
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Agata Lewandowska | -- | 1940 | 2022-04-21 12:07:20 | | | |
| 2 | Peter Tang | Meta information modification | 1940 | 2022-04-22 03:23:40 | | |
Video Upload Options
The COVID-19 pandemic caused by the SARS-CoV-2 virus made it necessary to search for new options for both causal treatment and mitigation of its symptoms. Scientists and researchers around the world are constantly looking for the best therapeutic options. These difficult circumstances have also spurred the re-examination of the potential of natural substances contained in Cannabis sativa L. Cannabinoids, apart from CB1 and CB2 receptors, may act multifacetedly through a number of other receptors, such as the GPR55, TRPV1, PPARs, 5-HT1A, adenosine and glycine receptors. The complex anti-inflammatory and antiviral effects of cannabinoids have been confirmed by interactions with various signaling pathways. Considering the fact that the SARS-CoV-2 virus causes excessive immune response and triggers an inflammatory cascade, and that cannabinoids have the ability to regulate these processes, it can be assumed that they have potential to be used in the treatment of COVID-19.
1. Introduction
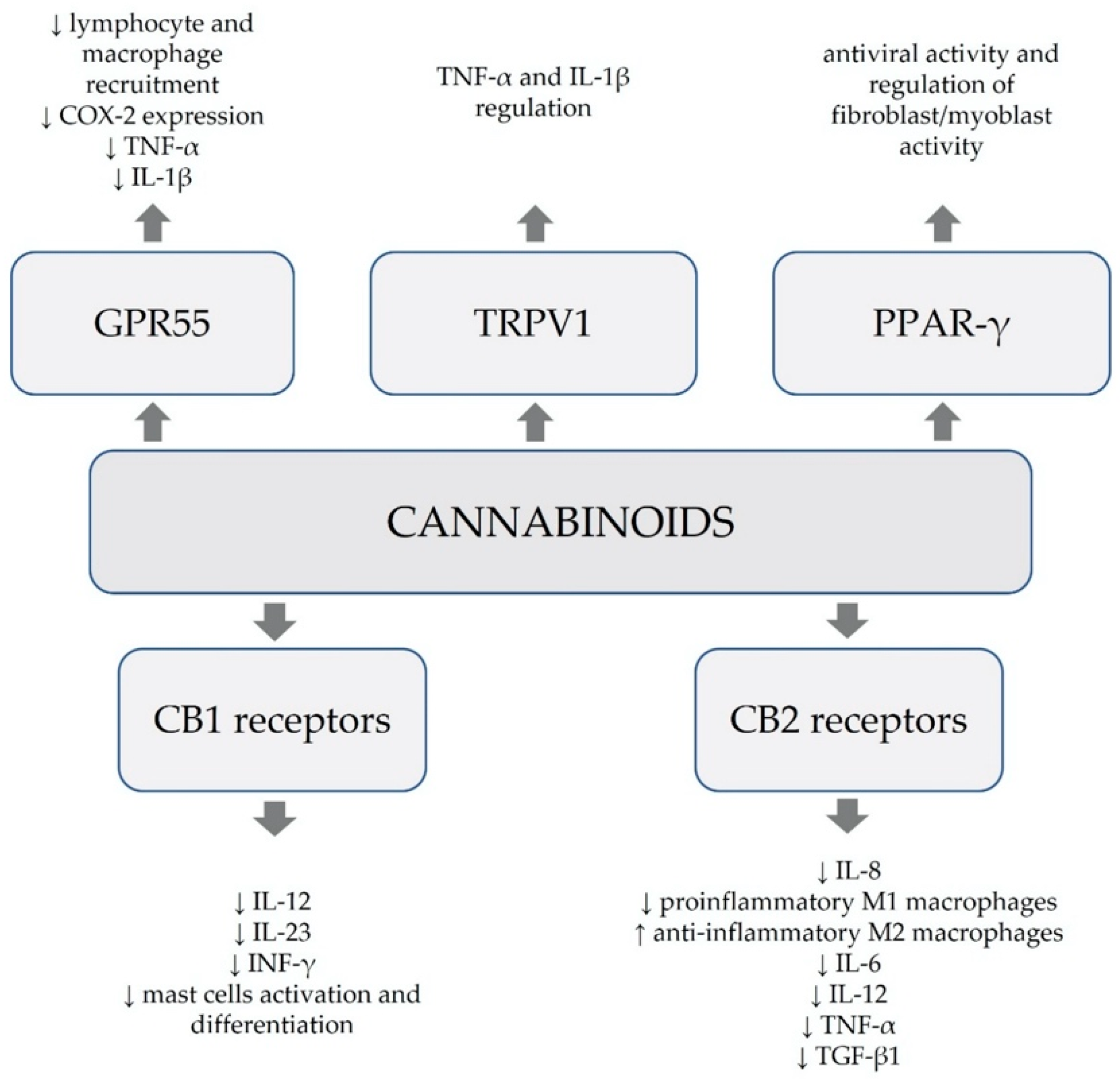
2. Antiviral Properties of Cannabinoids
|
CBD, THC |
Inhibiting the virus translation and replication by supressing SARS-CoV-2 Mpro protease. |
[5] |
|
CBD |
Preventing entry into host cells by inhibition of TMPRSS2. |
|
|
High-CBD cannabis extract, CBD |
Reducing susceptibility to infection by downregulation of the expression of ACE2 receptors. |
|
|
CBD |
Eradicating viral RNA expression in cells by preventing protein translation and other cellular alterations—potentially through activation of the interferon signalling pathway. |
[18] |
|
CBGA, CBDA |
Preventing the SARS-CoV-2 infection at the point of cell entry. |
[27] |
CBD, cannabidiol; THC, Δ9-tetrahydrocannabinol; TMPRSS2, transmembrane serine protease 2; ACE2, angiotensin converting enzyme 2; CBGA, cannabigerolic acid; CBDA, cannabidiolic acid.
