| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sandra van Wilpe | + 4040 word(s) | 4040 | 2020-10-01 12:31:37 | | | |
| 2 | Rita Xu | -2084 word(s) | 1956 | 2020-10-12 08:49:17 | | |
Video Upload Options
The prognosis and responsiveness to chemotherapy and checkpoint inhibitors differs substantially among patients with bladder cancer (BC). There is an unmet need for biomarkers that can accurately predict prognosis and treatment outcome. Here, we describe the available literature on the prognostic and predictive value of tumor-infiltrating immune cells in BC. Current evidence indicates that a high density of tumor-infiltrating CD8+ T cells is a favorable prognostic factor, whereas PD-L1 expression and tumor-associated macrophages are unfavorable prognostic features. While PD-L1 expression appears unsuccessful as biomarker for response to checkpoint inhibitors, there are some indications that high CD8+ T cell infiltration, low transforming growth factor-beta signaling and low densities of myeloid-derived suppressor cells are associated with response. Future studies should focus on combinations of biomarkers to accurately predict survival and response to treatment.
1. Introduction
The immune system plays a pivotal role in the development of cancer. During malignant transformation, cells acquire tumor-associated antigens (TAAs) due to mutations in protein-encoding genes or epigenetic alterations. T cells are able to recognize these TAAs and selectively eradicate cancer cells. During progression to clinically manifest cancer, tumor cells acquire mechanisms to escape immune surveillance. Tumors may lose antigenicity through the acquisition of defects in antigen processing and the presentation or loss of immunogenic TAAs. The upregulation of inhibitory checkpoint molecules, such as programmed cell death protein-1 (PD-1) or its ligand PD-L1, provides another way to avert an effective antitumor immune response. Additionally, tumors may escape immunity by attracting immune suppressive cells, such as regulatory T cells (Tregs), tumor-associated macrophages (TAMs) and myeloid-derived suppressor cells (MDSCs). Among various cancer types, high T cell infiltration has been associated with a favorable prognosis, whereas immune suppressive cells and inhibitory immune checkpoints have been associated with a poor clinical outcome [1].
Bladder cancer (BC) is considered an immunogenic tumor, due to its relatively high tumor mutational burden and its responsiveness to Bacillus Calmette–Guerin (BCG) bladder instillations and checkpoint inhibitors. BC is the tenth most common malignancy worldwide, with urothelial carcinoma (UC) being by far the most common histological type of BC (>90%) [2]. Approximately 75% of patients present with non-muscle invasive bladder cancer (NMIBC). NMIBC is treated primarily by the transurethral resection of the tumor, followed by chemotherapy or BCG bladder instillations, depending on additional risk factors. Unfortunately, many patients experience disease recurrence or progression to muscle-invasive disease (MIBC) despite bladder instillations [3]. For localized MIBC, the gold standard is neoadjuvant cisplatin-based chemotherapy followed by radical cystectomy. Neoadjuvant chemotherapy induces downstaging in 44.8% of patients and pathological complete responses in 25.7%. Nevertheless, the five-year overall survival (OS) benefit is only 8%, with five-year OS increasing from 45% to 53% [4]. Recent phase II trials evaluating the role of checkpoint inhibitors in MIBC have shown that pembrolizumab and atezolizumab induce remarkable pathological complete response rates in the neoadjuvant setting (42% and 29%, resp.), suggesting that this might be an alternative treatment option for MIBC [5][6]. Long-term follow-up data, however, are still awaited. For metastatic BC (mBC), cisplatin-based chemotherapy is currently the first line of treatment. Anti-PD-(L)1 is registered for the treatment of mBC patients who progressed on platinum-based chemotherapy and patients who are cisplatin-ineligible and have a high PD-L1 expression. Although anti-PD-(L)1 is able to induce durable responses in mBC patients, objective responses and disease control (objective response or stable disease) are achieved in only 21.1% and 38.5% of patients, respectively [7].
There is a need for biomarkers that predict the prognosis and responsiveness to the available treatments in BC. Strong prognostic biomarkers could be helpful for the selection of patients who could benefit from (neo)adjuvant treatments, such as bladder instillations and chemotherapy, and to determine the intensity of the follow-up. Predictive biomarkers, on the other hand, can be used to select the most effective drugs.
