| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Giorgio Ricci | + 1102 word(s) | 1102 | 2020-09-27 06:03:26 |
Video Upload Options
Protein cysteines are involved in many critical structural and functional roles that can be performed thanks to the peculiar properties of their sulfhydryl group, which, in its deprotonated form, becomes an active nucleophile. Due to the physiological importance of this residue, it represents an attractive and emerging target for the development and design of covalent ligands, which are able to modulate the function of specific proteins and enzymes.
1. Abstract
Protein cysteines often play crucial functional and structural roles, so they are emerging targets to design covalent thiol ligands that are able to modulate enzyme or protein functions. The data from the molten globule-like structures of ribonuclease, lysozyme, bovine serum albumin and chymotrypsinogen identified new speeding agents, i.e., hydrophobic/electrostatic interactions and productive complex formations involving the protein and thiol reagent, which were able to confer exceptional reactivity to structural cysteines that form disulfide bridges. Interestingly, the pKa perturbations can only produce two-four times increased reactivity at physiological pH values. These properties may shed light on the mechanisms involved in the disulfide formation during the oxidative folding of many proteins.
2. Introduction
Protein cysteines are involved in many critical structural and functional roles that can be performed thanks to the peculiar properties of their sulfhydryl group, which, in its deprotonated form, becomes an active nucleophile. Due to the physiological importance of this residue, it represents an attractive and emerging target for the development and design of covalent ligands, which are able to modulate the function of specific proteins and enzymes. Therefore, it is of paramount importance that a precise knowledge of the factors that influence the reactivity of these residues is obtained.[1][2] It is a common opinion (universally accepted) that this is mainly controlled by their pKa and by the accessibility of a given reagent.