Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Shazia Nakhoda | -- | 1970 | 2022-07-09 15:04:37 | | | |
| 2 | Conner Chen | Meta information modification | 1970 | 2022-07-11 02:59:30 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Nakhoda, S.; Rizwan, F.; Vistarop, A.; Nejati, R. Immunotherapy for Hodgkin's lymphoma. Encyclopedia. Available online: https://encyclopedia.pub/entry/24970 (accessed on 08 May 2026).
Nakhoda S, Rizwan F, Vistarop A, Nejati R. Immunotherapy for Hodgkin's lymphoma. Encyclopedia. Available at: https://encyclopedia.pub/entry/24970. Accessed May 08, 2026.
Nakhoda, Shazia, Farsha Rizwan, Aldana Vistarop, Reza Nejati. "Immunotherapy for Hodgkin's lymphoma" Encyclopedia, https://encyclopedia.pub/entry/24970 (accessed May 08, 2026).
Nakhoda, S., Rizwan, F., Vistarop, A., & Nejati, R. (2022, July 09). Immunotherapy for Hodgkin's lymphoma. In Encyclopedia. https://encyclopedia.pub/entry/24970
Nakhoda, Shazia, et al. "Immunotherapy for Hodgkin's lymphoma." Encyclopedia. Web. 09 July, 2022.
Copy Citation
The introduction of immunotherapy into the treatment options for Hodgkin’s lymphoma has improved survival in patients with recurrence of their cancer. These agents help the body’s immune system respond and clear cancer cells.
checkpoint inhibitors
immunotherapy
Hodgkin lymphoma
1. Introduction
Classical Hodgkin’s lymphoma (cHL) accounts for roughly 11% of all lymphomas in the United States [1] and is defined by its distinct pathologic features consistent with Reed Sternberg cells in a background of inflammatory cells, which enable the evasion of immune-mediated antitumor mechanisms [2][3][4]. Although the majority of patients with cHL can be cured with standard chemoradiation, patients who are unresponsive to initial treatment or who are not candidates for curative regimens have poor outcomes [5][6][7]. Immune checkpoint inhibitors (CPIs) enable the reconstitution of the immune response to target malignant cells, offering an alternative mechanism of action from traditional cytotoxic chemotherapy and have thus transformed the treatment landscape for relapsed/refractory (R/R) cHL.
Approximately 10–25% of patients with advanced or early-stage unfavorable risk disease have disease relapse [8][9]. The standard of care on relapse consists of salvage chemotherapy followed by autologous stem cell transplant (ASCT). This approach cures only about 50% of patients and is associated with significant risk for treatment-related toxicity and secondary malignancy [10][11]. Older patients and those with medical comorbidities who are not candidates for intensive therapy remain at a particularly high risk for relapse and inferior survival [12]. With modern frontline treatment regimens, patients over the age of 60 years had a ~15% worse 3-year progression-free survival (PFS) compared with younger patients [13].
Although the addition of the anti-CD30 monoclonal antibody drug conjugate brentuximab vedotin (BV) to the cHL treatment armamentarium has improved outcomes, the rate of relapse in patients treated with BV-containing frontline therapy is still 18% at 5 year follow-up [14]. In the high-risk R/R population who received BV as maintenance therapy after ASCT, 41% of patients have progression of their disease [15]. In the multiple-relapse setting, these responses are not durable, with only 38% of patients treated with BV maintaining their complete response (CR) at 5 year follow-up [16][17].
2. Immune Checkpoints and the Tumor Microenvironment (TME) in cHL
The interaction of the programmed death receptor ligands 1 and 2 (PDL1/CD274 and PDL2/CD273) on tumor cells with PD-1 receptors on T cells leads to reduced T-cell activation and proliferation, thus enabling tumors to evade immune response (see Figure 1) [18]. The amplification of PDL1 and PDL2 on tumor cells, coded on chromosome 9p24.1, is mediated by PIM serine/threonine kinases on tumor cells via the constitutive activation of the NFkB and JAK-STAT signaling pathways [19].
