Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ankit Srivastava | -- | 1884 | 2022-04-21 19:26:03 | | | |
| 2 | Jason Zhu | -24 word(s) | 1860 | 2022-04-22 03:31:16 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Srivastava, A.; Alam, P.; Caughey, B. Seed Amplification Assays for α-Synuclein. Encyclopedia. Available online: https://encyclopedia.pub/entry/22138 (accessed on 21 May 2026).
Srivastava A, Alam P, Caughey B. Seed Amplification Assays for α-Synuclein. Encyclopedia. Available at: https://encyclopedia.pub/entry/22138. Accessed May 21, 2026.
Srivastava, Ankit, Parvez Alam, Byron Caughey. "Seed Amplification Assays for α-Synuclein" Encyclopedia, https://encyclopedia.pub/entry/22138 (accessed May 21, 2026).
Srivastava, A., Alam, P., & Caughey, B. (2022, April 21). Seed Amplification Assays for α-Synuclein. In Encyclopedia. https://encyclopedia.pub/entry/22138
Srivastava, Ankit, et al. "Seed Amplification Assays for α-Synuclein." Encyclopedia. Web. 21 April, 2022.
Copy Citation
Various disease-associated forms or strains of α-synuclein (αSynD) can spread and accumulate in a prion-like fashion during synucleinopathies such as Parkinson’s disease (PD), Lewy body dementia (DLB), and multiple system atrophy (MSA). This capacity for self-propagation has enabled the development of seed amplification assays (SAAs) that can detect αSynD in clinical samples. Notably, α-synuclein real-time quaking-induced conversion (RT-QuIC) and protein misfolding cyclic amplification (PMCA) assays have evolved as ultrasensitive, specific, and relatively practical methods for detecting αSynD in a variety of biospecimens including brain tissue, CSF, skin, and olfactory mucosa from synucleinopathy patients.
α-synuclein
prion
seed amplification assays
Parkinson’s disease
synucleinopathies
1. Introduction
Multiple neurodegenerative diseases (NDDs) are associated with accumulation of pathological aggregates of the protein α synuclein (αSyn). In Parkinson’s disease (PD) and dementia with Lewy bodies (DLB), disease-associated forms of αSyn (αSynD) are major components of neuronal Lewy bodies (LB) and Lewy neurites, and in multiple system atrophy (MSA), αSynD accumulates in oligodendrocytes as glial cytoplasmic inclusions (GCIs) [1].
αSyn is normally a presynaptic neuronal protein that exists primarily as an intrinsically disordered monomer within the cytoplasm. However, in synucleinopathies, αSyn can be converted to β-sheet-rich, protease-resistant αSynD aggregates that grow by refolding and incorporating additional monomers [2][3][4]. Multiple studies have shown that αSynD can replicate and spread in a prion-like fashion within and between cells [5][6] and is considered the major culprit in the molecular pathology of synucleinopathies. The ‘dual-hit’ hypothesis postulates that an unknown trigger (e.g., an exogenous pathogen) is responsible for initiating the misfolding of native αSyn to yield assemblies that may then propagate via cellular communication mechanisms including passive diffusion, endocytosis, or exosomes [7]. Braak and colleagues described a pathological staging system in PD based on αSynD immunopositivity in the brain and other anatomical regions [8]. They proposed two plausible routes, i.e., nasal and gastric, for the spreading of αSynD and progression of PD.
This scenario may be analogous to the propagation mechanism(s) in prion diseases in which PrPSc (scrapie isoform of the prion protein) spreads between the tissues of infected hosts. Early evidence for prion-like spreading of αSyn aggregates was observed in the grafted dopaminergic neurons of patients who had undergone neuronal replacement therapy [9]. The presence of LB pathology within newly grafted neurons upon postmortem analysis supported the concept that αSynD can propagate between cells in the human brain. Also, cell-to-cell transfer of αSynD was observed in grafted neurons in a rat model [10].
The initiation and spread of LB pathology have also been observed using synthetic preformed αSyn fibrils (PFFs) in both cell and animal models [11][12]. Interestingly, PFFs convert into a GCI-like strain inside oligodendrocytes that maintains high seeding activity, even when propagated in neurons [13]. Furthermore, PFFs and other recombinant α-synuclein forms injected into the gastrointestinal tract of mice and rats induce LB-like aggregates in the brainstem via the vagus nerve [14][15][16]. Recently, Challis et al. observed that inoculating the duodenal wall of mice with PFFs led to changes in the enteric nervous system and gastrointestinal deficits [17]. Ultimately, inoculation of αSyn PFFs resulted in progression of αSyn histopathology to the midbrain in aged mice. Related, but not identical, pathological spreading has been seen in MSA, with αSynD having properties of infectious prions in cell cultures and animal models [18][19][20][21].
αSyn fibrils obtained from brain tissue or formed in vitro from recombinant protein [22][23][24] have been found to be structurally heterogenous [25]. Such αSyn fibril conformational polymorphs appear to be analogous to different prion strains and may be determinants of phenotypic diversity in synucleinopathies. Together, these observations support the one disease, one strain hypothesis for phenotypically distinct synucleinopathies [23][26]. To aid in synucleinopathy research and diagnostics, ultrasensitive seed amplification assays (SAAs) [27], including αSyn RT-QuIC [28][29] and the similar αSyn PMCA [30], have been developed to detect αSynD seeds in brain tissue, CSF, and other biospecimens.
2. Diagnostic Potential of αSynD
The clinical diagnosis of synucleinopathies and other NDDs has long been complicated by variable and overlapping symptoms, especially early in the pathologic process. Assessments of parkinsonism in patients can be helpful, but not definitive, with phenotypic manifestations including bradykinesia, resting tremor, rigidity, and postural instability. Parkinsonian traits are largely associated with PD and DLB and are less apparent in MSA [31]. Interestingly, DLB patients with parkinsonian traits are reportedly less responsive to L-DOPA (Levodopa) or similar treatments as compared with PD patients [32]. Another major difference is that DLB pathology involves a cognitive decline that is not found in PD patients, at least in the early and middle phases of disease. Thus, clinical diagnosis often involves a ‘one-year rule’, i.e., if cognitive alterations appear concurrent with, or before, movement symptoms, then the diagnosis is DLB and not late-phase PD dementia (PDD) [33].
