
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Junjie Luo | -- | 1611 | 2022-04-13 04:57:16 | | | |
| 2 | Lindsay Dong | Meta information modification | 1611 | 2022-04-13 08:21:27 | | |
Video Upload Options
Ferrous sulfate is a commonly used iron supplement for the correction of iron-deficiency anemia but with frequent gastrointestinal side effects. Milk-derived iron-binding glycoprotein lactoferrin possesses well gastrointestinal tolerance and fewer side effects caused by the intake of high-dose iron. However, the underlying mechanism of the iron-enhancing effect of lactoferrin remains unclear. In addition, the comparative efficacies between lactoferrin and ferrous sulfate are also remained to be determined. Studies support lactoferrin as a superior supplement to ferrous sulfate regarding the improvement in serum iron parameters and hemoglobin levels.
1. Introduction
2. Lactoferrin and Ferrous Sulfate Supplementation in Iron-Deficiency Anemia
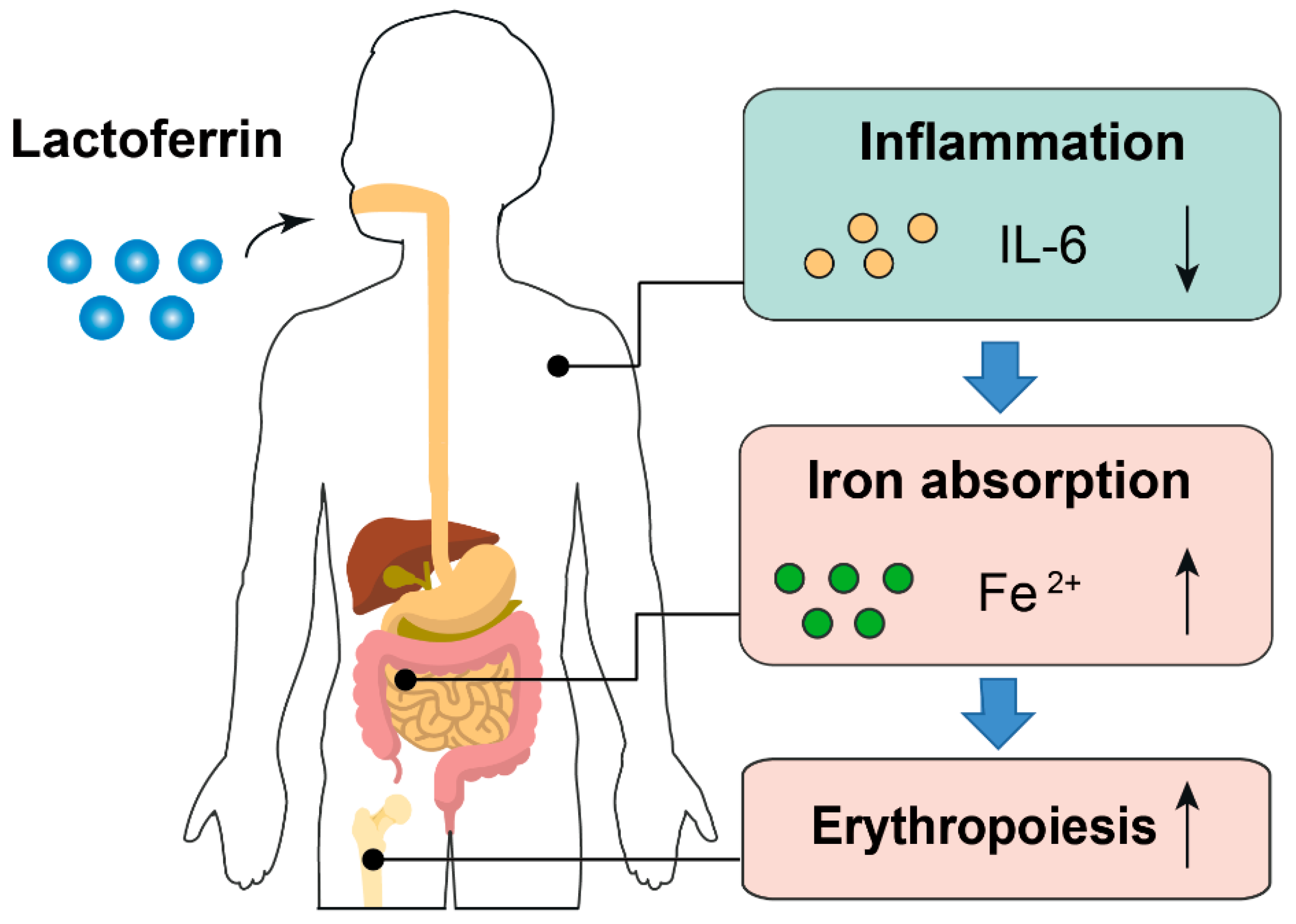
Lactoferrin is a multifunctional iron-binding protein possessing anti-inflammatory and anti-microbial effects. The major advantages of lactoferrin compared with traditional iron supplement ferrous sulfate are well gastrointestinal tolerance and fewer side effects caused by the intake of high-dose iron. Lactoferrin supplementation displayed better improvement for blood iron parameters including serum iron and ferritin when compared with ferrous sulfate. Moreover, a superior erythropoiesis-improving effect was observed in individuals receiving lactoferrin. Participants receiving lactoferrin had reduced IL-6 levels compared with those receiving ferrous sulfate, indicating lactoferrin mitigated inflammation.