[1][3][4][5][6][7] In fact, the sulfhydryl group of cysteines is almost inert in its protonated form (except in free-radical reactions), while the thiolate form is the true reactive form. A relevant number of cysteines with functional roles in catalysis have been found to react a hundred and even thousand times faster than a free cysteine, but it is only rarely that a quantitative and reasoned analysis of the contribution of a low pKa to these unusual reactivities has been made. Novel findings show that pKa is not the main determinant, given that, at physiological pH values, the highest increment of the reactivity due to pKa variations cannot exceed two-four times. Thus, other important factors will be considered in order to discover that, in a few proteins, they assume an almost exclusive prevalence in modulating the reactivity of these residues. Moreover, the hyper-reactivity is not an exclusive feature of functional cysteines and even structural cysteines may have extraordinary reactivity toward many thiol reagents or natural disulfides, which are possibly finalized to a correct and rapid formation of native disulfide bridges during the nascent phase.
3. Discussion
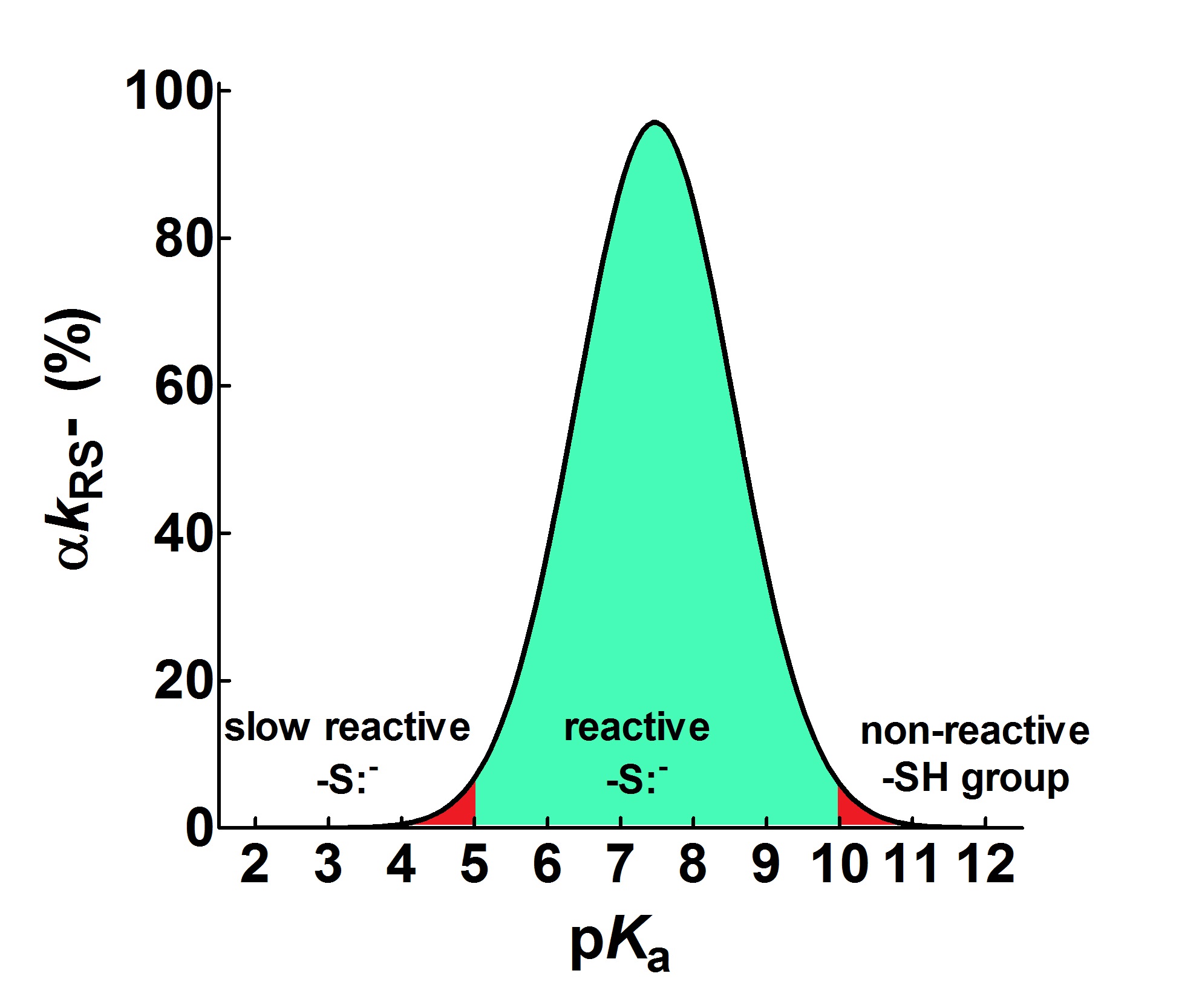
The pKa is not the main determinant in the enhancement of the reactivity of protein cysteines toward various reagents. Conversely, a very low pKa, as well as a very high pKa, may render unreactive these residues (see Figure). What is the utility of some functional cysteine showing very low pKa, such as selected residues in DsbA (pKa = 3.5), DsbC (pKa = 4.1) and Grx1 (pKa = 3.5)?[8] One reasonable explanation is that this property accelerates the reaction of the oxidized form of these enzymes with the thiol substrates stabilizing the products.
Another possibility is that a very low pKa that makes the thiolate less reactive and may preserve it against some un-proper modifications. This may be the case for GSTP1-1, where the thiolate of Cys47 (pKa = 3.5) is bound to Lys54 in an ion-pair, which is important for the enzyme mechanism and a correct binding of the substrate.[9]
Cysteine hyper-reactivity is not an exclusive property of functional cysteines involved in catalysis and even structural cysteines devoted to the formation of disulfides may display hundred or thousand times increased reactivity toward oxidized glutathione (GSSG) and various thiol reagents.