2. Prognostic Value of Tumor-Infiltrating Immune Cells
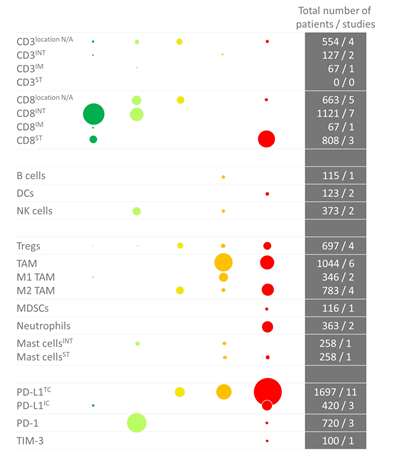
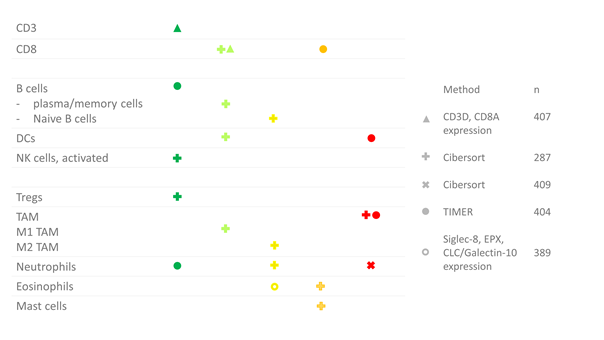
In our paper, we review the available evidence on the prognostic value of several immune types, i.e. CD3+ and CD8+ T cells, B cells, dendritic cells, natural killer cells, regulatory T cells, tumor-associated macrophages, myeloid-derived suppressor cells, neutrophils, eosinophils and mast cells. An overview of the available data is given in Figure 1 (IHC) and Figure 2 (RNA sequencing). In summary, there is plentiful evidence that high CD8+ T cell density in the tumor or invasive margin [8][9][10][11][12][13][14][15][16][17][18][19][20][21][22][23][24][25][26][27][28][29], low TAM infiltration [8][10][12][24][30][31][32][33][34][35][36][37][38][39] and low PD-L1 expression on tumor cells [14][40][41][42][43][44][45] are favorable prognostic factors in localized BC. Nevertheless, these markers have not yet been implemented in the clinic, likely due to the limited prognostic value of the individual markers. A few small studies have investigated the prognostic value of combinations of markers, i.e. FoxP3/CD3 ratio [15], CD68/CD3 ratio [8] and NLR [11], and found that the combination had stronger prognostic value compared to either marker alone.
Figure 1. Overview of immunohistochemistry studies in bladder cancer per immune cell type. The color (and position) indicate whether there was a positive or negative correlation. Dark green: significant positive correlation; light green: trend towards a positive correlation; yellow: no trend; orange: trend towards a negative correlation; red: negative correlation. The size of the dots indicates the number of studied patients. As some studies performed an analysis in more than one cohort, the number of dots is sometimes higher than the total number of studies (i.e., for the Tregs). Abbreviations: DCs: dendritic cells; IC: immune cell; INT: intraepithelial; IM: invasive margin; MDSCs: myeloid-derived mononuclear cells; N/A: not applicable; NK cells: natural killer cells; PD-(L)1: programmed death (ligand)-1; ST: stroma; TAM: tumor-associated macrophage; TC: tumor cell; TIM-3: T-cell immunoglobulin mucin-3; Tregs: regulatory T cells.
Figure 2. Overview of RNA sequencing studies in bladder cancer per immune cell type. The color (and position) indicate whether there was a positive or negative correlation. Dark green: significant positive correlation; light green: trend towards a positive correlation; yellow: no trend; orange: trend towards a negative correlation; red: negative correlation. The shape indicates the study where the data was derived from: ∆ [13], + [24], x [57], ● [12], o [58].
3. The predictive value of tumor-infiltrating immune cells for response to checkpoint inhibitors
Since 2017, immune checkpoint inhibitors are used for the treatment of mBC. The use of checkpoint inhibitors in BC is expanding. In January 2020, pembrolizumab was approved by the FDA for treatment of BCG-unresponsive, high-risk NMIBC, based on the results of the KEYNOTE-057 [46]. Additionally, phase II clinical trials evaluating the efficacy of neoadjuvant checkpoint inhibitor therapy in localized MIBC have shown promising results [5][6]. Unfortunately, not all patients benefit from checkpoint inhibitors. In mBC, objective responses and disease control are seen in only 21.1% and 38.5% of patients, respectively [7]. There is a need for predictive biomarkers that can be used to identify these patients.
