Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kariem Ezzat | -- | 2059 | 2022-04-08 19:16:33 | | | |
| 2 | Amina Yu | -18 word(s) | 2041 | 2022-04-11 03:17:16 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Ezzat, K.; Sturchio, A.; Espay, A. Amyloid Pathologies. Encyclopedia. Available online: https://encyclopedia.pub/entry/21527 (accessed on 26 March 2026).
Ezzat K, Sturchio A, Espay A. Amyloid Pathologies. Encyclopedia. Available at: https://encyclopedia.pub/entry/21527. Accessed March 26, 2026.
Ezzat, Kariem, Andrea Sturchio, Alberto Espay. "Amyloid Pathologies" Encyclopedia, https://encyclopedia.pub/entry/21527 (accessed March 26, 2026).
Ezzat, K., Sturchio, A., & Espay, A. (2022, April 08). Amyloid Pathologies. In Encyclopedia. https://encyclopedia.pub/entry/21527
Ezzat, Kariem, et al. "Amyloid Pathologies." Encyclopedia. Web. 08 April, 2022.
Copy Citation
Protein aggregation into amyloid fibrils affects many proteins in a variety of diseases, including neurodegenerative disorders, diabetes, and cancer. Physicochemically, amyloid formation is a phase transition process, where soluble proteins are transformed into solid fibrils with the characteristic cross-β conformation responsible for their fibrillar morphology.
amyloid
prion
Alzheimer’s
Parkinson’s
Creutzfeldt–Jakob disease (CJD)
1. What Are Amyloids?
Proteins, like any other molecules, can exist in different states or phases depending on their packing density. Similar to gas, liquid, and solid phases of water, for example, (water vapor, liquid water, and ice), proteins can be soluble or colloidally dispersed in the aqueous biological environment, concentrated in liquid droplets that form a separate liquid phase within the aqueous environment, or in a tightly packed solid state. The liquid–liquid phase separation of proteins has been intensively investigated recently [1][2]. It was focused on amyloids as specific form of protein solids.
There are two main types of protein solids that form in-vivo:
-
Fibrous proteins, such as actin, elastin, and collagen;
-
Amyloids, which are associated with many human diseases.
While both types of in vivo protein solids share similar physicochemical mechanisms of formation (nucleation and growth, see below), they differ in many fundamental ways. Fibrous proteins, such as actin or collagen fibers, are formed from a specific group of proteins, where the monomers (for examples G-actin and tropocollagen for actin and collagen fibers, respectively) are natively folded before assembling into fibers in a controlled and reversible manner, which involves energy-dependent processes (enzymes and ATP) [3][4][5]. In contrast, amyloid fibrils can be formed by almost any amino acid sequence, from globular nonfibrous proteins, such as myoglobin [6], insulin [7], and albumin, [8] to simple polylysines, polyglutamates, and polythreonines sequences [9]. Such generic nature- and sequence-independence indicate that the architecture of the amyloid state is not encoded in the primary sequence of proteins [10][11][12][13]. Unlike native protein folding, which depends on specific intramolecular interactions between the side chains of a particular sequence, the structure of the amyloid state is dominated by intermolecular interactions via the backbone that is common to all proteins. Consequently, amyloids from different proteins possess a common core conformation, the cross-β conformation, where intermolecular β-sheets pair tightly together with their side chains interdigitating (like zipper teeth) excluding water to form the so-called “dry steric zipper” (Figure 1) [14][15][16][17][18]. The intermolecular β-sheets form via a generic interbackbone hydrogen-bonding network between the amide N-H and C=O groups of adjacent protein molecules [19] and can comprise up to thousands of molecules extending for µm distances [14]. Within the β-sheet ladder, β strands are spaced 4.8 A° and the distance between opposite β sheets are in the range of 6–12 A° (Figure 1), which gives rise to the characteristic amyloid X-ray diffraction pattern with meridional and equatorial reflections of similar values, respectively [16][18]. Extended ladders of interdigitating β-sheet pairs form the core spine of the superstructural subunit of amyloids, the protofilament. A protofilament can accommodate a single or multiple steric zippers in different arrangements with different mating/interdigitation options between the side chains of the constituting ladders [15]. Protofilaments further associate laterally into fibrils, which further associate and precipitate as insoluble plaques, characteristic of tissues affected by amyloidosis [20].
