influences land fertility [8], and therefore comprises a huge effect on food security and economy at local, regional, and global levels[7]. Although plant tolerance to salinity varies with species and even varieties as well as with biotic and abiotic factors, the growth of most of the plant cultures is negatively influenced by salinity[9][10]. Excess accumulation of salts in the root zone often deteriorates the soil properties, viz. physical, chemical, and biological to such an extent that crop production is adversely affected[11]. In addition, excessive salinity in soil suppresses growth, decreases species diversity, and alters the community composition of plants; however, the effect of salinity on soil microbial communities is poorly understood[12].
Some of the microorganisms that live in close combination with the plant roots ensure a significant function in improving plant growth[13], and/or in stress mitigation of crops cultivated under salinity conditions[14]. Plant growth-promoting bacteria (PGPB) may be active through a diversity of processes[15] including the regulation of the transcription of various genes and cellular communication through quorum sensing[16], and mainly by production of various secondary metabolites[17]. For instance, through the production of exopolysaccharides (EPS), indole acetic acid (IAA), nitrogen fixation, and P-solubilization[18]. PGPB may form symbiotic relations with plants and improve their performance in the face of salinity stress [19][20]. Moreover, PGPB are an environmentally sustainable tool to mitigate the impacts of salinity on crop yields[2].
2. Halophile PGPB for Sustainable Agriculture
In the rhizosphere, plant–microorganism relationships are indicators of plant health, production, and soil fertility. Particularly, plant growth-promoting bacteria (PGPB) are microorganisms that could also promote plant increase and prevent plants from infection and abiotic factors via a wide range of processes. Those PGPBs that develop more intimate interactions with plants, like the endophytes, may be more effective in plant growth improvement [16]. Usage of PGPB as an important part of farming activity is an innovation whose time has arrived[1]. More particularly, some halophilic bacteria were proved able to alleviate plant salt stress, improving the development and productivity of crop production under salinity [21].
PGPB represent a possible solution to mitigate salinity-induced plant stress[22]. The impact of five plant growth stimulating halotolerant bacteria on wheat growth[21] revealed that bio-formulation of these halotolerant bacterial strains can improve salinity stress (80, 160, and 320 mM) in wheat seedlings, resulting in a 70% improvement of root length comparatively to negative controls. In addition, variations in the bacterial community formulation with the soil salinity were observed, and attention is growing in experiments regarding functional interactions between plants and microbes that lead to salt stress tolerance[25]. The use of microorganisms for saline soil restoration may be an environmentally sustainable, safer, and more efficient method, as the halophilic microorganisms have the potential to eliminate salt from saline soils[23]. Such microbes also provide exceptional prototypes for studying the stress resistance, adaptation, and response processes which might consequently be integrated into agricultural crops to cope with the pressures caused by climate modification[24].
2.1. Biofertilization
PGPB can successfully be manipulated in the agro-farming system as alternative strategies to control most of the stated abiotic stresses faced by crops and to increase their yields and would lead to the minimal use of synthetic fertilizers[25]. Arbuscular mycorhizae fungi and plant growth promoting bacteria are beneficial microbes in the soil which provide assistance to plants in many ways. Directly they are concerned with hormones and organic compounds synthesis while indirectly they reduce salinity stress. They can be employed in agriculture as biofertilizers[26]. Improved plant growth under saline soils has been attained with the use of PGPB[27]. Significant increase in growth attributes of rice plants with Bacillus amyloliquefaciens as compared to non-inoculated control plants under salinity stress[28]. Bacillus licheniformis SA03 inoculated with Chrysanthemum plants grown under saline-alkaline conditions significantly decreased saline-alkaline stress in plants along with augmented biomass and survival rates via mediating cellular ABA (abscisic acid) levels[29].
2.2. Biopesticides
Many strains of PGPB have been found to be positively linked with suppressing diverse plant pathogens (both in the rhizosphere and above ground) by producing antagonistic metabolites and enhancing the immunity potential of crops to pathogenic stress[30]. Moreover, Nadeem et al.[31] have comprehensively provided evidence about the growth promotion and biocontrol potentials of AMF and PGPR in agriculture. Egamberdieva et al.[32] found that Pseudomonas chlororaphis TSAU13 and P. extremorientalis TSAU20 reduced the Fusarium infection by 23% and 14% under non-saline conditions and by 42% and 25% under saline conditions, respectively. Tewari and Arora [33] reported EPS (exopolysaccharides) and salicylic acid producing P. aeruginosa PF23EPS+ showed biocontrol against M. phaseolina up to 500 mM NaCl.