It is also difficult to clinically differentiate synucleinopathies from other NDDs such as AD, progressive supranuclear palsy (PSP), corticobasal degeneration (CBD), and Creutzfeldt-Jakob disease (CJD) [34][35][36][37][38]. Even combined analyses of imaging (MRI, PET, and EMG) and fluid biomarkers (CSF Aβ42, NfL, p-tau, t-tau, αSyn, and HVA) do not routinely improve diagnostic accuracy for parkinsonism [39]. Thus, given the inconsistency and variability associated with current imaging and fluid biomarkers, the identification of more definitive biomarkers may help navigate these diagnostic ambiguities. Increasing evidence indicates that the detection of αSynD using SAAs can improve the clinical diagnosis of synucleinopathies [28][29][40][41][42][43][44], even in prodromal phases [27][40][45].
3. SAAs for Detecting Pathological Prion Aggregates
SAA platforms such as PMCA and RT-QuIC were initially developed to amplify, detect, and quantify pathological TSE (transmissible spongiform encephalopathy) prion aggregates in a variety of biospecimens. These assays were built on earlier observations that infectious prion protein aggregates, e.g., PrPSc, induce the conversion of a normal cellular prion protein (PrPC) into an abnormal protease-resistant form in cell-free reactions with striking species and strain specificities [46][47][48][49][50][51]. PMCA reactions showed that such seeded conformational conversion occurs continuously under suitable conditions and is accompanied by increases in prion infectivity [27][52].
The original PMCA reactions involved the cyclic sonication and incubation of infected samples with vast excesses of normal brain homogenates containing PrPC as the substrate for amplification [53]. PMCA products were then subjected to protease digestion and Western blotting to detect amplified conversion products. This protocol allowed the detection of prion aggregates with extraordinary sensitivity and selectivity [53][54]. However, limitations of this assay as a routine clinical test include the weeks-long reaction time for optimal sensitivity, the need for Western blotting, and the biohazardous infectivity of the amplified products.
The development of the amyloid seeding assay [55] and prion RT-QuIC [56][57] improved the practicality of SAAs by providing formats based on multiwell plates; shaking instead of sonication; recombinant, rather than brain-derived, PrPC substrate; fluorescence (Thioflavin T (ThT)) instead of Western blot readout; much shorter assay times (e.g., 1–2 days); and noninfectious amplification products [58] (Figure 1A,B). Further development of prion RT-QuIC assays has improved their sensitivity and specificity and their applicability to most prion diseases (e.g., [59]); many specimen types including brain tissue [57][60][61], CSF [56][62][63][64][65], skin [66][67][68], eye [69], and olfactory mucosa [70][71][72]; and the cervid pregnancy microenvironment [73][74][75]. In some sample matrices, analytical sensitivities in the atto- or femtogram ranges have been documented [54][56][57][60][61][76][77][78][79].
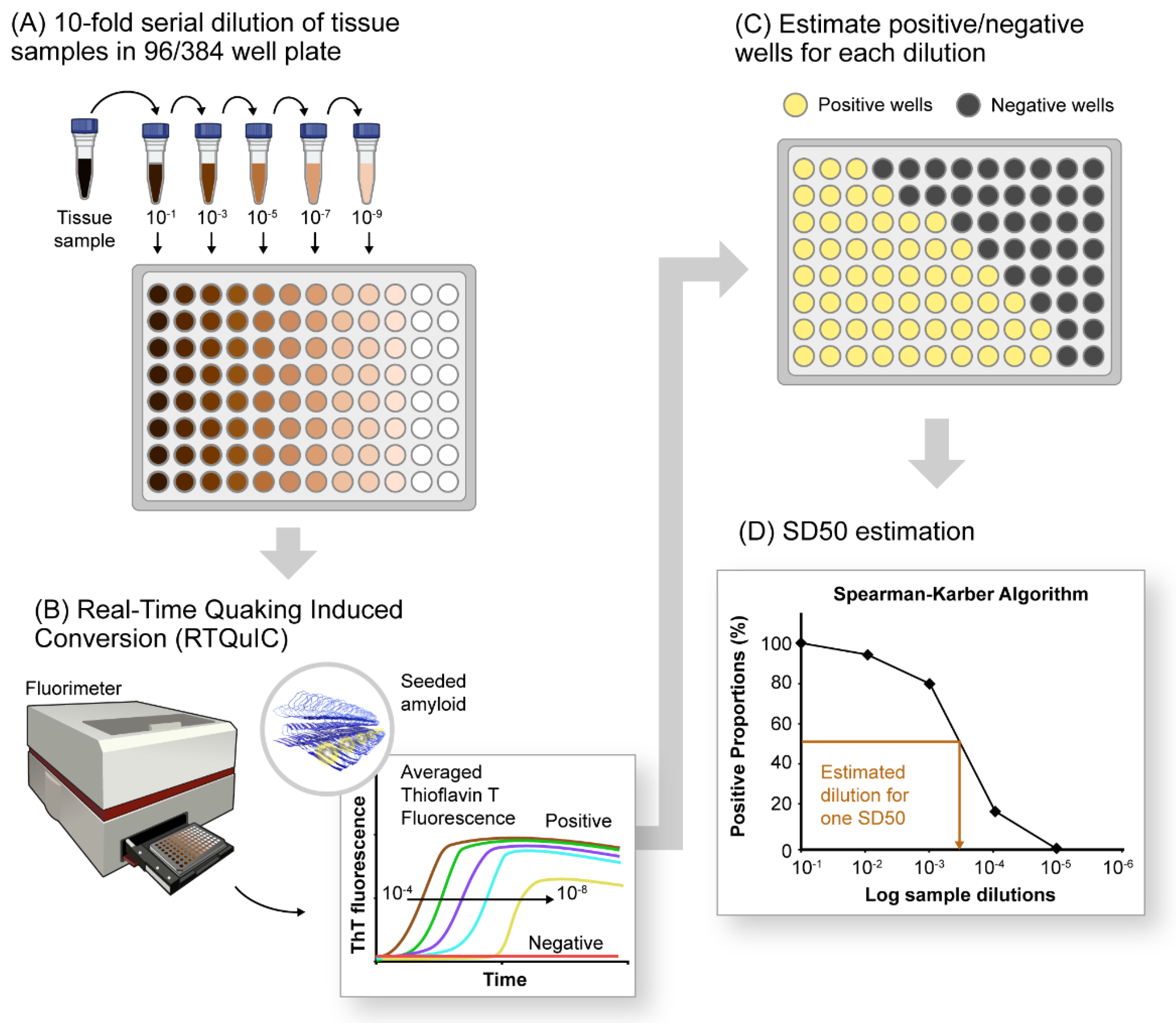
Figure 1. A schematic of αSyn RT-QuIC platform for detection and relative quantification of αSynD seeds in pathological tissues. (A) Serial dilutions (10−fold) of tissue sample (4 or 8 replicates) in a 96−well plate containing components of RT-QuIC reaction mixture including the recombinant, monomeric substrate protein and amyloid detection dye Thioflavin-T (ThT). (B) End-point estimation of serially diluted samples by the RT-QuIC assay. The resultant outcome is plotted as averaged ThT fluorescence versus time, showing declining fluorescence traces with increasing dilutions of positive samples. Negative samples do not exhibit any significant increase in ThT fluorescence under tested RT-QuIC conditions. (C) RT-QuIC outcomes for each dilution documented as number of positive and negative wells (for 4 or 8 replicates) per dilution tested. (D) Plot showing percentage of positive wells (shown as positive proportion percentages) for each sample dilution utilized for estimating seeding dose or sample dilutions in which 50% of the wells are ThT-positive (SD50).