3. Anti-Inflammatory Properties of Cannabinoids
References
- Gatta-Cherifi, B.; Cota, D. New insights on the role of the endocannabinoid system in the regulation of energy balance. Int. J. Obes. 2016, 40, 210–219.
- Di Marzo, V. The endocannabinoid system: Its general strategy of action, tools for its pharmacological manipulation and potential therapeutic exploitation. Pharmacol. Res. 2009, 60, 77–84.
- Graczyk, M.; Lewandowska, A.A.; Dzierżanowski, T. The Therapeutic Potential of Cannabis in Counteracting Oxidative Stress and Inflammation. Molecules 2021, 26, 4551.
- Kendall, D.A.; Yudowski, G.A. Cannabinoid receptors in the central nervous system: Their signaling and roles in disease. Front. Cell Neurosci. 2017, 10, 294.
- Raj, V.; Park, J.G.; Cho, K.-H.; Choi, P.; Kim, T.; Ham, J.; Lee, J. Assessment of antiviral potencies of cannabinoids against SARS-CoV-2 using computational and in vitro approaches. Int. J. Biol. Macromol. 2021, 168, 474–485.
- Jha, N.K.; Sharma, C.; Hashiesh, H.M.; Arunachalam, S.; Meeran, M.N.; Javed, H.; Patil, C.R.; Goyal, S.N.; Ojha, S. β-Caryophyllene, A Natural Dietary CB2 Receptor Selective Cannabinoid can be a Candidate to Target the Trinity of Infection, Immunity, and Inflammation in COVID-19. Front. Pharmacol. 2021, 12, 590201.
- García-Gutiérrez, M.S.; Navarrete, F.; Gasparyan, A.; Austrich-Olivares, A.; Sala, F.; Manzanares, J. Cannabidiol: A potential new alternative for the treatment of anxiety, depression, and psychotic disorders. Biomolecules 2020, 10, 1575.
- El Biali, M.; Broers, B.; Besson, M.; Demeules, J. Cannabinoids and COVID-19. Med. Cannabis Cannabinoids 2020, 3, 111–115.
- Wood, J.S.; Gordon, W.H.; Morgan, J.B.; Williamson, R.T. Calculated and experimental 1H and 13C NMR assignments for cannabicitran. Magn. Reson. Chem. 2022, 60, 196–202.
- Sommano, S.R.; Chittasupho, C.; Ruksiriwanich, W.; Jantrawut, P. The Cannabis Terpenes. Molecules 2020, 25, 5792.
- Terence, N.; Vikas, G. Tetrahydrocannabinol (THC); StatPearls Publishing: Treasure Island, FL, USA, 2022.
- Sharma, P.; Murthy, P.; Bharath, M.S. Chemistry, metabolism, and toxicology of cannabis: Clinical implications. Iran. J. Psychiatry 2012, 7, 149–156.
- Meissner, H.; Cascella, M. Cannabidiol (CBD); StatPearls Publishing: Treasure Island, FL, USA, 2022.
- Nichols, J.M.; Kaplan, B.L.F. Immune Responses Regulated by Cannabidiol. Cannabis Cannabinoid Res. 2020, 5, 12–31.
- Kogan, N.M.; Mechoulam, R. Cannabinoids in health and disease. Dialogues Clin. Neurosci. 2007, 9, 413–430.
- Pascarella, G.; Strumia, A.; Piliego, C.; Bruno, F.; Del Buono, R.; Costa, F.; Scarlata, S.; Agrò, F.E. COVID-19 diagnosis and management: A comprehensive review. J. Intern. Med. 2020, 288, 192–206.