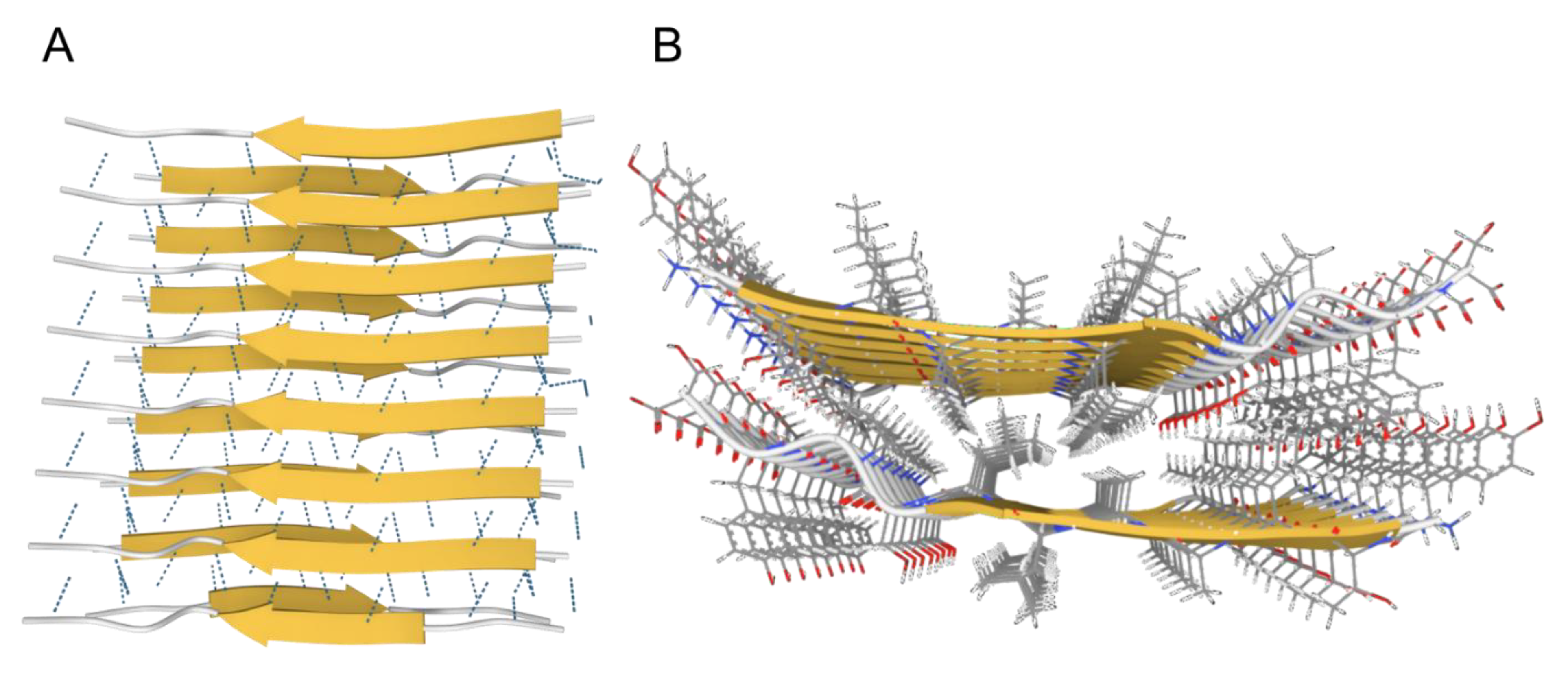
Figure 1. The cross-β conformation of amyloids. (A). Side-view showing the intermolecular β-sheets stabilized by hydrogen bonds (dotted lines). (B). Top-view showing the dry steric zipper between the two opposing β-sheets with interdigitating sidechains. Images created using Mol* [21] from PDB structure 2M5N from paper by Fitzpatrick et al., 2013 [22].