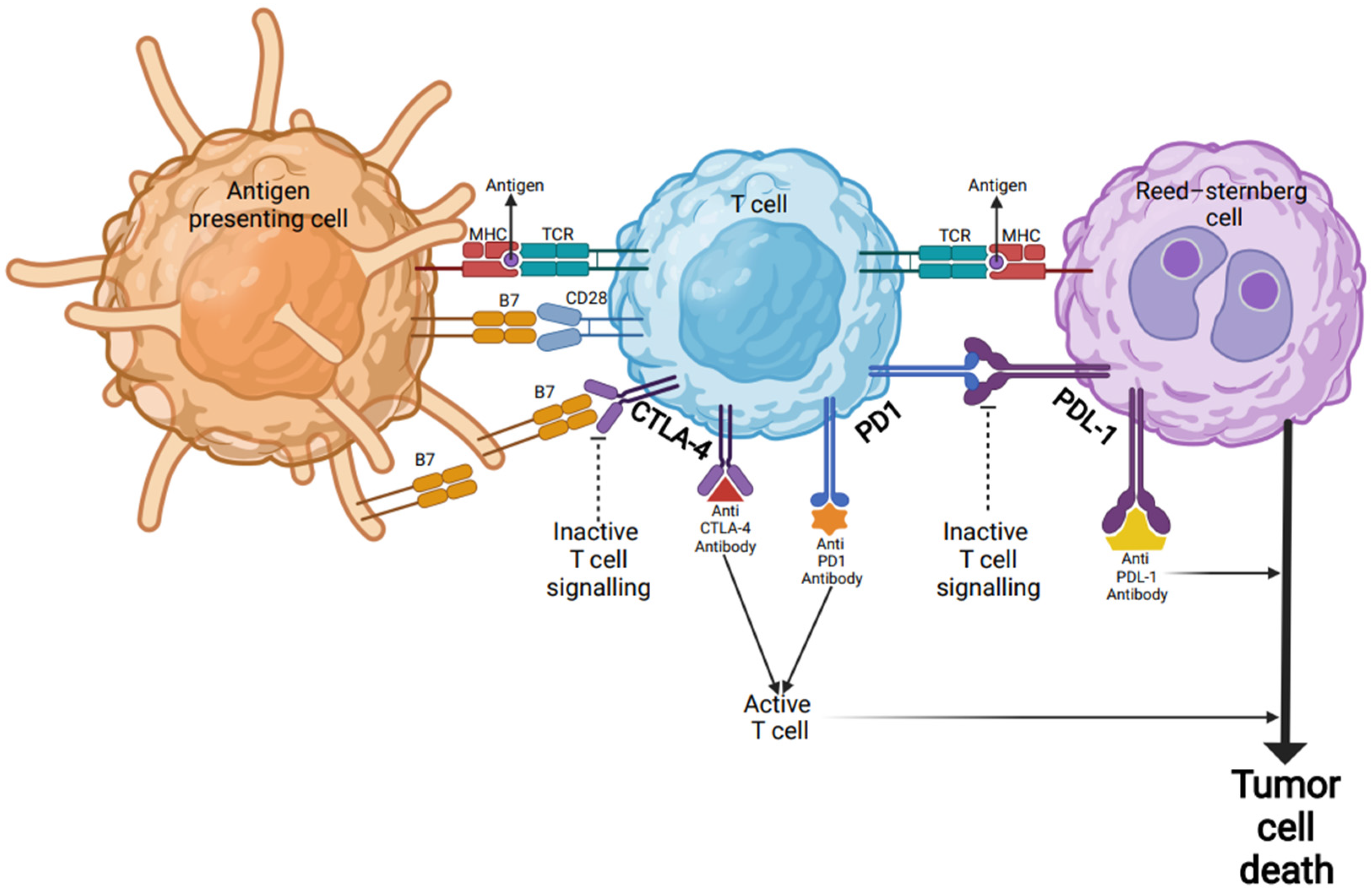
Figure 1. Checkpoint inhibitor immunotherapy in Hodgkin’s lymphoma. Inactive T cells are activated through their TCR by encountering antigenic peptides presented by the MHC complex on the surfaces of APCs or tumor cells (Reed-Sternberg cells). In addition to TCR-MHC engagement, a co-stimulatory signal via B7 protein is required for target-cell lysis and effector cell responses. B7 protein on activated APCs can pair with either a CD28 on the surface of a T-cell to produce a costimulatory signal to enhance the activity of TCR-MHC signal and T-cell activation, or it can pair with CTLA-4 to produce an inhibitory signal to keep the T cell in the inactive state. Blocking the binding of B7 to CTLA-4 with an anti-CTLA-4 antibody allows T cells to be activated and to kill tumor cells. Other immune checkpoint proteins such as PD-1 on the surfaces of T cells and PD-L1 on tumor cells can also prevent T cells from killing tumor cells. Immune checkpoint blockade via monoclonal antibodies (anti-PD-L1 or anti-PD-1) can lead T cells to kill tumor cells. Abbreviations: MHC: major histocompatibility complex; APC: antigen-presenting cells; TCR: T-cell receptor; CTLA-4: cytotoxic T-lymphocyte associated antigen 4; PD-1: programmed death 1; PD-L1: programmed death-ligand 1.
Anti-PD-1 monoclonal antibody (mAb) therapies reconstitute the immune surveillance of malignant cells, enabling a rapid reduction in Reed-Sternberg cells and PDL1+ inflammatory cells as well as a durable PD-1 blockade that persists even after treatment cessation based on patient samples during and after treatment [20]. A rise in CD4+ T cell receptors, dendritic cells, and mature, highly differentiated NK cells is seen in peripheral blood after anti-PD1 mAb treatment, correlating with earlier and more robust clinical response [20][21][22][23].
PDL-1 expression is a well-defined marker of response to anti-PD-1 mAb across multiple cancer subtypes. In addition to this, 9p24.1 copy number amplification and the expression of major histocompatibility complex class II are associated with improved response to CPI in cHL [24]. In the frontline setting, these biomarkers did not demonstrate an association with early response rates with anti-PD-1 mAb-based therapy [25]. Emerging data on the role of metabolic disorders and patient demographic factors such as sex, age, and gender on effector T cell activity and PD-1 expression suggest that these factors may also contribute to CPI efficacy [26]. The further investigation of these tumoral biomarkers and the effects of extra-tumoral risk factors on TME may help guide treatment decision-making in the future.
