
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Terisha Ghazi | + 1938 word(s) | 1938 | 2020-09-08 08:36:19 | | | |
| 2 | Dean Liu | -710 word(s) | 1228 | 2020-10-09 09:21:37 | | |
Video Upload Options
Cancer initiation and progression is an accumulation of genetic and epigenetic modifications. DNA methylation is a common epigenetic modification that regulates gene expression, and aberrant DNA methylation patterns are considered a hallmark of cancer. The human diet is a source of micronutrients, bioactive molecules, and mycotoxins that have the ability to alter DNA methylation patterns and are thus a contributing factor for both the prevention and onset of cancer. In this review, we summarize the literature on dietary micronutrients, bioactive compounds, and food-borne mycotoxins that affect DNA methylation patterns and identify their potential in the onset and treatment of cancer.
1.Introduction
Cancer is a public health concern and a major cause of mortality worldwide. According to the World Health Organization (WHO), the annual global cancer statistics indicated that an estimated 18.1 million new cases and 9.6 million deaths have occurred in the year 2018[1]. These statistics have increased dramatically over the past few years and are expected to double by the year 2040[1]. The increasing cancer burden is due to several factors including population growth and aging as well as changes in the prevalence and distribution of cancer risk factors, many of which are associated with social and economic development[1][2]. To date, significant advances have been made in the prevention and treatment of cancer; however, early detection, side effects, drug resistance, treatment costs, and lack of access to health care facilities and palliative care remain major challenges[1].
It is estimated that nearly 50% of all cancers can be avoided by dietary modification[1]. The Western diet coupled with physical inactivity is governed by convenience and comprises of an excessive intake of refined grains, processed meats, sugary desserts, fried foods, and high-fat dairy products that promote an oxidative and inflammatory environment ideal for cancer development[3][4][5][6]. In addition, the consumption of agricultural foods that are contaminated with mycotoxin-producing fungi can alter cellular functions, leading to genetic mutations and the onset of cancer[7][8][9][10][11].
Natural dietary micronutrients and bioactive compounds from fruits, vegetables, and spices have long been investigated, due to their wide availability and fewer side effects, for their potential to prevent and destroy cancer cells[12]. Previously, it was reported that 25–80% of cancer patients used dietary compounds and/or micronutrient supplementation as a therapeutic agent to replenish the body’s nutritional needs after surgery and/or chemotherapy, inhibit tumor growth, and prevent tumor recurrence. Despite this, the exact mechanism by which these compounds prevent and inhibit carcinogenesis remains unclear. Emerging evidence suggests that dietary micronutrients and bioactive molecules exhibit anti-cancer properties by reversing abnormal gene activation and inhibition through epigenetic modifications such as DNA methylation[13][14][15][16][17][18], and these changes in DNA methylation may provide insight into future therapeutic interventions. In this review, we summarize existing literature on natural dietary micronutrients, bioactive compounds, and food-borne mycotoxins that affect DNA methylation patterns and identify its potential in the onset and treatment of cancer.
2. Combinational Effects of Dietary Compounds on DNA Methylation in Cancer
Although the use of a single bioactive dietary compound has shown promise against the growth and risk of cancer as discussed in this review, evidence suggests that the combination of two or more bioactive dietary compounds can target multiple pathways to induce a more potent effect on DNA methylation and cancer. In one study, the treatment of prostate cancer (PC3 and DU145) cell lines with curcumin (5.0 µM) and quercetin (5.0 µM) for 48 h enhanced the decrease in cell proliferation and apoptosis by inhibiting DNMT activity and inducing global DNA hypomethylation as well as androgen receptor (AR) promoter hypomethylation and reactivation[19]. Exposure of the breast cancer (MDA-MB-231) cell line to a mixture of EGCG (20.0 µg/mL) and SFN (5.0 µM) for 72 h also enhanced cell growth inhibition and apoptosis by inducing global DNA hypomethylation as well as ERα promoter hypomethylation and expression[20]. Similarly, a 48 h treatment of the breast cancer (MCF7) cell line with a mixture of genistein (1 µM) and SFN (10 µM) synergistically increased CpG-site specific DNA methylation at the MB-COMT distal promoter and expression, leading to a decrease in cell proliferation, as measured by reduced BrdU (bromodeoxyuridine) incorporation[21].