2.3. Bioremediation
The concept of using PGPB to facilitate bioremediation of contaminated soil has only recently emerged. For example, Pacwa-Płociniczak et al.[34] proposed that two petroleum hydrocarbon-degrading Rhodococcus spp., with plant growth promoting traits, are likely to be good candidates for the phytoremediation of petroleum hydrocarbon contaminated sites. In recent years, the ecological functions of halophyte symbiotic microorganisms, including the growth promotion of host and crops, enhanced salt tolerance, and joint phytoremediation of contaminated soil, have been clarified by increasing studies[35][36]. As bioremediation agents, Streptomyces (in particular halophiles), participate in alleviating the toxic effect of xenobiotics on plants, using the strategies previously mentioned. Streptomyces are widely used in the remediation of soils contaminated with different pollutants. Moreover, they are studied as PGP in salt-affected soils, where they provide plants with salt resistance to face this major problem for agriculture[37][38].
Table 1. Various roles of PGPB (Plant growth promoting bacteria) in enhancing yield and growth of different plants/crops under salt stress.
| Crops | PGPB | NaCl (mM) | Beneficial Effects | Reference |
|---|---|---|---|---|
| Lettuce | Azotobacter chroococcum | 50 and 100 | Enhanced the radicle lengths and plumule of germinated seeds | [39] |
| Sunflowers | Pseudomonas fluorescens CECT 378T | 100 | Fresh weight significantly improved by more than 10%; K+/Na+ proportion | [40] |
| Strawberry | Bacilus sp. | 35 | Ameliorated fruit productivity, leaf water amount, ionic constitution, and membrane permeability | [41] |
| Wheat | Serratia sp. SL-12 | 150–200 | Improvement in plant increase, as determined by factors like root/shoot length, dry/fresh weight, and augmentation of photosynthetic pigment | [42] |
| Maize | Trichoderma harzianum Th-6 | 50–150 | Improves water composition and stomatal conductance, elevated pigment amounts, and increases photosynthetic performance | [43] |
| Barley | Azospirillum brasilense NO40 | 250 and 350 | Mitigate photosynthetic pigments in the roots and shoots, photosynthetic effectiveness, transpiration level, stomatal conductance, agglomeration of Mg, P, K, Fe, and Ca | [44] |
3. Mechanisms of PGPB (Plant Growth Promoting Bacteria) towards Salinity Tolerance
The key mechanisms involved in PGPR (plant growth promotion rhizobacteria) salinity tolerance include specific membrane or cell wall structures, draining ions out of the cell through salt efflux, or modification of their intracellular environment through accumulation of nontoxic organic osmolytes, and adapting enzymes and proteins to function under high solute ion concentrations. The ability of PGPB to improve crop yields during salt stress includes many direct and indirect pathways such as ferrous iron minerals and inorganic phosphate solubilization, exopolysaccharides and biofilms synthesis[45], production of phytohormones[46], increased ACC (1-aminocyclopropane-1-carboxylate) deaminase activity[47], and nitrogen fixation[48]. The Figure 1 shows PGPB (Plant growth promoting bacteria) salinity resistance strategies and mechanisms of plant growth improvement under salt stress. PGP bacteria may survive high salinity via three main mechanisms (pumping out the cell, intracellular adaptation processes, and cell wall construction). These bacteria stimulate plant growth during salt stress by means of several mechanisms including phytohormones production, amelioration of nutrients uptake, ACC deaminase, P solubilization, EPS synthesis, biofilms formation, and N2 fixation.
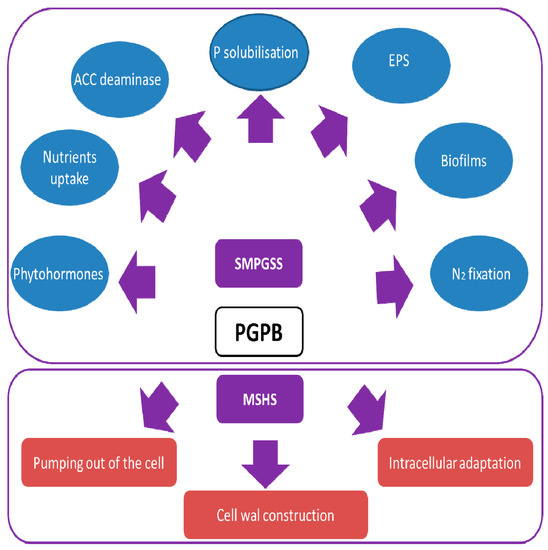
Figure 1. PGPB (Plant growth promoting bacteria) salinity resistance strategies and mechanisms of plant growth improvement under salt stress (SMPGSS: stimulation mechanisms of plant growth under salt stress; MSHS: mechanisms surviving high salinity, PGPB: plant growth-promoting bacteria, EPS: exopolysaccharides).