- Wang, B.; Kovalchuk, A.; Li, D.; Rodriguez-Juarez, R.; Ilnytskyy, Y.; Kovalchuk, I.; Kovalchuk, O. In search of preventive strategies: Novel high-CBD Cannabis sativa extracts modulate ACE2 expression in COVID-19 gateway tissues. Aging 2020, 12, 22425–22444.
- Nguyen, L.C.; Yang, D.; Nicolaescu, V.; Best, T.J.; Ohtsuki, T.; Chen, S.-N.; Friesen, J.B.; Drayman, N.; Mohamed, A.; Dann, C.; et al. Cannabidiol Inhibits SARS-CoV-2 Replication and Promotes the Host Innate Immune Response. bioRxiv Prepr. Serv. Biol. 2021, 3, 45.
- Suryavanshi, S.V.; Kovalchuk, I.; Kovalchuk, O. Cannabinoids as Key Regulators of Inflammasome Signaling: A Current Perspective. Front. Immunol. 2021, 11, 3638.
- Sureram, S.; Arduino, I.; Ueoka, R.; Rittà, M.; Francese, R.; Srivibool, R.; Darshana, D.; Piel, J.; Ruchirawat, S.; Muratori, L.; et al. The Peptide A-3302-B Isolated from a Marine Bacterium Micromonospora sp. Inhibits HSV-2 Infection by Preventing the Viral Egress from Host Cells. Int. J. Mol. Sci. 2022, 23, 947.
- Erukainure, O.L.; Matsabisa, M.G.; Muhammad, A.; Abarshi, M.M.; Amaku, J.F.; Katsayal, S.B.; Nde, A.L. Targeting of Protein’s Messenger RNA for Viral Replication, Assembly and Release in SARS-CoV-2 Using Whole Genomic Data From South Africa: Therapeutic Potentials of Cannabis Sativa L. Front. Pharmacol. 2021, 12, 2314.
- Gurung, A.B.; Ali, M.A.; Lee, J.; Farah, M.A.; Al-Anazi, K.M. Unravelling lead antiviral phytochemicals for the inhibition of SARS-CoV-2 Mpro enzyme through in silico approach. Life Sci. 2020, 255, 117831.
- Mahmud, S.; Hossain, M.S.; Ahmed, A.T.M.F.; Islam, Z.; Sarker, E.; Islam, R. Antimicrobial and antiviral (SARS-CoV-2) potential of cannabinoids and Cannabis sativa: A comprehensive review. Molecules 2021, 26, 7216.
- Esposito, G.; Pesce, M.; Seguella, L.; Sanseverino, W.; Lu, J.; Corpetti, C.; Sarnelli, G. The potential of cannabidiol in the COVID-19 pandemic. Br. J. Pharmacol. 2020, 177, 4967–4970.
- Malinowska, B.; Baranowska-Kuczko, M.; Kicman, A.; Schlicker, E. Opportunities, challenges and pitfalls of using cannabidiol as an adjuvant drug in COVID19. Int. J. Mol. Sci. 2021, 22, 1986.
- Anil, S.M.; Shalev, N.; Vinayaka, A.C.; Nadarajan, S.; Namdar, D.; Belausov, E.; Shoval, I.; Mani, K.A.; Mechrez, G.; Koltai, H. Cannabis compounds exhibit anti-inflammatory activity in vitro in COVID-19-related inflammation in lung epithelial cells and pro-inflammatory activity in macrophages. Sci. Rep. 2021, 11, 1462.
- van Breemen, R.B.; Muchiri, R.N.; Bates, T.A.; Weinstein, J.B.; Leier, H.C.; Farley, S.; Tafesse, F.G. Cannabinoids Block Cellular Entry of SARS-CoV-2 and the Emerging Variants. J. Nat. Prod. 2022, 85, 176–184.
- Ribeiro, A.; Almeida, V.I.; Costola-De-Souza, C.; Ferraz-De-Paula, V.; Pinheiro, M.L.; Vitoretti, L.B.; Junior, J.A.G.; Akamine, A.T.; Crippa, J.A.; Tavares-De-Lima, W.; et al. Cannabidiol improves lung function and inflammation in mice submitted to LPS-induced acute lung injury. Immunopharmacol. Immunotoxicol. 2015, 37, 35–41.
- Onaivi, E.S.; Sharma, V. Cannabis for COVID-19: Can cannabinoids quell the cytokine storm? Futur. Sci. OA 2020, 6, FSO625.
- Aragão, L.G.H.S.; Oliveira, J.T.; Temerozo, J.R.; Mendes, M.A.; Salerno, J.A.; Pedrosa, C.S.G.; Puig-Pijuan, T.; Veríssimo, C.P.; Ornelas, I.M.; Torquato, T.; et al. WIN 55,212-2 shows anti-inflammatory and survival properties in human iPSC-derived cardiomyocytes infected with SARS-CoV-2. PeerJ 2021, 9, e12262.