3. Clinical Trials with Bioactive Dietary Compounds and DNA Methylation in Cancer
Due to its promising role as both chemopreventative and chemotherapeutic agents, several bioactive dietary compounds have entered clinical trials. In a randomized clinical trial on 10 male Norwegian patients aged between 55 and 69 years who received either 30 mg genistein or placebo capsules daily for 3–6 weeks before prostatectomy, whole-genome methylation and expression profiling identified several differentially methylated sites and expressed genes between placebo and genistein groups. The majority of these genes were found to be involved in cell proliferation, thus highlighting the effects of genistein on global changes in gene expression in prostate cancer[22]. In another randomized clinical trial, 34 healthy premenopausal women (aged between 19 and 54 years) fed isoflavones including genistein at 40 or 140 mg daily throughout one menstrual cycle showed hypermethylation of 5 key cancer-related genes (p16INK4a, RASSF1A, RARβ2, ER, and CCND2)[23]. In contrast, resveratrol (5 or 50 mg) administered twice daily for 12 weeks to 39 women (median age of 59.5 years in the 5 mg group versus a median age of 54 years in the 50 mg group) with increased breast cancer risk displayed chemopreventative properties by inducing RASSF-1α demethylation and re-expression[24].
4. Conclusion
Cancer development is a continuous, progressive event, involving the activation of oncogenes and the inhibition of tumor suppressor genes by alterations of the epigenome. Lifestyle modification is a major step in preventing the disease and improving patient survival. The human diet has been shown to contain micronutrients, bioactive compounds, and mycotoxins that have the potential to alter the epigenome; this can either lead to cancer prevention or development. As discussed in this review, DNA methylation is a common epigenetic mechanism by which micronutrients, bioactive dietary compounds, and mycotoxins affect cancer cell growth, invasive capacities, and metastasis. Although micronutrients and bioactive dietary compounds have shown the potential to inhibit and prevent cancer, the main disadvantage is that the majority of these studies are conducted in vitro. In vitro models lack the complex biological functions and muticellularity observed in vivo and hence, the effects of these compounds in vivo may be different from those seen in vitro. This is further exaggerated by several studies in which changes in DNA methylation were both site- and cell-specific. Another disadvantage arises from the observation that the cancer-benefiting properties of these compounds occurred at varying concentrations, some of which may prove to be harmful in the long-term. Other challenges include the bioavailability of the exact concentration required to reverse the epigenetic modification and the effect of these micronutrients and bioactive compounds in combination with existing anti-cancer drugs. Likewise, some mycotoxins, which are common in agricultural foods (the same agricultural foods are a source of bioactive compounds), are known carcinogens. Figure 1 depicts an overview of the proposed mechanism for how food-borne mycotoxins affect the DNA methylation profile of the cell, thus leading to the development and progression of cancer.