Another major difference between native protein folding and the cross-β conformation of amyloids is that the interdigitation of side chains between β-sheet ladders generally deprives amyloids from any characteristic domains. Additionally, the extensive, hierarchical self-interaction (ladders within zippers, zippers within protofilaments, protofilaments within fibrils, and fibrils within plaques) makes amyloids very stable and, consequently, extremely difficult to solubilize and relatively inert. The aggregated, plaque nature of amyloids is, again, in contrast with functional fibrous proteins which assemble in well-defined networks by accessory proteins [23]. The difference between functional fibrous proteins and amyloids is summarized in Table 1.
Table 1. The differences between fibrous proteins and amyloid fibrils.
| Fibrous Proteins | Amyloid Fibrils |
|---|---|
| Specific proteins | Any protein sequence |
| Monomers assemble in their native conformation via specific intramolecular sidechain-based interactions | Proteins assemble into in cross-β conformation via generic intermolecular backbone interactions |
| Functional domains remain accessible | Majority of functional domains are buried in steric zippers |
| Form well-defined networks | Precipitate into plaques |
| Controlled nucleation and growth via structural elements (proline and glycine rich), capping proteins, specific nucleators, enzymes and ATP | Uncontrolled |
| Reversible | Irreversible |
Despite the universal cross-β conformation of any amyloid, the difference in number or arrangement of steric zippers within a protofilament and/or the number and arrangement of protofilaments within a fibril result in different polymorphs. Unlike the cross-β conformation which is structurally encoded in any protein sequence via generic inter-backbone hydrogen-bonding, polymorphism is dependent on environmental factors such as temperature, pH, concentration, and shaking; extrinsic factors that are not structurally encoded [24]. For example, with the same sequence, different polymorphic shapes of Aβ40 fibrils can be produced in quiescent versus agitating conditions [25], and the presence or absence of polyanions leads to the production of different polymorphs of α-synuclein fibrils [26]. The interaction between environmental factors and the protein sequence affects polymorphism by affecting the patterns of β-sheet ladder stacking and zipper interdigitation, which can also lead to different polymorphs of different sequences under same conditions [15].
As proteins have the information to fold natively in the primary sequence of side chains (Anfinsen’s dogma [27][28]), they also holds the necessary information to form the cross-β conformation based intermolecular backbone interactions, which requires molecular proximity. This is why amyloid formation requires supersaturated conditions and the likelihood of a protein forming an amyloid increases with concentration [29][30]. Above a certain concentration, the molecular proximity renders the intermolecular interactions more favorable than intramolecular interactions responsible for native folding, leading to amyloid formation. This intermolecular interaction will result in molecular packing, phase transition, and precipitation out of the aqueous biological environment.
2. How Do Amyloids Cause Toxicity?
Amyloid formation involves three pathological protein transformations: structural, from natively folded to the cross-β conformation; biophysical, from soluble to insoluble; and biological, from functional to non-functional [31]. The cross-β conformation buries the once-functional domains of the protein within the steric zipper architecture, which makes amyloids extremely stable [14], and, consequently, relatively inert. Additionally, the uncontrolled phase transition leads to loss of protein solubility and colloidal stability resulting in precipitation into plaques, which further buries any potential unpaired side chains via hierarchal self-interaction. Within plaques, amyloid protofilaments and fibrils adopt different polymorphic morphologies depending on environmental conditions. However, since all polymorphs are based on the same cross-β conformation, where the functional side chain domains are sequestered, they are expected to have similar, generic amyloid properties in terms of stability, insolubility, and low reactivity. Furthermore, the loss of protein solubility and colloidal stability favors precipitation and cluster formation over propagation of single fibrils, which requires colloidal dispersion. This is supported by clinical findings that plaques (heterogenous fibrillar clusters) are the hallmarks of amyloid pathologies.