References
- Glick, B.R. Plant growth-promoting bacteria: Mechanisms and applications. Scientifica 2012, 2012, 1–15.
- Chandra, D.; Srivastava, R.; Gupta, V.V.; Franco, C.M.; Sharma, A.K. Evaluation of ACC-deaminase-producing rhizobacteria to alleviate water-stress impacts in wheat (Triticum aestivum L.) plants. Can. J. Microbiol. 2019, 65, 387–403.
- Grime, J.P. Evidence for the existence of three primary strategies in plants and its relevance to ecological and evolutionary theory. Am. Nat. 1977, 111, 169–1194.
- Kumar, A.; Verma, J.P. Does plant–microbe interaction confer stress tolerance in plants: A review? Microbiol. Res. 2018, 207, 41–52.
- Machado, R.M.A.; Serralheiro, R.P. Soil salinity: Effect on vegetable crop growth. Management practices to prevent and mitigate soil salinization. Horticulturae 2017, 3, 30.
- Pimentel, D.; Berger, B.; Filiberto, D.; Newton, M.; Wolfe, B.; Karabinakis, E.; Clark, S.; Poon, E.; Abbett, E.; Nandaopal, S. Water Resources: Agricultural and Environmental Issues. BioScience 2004, 54, 909–918.
- Numan, M.; Bashir, S.; Khan, Y.; Mumtaz, R.; Shinwari, Z.K.; Khan, A.L.; Khan, A.; Ahmed, A.H. Plant growth promoting bacteria as an alternative strategy for salt tolerance in plants: A review. Microbiol. Res. 2018, 209, 21–32.
- Yadav, A.N.; Saxena, A.K. Biodiversity and biotechnological applications of halophilic microbes for sustainable agriculture. J. Appl. Biol. Biotechnol. 2018, 6, 48–55.
- Munns, R. Genes and salt tolerance: Bringing them together. New Phytol. 2005, 167, 645–663.
- Jamil, A.; Riaz, S.; Ashraf, M.; Foolad, M.R. Gene expression profiling of plants under salt stress. Crit. Rev. Plant Sci. 2011, 30, 435–458.
- Arora, S. Halotolerant Microbes for Amelioration of Salt-Affected Soils for Sustainable Agriculture. In Phyto-Microbiome in Stress Regulation; Kumar, M., Kumar, V., Prasad, R., Eds.; Springer: Singapore, 2020; pp. 323–343.
- Zhang, K.; Shi, Y.; Cui, X.; Yue, P.; Li, K.; Liu, X.; Tripathi, B.M.; Chu, H. Salinity is a key determinant for soil microbial communities in a desert ecosystem. MSystems 2019, 4, 1–11.
- Chu, T.N.; Tran, B.T.H.; Hoang, M.T.T. Plant growth-promoting rhizobacterium Pseudomonas PS01 induces salt tolerance in Arabidopsis thaliana. BMC Res. Notes 2019, 12, 1–7.
- Paul, D.; Lade, H. Plant-growth-promoting rhizobacteria to improve crop growth in saline soils: A review. Agron. Sustain. Dev. 2014, 34, 737–752.
- Vessey, J.K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 2003, 255, 571–586.
- Souza, R.D.; Ambrosini, A.; Passaglia, L.M. Plant growth-promoting bacteria as inoculants in agricultural soils. Genet. Mol. Biol. 2015, 38, 401–419.
- Bakka, K.; Challabathula, D. Amelioration of Salt Stress Tolerance in Plants by Plant Growth-Promoting Rhizobacteria: Insights from “Omics” Approaches. In Plant Microbe Symbiosis; Varma, A., Tripathi, S., Prasad, R., Eds.; Springer: Cham, Switzerland, 2020; pp. 303–330.