Contradictorily, lactoferrin has less FIA than ferrous sulfate based on 3 studies [29][30], suggesting that the iron-improving effect of lactoferrin is not completely brought by the iron within lactoferrin. The lower FIA of lactoferrin is probably due to its iron-binding activity, which sequesters iron rather than promotes iron absorption in the intestine [30]. This hypothesis can be supported by the phenotypes of lactoferrin knockout mice, in which increased serum transferrin saturation and liver iron stores were observed [31]. However, there is an inconsistent explanation about the effect of iron-binding capacity of lactoferrin on iron absorption. It regarded that lactoferrin may sequester dietary iron from binding to iron-chelating compounds in foods (such as polyphenols and phytic acid), which promotes extra iron absorption in foods. This hypothesis can be supported by the results from isotope-labeled iron absorption trial using apo-lactoferrin [30]. Higher FIA was observed in individuals receiving apo-lactoferrin than those who received holo-lactoferrin.
The anti-inflammatory effect of lactoferrin can partially explain why it had a better iron-improving effect even with lower FIA than ferrous sulfate. First, higher inflammatory cytokines will prevent dietary iron absorption and iron mobilization from reticuloendothelial system to meet the need of erythropoiesis because of increased hepcidin synthesis [32][33]. Second, amelioration of inflammation can improve erythropoiesis. Inflammatory cytokines such as IL-6 is a repressive factor for erythropoiesis [34] (Figure 1). Notably, the iron-binding capacity also confers lactoferrin the property of limiting the growth of gut pathogens, which requires iron to proliferate. Simultaneously, lactoferrin can also enhance the growth of gut-beneficial microorganisms such as Bifidobacterium and Lactobacillus [35]. These probiotics have been proven to reduce the adverse effects associated with iron supplementation [36][37].
References
- Donker, A.E.; van der Staaij, H. The critical roles of iron during the journey from fetus to adolescent: Developmental aspects of iron homeostasis. Blood Rev. 2021, 50, 100866.
- Lopez, A.; Cacoub, P. Iron deficiency anaemia. Lancet 2016, 387, 907–916.
- Dev, S.; Babitt, J.L. Overview of iron metabolism in health and disease. Hemodial. Int. 2017, 21 (Suppl. 1), S6–S20.
- Sundararajan, S.; Rabe, H. Prevention of iron deficiency anemia in infants and toddlers. Pediatr. Res. 2021, 89, 63–73.
- Camaschella, C. Iron deficiency. Blood 2019, 133, 30–39.
- Pasricha, S.-R.; Tye-Din, J.; Muckenthaler, M.U.; Swinkels, D.W. Iron deficiency. Lancet 2021, 397, 233–248.
- Vos, T.; Abajobir, A.A.; Abate, K.H.; Abbafati, C.; Abbas, K.M.; Abd-Allah, F.; Abdulkader, R.S.; Abdulle, A.M.; Abebo, T.A.; Abera, S.F.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1211–1259.
- World Health Organization. Global Anaemia Reduction Efforts among Women of Reproductive Age: Impact, Achievement of Targets and the Way forward for Optimizing Efforts; World Health Organization: Geneva, Switzerland, 2020; ISBN 978-924-001-220-2.
- Kassebaum, N.J.; Jasrasaria, R.; Naghavi, M.; Wulf, S.K.; Johns, N.; Lozano, R.; Regan, M.; Weatherall, D.; Chou, D.P.; Eisele, T.P.; et al. A systematic analysis of global anemia burden from 1990 to 2010. Blood 2014, 123, 615–624.