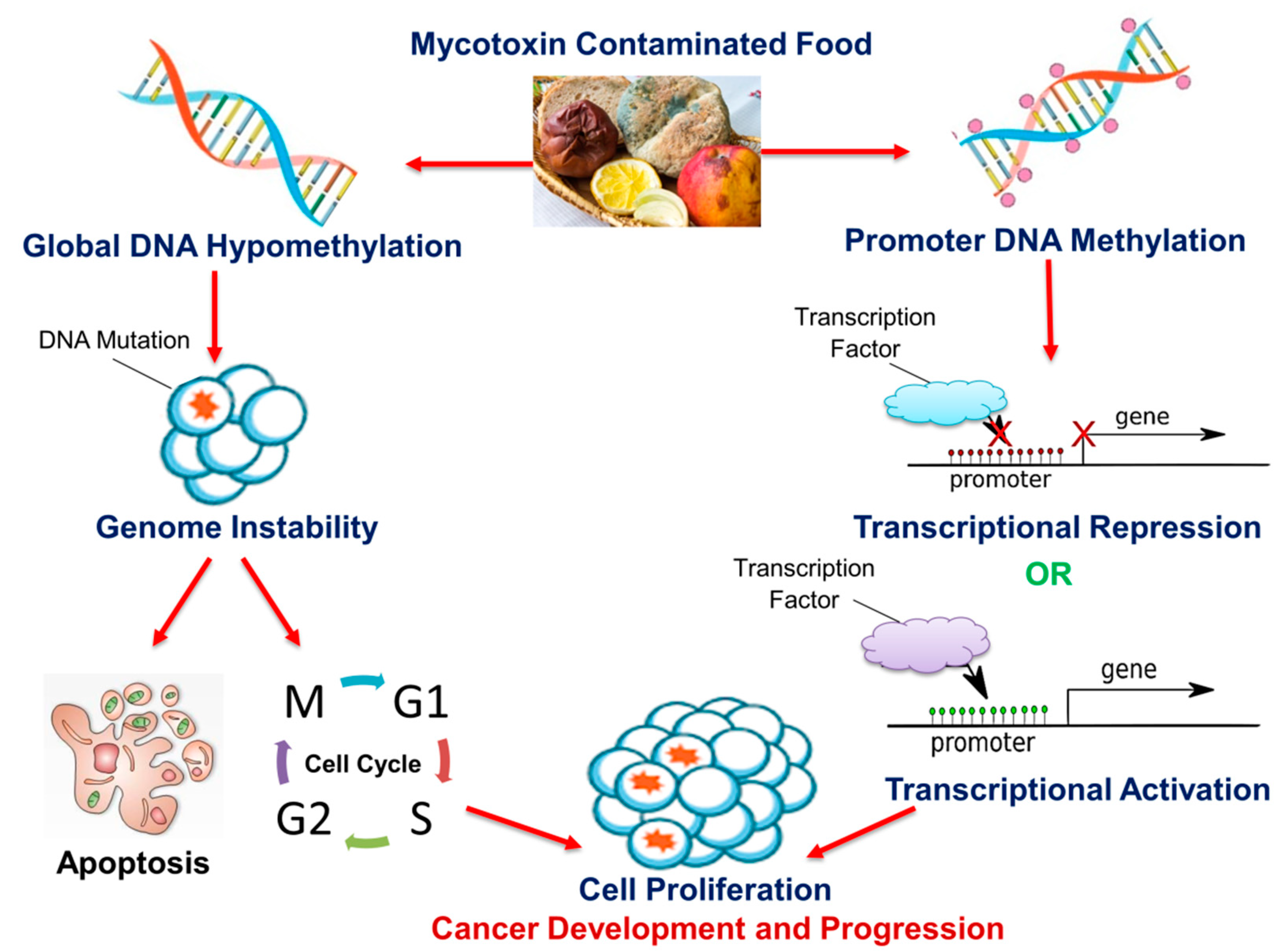 Figure 1. The effect of mycotoxins on DNA methylation and cancer. Mycotoxins can alter global DNA methylation and/or promoter DNA methylation in various cells both in vitro and in vivo. Global DNA hypomethylation leads to genome instability and increases the frequency of DNA mutations. Mutated cells are destroyed via apoptosis or evade cell cycle regulatory checkpoints and proliferate, leading to cancer development and progression. Promoter DNA methylation contributes to carcinogenesis via the transcriptional repression of tumor suppressor genes and/or transcriptional activation of oncogenes.
Figure 1. The effect of mycotoxins on DNA methylation and cancer. Mycotoxins can alter global DNA methylation and/or promoter DNA methylation in various cells both in vitro and in vivo. Global DNA hypomethylation leads to genome instability and increases the frequency of DNA mutations. Mutated cells are destroyed via apoptosis or evade cell cycle regulatory checkpoints and proliferate, leading to cancer development and progression. Promoter DNA methylation contributes to carcinogenesis via the transcriptional repression of tumor suppressor genes and/or transcriptional activation of oncogenes.
References
- World Health Organization. WHO Report on Cancer: Setting Priorities, Investing Wisely and Providing Care for all. Available online: https://apps.who.int/iris/handle/10665/330745 (accessed on 27 August 2020).
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2018, 68, 394–424.
- Adlercreutz, H.; Mousavi, Y.; Höckerstedt, K. Diet and breast cancer. Acta Oncol. 1992, 31, 175–181.
- Slattery, M.; Potter, J.; Ma, K.-N.; Caan, B.; Leppert, M.; Samowitz, W. Western diet, family history of colorectal cancer, NAT2, GSTM-1 and risk of colon cancer. Cancer Causes Control 2000, 11, 1–8.
- Stoll, B. Breast cancer and the western diet: Role of fatty acids and antioxidant vitamins. Eur. J. Cancer 1998, 34, 1852–1856.
- Stoll, B.A. Western diet, early puberty, and breast cancer risk. Breast Cancer Res. Treat. 1998, 49, 187–193.
- Peers, F.; Linsell, C. Dietary aflatoxins and liver cancer—A population based study in Kenya. Br. J. Cancer 1973, 27, 473.
- Yoshizawa, T.; Yamashita, A.; Luo, Y. Fumonisin occurrence in corn from high-and low-risk areas for human esophageal cancer in China. Appl. Environ. Microbiol. 1994, 60, 1626–1629.
- Gelderblom, W.; Abel, S.; Smuts, C.M.; Marnewick, J.; Marasas, W.; Lemmer, E.R.; Ramljak, D. Fumonisin-induced hepatocarcinogenesis: Mechanisms related to cancer initiation and promotion. Environ. Health Perspect. 2001, 109, 291–300.