Most evidence on biomarkers for checkpoint inhibitor response prediction in BC comes from clinical trials that included patients with UC of the bladder, urethra, ureter and renal pelvis. The biomarker that has been most extensively studied for its predictive value is PD-L1. Several tests have been developed to assess PD-L1 expression. Each therapeutic antibody has its own companion diagnostic with corresponding cut-off value and some variation can be observed between assays [47]. A recent meta-analysis, including 8 studies totaling 1436 patients, evaluated the use of PD-L1 as biomarker for response to PD-(L)1 inhibitors in advanced UC. Patients had received pembrolizumab, nivolumab, atezolizumab, durvalumab or avelumab. Patients with high PD-L1 expression had significantly higher objective response rates compared to patients with low PD-L1 expression (RR 0.53, p < 1x106). However, it did not accurately predict 1-year OS (RR 0.53, p = 0.304) [48]. To determine whether a biomarker is truly predictive for response rather than prognostic, a control arm is needed, consisting of patients that did not receive the treatment of interest. There are 2 studies available in UC that assessed PD-L1 expression and included a control arm. The IMvigor211 [49] is a randomized controlled, phase III trial comparing PD-L1 inhibitor atezolizumab with second-line chemotherapy in metastatic UC (mUC). Based on the hypothesis that efficacy of atezolizumab would be associated with PD-L1 expression, the investigators used a hierarchical study design where OS was first evaluated in the group of patients with PD-L1 expression on at least 5% of tumor-infiltrating immune cells (IC2/3). A significant difference would have allowed for further analysis in the intention-to-treat population, also including patients with lower PD-L1 expression. Atezolizumab, however, did not prolong OS in the IC2/3 population. High PD-L1 expression was associated with higher response rates in both the atezolizumab and chemotherapy arms. The other study including a control arm is the Keynote-045 [50], a randomized controlled, phase III trial comparing pembrolizumab with second-line chemotherapy. In the Keynote-045, the benefit of pembrolizumab appeared to be independent of PD-L1 expression on tumor and immune cells. These studies indicate that PD-L1 expression is not useful as predictive biomarker in mUC. However, in 2018, the use of pembrolizumab and atezolizumab as first-line treatment in cisplatin-ineligible mUC patients was restricted to patients with high PD-L1 expression, based on unpublished, interim analyses of the Keynote-361 (NCT02853305) and IMvigor130 (NCT02807636) performed by the data safety monitoring committees. Both trials compare the combination of checkpoint inhibitors and chemotherapy with checkpoint inhibitors or chemotherapy alone in treatment-naïve mUC patients and observed poor survival in patients that received first-line single-agent checkpoint inhibitors. With both studies still ongoing, it is uncertain whether PD-L1 expression is truly a predictive biomarker in this setting. It is also possible that anti-PD-(L)1 is inferior to chemotherapy in mUC regardless of PD-L1 expression in the first-line setting.
In addition to PD-L1 expression, several studies have evaluated the predictive value of CD8+ T cells for response to anti-PD-(L)1. In 212 mUC patients receiving nivolumab as second-line treatment, high CD8+ T cell infiltration was associated with favorable clinical outcome. Objective responses were observed in 25.5% and 11.3% of patients with high and low CD8+ T cell infiltration, respectively. Median OS was 11.3 and 5.72 months [51]. Similarly, in the IMvigor 210, a phase II trial evaluating the efficacy of atezolizumab in mUC, CD8+ T cell infiltration was higher in patients who responded to atezolizumab (p = 0.0265) [52]. Moreover, phase I studies evaluating the efficacy of atezolizumab have also described a positive association between effector T cell signatures and clinical outcome [53][54]. In line with the findings in mUC, results from the ABACUS trial, a phase II trial investigating the efficacy of neoadjuvant atezolizumab in UC of the bladder, indicate that CD8+ T cell infiltration is also associated with higher pathologic complete response rates to neoadjuvant atezolizumab (40% vs 20% (high vs low), p < 0.05). Moreover, a predefined eight-gene cytotoxic T cell signature was significantly increased in responders compared to patients with stable disease (p < 0.01) and patients who relapsed (p < 0.01) [55].