4. SAAs for Detecting αSyn Aggregates
The biochemical framework of prion PMCA assays was later adapted for amplifying both αSyn PFFs and exogenous seeds present in tissue homogenates of transgenic αSyn mice [80][81]. Several αSyn SAAs are available for the detection of αSynD in various biospecimens. Among them, CSF is most commonly used for immediate as well as longitudinal diagnostics for most NDDs including synucleinopathies. In 2016, Fairfoul and colleagues first demonstrated RT-QuIC detection of αSynD seeding in a panel of 99 CSFs from DLB and PD patients with sensitivities of 92% and 95%, respectively, and 100% specificity against controls [28]. They observed lower RT-QuIC sensitivities in mixed pathologies including DLB with AD and AD with incidental LBs. A follow-up study reported 84% accuracy in discriminating α-synucleinopathies from non-synucleinopathies in a cohort of patients with parkinsonism of unclear etiology [42].
The closely related αSyn PMCA assay was described in 2017 [30]. This assay is similar in format to RT-QuIC, rather than the classical sonicated, brain-homogenate-based PMCA assays described above. A blinded analysis of a panel of PD and control CSF samples using a modified αSyn PMCA obtained an overall sensitivity of 88.5% and specificity of 96.9% [30].
Thereafter, an improved and faster αSyn RT-QuIC assay (RT-QuICR) was reported by Groveman and colleagues that shortened the overall assay time to <2 days [29] as compared with 5–13 days for the earlier assays [28][30]. RT-QuICR utilizes a mutant K23Q recombinant αSyn substrate that is less prone to spontaneously fibrilizing than the wild-type substrate. RT-QuICR displayed a 93% diagnostic sensitivity for LB disorder (PD and DLB) CSF samples with 100% specificity against non-synucleinopathy and healthy controls. Intriguingly, researchers observed a difference in the average RT-QuIC fluorescence maxima obtained from PD and DLB CSFs, which they construed as possible strain differences analogous to those reported for different types of CJD cases in prion RT-QuIC reactions [59][64]. Importantly, this improved αSyn RT-QuIC assay also provides a quantification methodology for obtaining the median seeding units present in tested tissue sample that are designated as SD50, for ‘50% seeding dose’ (Figure 1C,D).
A similar RT-QuIC-based study detected αSyn seeds across a spectrum of LB-related disorders, including DLB and PD, with an overall sensitivity of 95.3% [82]. The use of a closely related αSyn RT-QuIC assay on a large panel of neuropathologically confirmed cases (n = 214) showed a 98% sensitivity for LB disorders and higher seeding activity in both brain homogenates and CSFs from DLB as compared with PD patients [83]. The above observations were corroborated by other studies showing faster RT-QuIC seeding kinetics for DLB compared with PD brain homogenates and CSFs [83][84]. A more recent RT-QuIC study showed that detergent-soluble fractions from PD frontal cortex had higher seeding efficiency compared with those from DLB frontal cortex [85]. Seeding differences in synucleinopathy samples were attributed to distinct αSyn fibril strains analogous to prion strains reported in other studies [22][24][86][87][88][89][90].
5. Conclusions
In summary, αSyn RT-QuIC and PMCA (SAA) assays exploit the self-propagation (seeding) activity of αSynD to allow detection in biospecimens. αSyn SAAs provide unprecedented diagnostic sensitivity and specificity for α-synucleinopathies using a variety of biospecimens including brain tissue, CSF, skin, nasal brushings, and other biological samples from synucleinopathy patients. Altogether, αSyn SAAs provide protocols that require minimal hands-on time and increasingly rapid assay times for the accurate and ultrasensitive detection of αSynD seeds from different tissues from synucleinopathy patients. Future studies might help establish the utility of αSyn SAAs in longitudinal assessments of synucleinopathy progression and therapeutics.