- Castegnaro, M.; Mohr, U.; Pfohl-Leszkowicz, A.; Estève, J.; Steinmann, J.; Tillmann, T.; Michelon, J.; Bartsch, H. Sex-and strain-specific induction of renal tumors by ochratoxin A in rats correlates with DNA adduction. Int. J. Cancer 1998, 77, 70–75.
- Belhassen, H.; Jiménez-Díaz, I.; Arrebola, J.; Ghali, R.; Ghorbel, H.; Olea, N.; Hedili, A. Zearalenone and its metabolites in urine and breast cancer risk: A case-control study in Tunisia. Chemosphere 2015, 128, 1–6.
- Khan, N.; Afaq, F.; Mukhtar, H. Cancer chemoprevention through dietary antioxidants: Progress and promise. Antioxid. Redox Signal. 2008, 10, 475–510.
- Ali Khan, M.; Kedhari Sundaram, M.; Hamza, A.; Quraishi, U.; Gunasekera, D.; Ramesh, L.; Goala, P.; Al Alami, U.; Ansari, M.Z.; Rizvi, T.A. Sulforaphane reverses the expression of various tumor suppressor genes by targeting DNMT3B and HDAC1 in human cervical cancer cells. Evid.-Based Complement. Altern. Med. 2015, 2015, 412149.
- Al-Yousef, N.; Shinwari, Z.; Al-Shahrani, B.; Al-Showimi, M.; Al-Moghrabi, N. Curcumin induces re-expression of BRCA1 and suppression of γ synuclein by modulating DNA promoter methylation in breast cancer cell lines. Oncol. Rep. 2020, 43, 827–838.
- Fang, M.Z.; Chen, D.; Sun, Y.; Jin, Z.; Christman, J.K.; Yang, C.S. Reversal of hypermethylation and reactivation of p16INK4a, RARβ, and MGMT genes by genistein and other isoflavones from soy. Clin. Cancer Res. 2005, 11, 7033–7041.
- Fudhaili, A.; Yoon, N.; Kang, S.; Ryu, J.; Jeong, J.Y.; Lee, D.H.; Kang, S.S. Resveratrol epigenetically regulates the expression of zinc finger protein 36 in non-small cell lung cancer cell lines. Oncol. Rep. 2019, 41, 1377–1386.
- Gamet-Payrastre, L.; Li, P.; Lumeau, S.; Cassar, G.; Dupont, M.-A.; Chevolleau, S.; Gasc, N.; Tulliez, J.; Tercé, F. Sulforaphane, a naturally occurring isothiocyanate, induces cell cycle arrest and apoptosis in HT29 human colon cancer cells. Cancer Res. 2000, 60, 1426–1433.
- Ji, Z.; Huo, C.; Yang, P. Genistein inhibited the proliferation of kidney cancer cells via CDKN2a hypomethylation: Role of abnormal apoptosis. Int. Urol. Nephrol. 2020, 1–7.
- Sharma, V.; Kumar, L.; Mohanty, S.K.; Maikhuri, J.P.; Rajender, S.; Gupta, G. Sensitization of androgen refractory prostate cancer cells to anti-androgens through re-expression of epigenetically repressed androgen receptor–synergistic action of quercetin and curcumin. Cell. Endocrinol. 2016, 431, 12–23.
- Meeran, S.M.; Patel, S.N.; Li, Y.; Shukla, S.; Tollefsbol, T.O. Bioactive dietary supplements reactivate ER expression in ER-negative breast cancer cells by active chromatin modifications. PLoS ONE 2012, 7, e37748.
- Wu, Q.; Odwin-Dacosta, S.; Cao, S.; Yager, J.D.; Tang, W.-y. Estrogen down regulates COMT transcription via promoter DNA methylation in human breast cancer cells. Appl. Pharmacol. 2019, 367, 12–22.
- Bilir, B.; Sharma, N.V.; Lee, J.; Hammarstrom, B.; Svindland, A.; Kucuk, O.; Moreno, C.S. Effects of genistein supplementation on genome-wide DNA methylation and gene expression in patients with localized prostate cancer. J. Oncol. 2017, 51, 223–234.
- Qin, W.; Zhu, W.; Shi, H.; Hewett, J.E.; Ruhlen, R.L.; MacDonald, R.S.; Rottinghaus, G.E.; Chen, Y.-C.; Sauter, E.R. Soy isoflavones have an antiestrogenic effect and alter mammary promoter hypermethylation in healthy premenopausal women. Cancer 2009, 61, 238–244.
- Zhu, W.; Qin, W.; Zhang, K.; Rottinghaus, G.E.; Chen, Y.-C.; Kliethermes, B.; Sauter, E.R. Trans-resveratrol alters mammary promoter hypermethylation in women at increased risk for breast cancer. Cancer 2012, 64, 393–400.