Lastly, other immune checkpoints have been identified as potential targets in the TME of cHL, including cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), lymphocyte-activation gene-3 (LAG-3), and T-cell immunoglobulin and mucin-domain containing 3 (TIM-3) [27][28]. The CD68 ligand that binds CTLA-4 is commonly expressed on HRS cells, and this expression persists after treatment with PD-1 blockade. This has therefore served as the rationale for targeting this protein with the monoclonal antibody ipilimumab to overcome anti-PD-1 mAb resistance [27]. Although LAG-3 and TIM-3 are not as frequently expressed on HRS cells, these proteins are commonly seen within the TME surrounding HRS cells [28]. However, ongoing investigation is needed to identify the clinical utility of targeting these alternative immune checkpoints.
3. Checkpoint Inhibitors in Multiple Relapsed/Refractory Disease
For patients with R/R disease after multiple lines of therapy including ASCT +/− BV, anti-PD-1 mAb monotherapy has an overall response rate (ORR) of ~70% with a median PFS of 14–15 months. Although only 20–30% of responders have CR in this setting, those who do achieve CR have a prolonged remission with a median duration of response (mDOR) of 37 months with nivolumab; additionally, mDOR was not reached with pembrolizumab at 5 year follow-up [29][30]. In a study of anti-PD-1 mAb tislelizumab in patients with relapse after ASCT or 2 systemic regimens in a Chinese population, 67% of patients achieved CR, and ~50% of patients had ongoing response at 30 month follow-up, confirming the correlation of depth of response to duration of response with this class of agents [31]. Novel combination regimens of immunotherapy combined with other targeted therapies have deepened response rates in the multiple-relapse setting. The combinations of BV with the checkpoint inhibitors nivolumab and ipilimumab achieved CRs of 61% and 57%, respectively. Although the combination of all three agents achieved the highest CR of 73%, the inclusion of ipilimumab resulted in higher toxicity than BV + Nivo in this phase I/II study. The rates of grade 3–4 adverse events were reported at 43% and 50%, respectively, in the BV-Ipi and triplet combination arms but only 16% in the BV-Nivo group. [32]. Therefore, anti-PD-1 mAb based therapies are preferred.
In early clinical investigation, the addition of hypomethylating agent to anti-PD-1 mAb improved CR from 32 to 71% in patients with R/R cHL to at least 2 prior lines of therapy, with 100% of patients still responding to combination therapy at 6 month follow-up. Investigational regimens evaluating nivolumab in combination with JAK2 inhibitor, and BTK inhibitor are also underway. Additionally, retreatment with anti PD-1 mAb has been described in a small number of patients with multiply relapsed disease, demonstrating ORRs of 70–100% and CRs of 33–73% [33][34][35][36]. Phase II investigation of combination BV + Nivo in patients with prior treatment of either agent is underway and may provide more insight into the roles of sequencing and retreatment as these agents are introduced to patients in earlier lines of therapy.
4. Immunotherapy as First Salvage and Bridge to ASCT
Treatment with standard chemotherapy options in first relapse followed by ASCT leads to long-term cure in only half of patients, with reported 6 year event-free survival and overall survival (OS) of 45% and 55%, respectively [37]. Standard cytotoxic chemotherapy salvage achieved an ORR of ~70–90% and a CR of ~20–30% [38][39][40][41][42]. In patients who fail to respond to initial salvage chemotherapy, the outcomes after ASCT are particularly dismal. At 10 year follow-up, the PFS and OS are only 7–23% and 11–17%, respectively [39][40] Achieving complete metabolic response (CMR) to salvage therapy prior to ASCT is associated with superior outcomes and has served as a rationale for incorporating targeted therapies into first salvage lines of therapy. While the addition of BV into first-line salvage regimens has improved CR to 70–80% [41][43][44][45], single-agent BV induces CRs of only 27–43% when assessed as monotherapy or as part of PET-adapted bridging therapy ASCT [46][47].
On the other hand, the investigation of anti-PD-1 mAb in the first relapse in several phase II studies has demonstrated increased depth of response both as a single agent and as combination therapy [48][49][50]. Combination BV and Nivo demonstrated ORR of 82–85% and CR of 59–67% [51][52][53] At 3 year follow-up after combination achieved a PFS of 77%, and in patients who responded to induction and proceeded to ASCT, PFS improved to 91% [48]. In a small phase II PET-adapted study evaluating nivolumab monotherapy followed by escalation to nivolumab with ifosfamide, carboplatin, and etoposide (ICE) in patients with positive interim PET scans, 71% of patients achieved CR to the single agent nivolumab and were eligible to proceed directly to ASCT, sparing 26 of the total 42 evaluable patients from requiring cytotoxic treatments prior to ASCT. A total of 9 of the 42 patients required escalation of their treatment to N-ICE, and of these, 89% achieved CR [50]. This approach demonstrated overall 2 year PFS and OS of 72% and 95%, respectively [50]. Larger studies are underway to evaluate nivolumab monotherapy as salvage prior to ASCT.