Hydrophobic interactions are the main determinant factors triggering hundred times increased reactivity toward alkylating reagents (CDNB and NBD-Cl) for some reduced proteins, like bovine serum albumin, chymotrypsinogen and lysozyme, while electrostatic interactions are the prominent factors for the reactivity of Ellman’s reagent (DTNB) toward reduced ribonuclease, lysozyme and chymotrypsinogen.[10][11][12][13]
A specific binding site for GSSG is also surprisingly present in the reduced molten globule-like conformations of albumin, lysozyme, ribonuclease and chymotrypsinogen.[10][11][12][13] It is the main determinant for the observed hundred and even thousand times increased reactivity of one specific cysteine i.e. Cys75, Cys94, Cys95 and Cys1 respectively. This phenomenon raises the question of whether a rapid glutathionylation may be the early step of their oxidative pathway.
Methods for the proteomic identification of cysteines, like the isoTOP-ABPP procedure,[1][14] should be used with caution, because they only identify hyper-reactive cysteines toward a specific reagent (i.e., a modified iodacetamide) and this property cannot be referred to an ‘intrinsic reactivity’ because it may be not present in reactions with different thiol reagents. Conversely, some protein cysteines, which are normo-reactive toward the modified iodoacetamide probe, can be hyper-reactive toward some natural intracellular compounds.
Data from literature likely indicate that one cysteine may have extraordinary hyper-reactivity toward a specific disulfide (GSSG) and normo-reactivity toward other small disulfides, like cystine and cystamine.[10][11][12][13] Conversely, many cysteines present in reduced bovine serum albumin and lysozyme are hyper-reactive toward hydrophobic reagents like CDNB and NBD-Cl, but (except for one residue) are normo-reactive toward GSSG and other small disulfides. In other words, the “intrinsic” reactivity for a protein cysteine is only determined by its pKa and by the nucleophilicity of its deprotonated form, but it cannot be increased more than three-four times. An evident hyper-reactivity can only be generated by “extrinsic” factors like the protein environment surrounding the cysteine, which may productively and often selectively bind a specific reagent through hydrophobic or electrostatic interactions.
In conclusion, this selective hyper-reactivity should be of particular interest in the elucidation of the early step of the oxidative folding of these proteins. The hyper-reactivity of protein cysteines appears to be an open puzzle whose pieces, even now, have not been completely identified.
Figure. Theoretical curve defining the range of useful pKa (αkRS− ≥ 5%) for a reacting thiolate group. The figure shows the pKa range that allows a sufficient rate of reaction of a protein cysteine with different reagents (green area) at pH = 7.4. Values with αkRS− < 5% are reported in red. Y-axis reports the second order kinetic constant of the thiolate (kRS−) multiplied by the active fraction of the thiolate (α).
References
- Aaron J Maurais; Eranthie Weerapana; Reactive-cysteine profiling for drug discovery. Current Opinion in Chemical Biology 2019, 50, 29-36, 10.1016/j.cbpa.2019.02.010.
- Darren A. E. Cross; Susan E. Ashton; Serban Ghiorghiu; Cath Eberlein; Caroline A. Nebhan; Paula J. Spitzler; Jonathon P. Orme; M. Raymond V. Finlay; Richard A. Ward; Martine J. Mellor; et al.Gareth HughesAmar RahiVivien N. JacobsMonica Red BrewerEiki IchiharaJing SunHailing JinPeter BallardKatherine Al-KadhimiRachel RowlinsonTeresa KlinowskaGraham H. P. RichmondMireille CantariniDong-Wan KimMalcolm R. RansonWilliam Pao AZD9291, an Irreversible EGFR TKI, Overcomes T790M-Mediated Resistance to EGFR Inhibitors in Lung Cancer. Cancer Discovery 2014, 4, 1046-1061, 10.1158/2159-8290.cd-14-0337.
- Ranieri Rossi; Donatella Barra; Andrea Bellelli; Giovanna Boumis; Silvia Canofeni; Paolo Di Simplicio; Lorenzo Lusini; Stefano Pascarella; Gino Amiconi; Fast-reacting thiols in rat hemoglobins can intercept damaging species in erythrocytes more efficiently than glutathione.. Journal of Biological Chemistry 1998, 273, 19198-19206, 10.1074/jbc.273.30.19198.