References
- Koga, S.; Sekiya, H.; Kondru, N.; Ross, O.A.; Dickson, D.W. Neuropathology and molecular diagnosis of Synucleinopathies. Mol. Neurodegener. 2021, 16, 83.
- Burre, J.; Vivona, S.; Diao, J.; Sharma, M.; Brunger, A.T.; Sudhof, T.C. Properties of native brain α-synuclein. Nature 2013, 498, E4–E6.
- Mehra, S.; Sahay, S.; Maji, S.K. α-Synuclein misfolding and aggregation: Implications in Parkinson’s disease pathogenesis. Biochim. Biophys. Acta Proteins Proteom. 2019, 1867, 890–908.
- Ullman, O.; Fisher, C.K.; Stultz, C.M. Explaining the structural plasticity of α-synuclein. J. Am. Chem. Soc. 2011, 133, 19536–19546.
- Aulic, S.; Le, T.T.; Moda, F.; Abounit, S.; Corvaglia, S.; Casalis, L.; Gustincich, S.; Zurzolo, C.; Tagliavini, F.; Legname, G. Defined α-synuclein prion-like molecular assemblies spreading in cell culture. BMC Neurosci. 2014, 15, 69.
- Tarutani, A.; Arai, T.; Murayama, S.; Hisanaga, S.I.; Hasegawa, M. Potent prion-like behaviors of pathogenic α-synuclein and evaluation of inactivation methods. Acta Neuropathol. Commun. 2018, 6, 29.
- Brundin, P.; Melki, R.; Kopito, R. Prion-like transmission of protein aggregates in neurodegenerative diseases. Nat. Rev. Mol. Cell Biol. 2010, 11, 301–307.
- Braak, H.; Ghebremedhin, E.; Rub, U.; Bratzke, H.; Del Tredici, K. Stages in the development of Parkinson’s disease-related pathology. Cell Tissue Res. 2004, 318, 121–134.
- Kurowska, Z.; Englund, E.; Widner, H.; Lindvall, O.; Li, J.Y.; Brundin, P. Signs of degeneration in 12–22-year old grafts of mesencephalic dopamine neurons in patients with Parkinson’s disease. J. Parkinsons Dis. 2011, 1, 83–92.
- Angot, E.; Steiner, J.A.; Lema Tome, C.M.; Ekstrom, P.; Mattsson, B.; Bjorklund, A.; Brundin, P. α-Synuclein cell-to-cell transfer and seeding in grafted dopaminergic neurons in vivo. PLoS ONE 2012, 7, e39465.
- Volpicelli-Daley, L.A.; Luk, K.C.; Patel, T.P.; Tanik, S.A.; Riddle, D.M.; Stieber, A.; Meaney, D.F.; Trojanowski, J.Q.; Lee, V.M. Exogenous α-synuclein fibrils induce Lewy body pathology leading to synaptic dysfunction and neuron death. Neuron 2011, 72, 57–71.
- Luk, K.C.; Kehm, V.; Carroll, J.; Zhang, B.; O’Brien, P.; Trojanowski, J.Q.; Lee, V.M. Pathological α-synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice. Science 2012, 338, 949–953.
- Peng, C.; Gathagan, R.J.; Covell, D.J.; Medellin, C.; Stieber, A.; Robinson, J.L.; Zhang, B.; Pitkin, R.M.; Olufemi, M.F.; Luk, K.C.; et al. Cellular milieu imparts distinct pathological α-synuclein strains in α-synucleinopathies. Nature 2018, 557, 558–563.
- Kim, S.; Kwon, S.H.; Kam, T.I.; Panicker, N.; Karuppagounder, S.S.; Lee, S.; Lee, J.H.; Kim, W.R.; Kook, M.; Foss, C.A.; et al. Transneuronal Propagation of Pathologic α-synuclein from the Gut to the Brain Models Parkinson’s Disease. Neuron 2019, 103, 627–641.e627.
- Holmqvist, S.; Chutna, O.; Bousset, L.; Aldrin-Kirk, P.; Li, W.; Bjorklund, T.; Wang, Z.Y.; Roybon, L.; Melki, R.; Li, J.Y. Direct evidence of Parkinson pathology spread from the gastrointestinal tract to the brain in rats. Acta Neuropathol. 2014, 128, 805–820.
- Uemura, N.; Yagi, H.; Uemura, M.T.; Hatanaka, Y.; Yamakado, H.; Takahashi, R. Inoculation of α-synuclein preformed fibrils into the mouse gastrointestinal tract induces Lewy body-like aggregates in the brainstem via the vagus nerve. Mol. Neurodegener. 2018, 13, 21.
- Challis, C.; Hori, A.; Sampson, T.R.; Yoo, B.B.; Challis, R.C.; Hamilton, A.M.; Mazmanian, S.K.; Volpicelli-Daley, L.A.; Gradinaru, V. Gut-seeded α-synuclein fibrils promote gut dysfunction and brain pathology specifically in aged mice. Nat. Neurosci. 2020, 23, 327–336.
- Woerman, A.L.; Stohr, J.; Aoyagi, A.; Rampersaud, R.; Krejciova, Z.; Watts, J.C.; Ohyama, T.; Patel, S.; Widjaja, K.; Oehler, A.; et al. Propagation of prions causing synucleinopathies in cultured cells. Proc. Natl. Acad. Sci USA 2015, 112, E4949–E4958.
- Prusiner, S.B.; Woerman, A.L.; Mordes, D.A.; Watts, J.C.; Rampersaud, R.; Berry, D.B.; Patel, S.; Oehler, A.; Lowe, J.K.; Kravitz, S.N.; et al. Evidence for α-synuclein prions causing multiple system atrophy in humans with parkinsonism. Proc. Natl. Acad. Sci. USA 2015, 112, E5308–E5317.
- Woerman, A.L.; Patel, S.; Kazmi, S.A.; Oehler, A.; Lee, J.; Mordes, D.A.; Olson, S.H.; Prusiner, S.B. Kinetics of α-synuclein prions preceding neuropathological inclusions in multiple system atrophy. PLoS Pathog. 2020, 16, e1008222.