Other small studies evaluating combination of immunotherapy with standard regimens as bridge to ASCT, including pembrolizumab combined with ICE or gemcitabine, vinorelbine, and pegylated liposomal doxorubicin (GVD), showed similarly high CR rates of 86–95% [54]. The active investigation of other anti-PD-1 mAb combination regimens via a PET-adapted approach using anti-PD1 mAb with GVD are underway to assess efficacy with less toxic or chemotherapy-sparing therapies.
5. Immunotherapy in Primary Refractory Disease
Historical data in patients with R/R disease within 1 year of frontline therapy who are treated with standard salvage chemotherapy followed by ASCT demonstrated a 10 year freedom from second failure (FF2F) and OS of 34 and 43%, respectively [9]. The incorporation of CPI has significantly improved outcomes in this population. In a subgroup analysis of R/R patients treated with pembrolizumab, patients with primary refractory disease achieved a CR of 35% with mDOR of 17 months at a median of 28 month follow-up, achieving 2 year PFS and OS of 32% and 94%, respectively. In a phase II study of pembrolizumab with chemotherapy (GVD) as initial salvage therapy as a bridge to ASCT, 79% of patients had either primary refractory disease or relapse within 1 year. All patients responded to this combination treatment with a remarkable CR of 95%, and 36 of the 39 patients enrolled proceeded to ASCT. All remained in remission at 13.5 month follow-up [49].
6. Immunotherapy as Maintenance after ASCT
Lastly, CPI has also been explored as consolidation therapy after ASCT similar to BV, as investigated in the phase III ATHERA study [15][55]. In the ATHERA study, high-risk patients, defined as primary refractory or relapse/progression of disease within 12 months of frontline therapy or extra-nodal involvement at relapse, were included. Extended follow-up of BV maintenance demonstrated a 5-year PFS of 59% vs 41% with placebo [55]. Phase II evaluation of pembrolizumab after ASCT demonstrated an estimated 18 month PFS of 82% overall, and among patients in the subgroup who met the criteria for high-risk disease per the ATHERA protocol, PFS was 85% [15][55].
In the absence of long-term follow-up and head-to-head comparison, the PFS with post-ASCT consolidative pembrolizumab compares favorably with that of BV. However, as BV has moved to the upfront setting as the standard of care for advanced-stage cHL and nivolumab is being used in earlier lines of therapy, the role of maintenance BV or anti-PD-1 mAb is less clear. Ongoing phase II investigation of the combination of BV and nivolumab in patients with relapse who previously received either agent may provide insight into the roles of sequencing and retreatment with these agents as consolidation post ASCT.
References
- National Cancer Institute. Cancer Stat Facts: Hodgkin Lymphoma. In Cancer Stat.; 2016. Available online: https://seer.cancer.gov/statfacts/html/hodg.html (accessed on 30 April 2022).
- Vardiman, J.W.; Harris, N.L.; Brunning, R.D. The World Health Organization (WHO) classification of the myeloid neoplasms. Blood 2002, 100, 2292–2302.
- Weniger, M.A.; Küppers, R. Molecular biology of Hodgkin lymphoma. Leukemia 2021, 35, 968–981.
- Aldinucci, D.; Gloghini, A.; Pinto, A.; De Filippi, R.; Carbone, A. The classical Hodgkin’s lymphoma microenvironment and its role in promoting tumour growth and immune escape. J. Pathol. 2010, 221, 248–263.
- Gordon, L.I.; Hong, F.; Fisher, R.I.; Bartlett, N.; Connors, J.M.; Gascoyne, R.D.; Wagner, H.; Stiff, P.J.; Cheson, B.D.; Gospodarowicz, M.; et al. Randomized Phase III Trial of ABVD Versus Stanford V With or Without Radiation Therapy in Locally Extensive and Advanced-Stage Hodgkin Lymphoma: An Intergroup Study Coordinated by the Eastern Cooperative Oncology Group (E2496). J. Clin. Oncol. 2013, 31, 684–691.
- Carde, P.; Karrasch, M.; Fortpied, C.; Brice, P.; Khaled, H.; Casasnovas, O.; Caillot, D.; Gaillard, I.; Bologna, S.; Ferme, C.; et al. Eight Cycles of ABVD Versus Four Cycles of BEACOPP escalated Plus Four Cycles of BEACOPP baseline in Stage III to IV, International Prognostic Score ≥ 3, High-Risk Hodgkin Lymphoma: First Results of the Phase III EORTC 20012 Intergroup Trial. J. Clin. Oncol. 2016, 34, 2028–2036.
- Sasse, S.; Bröckelmann, P.J.; Goergen, H.; Plütschow, A.; Müller, H.; Kreissl, S.; Buerkle, C.; Borchmann, S.; Fuchs, M.; Borchmann, P.; et al. Long-Term Follow-Up of Contemporary Treatment in Early-Stage Hodgkin Lymphoma: Updated Analyses of the German Hodgkin Study Group HD7, HD8, HD10, and HD11 Trials. J. Clin. Oncol. 2017, 35, 1999–2007.