- El-Akhdar, I.; Elsakhawy, T.; Abo-Koura, H.A. Alleviation of Salt Stress on Wheat (Triticum aestivum L.) by Plant Growth Promoting Bacteria strains Bacillus halotolerans MSR-H4 and Lelliottia amnigena MSR-M49. J. Adv. Appl. Microbiol. 2020, 20, 44–58.
- Obledo, E.N.; Barraga’n-Barraga’n, L.B.; Gutie’rrez-Gonza’lez, P.; Ramı’rez-Herna’ndez, B.C.; Ramı’rez, J.J.; Rodrı’guez-Garay, B. Increased photosyntethic efficiency generated by fungal symbiosis in Agave victoria-reginae. Plant Cell Tissue Organ Cult. 2003, 74, 237–241.
- Lynch, J.M. The Rhizosphere; Wiley-Interscience: Chichester, UK, 1990; p. 4.
- Ramadoss, D.; Lakkineni, V.K.; Bose, P.; Ali, S.; Annapurna, K. Mitigation of salt stress in wheat seedlings by halotolerant bacteria isolated from saline habitats. SpringerPlus 2013, 2, 1–7.
- Yao, L.; Wu, Z.; Zheng, Y.; Kaleem, I.; Li, C. Growth promotion and protection against salt stress by Pseudomonas putida Rs-198 on cotton. Eur. J. Soil Biol. 2010, 46, 49–54.
- Arora, S.; Trivedi, R.; Rao, G.G. Bioremediation of coastal and inland salt affected soils using halophyte plants and halophilic soil microbes. In CSSRI Annual Report 2012–13; CSSRI: Karnal, India, 2013; pp. 94–100.
- Grover, M.; Ali, S.Z.; Sandhya, V.; Rasul, A.; Venkateswarlu, B. Role of microorganisms in adaptation of agriculture crops to abiotic stresses. World J. Microbiol. Biotechnol. 2011, 27, 1231–1240.
- Majeed, A.; Muhammad, Z.; Ahmad, H. Plant growth promoting bacteria: Role in soil improvement, abiotic and biotic stress management of crops. Plant Cell Rep. 2018, 37, 1599–1609.
- Islam, S. Microorganisms in the Rhizosphere and their Utilization in Agriculture: A Mini Review. PSM Microbiol. 2018, 3, 105–110.
- Shrivastava, P.; Kumar, R. Soil salinity: A serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J. Biol. Sci. 2015, 22, 123–131.
- Shahzad, R.; Khan, A.L.; Bilal, S.; Waqas, M.; Kang, S.M.; Lee, I.J. Inoculation ofabscisic acid-producing endophytic bacteria enhances salinity stress tolerance in Oryza sativa. Environ. Exp. Bot. 2017, 136, 68–77.
- Zhou, C.; Fi, L.; Xie, Y.; Zhu, L.; Xiao, X.; Ma, Z.; Wang, J. Involvement of abscisic acid in microbe-induced saline-alkaline resistance in plants. Plant Signal. Behav. 2017, 12, 1–4.
- Berendsen, R.L.; Vismans, G.; Yu, K.; Song, Y.; Jonge, R.; Burgman, W.P.; Pieterse, C.M. The disease-induced assemblage of a plant beneficial bacterial consortium. ISME J. 2018, 12, 1496–1507.
- Nadeem, S.M.; Ahmad, M.; Zahir, Z.A.; Javaid, A.; Ashraf, M. The role of mycorrhizae and plant growth promoting rhizobacteria (PGPR) in improving crop productivity under stressful environments. Biotechnol. Adv. 2014, 32, 429–448.
- Egamberdieva, D.; Davranov, K.; Wirth, S.; Hashem, A.; Abd Allah, E.F. Impact of soil salinity on the plant-growth-promoting and biological control abilities of root associated bacteria. Saudi J. Biol. Sci. 2017, 24, 1601–1608.
- Tewari, S.; Arora, N.K. Role of salicylic acid from Pseudomonas aeruginosa PF23EPS+ ingrowth promotion of sunflower in saline soils infested with phytopathogen Macrophomina phaseolina. Environ. Sustain. 2018, 1, 49–59.
- Pacwa-Płociniczak, M.; Płociniczak, T.; Iwan, J.; Żarska, M.; Chorążewski, M.; Dzida, M.; Piotrowska-Seget, Z. Isolation of hydrocarbon-degrading and biosurfactant producing bacteria and assessment their plant growth-promoting traits. J. Environ. Manag. 2016, 168, 175–184.