Lack of response to atezolizumab in mUC has been associated with a signature of transforming growth factor-β (TGF-β), which was found to be particularly high in patients with immune-excluded tumors. Interestingly, in preclinical models the combination of anti-TGF-β and anti-PD-L1 was shown to reduce TGF-β signaling, increase intra-epithelial T cell penetration and significantly reduce tumor size in mice with high TGF-β signaling and immune-excluded tumors [29].
Finally, a phase I trial evaluating the efficacy of atezolizumab in 68 UC patients, found that patients with high baseline signatures of genes associated with myeloid cells (IL-1B, IL-8) had lower response rates to atezolizumab (p < 0.01) [54]. In line with these results, exploratory biomarker analysis in the Checkmate 275 trial, a single-arm phase II trial evaluating the efficacy of nivolumab after first-line chemotherapy in 220 mUC patients, suggests that low baseline circulating MDSC levels are indicative of a longer OS after nivolumab treatment(p < 0.05) [56].
To summarize, although PD-L1 expression has been associated with higher response rates to checkpoint inhibitors, it does not appear useful as a predictive biomarker in mUC. Studies have suggested that CD8+ T cell infiltration, TGF-β signaling and MDSCs are associated with response to checkpoint inhibitors. However, data are limited and due to the lack of a control arm, it is not possible to determine whether these biomarkers are truly predictive, or merely prognostic.
References
- Fridman, W.H.; Zitvogel, L.; Sautès–Fridman, C.; Kroemer, G. The immune contexture in cancer prognosis and treatment. Nat. Rev. Clin. Oncol. 2017, 14, 717–734.
- Chalasani, V.; Chin, J.L.; Izawa, J.I. Histologic variants of urothelial bladder cancer and nonurothelial histology in bladder cancer. J. Can. Urol. Assoc. 2009, 3, S193.
- Becker, R.E.N.; Kates, M.R.; Bivalacqua, T.J. Identification of Candidates for Salvage Therapy: The Past, Present, and Future of Defining Bacillus Calmette-Guérin Failure. Urol. Clin. 2020, 47, 15–21.
- Yin, M.; Joshi, M.; Meijer, R.P.; Glantz, M.; Holder, S.; Harvey, H.A.; Kaag, M.; Fransen van de Putte, E.E.; Horenblas, S.; Drabick, J.J. Neoadjuvant Chemotherapy for Muscle-Invasive Bladder Cancer: A Systematic Review and Two-Step Meta-Analysis. Oncologist 2016, 21, 708–715.
- Necchi, A.; Anichini, A.; Raggi, D.; Briganti, A.; Massa, S.; Lucianò, R.; Colecchia, M.; Giannatempo, P.; Mortarini, R.; Bianchi, M.; et al. Pembrolizumab as Neoadjuvant Therapy Before Radical Cystectomy in Patients With Muscle-Invasive Urothelial Bladder Carcinoma (PURE-01): An Open-Label, Single-Arm, Phase II Study. J. Clin. Oncol. 2018, 36, JCO1801148.
- Powles, T.; Rodriguez-Vida, A.; Duran, I.; Crabb, S.J.; Van der Heijden, M.S.; Font Pous, A.; Maillet, D. A phase II study investigating the safety and efficacy of neoadjuvant atezolizumab in muscle invasive bladder cancer (ABACUS). Ann. Oncol. 2018, 29, doi:10.1093/annonc/mdy283.108.
- Fradet, Y.; Bellmunt, J.; Vaughn, D.J.; Lee, J.L.; Fong, L.; Vogelzang, N.J.; Climent, M.A.; Petrylak, D.P.; Choueiri, T.K.; Necchi, A.; et al. Randomized phase III KEYNOTE-045 trial of pembrolizumab versus paclitaxel, docetaxel, or vinflunine in recurrent advanced urothelial cancer: Results of >2 years of follow-up. Ann. Oncol. 2019, 30, 970–976.
