| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jae-Sung Kwon | + 568 word(s) | 568 | 2020-09-27 03:43:44 | | | |
| 2 | Peter Tang | Meta information modification | 568 | 2020-09-29 04:57:19 | | | | |
| 3 | Peter Tang | -1 word(s) | 567 | 2020-10-27 09:30:45 | | | | |
| 4 | Peter Tang | Meta information modification | 567 | 2020-11-06 14:54:30 | | |
Video Upload Options
Osteogenic differentiations of mesenchymal stem cells are known to be influenced by both intracellular and extracellular factors. In terms of the extracellular factors, extracellular environment which would include biomaterials that are used for tissue regenerations play important role. Surface modifications of biomaterials in both phycial and chemical ways have been investigated to induce osteogenic differentiations. In terms of chemical modifications, induction of chemcial functionalities such as amine, would provide favorable environment for osteogenic differentiation of mesenchymal stem cells. Use of devices such as non-thermal atmospheric pressure plasma (also known as bio-plasma) have been investigated in relation to the osteogenic differentiations.
Stem cells are widely investigated in the field of tissue engineering with potential for tissue regenerations. Mesenchymal stem cells are obtained from bone marow, and have potential to differentiate into hard tissues. Hence, studies have been focused to induce osteogenesis of such mesenchymal stem cells.
Osteogenic differentiations of mesenchymal stem cells have known to be caused by both intra- and extra-cellular factors. Extra-cellular factors would include modifications of extracellular matrix with chemicals such as ascorbic acid or dexamethasone that have shown to influence osteogenic differentiations. Additionally, biomaterials that are present in extracellular environment would play imporant role on osteongenic differentiations of mesenchymal stem cells. Topographical features and elasticity of biomaterials have shown to influence osteongenic differentiations of mesenchymal stem cells. Recently focuse was with chemistry at functionality level in relation to osteongenic differentiations of mesenchymal stem cells.
The surface molecular chemistry, such as amine functionality, of biomaterials plays a crucial role in the osteogenic activity of relevant cells and tissues during hard tissue regeneration. Here, we examined the possibilities of creating amine functionalities on the surface of titanium by using the nonthermal atmospheric pressure plasma jet (NTAPPJ) method with humidified ammonia, and the effects on human mesenchymal stem cell (hMSC) were investigated. Titanium samples were subjected to NTAPPJ treatments using nitrogen (N-P), air (A-P), or humidified ammonia (NA-P) as the plasma gas, while control (C-P) samples were not subjected to plasma treatment. After plasma exposure, all treatment groups showed increased hydrophilicity and had more attached cells than the C-P. Among the plasma-treated samples, the A-P and NA-P showed surface oxygen functionalities and exhibited greater cell proliferation than the C-P and N-P. The NA-P additionally showed surface amine-related functionalities and exhibited a higher level of alkaline phosphatase activity and osteocalcin expression than the other samples. The results can be explained by increases in fibronectin absorption and focal adhesion kinase gene expression on the NA-P samples. These findings suggest that NTAPPJ technology with humidified ammonia as a gas source has clinical potential for hard tissue generation.
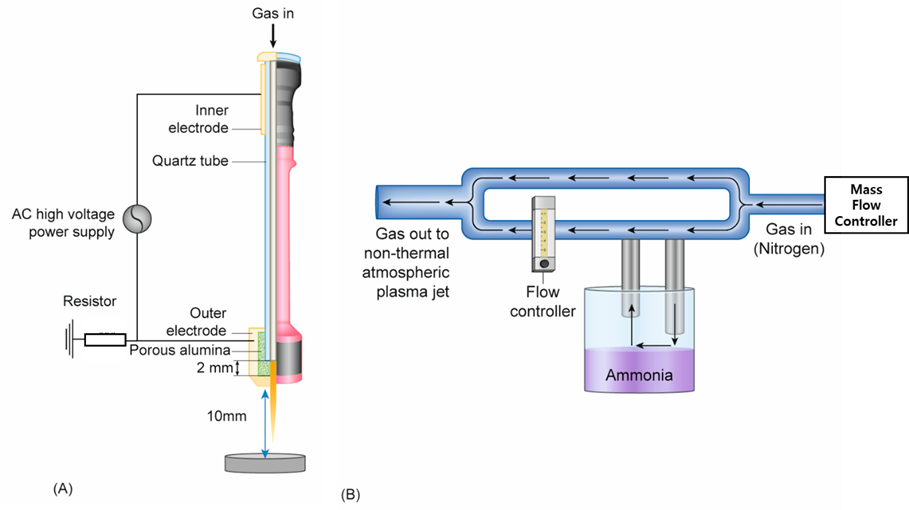
Figure 1. (A) Schematic diagram of the nonthermal atmospheric pressure plasma jet and (B). the design for a customized device that would supply nitrogen mixed with humidified ammonia.
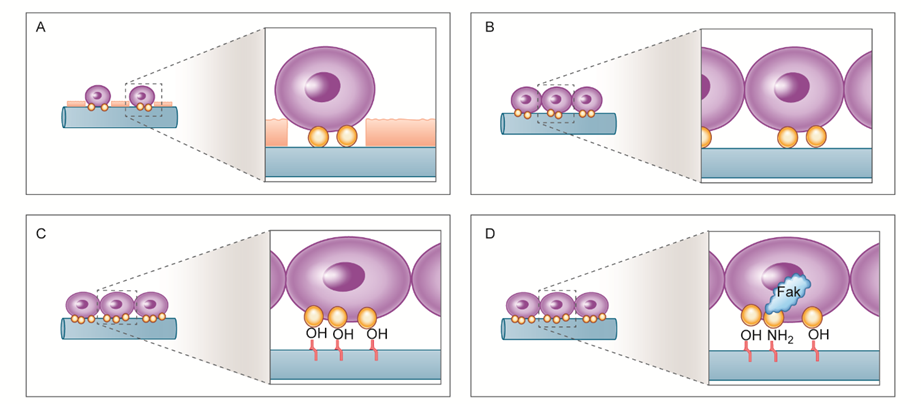
Figure 2. Schematic drawing of the putative mechanism underlying interactions between hMSCs and the (A) C-P, (B) N-P, (C) A-P, and (D) NA-P groups: For the N-P, A-P, and NA-P groups, more cell attachment was observed due to removal of hydrocarbon molecules from C-P. Higher levels of fibronectin absorption were observed on the A-P and NA-P groups, likely due to the presence of oxygen-related functional groups, which enhance osteogenic differentiation. The presence of nitrogen-related functional groups is considered to increase focal adhesion kinase (FAK) gene expression, further enhancing osteogenic differentiation on the NA-P samples.