- Mallick, I.; Bhattacharyya, C.; Mukherji, S.; Dey, D.; Sarkar, S.C.; Mukhopadhyay, U.K.; Ghosh, A. Effective rhizoinoculation and biofilm formation by arsenic immobilizing halophilic plant growth promoting bacteria (PGPB) isolated from mangrove rhizosphere: A step towards arsenic rhizoremediation. Sci. Total Environ. 2018, 610, 1239–1250.
- Kearl, J.; McNary, C.; Lowman, J.S.; Mei, C.; Aanderud, Z.T.; Smith, S.T.; West, J.; Colton, E.; Hamson, M.; Nielsen, B.L. Salt-tolerant halophyte rhizosphere bacteria stimulate growth of alfalfa in salty soil. Front. Microbiol. 2019, 10, 1–11.
- Thavamani, P.; Samkumar, R.A.; Satheesh, V.; Subashchandrabose, S.R.; Ramadass, K.; Naidu, R.; Venkateswarlu, K.; Megharaj, M. Microbes from mined sites: Harnessing their potential for reclamation of derelict mine sites. Environ. Pollut. 2017, 230, 495–505.
- Jaemsaeng, R.; Jantasuriyarat, C.; Thamchaipenet, A. Molecular interaction of1-aminocyclopropane-1-carboxylate deaminase (ACCD)-producing endophytic Streptomyces sp. GMKU 336 towards salt-stress resistance of Oryza sativa L. cv. KDML105. Sci. Rep. 2018, 8, 1–15.
- Hussein, K.A.; Joo, J.H. Plant growth-promoting rhizobacteria improved salinity tolerance of Lactuca sativa and Raphanus sativus. J. Microbiol. Biotechnol. 2018, 28, 938–945.
- Shilev, S.; Sancho, E.D.; Benlloch-Gonzalez, M. Rhizospheric bacteria alleviate salt-produced stress in sunflower. J. Environ. Manag. 2012, 95, S37–S41.
- Karlidag, H.; Esitken, A.; Yildirim, E.; Donmez, M.F.; Turan, M. Effects of plant growth promoting bacteria on yield, growth, leaf water content, membrane permeability, and ionic composition of strawberry under saline conditions. J. Plant Nutr. 2010, 34, 34–45.
- Singh, R.P.; Jha, P.N. Alleviation of salinity-induced damage on wheat plant by an ACC deaminase-producing halophilic bacterium Serratia sp. SL-12 isolated from a salt lake. Symbiosis 2016, 69, 101–111.
- Yasmeen, R.; Siddiqui, Z.S. Physiological responses of crop plants against Trichoderma harzianum in saline environment. Acta Bot. Croat. 2017, 76, 154–162.
- Omar, M.N.A.; Osman, M.E.H.; Kasim, W.A.; Abd El-Daim, I.A. Improvement of Salt Tolerance Mechanisms of Barley Cultivated Under Salt Stress Using Azospirillum brasilense. In Salinity and Water Stress: Improving Crop Efficiency; Ashraf, M., Ozturk, M., Athar, H., Eds.; Springer: Dordrecht, The Netherlands, 2009; Volume 44, pp. 133–147.
- Kasim, W.A.; Gaafar, R.M.; Abou-Ali, R.M.; Omar, M.N.; Hewait, H.M. Effect of biofilm forming plant growth promoting rhizobacteria on salinity tolerance in barley. Ann. Agric. Sci. 2016, 61, 217–227.
- Naz, I.; Bano, A.; Ul-Hassan, T. Isolation of phytohormones producing plant growth promoting rhizobacteria from weeds growing in Khewra salt range, Pakistan and their implication in providing salt tolerance to Glycine max L. Afr. J. Biotechnol. 2009, 8, 5762–5768.
- Bal, H.B.; Nayak, L.; Das, S.; Adhya, T.K. Isolation of ACC deaminase producing PGPR from rice rhizosphere and evaluating their plant growth promoting activity under salt stress. Plant Soil 2013, 366, 93–105.
- Sadiki, M.; Rabih, K. Selection of chickpea (Cicer arietinum) for yield and symbiotic nitrogen fixation ability under salt stress. Agronomie 2001, 21, 659–666.