- Sjödahl, G.; Lövgren, K.; Lauss, M.; Chebil, G.; Patschan, O.; Gudjonsson, S.; Månsson, W.; Fernö, M.; Leandersson, K.; Lindgren, D.; et al. Infiltration of CD3+ and CD68+ cells in bladder cancer is subtype specific and affects the outcome of patients with muscle-invasive tumors. Urol. Oncol. Semin. Orig. Investig. 2014, 32, 791–797.
- Li, X.-D.; Huang, C.-W.; Liu, Z.-F.; Jiang, L.-J.; Chen, J.-W.; Xie, D.; Zhou, F.-J.; Lu, H.-M.; Liu, Z.-W. Prognostic Role of the Immunoscore for Patients with Urothelial Carcinoma of the Bladder Who Underwent Radical Cystectomy. Ann. Surg. Oncol. 2019, doi:10.1245/s10434-019-07529-y.
- Krpina, K.; Babarović, E.; Jonjić, N. Correlation of tumor-infiltrating lymphocytes with bladder cancer recurrence in patients with solitary low-grade urothelial carcinoma. Virchows Arch. 2015, 467, 443–448.
- Liu, K.; Zhao, K.; Wang, L.; Sun, E. The prognostic values of tumor-infiltrating neutrophils, lymphocytes and neutrophil/lymphocyte rates in bladder urothelial cancer. Pathol. Res. Pract. 2018, 214, 1074–1080.
- Li, B.; Severson, E.; Pignon, J.-C.; Zhao, H.; Li, T.; Novak, J.; Jiang, P.; Shen, H.; Aster, J.C.; Rodig, S.; et al. Comprehensive analyses of tumor immunity: Implications for cancer immunotherapy. Genome Biol. 2016, 17, 174.
- Shi, M.J.; Meng, X.Y.; Wu, Q.J.; Zhou, X.H. High CD3D/CD4 ratio predicts better survival in muscle-invasive bladder cancer. Cancer Manag. Res. 2019, 11, 2987–2995.
- Wang, B.; Pan, W.; Yang, M.; Yang, W.; He, W.; Chen, X.; Bi, J.; Jiang, N.; Huang, J.; Lin, T. Programmed death ligand-1 is associated with tumor infiltrating lymphocytes and poorer survival in urothelial cell carcinoma of the bladder. Cancer Sci. 2019, 110, 489–498.
- Horn, T.; Laus, J.; Seitz, A.K.; Maurer, T.; Schmid, S.C.; Wolf, P.; Haller, B.; Winkler, M.; Retz, M.; Nawroth, R.; et al. The prognostic effect of tumour-infiltrating lymphocytic subpopulations in bladder cancer. World J. Urol. 2016, 34, 181–187.
- Wang, B.; Lin, J.; Yu, H.; Zeng, H.; Lin, T. Distribution and prognostic significance of CD8+ T cells in urothelial cell carcinoma of the bladder. Chin. J. Urol. 2015, doi:10.3760/CMA.J.ISSN.1000-6702.2015.07.007.
- Asano, T.; Ohnishi, K.; Shiota, T.; Motoshima, T.; Sugiyama, Y.; Yatsuda, J.; Kamba, T.; Ishizaka, K.; Komohara, Y. CD169-positive sinus macrophages in the lymph nodes determine bladder cancer prognosis. Cancer Sci. 2018, 109, 1723–1730.
- Yu, A.; Mansure, J.J.; Solanki, S.; Siemens, D.R.; Koti, M.; Dias, A.B.T.; Burnier, M.M.; Brimo, F.; Kassouf, W. Presence of lymphocytic infiltrate cytotoxic T lymphocyte CD3+, CD8+, and immunoscore as prognostic marker in patients after radical cystectomy. PLoS ONE 2018, 13, e0205746.
- Otto, W.; Denzinger, S.; Wieland, W.F.; Hartmann, A. First analysis of immune cell infiltration in stage pT1 urothelial bladder carcinoma: CD3 positivity as a prognostic marker for cancer-specific survival. World J. Urol. 2012, 30, 875–877.
- Winerdal, M.E.; Marits, P.; Winerdal, M.; Hasan, M.; Rosenblatt, R.; Tolf, A.; Selling, K.; Sherif, A.; Winqvist, O. FOXP3 and survival in urinary bladder cancer. BJU Int. 2011, 108, 1672–1678.