With the high stability and low reactivity, amyloids pose little direct toxicity unless they physically remodel a tissue, for example, in the rare situations of systemic amyloidosis such as immunoglobulin light chain amyloidosis and transthyretin amyloidosis [32][33]. This is especially true for amyloid accumulation within the muscle tissue (for example, cardiac amyloidosis), where amyloid infiltration physically impair muscle contractility [34]. However, in many others, amyloids exist as a benign mass similar to other benign masses, such as fibromas and lipomas. In insulin-derived amyloidosis, for example, repeated injection of insulin subcutaneously in the same spot leads to the creation of insulin amyloid lumps in some diabetic patients [35]. Despite the benign nature of such lumps, patients lose the ability to control glucose levels due to insulin sequestration in the form of amyloid aggregates [36]. Patients are, therefore, instructed to change the location of insulin injections to avoid local aggregation. The toxicity due to amyloid aggregation is due to loss-of-function (LOF) of the injected insulin and not due direct toxicity from the amyloid mass. Pathogenesis due to LOF is also demonstrated for p53 amyloid formation. P53 is a tumor suppressor protein whose dysregulation or inactivation is involved in more than 50% of all cancers [37]. It has been shown that p53 can form amyloid fibrils leading to enhanced cell proliferation due to its sequestration and LOF [38][39]. These findings clearly indicate that amyloids are not necessarily cytotoxic as they can enhance, not impair, cell proliferation via a LOF mechanism of p53. LOF is also the mechanism behind many phenotypes in yeast due to amyloid formation [40]. For example, amyloid formation of Sup35, which is an essential translation termination factor, induces lethality due to LOF as a result of its sequestration in the amyloid state [41]. Such an outcome can be reversed by supplying the yeast with a modified version of Sup35, where the residues more prone to amyloid formation are removed, while the domains involved in translation termination are maintained [42]. This replacement approach to overcome amyloid LOF toxicity is also utilized clinically in the treatment of diabetes mellites by using pramlintide, which is a less aggregating analogue of the peptide hormone amylin, whose amyloid aggregation in the pancreas and depletion in the circulation is common among diabetic patients [43]. The relatively benign nature of amyloid plaques can also be seen in neurodegenerative diseases such as AD, where up 30% of individuals who have plaques in their brains are cognitively normal [44]. It was recently shown that higher levels of soluble Aβ42 are associated with normal cognition and preservation of brain volume among amyloid positive individuals, regardless of and despite increasing levels of brain amyloid, indicating that LOF of the soluble Aβ42 is more detrimental to neurons than direct gain-of- function (GOF) toxicity from plaques [45]. This suggests that a replacement approach might also be feasible for AD treatment and other neurodegenerative diseases, an alternative to continuing with anti-amyloid strategies, which have invariably failed [46].
“Toxic oligomers”? Oligomers have been postulated to explain the lack of association between amyloid plaque load and toxicity, especially in AD. The term oligomers denotes low and medium molecular weight aggregates that are assumed to mediate the amyloid toxicity [47]. However, the evidence of their toxicity has been shown in vitro, not clinically, and the clinically relevant toxic oligomer remains unknown [48]. Moreover, under supersaturated conditions, which are necessary for amyloid formation, the distinction between oligomers and nuclei is hard to make, since the formation of any cluster stable enough will trigger phase transition into fibrils, whereas unstable clusters will dissociate back into monomers (see above). This has been demonstrated experimentally, where the majority of oligomers were shown to dissociate into monomers, in good accordance with the classical nucleation theory [49]. Moreover, the reduction of soluble Aβ42 levels during the disease course reduces the substrate for the oligomers, questioning the long-term effect of such species. This is supported by the fact that attempts to quantify oligomeric species of Aβ found that they are less in AD patients compared to controls [50][51][52].
“Ratios”. Despite the progressive decrease in the absolute levels of soluble Aβ42 in AD, the ratio of Aβ42 relative to other shorter versions of the peptide, such as Aβ40, was hypothesized to increase the likelihood of Aβ42 forming aggregates due to its more amyloidogenic nature [53]. However, amyloid aggregation is dependent on supersaturation, which is dependent on the absolute concentration of the peptide. Decreasing peptide concentration will decrease, not increase, its propensity to form any type of aggregates irrespective of its relative levels compared to other peptides. Moreover, it has been recently demonstrated that CSF Aβ42/Aβ40 ratio also decreases during the course of AD [54].