- Raemaekers, J.M.; André, M.P.; Federico, M.; Girinsky, T.; Oumedaly, R.; Brusamolino, E.; Brice, P.; Fermé, C.; Van Der Maazen, R.; Gotti, M.; et al. Omitting Radiotherapy in Early Positron Emission Tomography–Negative Stage I/II Hodgkin Lymphoma Is Associated With an Increased Risk of Early Relapse: Clinical Results of the Preplanned Interim Analysis of the Randomized EORTC/LYSA/FIL H10 Trial. J. Clin. Oncol. 2014, 32, 1188–1194.
- Sibon, D.; Morschhauser, F.; Resche-Rigon, M.; Ghez, D.; Dupuis, J.; Marcais, A.; Deau-Fischer, B.; Bouabdallah, R.; Sebban, C.; Salles, G.; et al. Single or tandem autologous stem-cell transplantation for first-relapsed or refractory Hodgkin lymphoma: 10-year follow-up of the prospective H96 trial by the LYSA/SFGM-TC study group. Haematologica 2015, 101, 474–481.
- Khera, N.; Storer, B.; Flowers, M.E.; Carpenter, P.A.; Inamoto, Y.; Sandmaier, B.M.; Martin, P.J.; Lee, S.J. Nonmalignant Late Effects and Compromised Functional Status in Survivors of Hematopoietic Cell Transplantation. J. Clin. Oncol. 2012, 30, 71–77.
- Schaapveld, M.; Aleman, B.M.P.; Van Eggermond, A.M.; Janus, C.P.M.; Krol, S.; Van Der Maazen, R.W.M.; Roesink, J.M.; Raemaekers, J.M.M.; De Boer, J.P.; Zijlstra, J.M.; et al. Second Cancer Risk Up to 40 Years after Treatment for Hodgkin’s Lymphoma. N. Engl. J. Med. 2015, 373, 2499–2511.
- Evens, A.M.; Hong, F.; Gordon, L.I.; Fisher, R.I.; Bartlett, N.; Connors, J.M.; Wagner, H.; Gospodarowicz, M.K.; Cheson, B.D.; Advani, R.; et al. Efficacy and tolerability of ABVD and Stanford V for elderly advanced-stage Hodgkin lymphoma (HL): Analysis from the phase III randomized U.S. Intergroup Trial E2496. J. Clin. Oncol. 2011, 29, 8035.
- Straus, D.J.; Długosz-Danecka, M.; Alekseev, S.; Illés, Á.; Picardi, M.; Lech-Maranda, E.; Feldman, T.; Smolewski, P.; Savage, K.J.; Bartlett, N.; et al. Brentuximab vedotin with chemotherapy for stage III/IV classical Hodgkin lymphoma: 3-year update of the ECHELON-1 study. Blood 2020, 135, 735–742.
- Straus, D.J.; Długosz-Danecka, M.; Connors, J.M.; Alekseev, S.; Illés, Á.; Picardi, M.; Lech-Maranda, E.; Feldman, T.; Smolewski, P.; Savage, K.J.; et al. Brentuximab vedotin with chemotherapy for stage III or IV classical Hodgkin lymphoma (ECHELON-1): 5-year update of an international, open-label, randomised, phase 3 trial. Lancet Haematol. 2021, 8, e410–e421.
- Moskowitz, C.H.; Walewski, J.; Nademanee, A.; Masszi, T.; Agura, E.; Holowiecki, J.; Abidi, M.H.; Chen, A.I.; Stiff, P.; Viviani, S.; et al. Five-year PFS from the AETHERA trial of brentuximab vedotin for Hodgkin lymphoma at high risk of progression or relapse. Blood 2018, 132, 2639–2642.
- Younes, A.; Gopal, A.K.; Smith, S.E.; Ansell, S.M.; Rosenblatt, J.D.; Savage, K.J.; Ramchandren, R.; Bartlett, N.L.; Cheson, B.D.; De Vos, S.; et al. Results of a Pivotal Phase II Study of Brentuximab Vedotin for Patients With Relapsed or Refractory Hodgkin’s Lymphoma. J. Clin. Oncol. 2012, 30, 2183–2189.
- Chen, R.; Gopal, A.K.; Smith, S.E.; Ansell, S.M.; Rosenblatt, J.D.; Savage, K.J.; Connors, J.M.; Engert, A.; Larsen, E.K.; Huebner, D.; et al. Five-year survival and durability results of brentuximab vedotin in patients with relapsed or refractory Hodgkin lymphoma. Blood 2016, 128, 1562–1566.
- Zou, W.; Wolchok, J.D.; Chen, L. PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: Mechanisms, response biomarkers, and combinations. Sci. Transl. Med. 2016, 8, 328rv4.
- Szydłowski, M.; Prochorec-Sobieszek, M.; Szumera-Ciećkiewicz, A.; Derezińska, E.; Hoser, G.; Wasilewska, D.; Szymańska-Giemza, O.; Jabłońska, E.; Białopiotrowicz, E.; Sewastianik, T.; et al. Expression of PIM kinases in Reed-Sternberg cells fosters immune privilege and tumor cell survival in Hodgkin lymphoma. Blood 2017, 130, 1418–1429.
- Merryman, R.W.; Redd, R.; Jeter, E.; Wong, J.L.; McHugh, K.; Reynolds, C.; Nazzaro, M.; Varden, A.; Brown, J.R.; Crombie, J.L.; et al. Immune Reconstitution following High-Dose Chemotherapy and Autologous Stem Cell Transplantation with or without Pembrolizumab Maintenance Therapy in Patients with Lymphoma. Transplant. Cell. Ther. 2021, 28, 32.e1–32.e10.