- Sharma, P.; Shen, Y.; Wen, S.; Yamada, S.; Jungbluth, A.A.; Gnjatic, S.; Bajorin, D.F.; Reuter, V.E.; Herr, H.; Old, L.J.; et al. CD8 tumor-infiltrating lymphocytes are predictive of survival in muscle-invasive urothelial carcinoma. Proc. Natl. Acad. Sci. USA 2007, 104, 3967–3972.
- Faraj, S.F.; Munari, E.; Guner, G.; Taube, J.; Anders, R.; Hicks, J.; Meeker, A.; Schoenberg, M.; Bivalacqua, T.; Drake, C.; et al. Assessment of tumoral PD-L1 expression and intratumoral CD8+ T cells in urothelial carcinoma. Urology 2015, 85, 703.
- Zhu, Y.; Fu, H.; Liu, Z.; Zhang, J.; Ye, D. Immune-desert, immune-excluded and inflamed phenotypes predict survival and adjuvant chemotherapy response in patients with MIBC. Eur. Urol. Suppl. 2018, 17, 128–130.
- Fu, H.; Zhu, Y.; Wang, Y.; Liu, Z.; Zhang, J.; Xie, H.; Fu, Q.; Dai, B.; Ye, D.; Xu, J. Identification and Validation of Stromal Immunotype Predict Survival and Benefit from Adjuvant Chemotherapy in Patients with Muscle-Invasive Bladder Cancer. Clin. Cancer Res. 2018, 24, 3069–3078.
- Wang, B.; Wu, S.; Zeng, H.; Liu, Z.; Dong, W.; He, W.; Chen, X.; Dong, X.; Zheng, L.; Lin, T.; et al. CD103 + Tumor Infiltrating Lymphocytes Predict a Favorable Prognosis in Urothelial Cell Carcinoma of the Bladder. J. Urol. 2015, 194, 556–562.
- Masson-Lecomte, A.; Maillé, P.; Pineda, S.; Soyeux, P.; Sagrera, A.; Rava, M.; Lopez De Maturana, E.; Márquez, M.; Tardón, A.; Carrato, A.; et al. CD8+ Cytotoxic Immune Infiltrate in Non-Muscle Invasive Bladder Cancer: A Standardized Methodology to Study Association with Clinico-Pathological Features and Prognosis. Bladder Cancer 2019, 5, 159–169.
- Zhang, Q.; Hao, C.; Cheng, G.; Wang, L.; Wang, X.; Li, C.; Qiu, J.; Ding, K. High CD4+ T cell density is associated with poor prognosis in patients with non-muscle-invasive bladder cancer. Int. J. Clin. Exp. Pathol. 2015, 8, 11510–11516.
- Pagès, F.; Mlecnik, B.; Marliot, F.; Bindea, G.; Ou, F.S.; Bifulco, C.; Lugli, A.; Zlobec, I.; Rau, T.T.; Berger, M.D.; et al. International validation of the consensus Immunoscore for the classification of colon cancer: A prognostic and accuracy study. Lancet 2018, 391, 2128–2139.
- Mariathasan, S.; Turley, S.J.; Nickles, D.; Castiglioni, A.; Yuen, K.; Wang, Y.; Kadel, E.E.; Koeppen, H.; Astarita, J.L.; Cubas, R.; et al. TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 2018, 554, 544–548.
- Ayari, C.; Larue, H.; Hovington, H.; Caron, A.; Bergeron, A.; Têtu, B.; Fradet, V.; Fradet, Y. High level of mature tumor-infiltrating dendritic cells predicts progression to muscle invasion in bladder cancer. Hum. Pathol. 2013, 44, 1630–1637.
- Ayari, C.; LaRue, H.; Hovington, H.; Decobert, M.; Harel, F.; Bergeron, A.; Têtu, B.; Lacombe, L.; Fradet, Y. Bladder tumor infiltrating mature dendritic cells and macrophages as predictors of response to bacillus Calmette-Guérin immunotherapy. Eur. Urol. 2009, 55, 1386–1395.
- Li, Z.; Ju, X.; Silveira, P.A.; Abadir, E.; Hsu, W.H.; Hart, D.N.J.; Clark, G.J. CD83: Activation marker for antigen presenting cells and its therapeutic potential. Front. Immunol. 2019, 10, 1312.