Another indication on the importance of LOF as a pathogenic mechanism in neurodegenerative diseases is the fact that animal models where the amyloidogenic proteins were knocked out or down display phenotypes that resemble those obtained by protein overexpression and aggregation. This has been demonstrated for Aβ42 [55][56] in AD, α-synuclein in Parkinson’s disease [57][58][59], and other neurodegeneration-related proteins that form amyloids such as Tau [60], PrP [61], SOD1 [62], and TDP43 [63]. The similarity of phenotype in both the presence and absence of aggregates can only be explained by LOF mechanism, where the sequestration of protein due to aggregation mimics the effects of gene knock down. Additionally, the phenotype can be rescued by restoration of normal soluble levels of these proteins [56][58]. For further discussion on LOF toxicity, these was referred to [31][64].
References
- Brangwynne, C.P. Liquid phase condensation in cell physiology and disease. Science 2017, 357, eaaf4382.
- Alberti, S.; Gladfelter, A.; Mittag, T. Considerations and challenges in studying liquid-liquid phase separation and biomolecular condensates. Cell 2019, 176, 419–434.
- Ozsvar, J.; Yang, C.; Cain, S.A.; Baldock, C.; Tarakanova, A.; Weiss, A.S. Tropoelastin and elastin assembly. Front. Bioeng. Biotechnol. 2021, 9, 138.
- Dominguez, R.; Holmes, K.C. Actin structure and function. Annu. Rev. Biophys. 2011, 40, 169–186.
- Shoulders, M.D.; Raines, R.T. Collagen structure and stability. Annu. Rev. Biochem. 2009, 78, 929–958.
- Fändrich, M.; Fletcher, M.A.; Dobson, C.M. Amyloid fibrils from muscle myoglobin. Nature 2001, 410, 165–166.
- Jiménez, J.L.; Nettleton, E.J.; Bouchard, M.; Robinson, C.V.; Dobson, C.M.; Saibil, H.R. The protofilament structure of insulin amyloid fibrils. Proc. Natl. Acad. Sci. USA 2002, 99, 9196–9201.
- Taboada, P.; Barbosa, S.; Castro, E.; Mosquera, V. Amyloid fibril formation and other aggregate species formed by human serum albumin association. J. Phys. Chem. B 2006, 110, 20733–20736.
- Fändrich, M.; Dobson, C.M. The behaviour of polyamino acids reveals an inverse side chain effect in amyloid structure formation. EMBO J. 2002, 21, 5682–5690.
- Dobson, C.M. The amyloid phenomenon and its links with human disease. Cold Spring Harb. Perspect. Biol. 2017, 9, a023648.
- Ross, E.D.; Edskes, H.K.; Terry, M.J.; Wickner, R.B. Primary sequence independence for prion formation. Proc. Natl. Acad. Sci. USA 2005, 102, 12825–12830.
- Christopher, M. Dobson Protein misfolding, evolution and disease. Trends Biochem. Sci. 1999, 24, 329–332.
- Dobson, C.M. Protein folding and misfolding. Nature 2003, 426, 884–890.
- Eisenberg, D.S.; Sawaya, M.R. Structural studies of amyloid proteins at the molecular level. Annu. Rev. Biochem. 2017, 86, 69–95.
- Sawaya, M.R.; Hughes, M.P.; Rodriguez, J.A.; Riek, R.; Eisenberg, D.S. The expanding amyloid family: Structure, stability, function, and pathogenesis. Cell 2021, 184, 4857–4873.
- Riek, R.; Eisenberg, D.S. The activities of amyloids from a structural perspective. Nature 2016, 539, 227–235.
- Eisenberg, D.; Jucker, M. The amyloid state of proteins in human diseases. Cell 2012, 148, 1188–1203.
- Ke, P.C.; Zhou, R.; Serpell, L.C.; Riek, R.; Knowles, T.P.J.; Lashuel, H.A.; Gazit, E.; Hamley, I.W.; Davis, T.P.; Fa, M.; et al. Half a century of amyloids: Past, present and future. Chem. Soc. Rev. 2020, 49, 5473–5509.