- Stefano M. Marino; Vadim N. Gladyshev; Analysis and Functional Prediction of Reactive Cysteine Residues. Journal of Biological Chemistry 2011, 287, 4419-4425, 10.1074/jbc.r111.275578.
- P. J. Britto; Leslie Knipling; J. Wolff; The Local Electrostatic Environment Determines Cysteine Reactivity of Tubulin. Journal of Biological Chemistry 2002, 277, 29018-29027, 10.1074/jbc.m204263200.
- F. Peter Guengerich; Qingming Fang; Liping Liu; David L. Hachey; Anthony E. Pegg; O6-Alkylguanine-DNA Alkyltransferase: Low pKaand High Reactivity of Cysteine 145†. Biochemistry 2003, 42, 10965-10970, 10.1021/bi034937z.
- Nicholas J. Fowler; Christopher F. Blanford; Sam P. De Visser; Jim Warwicker; Features of reactive cysteines discovered through computation: from kinase inhibition to enrichment around protein degrons. Scientific Reports 2017, 7, 16338, 10.1038/s41598-017-15997-z.
- Péter Nagy; Kinetics and Mechanisms of Thiol–Disulfide Exchange Covering Direct Substitution and Thiol Oxidation-Mediated Pathways. Antioxidants & Redox Signaling 2013, 18, 1623-1641, 10.1089/ars.2012.4973.
- M Lo Bello; M W Parker; A Desideri; F Polticelli; M Falconi; G Del Boccio; A Pennelli; G Federici; G Ricci; Peculiar spectroscopic and kinetic properties of Cys-47 in human placental glutathione transferase. Evidence for an atypical thiolate ion pair near the active site.. Journal of Biological Chemistry 1993, 268, 19033-8.
- Alessio Bocedi; Raffaele Fabrini; Jens Z. Pedersen; Giorgio Federici; Federica Iavarone; Claudia Martelli; Massimo Castagnola; Giorgio Ricci; The extreme hyper-reactivity of selected cysteines drives hierarchical disulfide bond formation in serum albumin. The FEBS Journal 2016, 283, 4113-4127, 10.1111/febs.13909.
- Alessio Bocedi; Giada Cattani; Claudia Martelli; Flora Cozzolino; Massimo Castagnola; Pietro Pucci; Giorgio Ricci; The extreme hyper-reactivity of Cys94 in lysozyme avoids its amorphous aggregation. Scientific Reports 2018, 8, 16050, 10.1038/s41598-018-34439-y.
- Alessio Bocedi; Giada Cattani; Giorgia Gambardella; Silvia Ticconi; Flora Cozzolino; Ornella Di Fusco; Pietro Pucci; Giorgio Ricci; Di Fusco; Ultra-Rapid Glutathionylation of Ribonuclease: Is this the Real Incipit of its Oxidative Folding?. International Journal of Molecular Sciences 2019, 20, 5440, 10.3390/ijms20215440.
- Alessio Bocedi; Giorgia Gambardella; Giada Cattani; Simonetta Bartolucci; Danila Limauro; Emilia Pedone; Federica Iavarone; Massimo Castagnola; Giorgio Ricci; Ultra-rapid glutathionylation of chymotrypsinogen in its molten globule-like conformation: A comparison to archaeal proteins. Scientific Reports 2020, 10, 8943, 10.1038/s41598-020-65696-5.
- Eranthie Weerapana; Chu Wang; Gabriel M. Simon; Florian Richter; Sagar Khare; Myles B. D. Dillon; Daniel A. Bachovchin; Kerri Mowen; David Baker; Benjamin F. Cravatt; et al. Quantitative reactivity profiling predicts functional cysteines in proteomes. Nature Cell Biology 2010, 468, 790-795, 10.1038/nature09472.