- Stevens, G.A.; Finucane, M.M.; De-Regil, L.M.; Paciorek, C.J.; Flaxman, S.R.; Branca, F.; Peña-Rosas, J.P.; Bhutta, Z.A.; Ezzati, M.; on behalf of Nutrition Impact Model Study Group (Anaemia). Global, regional, and national trends in haemoglobin concentration and prevalence of total and severe anaemia in children and pregnant and non-pregnant women for 1995–2011: A systematic analysis of population-representative data. Lancet Glob. Health 2013, 1, e16–e25.
- World Health Organization. The Global Prevalence of Anaemia in 2011; World Health Organization: Geneva, Switzerland, 2015.
- Daru, J.; Zamora, J.; Fernández-Félix, B.M.; Vogel, J.; Oladapo, O.T.; Morisaki, N.; Tunçalp, Ö.; Torloni, M.R.; Mittal, S.; Jayaratne, K.; et al. Risk of maternal mortality in women with severe anaemia during pregnancy and post partum: A multilevel analysis. Lancet Glob. Health 2018, 6, e548–e554.
- Breymann, C. Iron Deficiency Anemia in Pregnancy. Semin Hematol. 2015, 52, 339–347.
- Global Nutrition Targets 2025: Anaemia Policy Brief; World Health Organization: Geneva, Switzerland, 2019.
- Azzopardi, P.S.; Hearps, S.J.; Francis, K.L.; Kennedy, E.C.; Mokdad, A.H.; Kassebaum, N.J.; Lim, S.; Irvine, C.M.S.; Vos, T.; Brown, A.D.; et al. Progress in adolescent health and wellbeing: Tracking 12 headline indicators for 195 countries and territories, 1990–2016. Lancet 2019, 393, 1101–1118.
- Donovan, A.; Brownlie, A.; Zhou, Y.; Shepard, J.; Pratt, S.J.; Moynihan, J.; Paw, B.H.; Drejer, A.; Barut, B.; Zapata, A.; et al. Positional cloning of zebrafish ferroportin1 identifies a conserved vertebrate iron exporter. Nature 2000, 403, 776–781.
- Pigeon, C.; Ilyin, G.; Courselaud, B.; Leroyer, P.; Turlin, B.; Brissot, P.; Loréal, O. A new mouse liver-specific gene, encoding a protein homologous to human antimicrobial peptide hepcidin, is overexpressed during iron overload. J. Biol. Chem. 2001, 276, 7811–7819.
- Nemeth, E.; Tuttle, M.S.; Powelson, J.; Vaughn, M.B.; Donovan, A.; Ward, D.M.V.; Ganz, T.; Kaplan, J. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science 2004, 306, 2090–2093.
- Muñoz, M.; García-Erce, J.A.; Remacha, Á.F. Disorders of iron metabolism. Part 1: Molecular basis of iron homoeostasis. J. Clin. Pathol. 2011, 64, 281–286.
- Lee, P.; Peng, H.; Gelbart, T.; Wang, L.; Beutler, E. Regulation of hepcidin transcription by interleukin-1 and interleukin-6. Proc. Natl. Acad. Sci. USA 2005, 102, 1906–1910.
- Wrighting, D.M.; Andrews, N.C. Interleukin-6 induces hepcidin expression through STAT3. Blood 2006, 108, 3204–3209.
- Das, N.K.; Schwartz, A.J.; Barthel, G.; Inohara, N.; Liu, Q.; Sankar, A.; Hill, D.R.; Ma, X.; Lamberg, O.; Schnizlein, M.K.; et al. Microbial Metabolite Signaling Is Required for Systemic Iron Homeostasis. Cell Metab. 2020, 31, 115–130.e116.
- Griffin, I.J. The Effects of Different Forms of Lactoferrin on Iron Absorption. J. Nutr. 2020, 150, 3053–3054.
- Ke, C.; Lan, Z.; Hua, L.; Ying, Z.; Humina, X.; Jia, S.; Weizheng, T.; Ping, Y.; Lingying, C.; Meng, M. Iron metabolism in infants: Influence of bovine lactoferrin from iron-fortified formula. Nutrition 2015, 31, 304–309.