- Knowles, T.P.; Fitzpatrick, A.W.; Meehan, S.; Mott, H.R.; Vendruscolo, M.; Dobson, C.M.; Welland, M.E. Role of intermolecular forces in defining material properties of protein nanofibrils. Science 2007, 318, 1900–1903.
- Chiti, F.; Dobson, C.M. Protein misfolding, amyloid formation, and human disease: A summary of progress over the last decade. Annu. Rev. Biochem. 2017, 86, 27–68.
- Sehnal, D.; Bittrich, S.; Deshpande, M.; Svobodová, R.; Berka, K.; Bazgier, V.; Velankar, S.; Burley, S.K.; Koča, J.; Rose, A.S. Mol* viewer: Modern web app for 3D visualization and analysis of large biomolecular structures. Nucleic Acids Res. 2021, 49, W431–W437.
- Fitzpatrick, A.W.P.; Debelouchina, G.T.; Bayro, M.J.; Clare, D.K.; Caporini, M.A.; Bajaj, V.S.; Jaroniec, C.P.; Wang, L.; Ladizhansky, V.; Müller, S.A.; et al. Atomic structure and hierarchical assembly of a cross-β amyloid fibril. Proc. Natl. Acad. Sci. USA 2013, 110, 5468–5473.
- Lieleg, O.; Claessens, M.M.A.E.; Bausch, A.R. Structure and dynamics of cross-linked actin networks. Soft Matter 2010, 6, 218–225.
- Fändrich, M.; Schmidt, M.; Grigorieff, N. Recent progress in understanding Alzheimer’s β-amyloid structures. Trends Biochem. Sci. 2011, 36, 338–345.
- Petkova, A.T.; Leapman, R.D.; Guo, Z.; Yau, W. Self-propagating, molecular-level polymorphism in Alzheimer’s ß-amyloid fibrils. Science 2005, 307, 262–266.
- Guerrero-Ferreira, R.; Taylor, N.M.I.; Arteni, A. Two new polymorphic structures of human full-length alpha-synuclein fibrils solved by cryo-electron microscopy. eLife 2019, 8, e48907.
- Kresge, N.; Simoni, R.D.; Hill, R.L. The thermodynamic hypothesis of protein folding: The work of Christian Anfinsen. J. Biol. Chem. 2006, 281, e11–e13.
- Anfinsen, C.B. Principles that govern the folding of protein chains. Science 1973, 181, 223–230.
- Ciryam, P.; Kundra, R.; Morimoto, R.I.; Dobson, C.M.; Vendruscolo, M. Supersaturation is a major driving force for protein aggregation in neurodegenerative diseases. Trends Pharmacol. Sci. 2015, 36, 72–77.
- Jarrett, J.T.; Lansbury, P.T. Seeding “one dimensional cristallization” of amyloid: A pathogenic mechanism in Alzheimer’s disease and scrapie? Cell 1993, 73, 1055–1058.
- Malmberg, M.; Malm, T.; Gustafsson, O.; Wright, A.; El Andaloussi, S.; Ezzat, K. Disentangling the amyloid pathways: A mechanistic approach to etiology. Front. Neurosci. 2020, 14, 256.
- D’Aguanno, V.; Ralli, M.; Artico, M.; Russo, F.Y.; Scarpa, A.; Fiore, M.; Tirassa, P.; Severini, C.; de Vincentiis, M.; Greco, A. Systemic amyloidosis: A contemporary overview. Clin. Rev. Allergy Immunol. 2019, 59, 304–322.
- Gertz, M.A.; Dispenzieri, A. Systemic amyloidosis recognition, prognosis, and therapy: A systematic review. JAMA 2020, 324, 79–89.
- Shah, K.B.; Inoue, Y.; Mehra, M.R. Amyloidosis and the heart: A comprehensive review. Arch. Intern. Med. 2006, 166, 1805–1813.
- Surin, A.K.; Grishin, S.Y.; Galzitskaya, O.V. Determination of amyloid core regions of insulin analogues fibrils. Prion 2020, 14, 149–162.