- Woerman, A.L.; Oehler, A.; Kazmi, S.A.; Lee, J.; Halliday, G.M.; Middleton, L.T.; Gentleman, S.M.; Mordes, D.A.; Spina, S.; Grinberg, L.T.; et al. Multiple system atrophy prions retain strain specificity after serial propagation in two different Tg(SNCA*A53T) mouse lines. Acta Neuropathol. 2019, 137, 437–454.
- Tuttle, M.D.; Comellas, G.; Nieuwkoop, A.J.; Covell, D.J.; Berthold, D.A.; Kloepper, K.D.; Courtney, J.M.; Kim, J.K.; Barclay, A.M.; Kendall, A.; et al. Solid-state NMR structure of a pathogenic fibril of full-length human α-synuclein. Nat. Struct. Mol. Biol. 2016, 23, 409–415.
- Burger, D.; Fenyi, A.; Bousset, L.; Stahlberg, H.; Melki, R. Cryo-EM structure of α-synuclein fibrils amplified by PMCA from PD and MSA patient brains. bioRxiv 2021.
- Guerrero-Ferreira, R.; Taylor, N.M.; Mona, D.; Ringler, P.; Lauer, M.E.; Riek, R.; Britschgi, M.; Stahlberg, H. Cryo-EM structure of α-synuclein fibrils. Elife 2018, 7, e36402.
- Holec, S.A.M.; Woerman, A.L. Evidence of distinct α-synuclein strains underlying disease heterogeneity. Acta Neuropathol. 2021, 142, 73–86.
- Strohaker, T.; Jung, B.C.; Liou, S.H.; Fernandez, C.O.; Riedel, D.; Becker, S.; Halliday, G.M.; Bennati, M.; Kim, W.S.; Lee, S.J.; et al. Structural heterogeneity of α-synuclein fibrils amplified from patient brain extracts. Nat. Commun. 2019, 10, 5535.
- Russo, M.J.; Orru, C.D.; Concha-Marambio, L.; Giaisi, S.; Groveman, B.R.; Farris, C.M.; Holguin, B.; Hughson, A.G.; LaFontant, D.E.; Caspell-Garcia, C.; et al. High diagnostic performance of independent α-synuclein seed amplification assays for detection of early Parkinson’s disease. Acta Neuropathol. Commun. 2021, 9, 179.
- Fairfoul, G.; McGuire, L.I.; Pal, S.; Ironside, J.W.; Neumann, J.; Christie, S.; Joachim, C.; Esiri, M.; Evetts, S.G.; Rolinski, M.; et al. α-Synuclein RT-QuIC in the CSF of patients with α-synucleinopathies. Ann. Clin. Transl. Neurol. 2016, 3, 812–818.
- Groveman, B.R.; Orru, C.D.; Hughson, A.G.; Raymond, L.D.; Zanusso, G.; Ghetti, B.; Campbell, K.J.; Safar, J.; Galasko, D.; Caughey, B. Rapid and ultra-sensitive quantitation of disease-associated α-synuclein seeds in brain and cerebrospinal fluid by alphaSyn RT-QuIC. Acta Neuropathol. Commun. 2018, 6, 7.
- Shahnawaz, M.; Tokuda, T.; Waragai, M.; Mendez, N.; Ishii, R.; Trenkwalder, C.; Mollenhauer, B.; Soto, C. Development of a Biochemical Diagnosis of Parkinson Disease by Detection of α-Synuclein Misfolded Aggregates in Cerebrospinal Fluid. JAMA Neurol. 2017, 74, 163–172.
- Gilman, S.; Wenning, G.K.; Low, P.A.; Brooks, D.J.; Mathias, C.J.; Trojanowski, J.Q.; Wood, N.W.; Colosimo, C.; Durr, A.; Fowler, C.J.; et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology 2008, 71, 670–676.
- Lucetti, C.; Logi, C.; Del Dotto, P.; Berti, C.; Ceravolo, R.; Baldacci, F.; Dolciotti, C.; Gambaccini, G.; Rossi, G.; Bonuccelli, U. Levodopa response in dementia with lewy bodies: A 1-year follow-up study. Parkinsonism. Relat. Disord. 2010, 16, 522–526.
- McKeith, I.G.; Dickson, D.W.; Lowe, J.; Emre, M.; O’Brien, J.T.; Feldman, H.; Cummings, J.; Duda, J.E.; Lippa, C.; Perry, E.K.; et al. Diagnosis and management of dementia with Lewy bodies: Third report of the DLB Consortium. Neurology 2005, 65, 1863–1872.
- Hoglinger, G.U.; Respondek, G.; Stamelou, M.; Kurz, C.; Josephs, K.A.; Lang, A.E.; Mollenhauer, B.; Muller, U.; Nilsson, C.; Whitwell, J.L.; et al. Clinical diagnosis of progressive supranuclear palsy: The movement disorder society criteria. Mov. Disord. 2017, 32, 853–864.
- McKeith, I.G.; Boeve, B.F.; Dickson, D.W.; Halliday, G.; Taylor, J.P.; Weintraub, D.; Aarsland, D.; Galvin, J.; Attems, J.; Ballard, C.G.; et al. Diagnosis and management of dementia with Lewy bodies: Fourth consensus report of the DLB Consortium. Neurology 2017, 89, 88–100.
- Geschwind, M.D.; Murray, K. Differential diagnosis with other rapid progressive dementias in human prion diseases. Handb Clin. Neurol. 2018, 153, 371–397.
- Zerr, I.; Hermann, P. Diagnostic challenges in rapidly progressive dementia. Expert Rev. Neurother. 2018, 18, 761–772.
- Fabbrini, G.; Fabbrini, A.; Suppa, A. Progressive supranuclear palsy, multiple system atrophy and corticobasal degeneration. Handb. Clin. Neurol. 2019, 165, 155–177.