- Macciò, A.; Madeddu, C. Efficacy and Safety of Oral Lactoferrin Supplementation in Combination with rHuEPO-β for the Treatment of Anemia in Advanced Cancer Patients Undergoing Chemotherapy: Open-Label, Randomized Controlled Study. Oncologist 2010, 15, 894–902.
- Rosa, L.; Lepanto, M.S.; Cutone, A.; Siciliano, R.A.; Paesano, R.; Costi, R.; Musci, G.; Valenti, P. Influence of oral administration mode on the efficacy of commercial bovine Lactoferrin against iron and inflammatory homeostasis disorders. Biometals 2020, 33, 159–168.
- Lambert, L.A.; Perri, H.; Meehan, T. Evolution of duplications in the transferrin family of proteins. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2005, 140, 11–25.
- Kruzel, M.L.; Olszewska, P.; Pazdrak, B.; Krupinska, A.M.; Actor, J.K. New insights into the systemic effects of oral lactoferrin: Transcriptome profiling. Biochem. Cell Biol. 2021, 99, 47–53.
- Lönnerdal, B.; Bryant, A. Absorption of iron from recombinant human lactoferrin in young US women. Am. J. Clin. Nutr. 2006, 83, 305–309.
- Mikulic, N.; Uyoga, M.A.; Mwasi, E.; Stoffel, N.U.; Zeder, C.; Karanja, S.; Zimmermann, M.B. Iron Absorption is Greater from Apo-Lactoferrin and is Similar Between Holo-Lactoferrin and Ferrous Sulfate: Stable Iron Isotope Studies in Kenyan Infants. J. Nutr. 2020, 150, 3200–3207.
- Ward, P.P.; Mendoza-Meneses, M.; Cunningham, G.A.; Conneely, O.M. Iron status in mice carrying a targeted disruption of lactoferrin. Mol. Cell Biol. 2003, 23, 178–185.
- Roy, C.N.; Mak, H.H.; Akpan, I.; Losyev, G.; Zurakowski, D.; Andrews, N.C. Hepcidin antimicrobial peptide transgenic mice exhibit features of the anemia of inflammation. Blood 2007, 109, 4038–4044.
- Nicolas, G.; Bennoun, M.; Porteu, A.; Mativet, S.; Beaumont, C.; Grandchamp, B.; Sirito, M.; Sawadogo, M.; Kahn, A.; Vaulont, S. Severe iron deficiency anemia in transgenic mice expressing liver hepcidin. Proc. Natl. Acad. Sci. USA 2002, 99, 4596–4601.
- Jeong, J.Y.; Silver, M.; Parnes, A.; Nikiforow, S.; Berliner, N.; Vanasse, G.J. Resveratrol ameliorates TNFα-mediated suppression of erythropoiesis in human CD34+ cells via modulation of NF-κB signalling. Br. J. Haematol. 2011, 155, 93–101.
- Vega-Bautista, A.; de la Garza, M.; Carrero, J.C.; Campos-Rodríguez, R.; Godínez-Victoria, M.; Drago-Serrano, M.E. The Impact of Lactoferrin on the Growth of Intestinal Inhabitant Bacteria. Int. J. Mol. Sci. 2019, 20, 4707.
- Paganini, D.; Uyoga, M.A.; Kortman, G.A.M.; Cercamondi, C.I.; Moretti, D.; Barth-Jaeggi, T.; Schwab, C.; Boekhorst, J.; Timmerman, H.M.; Lacroix, C.; et al. Prebiotic galacto-oligosaccharides mitigate the adverse effects of iron fortification on the gut microbiome: A randomised controlled study in Kenyan infants. Gut 2017, 66, 1956–1967.
- Paganini, D.; Uyoga, M.A.; Cercamondi, C.I.; Moretti, D.; Mwasi, E.; Schwab, C.; Bechtler, S.; Mutuku, F.M.; Galetti, V.; Lacroix, C.; et al. Consumption of galacto-oligosaccharides increases iron absorption from a micronutrient powder containing ferrous fumarate and sodium iron EDTA: A stable-isotope study in Kenyan infants. Am. J. Clin. Nutr. 2017, 106, 1020–1031.
- Main Elements of Proposed Model. Available online: https://www.freepik.com/ (accessed on 20 January 2022).
- Main Elements of Proposed Model. Available online: http://smart.servier.com/ (accessed on 20 January 2022).
- Muñoz, M.; García-Erce, J.A.; Remacha, F. Disorders of iron metabolism. Part II: Iron deficiency and iron overload. J. Clin. Pathol. 2011, 64, 287–296.