- Nagase, T.; Iwaya, K.; Iwaki, Y.; Kotake, F.; Uchida, R.; Oh-I, T.; Sekine, H.; Miwa, K.; Murakami, S.; Odaka, T.; et al. Insulin-derived amyloidosis and poor glycemic control: A case series. Am. J. Med. 2014, 127, 450–454.
- Levine, A.J.; Oren, M. The first 30 years of p53: Growing ever more complex. Nat. Rev. Cancer 2009, 9, 749–758.
- Silva, J.L.; Gallo, C.V.D.M.; Costa, D.C.F.; Rangel, L.P.; De Moura Gallo, C.V.; Costa, D.C.F.; Rangel, L.P. Prion-like aggregation of mutant p53 in cancer. Trends Biochem. Sci. 2014, 39, 260–267.
- Navalkar, A.; Ghosh, S.; Pandey, S.; Paul, A.; Datta, D.; Maji, S.K. Prion-like p53 amyloids in cancer. Biochemistry 2019, 59, 146–155.
- Liebman, S.W.; Chernoff, Y.O. Prions in yeast. Genetics 2012, 191, 1041–1072.
- Wickner, R.B.; Edskes, H.K.; Son, M.; Bezsonov, E.E.; DeWilde, M.; Ducatez, M. Yeast prions compared to functional prions and amyloids. J. Mol. Biol. 2018, 430, 3707–3719.
- McGlinchey, R.P.; Kryndushkin, D.; Wickner, R.B. Suicidal is a lethal yeast prion. Proc. Natl. Acad. Sci. USA 2011, 108, 5337–5341.
- Denroche, H.C.; Verchere, C.B. IAPP and type 1 diabetes: Implications for immunity, metabolism and islet transplants. J. Mol. Endocrinol. 2018, 60, R57–R75.
- Chételat, G.; La Joie, R.; Villain, N.; Perrotin, A.; De La Sayette, V.; Eustache, F.; Vandenberghe, R. Amyloid imaging in cognitively normal individuals, at-risk populations and preclinical Alzheimer’s disease. NeuroImage Clin. 2013, 2, 356–365.
- Sturchio, A.; Dwivedi, A.K.; Young, C.B.; Malm, T.; Marsili, L.; Sharma, J.S.; Mahajan, A.; Hill, E.J.; El Andaloussi, S.; Poston, K.L.; et al. High cerebrospinal amyloid-β 42 is associated with normal cognition in individuals with brain amyloidosis. EClinicalMedicine 2021, 38, 100988.
- Panza, F.; Lozupone, M.; Logroscino, G.; Imbimbo, B.P. A critical appraisal of amyloid-β-targeting therapies for Alzheimer disease. Nat. Rev. Neurol. 2019, 15, 73–88.
- Selkoe, D.J.; Hardy, J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol. Med. 2016, 8, 595–608.
- Walsh, D.M.; Selkoe, D.J. Amyloid b-protein and beyond: The path forward in Alzheimer’s disease. Curr. Opin. Neurobiol. 2020, 61, 116–124.
- Dear, A.J.; Michaels, T.C.T.; Meisl, G.; Klenerman, D.; Wu, S.; Perrett, S.; Linse, S.; Dobson, C.M.; Knowles, T.P.J. Kinetic diversity of amyloid oligomers. Proc. Natl. Acad. Sci. USA 2020, 117, 201922267.
- Sancesario, G.M.; Cencioni, M.T.; Esposito, Z.; Borsellino, G.; Nuccetelli, M.; Martorana, A.; Battistini, L.; Sorge, R.; Spalletta, G.; Ferrazzoli, D.; et al. The load of amyloid-β oligomers is decreased in the cerebrospinal fluid of Alzheimer’s disease patients. J. Alzheimers Dis. 2012, 31, 865–878.
- Jongbloed, W.; Bruggink, K.A.; Kester, M.I.; Visser, P.J.; Scheltens, P.; Blankenstein, M.A.; Verbeek, M.M.; Teunissen, C.E.; Veerhuis, R. Amyloid-β oligomers relate to cognitive decline in alzheimer’s disease. J. Alzheimers Dis. 2015, 45, 35–43.