- Aerts, M.B.; Esselink, R.A.; Abdo, W.F.; Meijer, F.J.; Drost, G.; Norgren, N.; Janssen, M.J.; Borm, G.F.; Bloem, B.R.; Verbeek, M.M. Ancillary investigations to diagnose parkinsonism: A prospective clinical study. J. Neurol. 2015, 262, 346–356.
- Iranzo, A.; Fairfoul, G.; Ayudhaya, A.C.N.; Serradell, M.; Gelpi, E.; Vilaseca, I.; Sanchez-Valle, R.; Gaig, C.; Santamaria, J.; Tolosa, E.; et al. Detection of α-synuclein in CSF by RT-QuIC in patients with isolated rapid-eye-movement sleep behaviour disorder: A longitudinal observational study. Lancet Neurol. 2021, 20, 203–212.
- Kang, U.J.; Boehme, A.K.; Fairfoul, G.; Shahnawaz, M.; Ma, T.C.; Hutten, S.J.; Green, A.; Soto, C. Comparative study of cerebrospinal fluid α-synuclein seeding aggregation assays for diagnosis of Parkinson’s disease. Mov. Disord. 2019, 34, 536–544.
- van Rumund, A.; Green, A.J.E.; Fairfoul, G.; Esselink, R.A.J.; Bloem, B.R.; Verbeek, M.M. α-Synuclein real-time quaking-induced conversion in the cerebrospinal fluid of uncertain cases of parkinsonism. Ann. Neurol. 2019, 85, 777–781.
- Bongianni, M.; Ladogana, A.; Capaldi, S.; Klotz, S.; Baiardi, S.; Cagnin, A.; Perra, D.; Fiorini, M.; Poleggi, A.; Legname, G.; et al. α-Synuclein RT-QuIC assay in cerebrospinal fluid of patients with dementia with Lewy bodies. Ann. Clin. Transl. Neurol. 2019, 6, 2120–2126.
- De Luca, C.M.G.; Elia, A.E.; Portaleone, S.M.; Cazzaniga, F.A.; Rossi, M.; Bistaffa, E.; De Cecco, E.; Narkiewicz, J.; Salzano, G.; Carletta, O.; et al. Efficient RT-QuIC seeding activity for α-synuclein in olfactory mucosa samples of patients with Parkinson’s disease and multiple system atrophy. Transl. Neurodegener. 2019, 8, 24.
- Rossi, M.; Baiardi, S.; Teunissen, C.E.; Quadalti, C.; van de Beek, M.; Mammana, A.; Maserati, M.S.; Van der Flier, W.M.; Sambati, L.; Zenesini, C.; et al. Diagnostic Value of the CSF α-Synuclein Real-Time Quaking-Induced Conversion Assay at the Prodromal MCI Stage of Dementia With Lewy Bodies. Neurology 2021, 97, e930–e940.
- Raymond, G.J.; Hope, J.; Kocisko, D.A.; Priola, S.A.; Raymond, L.D.; Bossers, A.; Ironside, J.; Will, R.G.; Chen, S.G.; Petersen, R.B.; et al. Molecular assessment of the transmissibilities of BSE and scrapie to humans. Nature 1997, 388, 285–288.
- Kocisko, D.A.; Priola, S.A.; Raymond, G.J.; Chesebro, B.; Lansbury, P.T., Jr.; Caughey, B. Species specificity in the cell-free conversion of prion protein to protease-resistant forms: A model for the scrapie species barrier. Proc. Natl. Acad. Sci. USA 1995, 92, 3923–3927.
- Bessen, R.A.; Kocisko, D.A.; Raymond, G.J.; Nandan, S.; Lansbury, P.T., Jr.; Caughey, B. Nongenetic propagation of strain-specific phenotypes of scrapie prion protein. Nature 1995, 375, 698–700.
- Kocisko, D.A.; Come, J.H.; Priola, S.A.; Chesebro, B.; Raymond, G.J.; Lansbury, P.T.; Caughey, B. Cell-free formation of protease-resistant prion protein. Nature 1994, 370, 471–474.
- Raymond, G.J.; Bossers, A.; Raymond, L.D.; O’Rourke, K.I.; McHolland, L.E.; Bryant, P.K., III; Miller, M.W.; Williams, E.S.; Smits, M.; Caughey, B. Evidence of a molecular barrier limiting susceptibility of humans, cattle and sheep to chronic wasting disease. EMBO J. 2000, 19, 4425–4430.
- Bossers, A.; Belt, P.B.G.M.; Raymond, G.J.; Caughey, B.; de Vries, R.; Smits, M.A. Scrapie susceptibility-linked polymorphisms modulate the in vitro conversion of sheep prion protein to protease-resistant forms. Proc. Natl. Acad. Sci. USA 1997, 94, 4931–4936.
- Castilla, J.; Saa, P.; Hetz, C.; Soto, C. In vitro generation of infectious scrapie prions. Cell 2005, 121, 195–206.
- Saborio, G.P.; Permanne, B.; Soto, C. Sensitive detection of pathological prion protein by cyclic amplification of protein misfolding. Nature 2001, 411, 810–813.
- Saa, P.; Castilla, J.; Soto, C. Ultra-efficient replication of infectious prions by automated protein misfolding cyclic amplification. J. Biol. Chem. 2006, 281, 35245–35252.
- Colby, D.W.; Zhang, Q.; Wang, S.; Groth, D.; Legname, G.; Riesner, D.; Prusiner, S.B. Prion detection by an amyloid seeding assay. Proc. Natl. Acad. Sci. USA 2007, 104, 20914–20919.
- Atarashi, R.; Satoh, K.; Sano, K.; Fuse, T.; Yamaguchi, N.; Ishibashi, D.; Matsubara, T.; Nakagaki, T.; Yamanaka, H.; Shirabe, S.; et al. Ultrasensitive human prion detection in cerebrospinal fluid by real-time quaking-induced conversion. Nat. Med. 2011, 17, 175–178.