- Guolo, F.; Minetto, P.; Pesce, S.; Ballerini, F.; Clavio, M.; Cea, M.; Frello, M.; Garibotto, M.; Greppi, M.; Bozzo, M.; et al. Post-Transplant Nivolumab Plus Unselected Autologous Lymphocytes in Refractory Hodgkin Lymphoma: A Feasible and Promising Salvage Therapy Associated With Expansion and Maturation of NK Cells. Front. Immunol. 2021, 12, 4535.
- Cader, F.Z.; Hu, X.; Goh, W.L.; Wienand, K.; Ouyang, J.; Mandato, E.; Redd, R.; Lawton, L.N.; Chen, P.-H.; Weirather, J.L.; et al. A peripheral immune signature of responsiveness to PD-1 blockade in patients with classical Hodgkin lymphoma. Nat. Med. 2020, 26, 1468–1479.
- Garcia-Marquez, M.A.; Thelen, M.; Reinke, S.; Keller, D.; Wennhold, K.; Lehmann, J.; Veldman, J.; Borchmann, S.; Rosenwald, A.; Sasse, S.; et al. Reverted exhaustion phenotype of circulating lymphocytes as immune correlate of anti-PD1 first-line treatment in Hodgkin lymphoma. Leukemia 2021, 36, 760–771.
- Roemer, M.G.; Redd, R.A.; Cader, F.Z.; Pak, C.J.; Abdelrahman, S.; Ouyang, J.; Sasse, S.; Younes, A.; Fanale, M.; Santoro, A.; et al. Major Histocompatibility Complex Class II and Programmed Death Ligand 1 Expression Predict Outcome After Programmed Death 1 Blockade in Classic Hodgkin Lymphoma. J. Clin. Oncol. 2018, 36, 942–950.
- Gerhard-Hartmann, E.; Goergen, H.; Bröckelmann, P.J.; Mottok, A.; Steinmüller, T.; Grund, J.; Zamò, A.; Ben-Neriah, S.; Sasse, S.; Borchmann, S.; et al. 9p24.1 alterations and programmed cell death 1 ligand 1 expression in early stage unfavourable classical Hodgkin lymphoma: An analysis from the German Hodgkin Study Group NIVAHL trial. Br. J. Haematol. 2021, 196, 116–126.
- Deshpande, R.P.; Sharma, S.; Watabe, K. The Confounders of Cancer Immunotherapy: Roles of Lifestyle, Metabolic Disorders and Sociological Factors. Cancers 2020, 12, 2983.
- Patel, S.S.; Weirather, J.L.; Lipschitz, M.; Lako, A.; Chen, P.-H.; Griffin, G.K.; Armand, P.; Shipp, M.A.; Rodig, S.J. The microenvironmental niche in classic Hodgkin lymphoma is enriched for CTLA-4-positive T cells that are PD-1-negative. Blood 2019, 134, 2059–2069.
- El Halabi, L.; Adam, J.; Gravelle, P.; Marty, V.; Danu, A.; Lazarovici, J.; Ribrag, V.; Bosq, J.; Camara-Clayette, V.; Laurent, C.; et al. Expression of the Immune Checkpoint Regulators LAG-3 and TIM-3 in Classical Hodgkin Lymphoma. Clin. Lymphoma Myeloma Leuk. 2020, 21, 257–266.
- Armand, P.; Zinzani, P.L.L.; Lee, H.J.; Johnson, N.; Brice, P.; Radford, J.; Ribrag, V.; Molin, D.; Vassilakopoulos, T.P.; Tomita, A.; et al. Five-Year Follow-up of Keynote-087: Pembrolizumab Monotherapy in Relapsed/Refractory Classical Hodgkin Lymphoma (R/R cHL). Blood 2021, 138, 1366.
- Ansell, S.M.; Bröckelmann, P.J.; von Keudell, G.; Lee, H.J.; Santoro, A.; Zinzani, P.L.; Collins, G.P.; Cohen, J.B.; De Boer, J.P.; Kuruvilla, J.; et al. Nivolumab for Relapsed or Refractory (R/R) Classical Hodgkin Lymphoma (CHL) after Autologous Transplantation: 5-year Overall Survival from the Phase 2 Checkmate 205 Study. Hematol. Oncol. 2021, 39.
- Song, Y.; Gao, Q.; Zhang, H.; Fan, L.; Zhou, J.; Zou, D.; Li, W.; Yang, H.; Liu, T.; Wang, Q.; et al. Tislelizumab for Relapsed/Refractory Classical Hodgkin Lymphoma: 3-Year Follow-up and Correlative Biomarker Analysis. Clin. Cancer Res. 2022, 28, 1147–1156.
- Diefenbach, C.S.; Hong, F.; Ambinder, R.F.; Cohen, J.B.; Robertson, M.J.; David, A.K.; Advani, R.H.; Fenske, T.S.; Barta, S.K.; Palmisiano, N.D.; et al. Ipilimumab, nivolumab, and brentuximab vedotin combination therapies in patients with relapsed or refractory Hodgkin lymphoma: Phase 1 results of an open-label, multicentre, phase 1/2 trial. Lancet Haematol. 2020, 7, e660–e670.