- Sturm, G.; Finotello, F.; Petitprez, F.; Zhang, J.D.; Baumbach, J.; Fridman, W.H.; List, M.; Aneichyk, T. Comprehensive evaluation of transcriptome-based cell-type quantification methods for immuno-oncology. Bioinformatics 2019, 35, 436–445.
- Mukherjee, N.; Ji, N.; Hurez, V.; Curiel, T.J.; Montgomery, M.O.; Braun, A.J.; Nicolas, M.; Aguilera, M.; Kaushik, D.; Liu, Q.; et al. Intratumoral CD56bright natural killer cells are associated with improved survival in bladder cancer. Oncotarget 2018, 9, 36492–36502.
- Michel, T.; Poli, A.; Cuapio, A.; Briquemont, B.; Iserentant, G.; Ollert, M.; Zimmer, J. Human CD56 bright NK Cells: An Update. J. Immunol. 2016, 196, 2923–2931.
- Shang, B.; Liu, Y.; Jiang, S.J.; Liu, Y. Prognostic value of tumor-infiltrating FoxP3+ regulatory T cells in cancers: A systematic review and meta-analysis. Sci Rep. 2015, 5, 15179.
- Pollard, J.W.Tumour-educated macrophages promote tumour progression and metastasis. Nat. Rev. Cancer 2004, 4, 71–78.
- Komohara, Y.; Jinushi, M.; Takeya, M. Clinical significance of macrophage heterogeneity in human malignant tumors. Cancer Sci. 2014, 105, 1–8.
- Asano, K.; Nabeyama, A.; Miyake, Y.; Qiu, C.H.; Kurita, A.; Tomura, M.; Kanagawa, O.; Shin-ichiro, F.; Tanaka, M. CD169-Positive Macrophages Dominate Antitumor Immunity by Crosspresenting Dead Cell-Associated Antigens. Immunity 2011, 34, 85–95.
- Wang, B.; Liu, H.; Dong, X.; Wu, S.; Zeng, H.; Liu, Z.; Wan, D.; Dong, W.; He, W.; Chen, X.; et al. High CD204+ tumor-infiltrating macrophage density predicts a poor prognosis in patients with urothelial cell carcinoma of the bladder. Oncotarget 2015, 6, 20204–202014.
- Takeuchi, H.; Tanaka, M.; Tanaka, A.; Tsunemi, A.; Yamamoto, H. Predominance of M2-polarized macrophages in bladder cancer affects angiogenesis, tumor grade and invasiveness. Oncol. Lett. 2016, 11, 3403–3408.
- Yang, G.; Zhang, L.; Liu, M.; Liu, Q.; Duan, X.; Bo, J. CD163+ macrophages predict a poor prognosis in patients with primary T1 high-grade urothelial carcinoma of the bladder. World J. Urol. 2019, doi:10.1007/s00345-018-02618-1.
- Qi, Y.; Chang, Y.; Wang, Z.; Chen, L.; Kong, Y.; Zhang, P.; Liu, Z.; Zhou, Q.; Chen, Y.; Wang, J.; et al. Tumor-associated macrophages expressing galectin-9 identify immunoevasive subtype muscle-invasive bladder cancer with poor prognosis but favorable adjuvant chemotherapeutic response. Cancer Immunol. Immunother. 2019, 68, 2067–2080.
- Xu, Z.; Wang, L.; Tian, J.; Man, H.; Li, P.; Shan, B. High expression of B7-H3 and CD163 in cancer tissues indicates malignant clinicopathological status and poor prognosis of patients with urothelial cell carcinoma of the bladder. Oncol. Lett. 2018, 15, 6519–6526.
- Boström, M.M.; Irjala, H.; Mirtti, T.; Taimen, P.; Kauko, T.; Ålgars, A.; Jalkanen, S.; Boström, P.J. Tumor-Associated Macrophages Provide Significant Prognostic Information in Urothelial Bladder Cancer. PLoS ONE 2015, 10, e0133552.
- Zhang, H.; Ye, Y.-L.; Li, M.-X.; Ye, S.-B.; Huang, W.-R.; Cai, T.-T.; He, J.; Peng, J.-Y.; Duan, T.-H.; Cui, J.; et al. CXCL2/MIF-CXCR2 signaling promotes the recruitment of myeloid-derived suppressor cells and is correlated with prognosis in bladder cancer. Oncogene 2017, 36, 2095–2104.