- Wilham, J.M.; Orrú, C.D.; Bessen, R.A.; Atarashi, R.; Sano, K.; Race, B.; Meade-White, K.D.; Taubner, L.M.; Timmes, A.; Caughey, B. Rapid End-Point Quantitation of Prion Seeding Activity with Sensitivity Comparable to Bioassays. PLoS Pathog. 2010, 6, e1001217.
- Raymond, G.J.; Race, B.; Orru, C.D.; Raymond, L.D.; Bongianni, M.; Fiorini, M.; Groveman, B.R.; Ferrari, S.; Sacchetto, L.; Hughson, A.G.; et al. Transmission of CJD from nasal brushings but not spinal fluid or RT-QuIC product. Ann. Clin. Transl. Neurol. 2020, 7, 932–944.
- Orru, C.D.; Groveman, B.R.; Raymond, L.D.; Hughson, A.G.; Nonno, R.; Zou, W.; Ghetti, B.; Gambetti, P.; Caughey, B. Bank Vole Prion Protein As an Apparently Universal Substrate for RT-QuIC-Based Detection and Discrimination of Prion Strains. PLoS Pathog. 2015, 11, e1004983.
- Atarashi, R.; Wilham, J.M.; Christensen, L.; Hughson, A.G.; Moore, R.A.; Johnson, L.M.; Onwubiko, H.A.; Priola, S.A.; Caughey, B. Simplified ultrasensitive prion detection by recombinant PrP conversion with shaking. Nat. Methods 2008, 5, 211–212.
- Atarashi, R.; Moore, R.A.; Sim, V.L.; Hughson, A.G.; Dorward, D.W.; Onwubiko, H.A.; Priola, S.A.; Caughey, B. Ultrasensitive detection of scrapie prion protein using seeded conversion of recombinant prion protein. Nat. Methods 2007, 4, 645–650.
- McGuire, L.I.; Peden, A.H.; Orru, C.D.; Wilham, J.M.; Appleford, N.E.; Mallinson, G.; Andrews, M.; Head, M.W.; Caughey, B.; Will, R.G.; et al. RT-QuIC analysis of cerebrospinal fluid in sporadic Creutzfeldt-Jakob disease. Ann. Neurol. 2012, 72, 278–285.
- Orru, C.D.; Groveman, B.R.; Hughson, A.G.; Zanusso, G.; Coulthart, M.B.; Caughey, B. Rapid and sensitive RT-QuIC detection of human Creutzfeldt-Jakob disease using cerebrospinal fluid. MBio 2015, 6, e02451-14.
- Foutz, A.; Appleby, B.S.; Hamlin, C.; Liu, X.; Yang, S.; Cohen, Y.; Chen, W.; Blevins, J.; Fausett, C.; Wang, H.; et al. Diagnostic and prognostic value of human prion detection in cerebrospinal fluid. Ann. Neurol. 2017, 81, 79–92.
- Franceschini, A.; Baiardi, S.; Hughson, A.G.; McKenzie, N.; Moda, F.; Rossi, M.; Capellari, S.; Green, A.; Giaccone, G.; Caughey, B.; et al. High diagnostic value of second generation CSF RT-QuIC across the wide spectrum of CJD prions. Sci. Rep. 2017, 7, 10655.
- Wang, Z.; Manca, M.; Foutz, A.; Camacho, M.V.; Raymond, G.J.; Race, B.; Orru, C.D.; Yuan, J.; Shen, P.; Li, B.; et al. Early preclinical detection of prions in the skin of prion-infected animals. Nat. Commun. 2019, 10, 247.
- Orru, C.D.; Yuan, J.; Appleby, B.S.; Li, B.; Li, Y.; Winner, D.; Wang, Z.; Zhan, Y.A.; Rodgers, M.; Rarick, J.; et al. Prion seeding activity and infectivity in skin samples from patients with sporadic Creutzfeldt-Jakob disease. Sci. Transl. Med. 2017, 9, eaam7785.
- Mammana, A.; Baiardi, S.; Rossi, M.; Franceschini, A.; Donadio, V.; Capellari, S.; Caughey, B.; Parchi, P. Detection of prions in skin punch biopsies of Creutzfeldt-Jakob disease patients. Ann. Clin. Transl. Neurol. 2020, 7, 559–564.
- Orru, C.D.; Soldau, K.; Cordano, C.; Llibre-Guerra, J.; Green, A.J.; Sanchez, H.; Groveman, B.R.; Edland, S.D.; Safar, J.G.; Lin, J.H.; et al. Prion Seeds Distribute throughout the Eyes of Sporadic Creutzfeldt-Jakob Disease Patients. MBio 2018, 9, e02095-18.
- Orru, C.D.; Bongianni, M.; Tonoli, G.; Ferrari, S.; Hughson, A.G.; Groveman, B.R.; Fiorini, M.; Pocchiari, M.; Monaco, S.; Caughey, B.; et al. A test for Creutzfeldt-Jakob disease using nasal brushings. N. Engl. J. Med. 2014, 371, 519–529.
- Bongianni, M.; Orrù, C.D.; Groveman, B.R.; Sacchetto, L.; Fiorini, M.; Tonoli, G.; Triva, G.; Capaldi, S.; Testi, S.; Ferrari, S.; et al. Diagnosis of Human Prion Disease Using Real-Time Quaking-Induced Conversion Testing of Olfactory Mucosa and Cerebrospinal Fluid Samples. JAMA Neurol. 2017, 74, 155–162.
- Orru, C.D.; Groveman, B.R.; Foutz, A.; Bongianni, M.; Cardone, F.; McKenzie, N.; Culeux, A.; Poleggi, A.; Grznarova, K.; Perra, D.; et al. Ring trial of 2nd generation RT-QuIC diagnostic tests for sporadic CJD. Ann. Clin. Transl. Neurol. 2020, 7, 2262–2271.
- Nalls, A.V.; McNulty, E.E.; Mayfield, A.; Crum, J.M.; Keel, M.K.; Hoover, E.A.; Ruder, M.G.; Mathiason, C.K. Detection of Chronic Wasting Disease Prions in Fetal Tissues of Free-Ranging White-Tailed Deer. Viruses 2021, 13, 2430.