- Fedorova, L.V.; Lepik, K.V.; Mikhailova, N.B.; Kondakova, E.V.; Zalyalov, Y.R.; Baykov, V.V.; Babenko, E.V.; Kozlov, A.V.; Moiseev, I.S.; Afanasyev, B.V. Nivolumab discontinuation and retreatment in patients with relapsed or refractory Hodgkin lymphoma. Ann. Hematol. 2021, 100, 691–698.
- Manson, G.; Brice, P.; Herbaux, C.; Bouabdallah, K.; Antier, C.; Poizeau, F.; Dercle, L.; Houot, R. Efficacy of anti-PD1 re-treatment in patients with Hodgkin lymphoma who relapsed after anti-PD1 discontinuation. Haematologica 2020, 105, 2664–2666.
- Lepik, K.V.; Fedorova, L.V.; Kondakova, E.V.; Zalyalov, Y.R.; Babenko, E.V.; Lepik, E.E.; Kotselyabina, P.V.; Beynarovich, A.V.; Popova, M.O.; Volkov, N.P.; et al. A Phase 2 Study of Nivolumab Using a Fixed Dose of 40 mg (Nivo40) in Patients With Relapsed/Refractory Hodgkin Lymphoma. HemaSphere 2020, 4, e480.
- Chan, T.S.Y.; Hwang, Y.; Khong, P.; Leung, A.Y.H.; Chim, C.; Tse, E.W.C.; Kwong, Y. Low-dose pembrolizumab and nivolumab were efficacious and safe in relapsed and refractory classical Hodgkin lymphoma: Experience in a resource-constrained setting. Hematol. Oncol. 2020, 38, 726–736.
- Smith, S.D.; Moskowitz, C.H.; Dean, R.; Pohlman, B.; Sobecks, R.; Copelan, E.; Andresen, S.; Bolwell, B.; Maragulia, J.C.; Vanak, J.M.; et al. Autologous stem cell transplant for early relapsed/refractory Hodgkin lymphoma: Results from two transplant centres. Br. J. Haematol. 2011, 153, 358–363.
- Baetz, T.; Belch, A.; Couban, S.; Imrie, K.; Yau, J.; Myers, R.; Ding, K.; Paul, N.; Shepherd, L.; Iglesias, J.; et al. Gemcitabine, dexamethasone and cisplatin is an active andnon-toxic chemotherapy regimen in relapsed or refractory Hodgkin’s disease: A phase II study by the National Cancer Institute of Canada Clinical Trials Group. Ann. Oncol. 2003, 14, 1762–1767.
- Sirohi, B.; Cunningham, D.; Powles, R.; Murphy, F.; Arkenau, T.; Norman, A.; Oates, J.; Wotherspoon, A.; Horwich, A. Long-term outcome of autologous stem-cell transplantation in relapsed or refractory Hodgkin’s lymphoma. Ann. Oncol. 2008, 19, 1312–1319.
- Moskowitz, C.H.; KewalRamani, T.; Nimer, S.D.; Gonzalez, M.; Zelenetz, A.; Yahalom, J. Effectiveness of high dose chemoradiotherapy and autologous stem cell transplantation for patients with biopsy-proven primary refractory Hodgkin’s disease. Br. J. Haematol. 2004, 124, 645–652.
- Stamatoullas, A.; Ghesquieres, H.; Filliatre, L.C.; Quittet, P.; Morschhauser, F.; Ribrag, V.; Edeline, V.; Brice, P. Brentuximab Vedotin in First Refractory/Relapsed Classical Hodgkin Lymphoma Patients Treated By Chemotherapy (ICE) before Autologous Transplantation. Final Analysis of Phase II Study. Blood 2019, 134, 132.
- Bartlett, N.; Niedzwiecki, D.; Johnson, J.; Friedberg, J.; Johnson, K.; van Besien, K.; Zelenetz, A.; Cheson, B.; Canellos, G. Gemcitabine, vinorelbine, and pegylated liposomal doxorubicin (GVD), a salvage regimen in relapsed Hodgkin’s lymphoma: CALGB. Ann. Oncol. 2007, 18, 1071–1079.
- LaCasce, A.S.; Bociek, R.G.; Sawas, A.; Caimi, P.; Agura, E.; Matous, J.; Ansell, S.M.; Crosswell, H.E.; Islas-Ohlmayer, M.; Behler, C.; et al. Brentuximab vedotin plus bendamustine: A highly active first salvage regimen for relapsed or refractory Hodgkin lymphoma. Blood 2018, 132, 40–48.
- Hagenbeek, A.; Zijlstra, J.M.; Plattel, W.J.; Morschhauser, F.; Lugtenburg, P.J.; Brice, P.; Hutchings, M.; Gastinne, T.; Liu, R.D.; Burggraaf, C.N.; et al. Combining Brentuximab Vedotin with DHAP as Salvage Treatment in Relapsed/Refractory Hodgkin Lymphoma: The Phase II HOVON/LLPC Transplant BRaVE study. Blood 2018, 132, 2923.