- Ornstein, M.C.; Diaz-Montero, C.M.; Rayman, P.; Elson, P.; Haywood, S.; Finke, J.H.; Kim, J.S.; Pavicic, P.G.; Lamenza, M.; Devonshire, S.; et al. Myeloid-derived suppressors cells (MDSC) correlate with clinicopathologic factors and pathologic complete response (pCR) in patients with urothelial carcinoma (UC) undergoing cystectomy. Urol. Oncol. 2018, 36, 405–412.
- Yang, G.; Shen, W.; Zhang, Y.; Liu, M.; Zhang, L.; Liu, Q.; Lu, H.H.; Bo, J. Accumulation of myeloid-derived suppressor cells (MDSCs) induced by low levels of IL-6 correlates with poor prognosis in bladder cancer. Oncotarget 2017, 8, 38378–38388.
- Chevalier, M.F.; Trabanelli, S.; Racle, J.; Salomé, B.; Cesson, V.; Gharbi, D.; Bohner, P.; Domingos-Pereira, S.; Dartiguenave, F.; Fritschi, A.-S.; et al. ILC2-modulated T cell-to-MDSC balance is associated with bladder cancer recurrence. J. Clin. Investig. 2017, 127, 2916–2929.
- Shen, M.; Hu, P.; Donskov, F.; Wang, G.; Liu, Q.; Du, J. Tumor-associated neutrophils as a new prognostic factor in cancer: A systematic review and meta-analysis. PLoS ONE 2014, 9, e98259.
- Zhou, L.; Xu, L.; Chen, L.; Fu, Q.; Liu, Z.; Chang, Y.; Lin, Z.; Xu, J. Tumor-infiltrating neutrophils predict benefit from adjuvant chemotherapy in patients with muscle invasive bladder cancer. Oncoimmunology 2017, 6, 1293211.
- Varricchi, G.; Galdiero, M.R.; Loffredo, S.; Lucarini, V.; Marone, G.; Mattei, F.; Marone, G.; Schiavonid, G. Eosinophils: The unsung heroes in cancer? Oncoimmunology 2018, 7, 1393134.
- Sari, A.; Calli, A.; Cakalagaoglu, F.; Altınboga, A.A.; Bal, K. Association of mast cells with microvessel density in urothelial carcinomas of the urinary bladder. Ann. Diagn. Pathol. 2012, 16, 1–6.
- Rao, Q.; Chen, Y.; Yeh, C.-R.; Ding, J.; Li, L.; Chang, C.; Yeh, S. Recruited mast cells in the tumor microenvironment enhance bladder cancer metastasis via modulation of ERβ/CCL2/CCR2 EMT/MMP9 signals. Oncotarget 2016, 7, 7842–7855.
- Zhu, L.; Sun, J.; Wang, L.; Li, Z.; Wang, L.; Li, Z. Prognostic and Clinicopathological Significance of PD-L1 in Patients With Bladder Cancer: A Meta-Analysis. Front. Pharmacol. 2019, 10, 962.
- Xylinas, E.; Robinson, B.D.; Kluth, L.A.; Volkmer, B.G.; Hautmann, R.; Küfer, R.; Zerbib, M.; Kwon, E.; Thompson, R.H.; Boorjian, S.A.; et al. Association of T-cell co-regulatory protein expression with clinical outcomes following radical cystectomy for urothelial carcinoma of the bladder. Eur. J. Surg. Oncol. 2014, 40, 121–127.
- Boorjian, S.A.; Sheinin, Y.; Crispen, P.L.; Farmer, S.A.; Lohse, C.M.; Kuntz, S.M.; Leibovich, B.C.; Kwon, E.D.; Frank, I. T-Cell coregulatory molecule expression in urothelial cell carcinoma: Clinicopathologic correlations and association with survival. Clin. Cancer Res. 2008, 14, 4800–4808.
- Pichler, R.; Fritz, J.; Lackner, F.; Sprung, S.; Brunner, A.; Horninger, W.; Loidl, W.; Pircher, A.; Heidegger, I. Prognostic Value of Testing PD-L1 Expression After Radical Cystectomy in High-risk Patients. Clin. Genitourin. Cancer 2018, 16, 1015–1024.