- Nalls, A.V.; McNulty, E.; Hoover, C.E.; Pulscher, L.A.; Hoover, E.A.; Mathiason, C.K. Infectious Prions in the Pregnancy Microenvironment of Chronic Wasting Disease-Infected Reeves’ Muntjac Deer. J. Virol. 2017, 91, e00501-17.
- Selariu, A.; Powers, J.G.; Nalls, A.; Brandhuber, M.; Mayfield, A.; Fullaway, S.; Wyckoff, C.A.; Goldmann, W.; Zabel, M.M.; Wild, M.A.; et al. In utero transmission and tissue distribution of chronic wasting disease-associated prions in free-ranging Rocky Mountain elk. J. Gen. Virol. 2015, 96, 3444–3455.
- Chen, B.; Morales, R.; Barria, M.A.; Soto, C. Estimating prion concentration in fluids and tissues by quantitative PMCA. Nat. Methods 2010, 7, 519–520.
- Orru, C.D.; Wilham, J.M.; Raymond, L.D.; Kuhn, F.; Schroeder, B.; Raeber, A.J.; Caughey, B. Prion disease blood test using immunoprecipitation and improved quaking-induced conversion. mBio 2011, 2, e00078-11.
- Morales, R.; Duran-Aniotz, C.; Diaz-Espinoza, R.; Camacho, M.V.; Soto, C. Protein misfolding cyclic amplification of infectious prions. Nat. Protoc. 2012, 7, 1397–1409.
- Concha-Marambio, L.; Pritzkow, S.; Moda, F.; Tagliavini, F.; Ironside, J.W.; Schulz, P.E.; Soto, C. Detection of prions in blood from patients with variant Creutzfeldt-Jakob disease. Sci. Transl. Med. 2016, 8, 370ra183.
- Jung, B.C.; Lim, Y.J.; Bae, E.J.; Lee, J.S.; Choi, M.S.; Lee, M.K.; Lee, H.J.; Kim, Y.S.; Lee, S.J. Amplification of distinct α-synuclein fibril conformers through protein misfolding cyclic amplification. Exp. Mol. Med. 2017, 49, e314.
- Herva, M.E.; Zibaee, S.; Fraser, G.; Barker, R.A.; Goedert, M.; Spillantini, M.G. Anti-amyloid compounds inhibit α-synuclein aggregation induced by protein misfolding cyclic amplification (PMCA). J. Biol. Chem. 2014, 289, 11897–11905.
- Rossi, M.; Candelise, N.; Baiardi, S.; Capellari, S.; Giannini, G.; Orru, C.D.; Antelmi, E.; Mammana, A.; Hughson, A.G.; Calandra-Buonaura, G.; et al. Ultrasensitive RT-QuIC assay with high sensitivity and specificity for Lewy body-associated synucleinopathies. Acta Neuropathol. 2020, 140, 49–62.
- Bargar, C.; Wang, W.; Gunzler, S.A.; LeFevre, A.; Wang, Z.; Lerner, A.J.; Singh, N.; Tatsuoka, C.; Appleby, B.; Zhu, X.; et al. Streamlined α-synuclein RT-QuIC assay for various biospecimens in Parkinson’s disease and dementia with Lewy bodies. Acta Neuropathol. Commun. 2021, 9, 62.
- Candelise, N.; Schmitz, M.; Llorens, F.; Villar-Pique, A.; Cramm, M.; Thom, T.; da Silva Correia, S.M.; da Cunha, J.E.G.; Mobius, W.; Outeiro, T.F.; et al. Seeding variability of different alpha synuclein strains in synucleinopathies. Ann. Neurol. 2019, 85, 691–703.
- Poggiolini, I.; Erskine, D.; Vaikath, N.N.; Ponraj, J.; Mansour, S.; Morris, C.M.; El-Agnaf, O.M.A. RT-QuIC Using C-Terminally Truncated α-Synuclein Forms Detects Differences in Seeding Propensity of Different Brain Regions from Synucleinopathies. Biomolecules 2021, 11, 820.
- Ayers, J.I.; Lee, J.; Monteiro, O.; Woerman, A.L.; Lazar, A.A.; Condello, C.; Paras, N.A.; Prusiner, S.B. Different α-synuclein prion strains cause dementia with Lewy bodies and multiple system atrophy. Proc. Natl. Acad. Sci. USA 2022, 119, e2113489119.
- Schweighauser, M.; Shi, Y.; Tarutani, A.; Kametani, F.; Murzin, A.G.; Ghetti, B.; Matsubara, T.; Tomita, T.; Ando, T.; Hasegawa, K.; et al. Structures of α-synuclein filaments from multiple system atrophy. Nature 2020, 585, 464–469.
- Li, Y.; Zhao, C.; Luo, F.; Liu, Z.; Gui, X.; Luo, Z.; Zhang, X.; Li, D.; Liu, C.; Li, X. Amyloid fibril structure of α-synuclein determined by cryo-electron microscopy. Cell Res. 2018, 28, 897–903.
- Li, B.; Ge, P.; Murray, K.A.; Sheth, P.; Zhang, M.; Nair, G.; Sawaya, M.R.; Shin, W.S.; Boyer, D.R.; Ye, S.; et al. Cryo-EM of full-length α-synuclein reveals fibril polymorphs with a common structural kernel. Nat. Commun. 2018, 9, 3609.
- Boyer, D.R.; Li, B.; Sun, C.; Fan, W.; Sawaya, M.R.; Jiang, L.; Eisenberg, D.S. Structures of fibrils formed by α-synuclein hereditary disease mutant H50Q reveal new polymorphs. Nat. Struct. Mol. Biol. 2019, 26, 1044–1052.
More
Information
Subjects:
Clinical Neurology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.6K
Revisions:
2 times
(View History)
Update Date:
22 Apr 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