- Van Helmond, Z.; Miners, J.S.; Kehoe, P.G.; Love, S. Higher soluble amyloid β concentration in frontal cortex of young adults than in normal elderly or Alzheimer’s disease. Brain Pathol. 2010, 20, 787–793.
- Wolfe, M.S. When loss is gain: Reduced presenilin proteolytic function leads to increased Aβ42/Aβ40. Talking Point on the role of presenilin mutations in Alzheimer disease. EMBO Rep. 2007, 8, 136–140.
- Fagan, A.M.; Henson, R.L.; Li, Y.; Boerwinkle, A.H.; Xiong, C.; Bateman, R.J.; Goate, A.; Ances, B.M.; Doran, E.; Christian, B.T.; et al. Comparison of CSF biomarkers in down syndrome and autosomal dominant Alzheimer’s disease: A cross-sectional study. Lancet Neurol. 2021, 20, 615–626.
- Southam, K.A.; Stennard, F.; Pavez, C.; Small, D.H. Knockout of amyloid β protein precursor (APP) expression alters synaptogenesis, neurite branching and axonal morphology of hippocampal neurons. Neurochem. Res. 2019, 44, 1346–1355.
- Puzzo, D.; Privitera, L.; Fa, M.; Staniszewski, A.; Hashimoto, G.; Aziz, F.; Sakurai, M.; Ribe, E.M.; Troy, C.M.; Mercken, M.; et al. Endogenous amyloid-β is necessary for hippocampal synaptic plasticity and memory. Ann. Neurol. 2011, 69, 819–830.
- Benskey, M.J.; Sellnow, R.C.; Sandoval, I.M.; Sortwell, C.E.; Lipton, J.W.; Manfredsson, F.P. Silencing alpha synuclein in mature nigral neurons results in rapid neuroinflammation and subsequent toxicity. Front. Mol. Neurosci. 2018, 11, 36.
- Gorbatyuk, O.S.; Li, S.; Nash, K.; Gorbatyuk, M.; Lewin, A.S.; Sullivan, L.F.; Mandel, R.J.; Chen, W.; Meyers, C.; Manfredsson, F.P.; et al. In vivo RNAi-mediated α-synuclein silencing induces nigrostriatal degeneration. Mol. Ther. 2010, 18, 1450–1457.
- Collier, T.J.; Eugene Redmond, D.; Steece-Collier, K.; Lipton, J.W.; Manfredsson, F.P. Is alpha-synuclein loss-of-function a contributor to parkinsonian pathology? Evidence from non-human primates. Front. Neurosci. 2016, 10, 12.
- Velazquez, R.; Ferreira, E.; Tran, A.; Turner, E.C.; Belfiore, R.; Branca, C.; Oddo, S. Acute tau knockdown in the hippocampus of adult mice causes learning and memory deficits. Aging Cell 2018, 17, e12775.
- Bremer, J.; Baumann, F.; Tiberi, C.; Wessig, C.; Fischer, H.; Schwarz, P.; Steele, A.D.; Toyka, K.V.; Nave, K.A.; Weis, J.; et al. Axonal prion protein is required for peripheral myelin maintenance. Nat. Neurosci. 2010, 13, 310–318.
- Saccon, R.A.; Bunton-Stasyshyn, R.K.A.; Fisher, E.M.C.; Fratta, P. Is SOD1 loss of function involved in amyotrophic lateral sclerosis? Brain 2013, 136, 2342–2358.
- Kabashi, E.; Lin, L.; Tradewell, M.L.; Dion, P.A.; Bercier, V.; Bourgouin, P.; Rochefort, D.; Bel Hadj, S.; Durham, H.D.; Velde, C.V.; et al. Gain and loss of function of ALS-related mutations of TARDBP (TDP-43) cause motor deficits in vivo. Hum. Mol. Genet. 2009, 19, 671–683.
- Espay, A.J.; Sturchio, A.; Schneider, L.S.; Ezzat, K. Soluble Amyloid-β Consumption in Alzheimer’s Disease. J. Alzheimers Dis. 2021, 82, 1403–1415.
More
Information
Subjects:
Biophysics
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
808
Revisions:
2 times
(View History)
Update Date:
11 Apr 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