- Cassaday, R.D.; Fromm, J.; Cowan, A.J.; Libby, E.N.; Philip, M.; Behnia, S.; Nartea, M.; Press, O.; Gopal, A.K. Safety and Activity of Brentuximab Vedotin (BV) Plus Ifosfamide, Carboplatin, and Etoposide (ICE) for Relapsed/Refractory (Rel/Ref) Classical Hodgkin Lymphoma (cHL): Initial Results of a Phase I/II Trial. Blood 2016, 128, 1834.
- Herrera, A.; Palmer, J.; Martin, P.; Armenian, S.; Tsai, N.-C.; Kennedy, N.; Sahebi, F.; Cao, T.; Budde, L.; Mei, M.; et al. Autologous stem-cell transplantation after second-line brentuximab vedotin in relapsed or refractory Hodgkin lymphoma. Ann. Oncol. 2017, 29, 724–730.
- Moskowitz, A.J.; Schöder, H.; Yahalom, J.; McCall, S.J.; Fox, S.Y.; Gerecitano, J.; Grewal, R.; Hamlin, P.; Horwitz, S.; Kobos, R.; et al. PET-adapted sequential salvage therapy with brentuximab vedotin followed by augmented ifosamide, carboplatin, and etoposide for patients with relapsed and refractory Hodgkin’s lymphoma: A non-randomised, open-label, single-centre, phase 2 study. Lancet Oncol. 2015, 16, 284–292.
- Advani, R.H.; Moskowitz, A.J.; Bartlett, N.L.; Vose, J.M.; Ramchandren, R.; Feldman, T.A.; LaCasce, A.S.; Christian, B.A.; Ansell, S.M.; Moskowitz, C.H.; et al. Brentuximab vedotin in combination with nivolumab in relapsed or refractory Hodgkin lymphoma: 3-year study results. Blood 2021, 138, 427–438.
- Moskowitz, A.J.; Shah, G.; Schöder, H.; Ganesan, N.; Drill, E.; Hancock, H.; Davey, T.; Perez, L.; Ryu, S.; Sohail, S.; et al. Phase II Trial of Pembrolizumab Plus Gemcitabine, Vinorelbine, and Liposomal Doxorubicin as Second-Line Therapy for Relapsed or Refractory Classical Hodgkin Lymphoma. J. Clin. Oncol. 2021, 39, 3109–3117.
- Mei, M.G.; Lee, H.J.; Palmer, J.; Chen, R.W.; Tsai, N.-C.; Chen, L.; McBride, K.; Smith, D.L.; Melgar, I.; Song, J.Y.; et al. Response-adapted anti-PD1 based salvage therapy for Hodgkin lymphoma with nivolumab +/− ICE (NICE). Blood 2022.
- Lepik, K.V.; Mikhailova, N.B.; Kondakova, E.V.; Zalyalov, Y.R.; Fedorova, L.V.; Tsvetkova, L.A.; Kotselyabina, P.V.; Borzenkova, E.S.; Babenko, E.V.; Popova, M.O.; et al. A Study of Safety and Efficacy of Nivolumab and Bendamustine (NB) in Patients With Relapsed/Refractory Hodgkin Lymphoma After Nivolumab Monotherapy Failure. HemaSphere 2020, 4, e401.
- Herrera, A.F.; Moskowitz, A.J.; Bartlett, N.L.; Vose, J.M.; Ramchandren, R.; Feldman, T.A.; LaCasce, A.S.; Ansell, S.M.; Moskowitz, C.H.; Fenton, K.; et al. Interim results of brentuximab vedotin in combination with nivolumab in patients with relapsed or refractory Hodgkin lymphoma. Blood 2018, 131, 1183–1194.
- Cole, P.D.; Mauz-Körholz, C.; Mascarin, M.; Michel, G.; Cooper, S.; Beishuizen, A.; Leger, K.J.; Amoroso, L.; Buffardi, S.; Rigaud, C.; et al. Nivolumab and brentuximab vedotin (BV)-based, response-adapted treatment in children, adolescents, and young adults (CAYA) with standard-risk relapsed/refractory classical Hodgkin lymphoma (R/R cHL): Primary analysis. J. Clin. Oncol. 2020, 38, 8013.
- Bryan, L.J.; Casulo, C.; Allen, P.; Smith, S.E.; Savas, H.; Karmali, R.; Pro, B.; O’Shea, K.; Chmiel, J.; Palmer, B.A.; et al. Pembrolizumab (PEM) Added to ICE Chemotherapy Results in High Complete Metabolic Response Rates in Relapsed/Refractory Classic Hodgkin Lymphoma (cHL): A Multi-Institutional Phase II Trial. Blood 2021, 138, 229.
- Armand, P.; Chen, Y.-B.; Redd, R.A.; Joyce, R.M.; Bsat, J.; Jeter, E.; Merryman, R.; Coleman, K.C.; Dahi, P.B.; Nieto, Y.; et al. PD-1 blockade with pembrolizumab for classical Hodgkin lymphoma after autologous stem cell transplantation. Blood 2019, 134, 22–29.
More
Information
Subjects:
Hematology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
991
Revisions:
2 times
(View History)
Update Date:
11 Jul 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





